Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (19): 2968-2974.doi: 10.3969/j.issn.2095-4344.2014.19.003
Previous Articles Next Articles
Melatonin inhibits adipogenic differentiation of bone marrow mesenchymal stem cells through retinoid-related orphan receptor alpha
Zhang Liang-ming1, Wang Xuan1, Gao Wen-jie2, Huang Dong-sheng2, Rong Li-min1
- 1Department of Spine Surgery, the Third Affiliated Hospital, Sun Yat-sen University, Guangzhou 510630, Guangdong Province, China
2Department of Spine Surgery, Sun Yat-sen Memorial Hospital, Sun Yat-sen University, Guangzhou 510120, Guangdong Province, China
-
Revised:2014-03-07Online:2014-05-07Published:2014-05-07 -
Contact:Rong Li-min, M.D., Professor, Chief physician, Doctoral supervisor, Department of Spine Surgery, the Third Affiliated Hospital, Sun Yat-sen University, Guangzhou 510630, Guangdong Province, China -
About author:Zhang Liang-ming, M.D., Physician, Department of Spine Surgery, the Third Affiliated Hospital, Sun Yat-sen University, Guangzhou 510630, Guangdong Province, China -
Supported by:the National Natural Science Foundation of China, No. 81301524, 81171674; the Natural Science Foundation of Guangdong Province, No. S2013040015484; the Specialized Research Fund for the Doctoral Program of Higher Education, No. 20110171110067
CLC Number:
Cite this article
Zhang Liang-ming, Wang Xuan, Gao Wen-jie, Huang Dong-sheng, Rong Li-min . Melatonin inhibits adipogenic differentiation of bone marrow mesenchymal stem cells through retinoid-related orphan receptor alpha[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(19): 2968-2974.
share this article
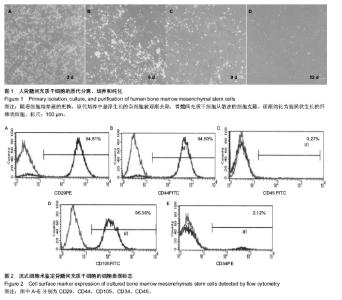
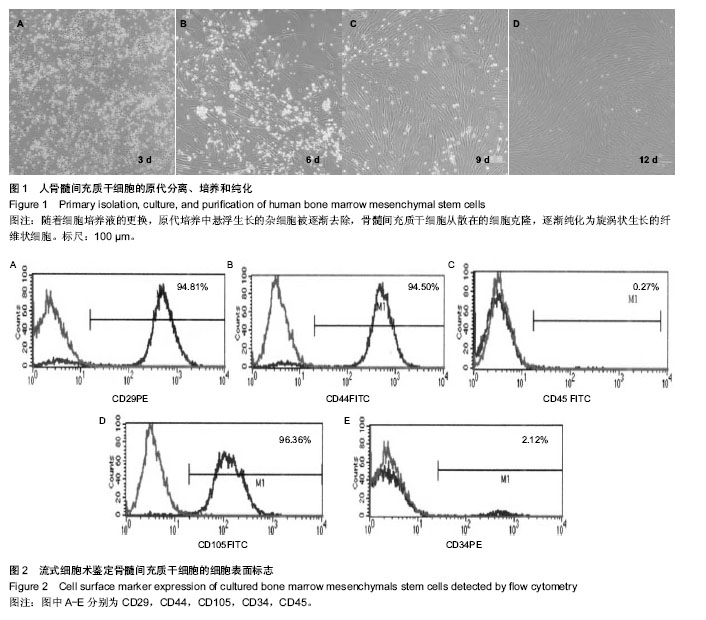
2.1 原代骨髓间充质干细胞的形态与鉴定 骨髓稀释液经过密度梯度离心后,骨髓稀释液被分成数层:血清、单个核细胞层、淋巴细胞分离液、血细胞层。小心吸取呈云雾状单个核细胞层进行原代细胞培养。培养早期以悬浮、折光性强的圆形细胞为主,通过换液悬浮细胞逐渐被清除。骨髓间充质干细胞开始时呈散在细胞克隆生长,为梭形纤维状,数量为3-10个不等。细胞克隆逐渐铺长,最后多个克隆细胞融合在一起,形成梭形纤维状细胞单层,细胞排列有方向性,呈平行、漩涡状生长。一般在10-12 d,细胞可以达到80%-90%融合(图1)。体外培养的第3代骨髓间充质干细胞,采用流式细胞仪计算荧光表达率,检测骨髓间充质干细胞的细胞表面CD分子表达情况。造血干细胞表面标记:CD34表达率2.12%,CD45表达率0.27%;间充质干细胞细胞表面标记:CD29表达率94.81%,CD44表达率94.50%,CD105表达率96.36%(图2)。"
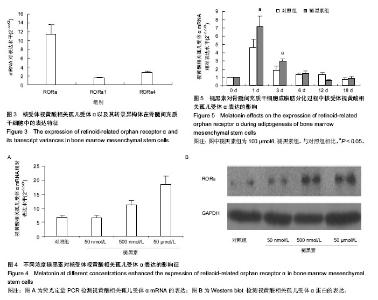
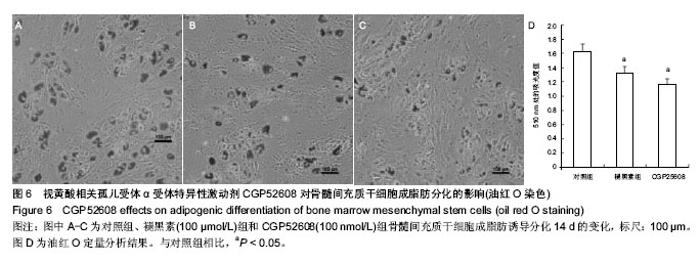
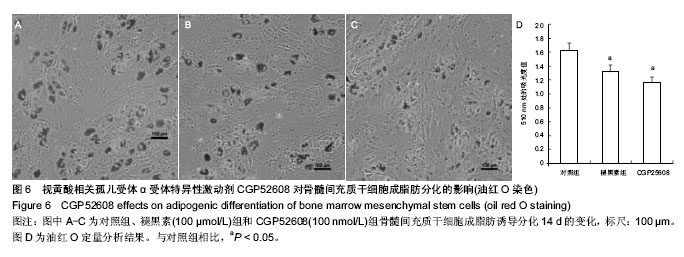
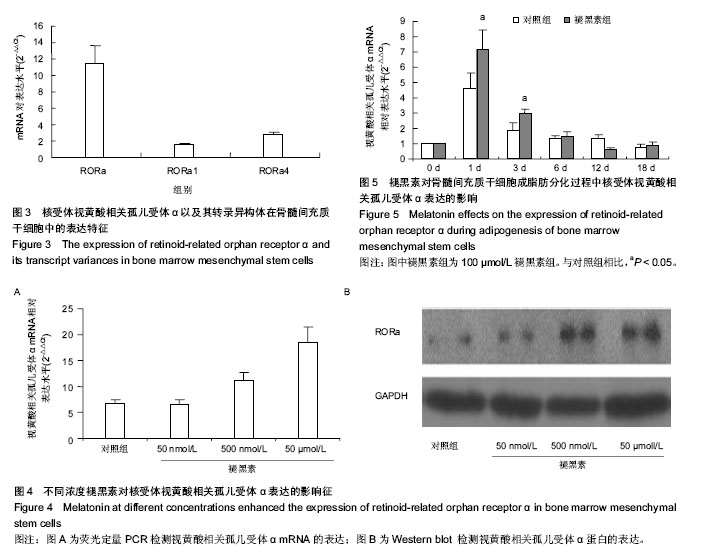
2.2 视黄酸相关孤儿受体α在骨髓间充质干细胞以及其成脂肪分化过程中的表达特征 荧光定量PCR发现骨髓间充质干细胞高表达视黄酸相关孤儿受体α核受体。视黄酸相关孤儿受体α的4种转录异构体中,只有视黄酸相关孤儿受体α1和视黄酸相关孤儿受体α4在骨髓间充质干细胞上表达,其中以视黄酸相关孤儿受体α4表达最为丰富,其表达量约为视黄酸相关孤儿受体α1的2倍(图3)。视黄酸相关孤儿受体α2和视黄酸相关孤儿受体α3在骨髓间充质干细胞没有表达。 在不同浓度的褪黑素的作用下,骨髓间充质干细胞表达视黄酸相关孤儿受体α的量被上调,而且这种作用随着褪黑素的浓度上升而增强(图4A)。进一步,用Western blot检测视黄酸相关孤儿受体α蛋白水平表达量也证实了褪黑素的这种浓度依赖性作用(图4B)。 在骨髓间充质干细胞成脂肪分化过程中,视黄酸相关孤儿受体α在早期(1,3,6 d)有明显的上调表达(P < 0.05),在中后期就维持在相对低水平(图5)。褪黑素抑制成脂肪分化的作用可能与此相关。"
