Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (31): 5030-5037.doi: 10.3969/j.issn.2095-4344.2138
Previous Articles Next Articles
Melatonin promotes Schwann cell migration by activating a typical Wnt/β-catenin signaling pathway
Lü Jianwei1, Ma Jianxiong2, Ma Xinlong1
- 1Department of Orthopedics, Tianjin Hospital, Tianjin 300211, China; 2Orthopedics Research Institute, Tianjin Hospital, Tianjin 300050, China
-
Received:2020-03-10Revised:2020-03-14Accepted:2020-04-15Online:2020-11-08Published:2020-09-04 -
Contact:Ma Xinlong, Master, Professor, Department of Orthopedics, Tianjin Hospital, Tianjin 300211, China -
About author:Lü Jianwei, Master, Physician, Department of Orthopedics, Tianjin Hospital, Tianjin 300211, China -
Supported by:the National Natural Science Foundation of China, No. 81572154; the Natural Science Foundation of Tianjin, No. 17JCQNJC1090
CLC Number:
Cite this article
Lü Jianwei, Ma Jianxiong, Ma Xinlong. Melatonin promotes Schwann cell migration by activating a typical Wnt/β-catenin signaling pathway[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 5030-5037.
share this article
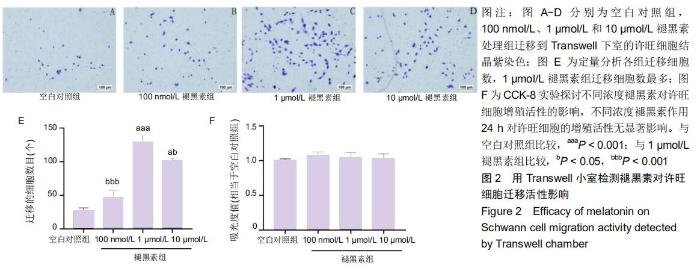
2.2 褪黑素对许旺细胞增殖活性的影响 如图2F所示,培养24 h后,不同浓度褪黑素组以及空白对照组两两比较,许旺细胞的吸光度值无显著性差异。表明不同浓度的褪黑素作用24 h,许旺细胞的增殖活性无显著改变,因此,接下来的Transwell细胞迁移实验选择20 h为检测时间点。 2.3 褪黑素促进许旺细胞迁移 用Transwell小室检测不同浓度褪黑素对许旺细胞迁移活性的影响。如图2所示,不同浓度褪黑素组迁移到Transwell下室的细胞数量均高于空白对照组,其中1,10 μmol/L褪黑素组细胞数相较于空白对照组差异有显著性意义(P < 0.001);此外,1 μmol/L褪黑素组迁移细胞数目显著多于10 μmol/L组(P=0.011)。因此,一定浓度褪黑素促进许旺细胞迁移,对其产生化学性吸引作用,然而随着浓度继续增高,褪黑素促进许旺细胞迁移的作用逐渐减弱。 "

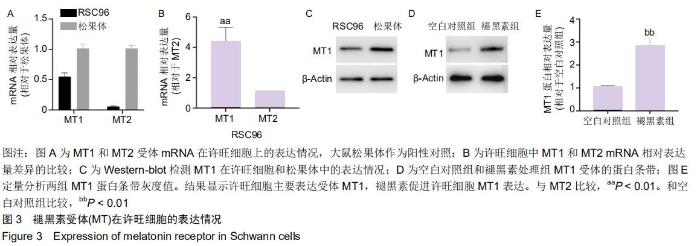
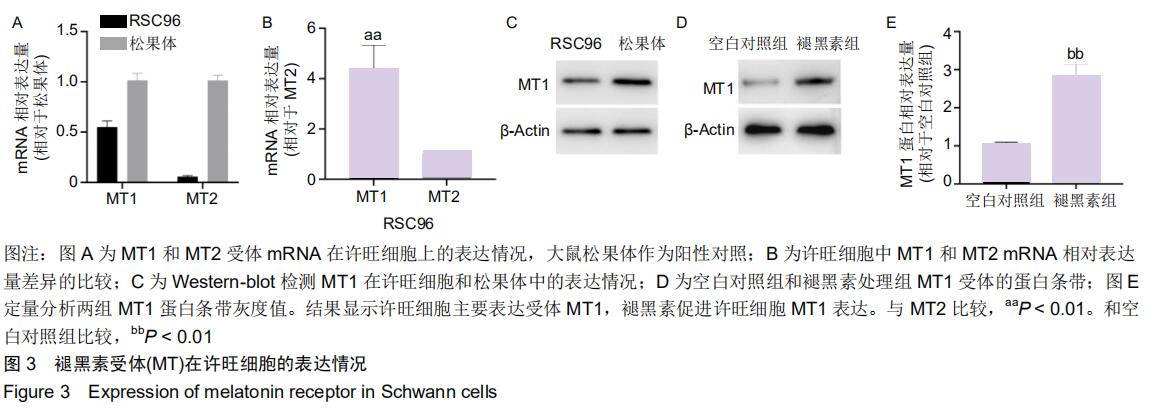
2.4 许旺细胞主要表达受体MT1 以大鼠松果体作为阳性对照,PCR结果显示,松果体和许旺细胞均表达MT1和MT2的mRNA,见图3A;许旺细胞中MT1和MT2的相对含量表示为各自循环阈值(Ct)与β-actin循环阈值的比值,进而以MT2相对Ct值为参照,比较许旺细胞中MT1和MT2的相对含量发现,许旺细胞中MT1的表达量显著高于MT2 (P=0.003),见图3B;Western-blot显示,松果体和许旺细胞均表达MT1蛋白,见图3C;此外,在褪黑素作用下,相较于空白对照组,MT1蛋白在许旺细胞中表达量显著增加(P=0.006),见图3D,E。因此,许旺细胞主要表达褪黑素受体MT1。 "
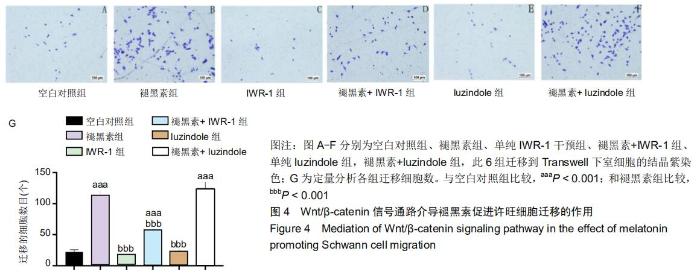
2.5 Wnt/β-catenin信号通路而非MT1介导褪黑素促进许旺细胞迁移的作用 接下来探究褪黑素是否通过MT1或Wnt通路促进许旺细胞迁移。分别用MT拮抗剂luzindole (50 μmol/L)或Wnt通路抑制剂IWR-1(2 μmol/L)预处理许旺细胞1 h,然后行Transwell实验。如图4所示:相对于单纯褪黑素组,褪黑素+IWR-1组细胞迁移数量明显降低(P < 0.001),但仍高于空白对照组(P < 0.001)。然而,褪黑素+ luzindole组相较于单纯褪黑素组迁移细胞数目无明显改变,差异无显著性意义。因此,褪黑素通过激活Wnt/ β-catenin信号通路促进许旺细胞迁移,而MT1不介导褪黑素对许旺细胞迁移活性的影响。 "
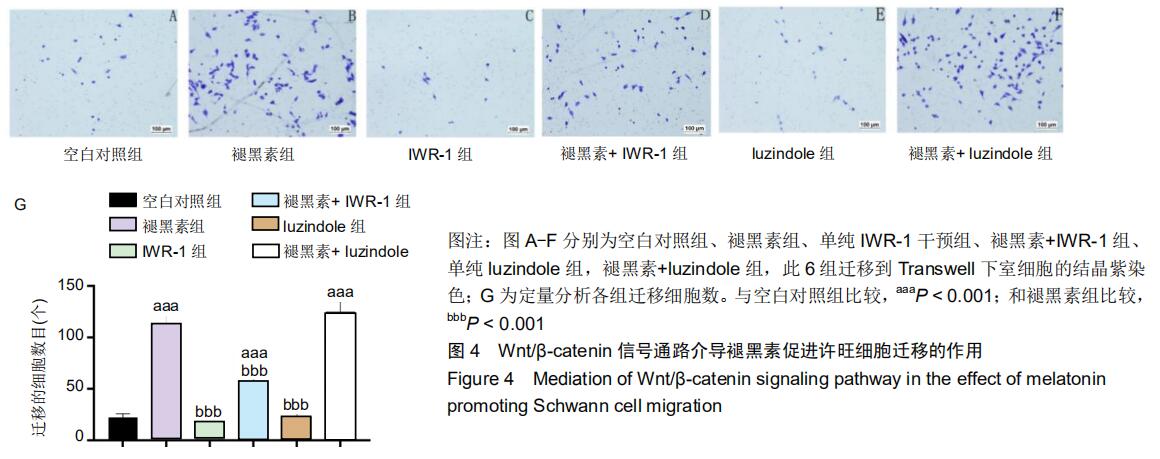
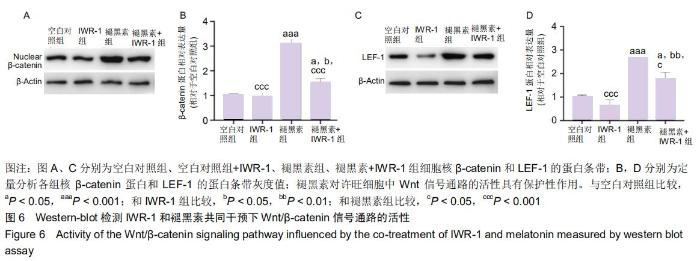
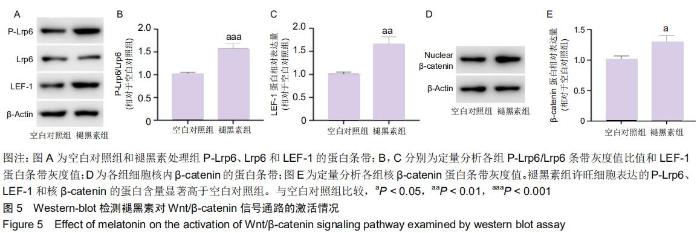
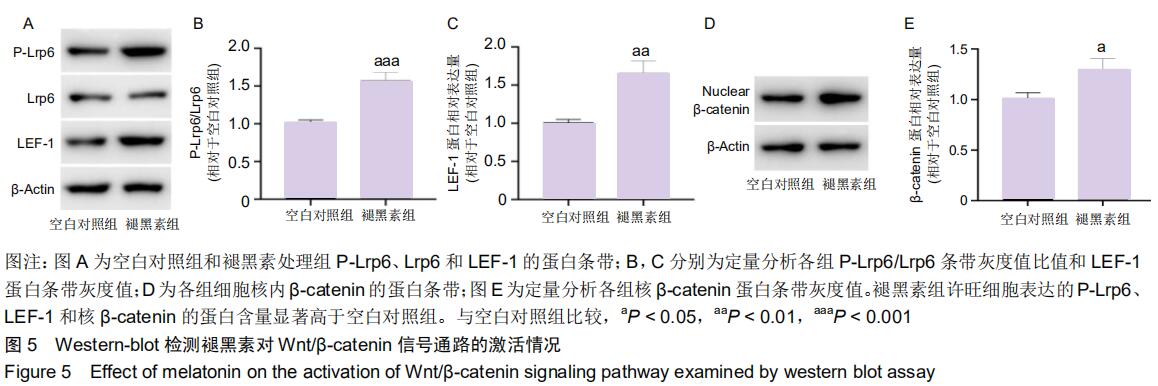
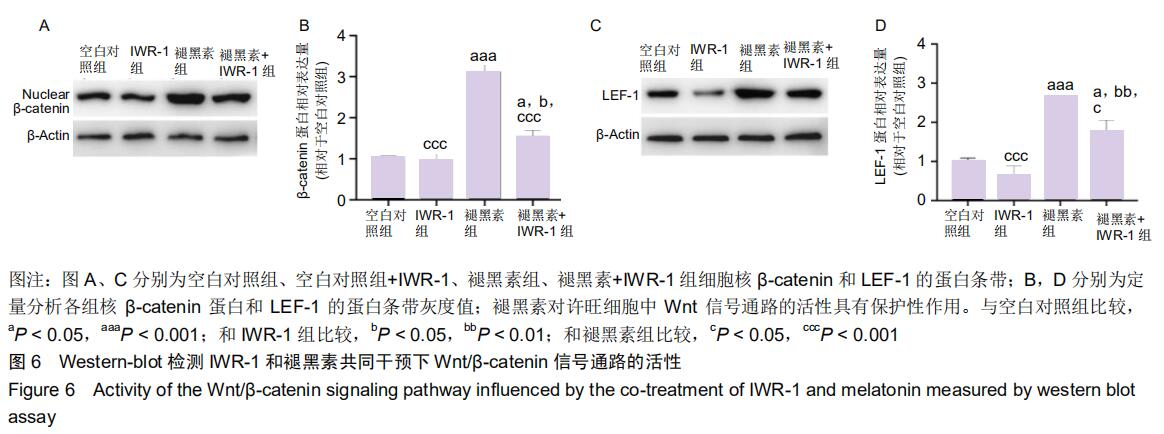