| [1] Reiter RJ. Melatonin: the chemical expression of darkness. Mol Cell Endocrinol. 1991;79(1-3):C153-158. [2] Korkmaz A, Sanchez-Barcelo EJ, Tan DX, et al. Role of melatonin in the epigenetic regulation of breast cancer. Breast Cancer Res Treat. 2009;115(1):13-27. [3] Sainz RM, Reiter RJ, Tan DX, et al. Critical role of glutathione in melatonin enhancement of tumor necrosis factor and ionizing radiation-induced apoptosis in prostate cancer cells in vitro. J Pineal Res. 2008;45(3):258-270. [4] Pedrosa AM, Weinlich R, Mognol GP, et al. Melatonin protects CD4+ T cells from activation-induced cell death by blocking NFAT-mediated CD95 ligand upregulation. J Immunol. 2010; 184(7):3487-3494. [5] Paredes SD, Barriga C, Reiter RJ, et al. Assessment of the potential role of tryptophan as the precursor of serotonin and melatonin for the aged sleep-wake cycle and immune function: streptopelia risoria as a model. Int J Tryptophan Res. 2009;2: 23-36. [6] Bonnefont-Rousselot D, Collin F, Jore D, et al. Reaction mechanism of melatonin oxidation by reactive oxygen species in vitro. J Pineal Res. 2011;50(3):328-335. [7] Conti A, Conconi S, Hertens E, et al. Evidence for melatonin synthesis in mouse and human bone marrow cells. J Pineal Res. 2000;28(4):193-202. [8] Tan DX, Manchester LC, Reiter RJ, et al. Identification of highly elevated levels of melatonin in bone marrow: its origin and significance. Biochim Biophys Acta. 1999;1472(1-2): 206-214. [9] Muruganandan S, Roman AA, Sinal CJ. Adipocyte differentiation of bone marrow-derived mesenchymal stem cells: cross talk with the osteoblastogenic program. Cell Mol Life Sci. 2009;66(2):236-253. [10] Rodríguez JP, Astudillo P, Ríos S, et al. Involvement of adipogenic potential of human bone marrow mesenchymal stem cells (MSCs) in osteoporosis. Curr Stem Cell Res Ther. 2008;3(3):208-218. [11] Bianco P. Minireview: The stem cell next door: skeletal and hematopoietic stem cell "niches" in bone. Endocrinology. 2011;152(8):2957-2962. [12] Zhang L, Su P, Xu C, et al. Melatonin inhibits adipogenesis and enhances osteogenesis of human mesenchymal stem cells by suppressing PPARγ expression and enhancing Runx2 expression. J Pineal Res. 2010;49(4):364-372. [13] Jetten AM, Kurebayashi S, Ueda E. The ROR nuclear orphan receptor subfamily: critical regulators of multiple biological processes. Prog Nucleic Acid Res Mol Biol. 2001;69:205-247. [14] Becker-André M, Wiesenberg I, Schaeren-Wiemers N, et al. Pineal gland hormone melatonin binds and activates an orphan of the nuclear receptor superfamily. J