2.7 褪黑素对许旺细胞Wnt/β-catenin信号通路的活性具有保护性作用 由于IWR-1能在一定程度上拮抗褪黑素促进许旺细胞迁移的作用,接下来通过Western-blot探究IWR-1干预下,许旺细胞中Wnt通路的激活情况。如图6所示,和单纯褪黑素组比较,IWR-1+褪黑素组许旺细胞中LEF-1蛋白表达显著下调(P=0.013),核β-catenin蛋白水平明显下降(P < 0.001),但是二者表达量均仍高于空白对照组和单纯IWR-1组(LEF-1分别为:P=0.035,P=0.004;β-catenin分别为:P=0.019,P=0.014)。因此,褪黑素对Wnt信号通路的活性具有保护性作用。 "
|
[1] QIAN Y, HAN Q, ZHAO X, et al. 3D melatonin nerve scaffold reduces oxidative stress and inflammation and increases autophagy in peripheral nerve regeneration. J Pineal Res. 2018;65(4):e12516.
[2] TRICAUD N, PARK HT. Wallerian demyelination: chronicle of a cellular cataclysm. Cell Mol Life Sci. 2017;74(22):4049-4057.
[3] WONG KM, BABETTO E, BEIROWSKI B. Axon degeneration: make the Schwann cell great again. Neural Regen Res. 2017; 12(4):518-524.
[4] 高伟,邵水金,胡琳娜,等.许旺细胞在周围神经损伤修复中的作用及其可能分子机制[J].中国组织工程研究与临床康复,2010, 14(36):6825-6827.
[5] YU F, WENG J, YUAN Y, et al. Wnt5a Affects Schwann Cell Proliferation and Regeneration via Wnt/c-Jun and PTEN Signaling Pathway. Chin Med J (Engl). 2018;131(21): 2623-2625.
[6] SHEFA U, JUNG J. Comparative study of microarray and experimental data on Schwann cells in peripheral nerve degeneration and regeneration: big data analysis. Neural Regen Res. 2019;14(6):1099-1104.
[7] HSU C, HUANG H, WU P, et al. Sesame oil improves functional recovery by attenuating nerve oxidative stress in a mouse model of acute peripheral nerve injury: role of Nrf-2. J Nutr Biochem. 2016;38:102-106.
[8] KAYA Y, SARIKCIOĞLU L, ASLAN M, et al. Comparison of the beneficial effect of melatonin on recovery after cut and crush sciatic nerve injury: a combined study using functional, electrophysiological, biochemical, and electron microscopic analyses. Childs Nerv Syst. 2013;29(3):389-401.
[9] GÜL S, CELIK SE, KALAYCI M, et al. Dose-dependent neuroprotective effects of melatonin on experimental spinal cord injury in rats. Surg Neurol. 2005;64(4):355-361.
[10] TORDJMAN S, CHOKRON S, DELORME R, et al. Melatonin: pharmacology, functions and therapeutic benefits. Curr Neuropharmacol. 2017;15(3):434-443.
[11] CUTANDO A, ANEIROS-FERNÁNDEZ J, LÓPEZ-VALVERDE A, et al. A new perspective in Oral health: potential importance and actions of melatonin receptors MT1, MT2, MT3, and RZR/ROR in the oral cavity. Arch Oral Biol. 2011;56(10):944-950.
[12] CHEN BH, PARK JH, LEE YL, et al. Melatonin improves vascular cognitive impairment induced by ischemic stroke by remyelination via activation of ERK1/2 signaling and restoration of glutamatergic synapses in the gerbil hippocampus. Biomed Pharmacother. 2018;108:687-697.