Biol Chem. 1994; 269(46):28531-28534. [15] Wiesenberg I, Missbach M, Kahlen JP, et al. Transcriptional activation of the nuclear receptor RZR alpha by the pineal gland hormone melatonin and identification of CGP 52608 as a synthetic ligand. Nucleic Acids Res. 1995;23(3):327-333. [16] Winczyk K, Pawlikowski M, Karasek M. Melatonin and RZR/ROR receptor ligand CGP 52608 induce apoptosis in the murine colonic cancer. J Pineal Res. 2001;31(2):179-182. [17] Pittenger MF, Mackay AM, Beck SC, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284(5411):143-147. [18] Rio DC, Ares M Jr, Hannon GJ, et al. Purification of RNA using TRIzol (TRI reagent). Cold Spring Harb Protoc. 2010; 2010(6):pdb.prot5439. [19] Connolly MA, Clausen PA, Lazar JG. Purification of RNA from animal cells using trizol. CSH Protoc. 2006;2006(1). pii: pdb. prot4104. [20] 甘淋,李娟,何涛,等.几种蛋白质含量测定方法的比较研究[J].泸州医学院学报,2004,27(6):500-502. [21] Bailey DM, Evans KA, James PE, et al. Altered free radical metabolism in acute mountain sickness: implications for dynamic cerebral autoregulation and blood-brain barrier function. J Physiol. 2009;587(Pt 1):73-85. [22] Lerner AB, Case JD, Takahashi Y. Isolation of melatonin and 5-methoxyindole-3-acetic acid from bovine pineal glands. J Biol Chem. 1960;235:1992-1997. [23] Lerner AB, CASE JD, Mori W, et al. Melatonin in peripheral nerve. Nature. 1959;183:1821. [24] Jung-Hynes B, Reiter RJ, Ahmad N. Sirtuins, melatonin and circadian rhythms: building a bridge between aging and cancer. J Pineal Res. 2010;48(1):9-19. [25] Reiter RJ, Tan DX, Rosales-Corral S, et al. The universal nature, unequal distribution and antioxidant functions of melatonin and its derivatives. Mini Rev Med Chem. 2013; 13(3): 373-384. [26] Proietti S, Cucina A, Reiter RJ, et al. Molecular mechanisms of melatonin's inhibitory actions on breast cancers. Cell Mol Life Sci. 2013;70(12):2139-2157. [27] Coto-Montes A, Boga JA, Rosales-Corral S, et al. Role of melatonin in the regulation of autophagy and mitophagy: a review. Mol Cell Endocrinol. 2012;361(1-2):12-23. [28] Galano A, Tan DX, Reiter RJ. Melatonin as a natural ally against oxidative stress: a physicochemical examination. J Pineal Res. 2011;51(1):1-16. [29] Borges-Silva CN, Fonseca-Alaniz MH, Alonso-Vale MI, et al. Reduced lipolysis and increased lipogenesis in adipose tissue from pinealectomized rats adapted to training. J Pineal Res. 2005;39(2):178-184. [30] Wolden-Hanson T, Mitton DR, McCants RL, et al. Daily melatonin administration to middle-aged male rats suppresses body weight, intraabdominal adiposity, and plasma leptin and insulin independent of food intake and total body fat. Endocrinology. 2000;141(2):487-497. [31] Brydon L, Petit L, Delagrange P, et al. Functional expression of MT2 (Mel1b) melatonin receptors in human PAZ6 adipocytes. Endocrinology. 2001;142(10):4264-4271. [32] Alonso-Vale MI, Andreotti S, Borges-Silva Cd, et al. Intermittent and rhythmic exposure to melatonin in primary cultured adipocytes enhances the insulin and dexamethasone effects on leptin expression. J Pineal Res. 2006;41(1):28-34. [33] Alonso-Vale MI, Peres SB, Vernochet C, et al. Adipocyte differentiation is inhibited by melatonin through the regulation of C/EBPbeta transcriptional activity. J Pineal Res. 2009;47(3):221-227. [34] Ekmekcioglu C. Melatonin receptors in humans: biological role and clinical relevance. Biomed Pharmacother. 2006;60(3): 97-108. [35] Slominski RM, Reiter RJ, Schlabritz-Loutsevitch N, et al. Melatonin membrane receptors in peripheral tissues: distribution and functions. Mol Cell Endocrinol. 2012; 351(2): 152-166. [36] Jetten AM. Retinoid-related orphan receptors (RORs): critical roles in development, immunity, circadian rhythm, and cellular metabolism. Nucl Recept Signal. 2009;7:e003. [37] Duez H, Duhem C, Laitinen S, et al. Inhibition of adipocyte differentiation by RORalpha. FEBS Lett. 2009;583(12): 2031-2036. [38] Ohoka N, Kato S, Takahashi Y, et al. The orphan nuclear receptor RORalpha restrains adipocyte differentiation through a reduction of C/EBPbeta activity and perilipin gene expression. Mol Endocrinol. 2009;23(6):759-771. |
| [1] | Li Cai, Zhao Ting, Tan Ge, Zheng Yulin, Zhang Ruonan, Wu Yan, Tang Junming. Platelet-derived growth factor-BB promotes proliferation, differentiation and migration of skeletal muscle myoblast [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1050-1055. |
| [2] | Zhou Wu, Wang Binping, Wang Yawen, Cheng Yanan, Huang Xieshan. Transforming growth factor beta combined with bone morphogenetic protein-2 induces the proliferation and differentiation of mouse MC3T3-E1 cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3630-3635. |
| [3] | Chen Qiang, Zhuo Hongwu, Xia Tian, Ye Zhewei . Toxic effects of different-concentration isoniazid on newborn rat osteoblasts in vitro [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(8): 1162-1167. |
| [4] | Zhang Wen, Lei Kun, Gao Lei, Li Kuanxin. Neuronal differentiation of rat bone marrow mesenchymal stem cells via lentivirus-mediated bone morphogenetic protein 7 transfection [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(7): 985-990. |
| [5] | Qin Xinyu, Zhang Yan, Zhang Ningkun, Gao Lianru, Cheng Tao, Wang Ze, Tong Shanshan, Chen Yu. Elabela promotes differentiation of Wharton’s jelly-derived mesenchymal stem cells into cardiomyocyte-like cells [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(7): 1046-1051. |
| [6] | Zhang Peigen, Heng Xiaolai, Xie Di, Wang Jin, Ma Jinglin, Kang Xuewen. Electrical stimulation combined with neurotrophin 3 promotes proliferation and differentiation of endogenous neural stem cells after spinal cord injury in rats [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(7): 1076-1082. |
| [7] | Huang Cheng, Liu Yuanbing, Dai Yongping, Wang Liangliang, Cui Yihua, Yang Jiandong. Transplantation of bone marrow mesenchymal stem cells overexpressing glial cell line derived neurotrophic factor gene for spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(7): 1037-1045. |
| [8] | Tang Jingfeng, Zhang Jun, You Qi, Liu Yi. The role and mechanism of graphene and its derivatives-related composites in cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(4): 619-624. |
| [9] | Lü Jianwei, Ma Jianxiong, Ma Xinlong. Melatonin promotes Schwann cell migration by activating a typical Wnt/β-catenin signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 5030-5037. |
| [10] | Wang Wenhong, Li Yanjun, Cui Caiyun. Factors influencing differentiation of stem cells from the apical papilla into odontoblasts [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 5071-5078. |
| [11] | Yao Shunhan, Wei Huacheng, Qin Jiagang, Liao Liang. Mogroside V stimulates osteoblast proliferation and differentiation by promoting LncRNA TUG1 expression [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(26): 4129-4134. |
| [12] |
Yang Jiayi, Zhang Guanghao, Zhang Cheng, Li Ke, Huo Xiaolin, Wu Changzhe.
Stem cell expansion and hepatic differentiation: measurement of related indicators and detection methods [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(25): 4039-4045. |
| [13] | Yang Fan, Liu Baoyi, Cao Meng, Zhu Xiaoshu, Zhang Yu, Qin Kairong, Zhao Dewei. Basic fibroblast growth factors protect chondrocytes by antagonizing extracellular inflammatory factors [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(23): 3621-3626. |
| [14] | Yin Lingni, Chen Dexuan. The role of PI3K/Akt pathway in hypoxia-induced cell proliferation and endothelial cell differentiation of adipose-derived stem cells [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(19): 3004-3009. |
| [15] | Zhu Haizhou, Zhao Zhankui, Wang Xinzhe, Liu Deqian, Yu Honglian. Urine-derived stem cells differentiate into urothelial cells and smooth muscle cells in vitro [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(19): 3010-3016. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||