[13] TURGUT M, KAPLAN S. Effects of melatonin on peripheral nerve regeneration. Recent Pat Endocr Metab Immune Drug Discov. 2011;5(2):100-108.
[14] SAYAN H, OZACMAK VH, Ozen OA, et al. Beneficial effects of melatonin on reperfusion injury in rat sciatic nerve. J Pineal Res. 2004;37(3):143-148.
[15] Edizer DT, Donmez Z, Gul M, et al. Effects of Melatonin and Dexamethasone on Facial Nerve Neurorrhaphy. J Int Adv Otol. 2019;15(1):43-50.
[16] CHIANG RP, HUANG C, TSAI Y. Melatonin reduces median nerve injury-induced mechanical hypersensitivity via inhibition of microglial p38 mitogen-activated protein kinase activation in rat cuneate nucleus. J Pineal Res. 2013;54(2):232-244. [17] SALEHI M, NASERI-NOSAR M, EBRAHIMI-BAROUGH S, et al. Polyurethane/Gelatin Nanofibrils Neural Guidance Conduit Containing Platelet-Rich Plasma and Melatonin for Transplantation of Schwann Cells. Cell Mol Neurobiol. 2018; 38(3):703-713.
[18] CHANG H, LIU C, HSU W, et al. Proliferative effects of melatonin on Schwann cells: implication for nerve regeneration following peripheral nerve injury. J Pineal Res. 2014;56(3):322-332.
[19] TIONG YL, NG KY, KOH RY, et al. Melatonin Prevents Oxidative Stress-Induced Mitochondrial Dysfunction and Apoptosis in High Glucose-Treated Schwann Cells via Upregulation of Bcl2, NF-κB, mTOR, Wnt Signalling Pathways. Antioxidants (Basel). 2019;8(7):198.
[20] GUTIERREZ-CUESTA J, TAJES M, JIMÉNEZ A, et al. Evaluation of potential pro-survival pathways regulated by melatonin in a murine senescence model. J Pineal Res. 2008;45(4):497-505.
[21] SHEN Z, ZHOU Z, GAO S, et al. Melatonin Inhibits Neural Cell Apoptosis and Promotes Locomotor Recovery via Activation of the Wnt/β-Catenin Signaling Pathway After Spinal Cord Injury. Neurochem Res. 2017;42(8):2336-2343.
[22] HU F, YAN Y, WANG C, et al. Article Effect and Mechanism of Ganoderma lucidum Polysaccharides on Human Fibroblasts and Skin Wound Healing in Mice. Chin J Integr Med. 2019; 25(3):203-209.
[23] LIAO B, CHEN R, LIN F, et al. Long noncoding RNA HOTTIP promotes endothelial cell proliferation and migration via activation of the Wnt/β-catenin pathway. J Cell Biochem. 2018;119(3):2797-2805.
[24] WANG Y, CHEN H, ZHANG H. Kaempferol promotes proliferation, migration and differentiation of MC3T3-E1 cells via up-regulation of microRNA-101. Artif Cells Nanomed Biotechnol. 2019;47(1):1050-1056.
[25] GALANO A, TAN DX, REITER RJ. On the free radical scavenging activities of melatonin's metabolites, AFMK and AMK. J Pineal Res. 2013;54(3):245-257.
[26] LIU D, WEI N, MAN H, et al. The MT2 receptor stimulates axonogenesis and enhances synaptic transmission by activating Akt signaling. Cell Death Differ. 2015;22(4): 583-596.
[27] LIU L, HOFFMAN GE, FEI X, et al. Delayed rectifier outward K+ current mediates the migration of rat cerebellar granule cells stimulated by melatonin. J Neurochem. 2007;102(2): 333-344.
[28] LEE S, JUNG YH, OH SY, et al. Melatonin enhances the human mesenchymal stem cells motility via melatonin receptor 2 coupling with Gαq in skin wound healing. J Pineal Res. 2014;57(4):393-407.
[29] CHAO C, CHEN P, CHIOU P, et al. Melatonin suppresses lung cancer metastasis by inhibition of epithelial-mesenchymal transition through targeting to Twist. Clin Sci (Lond). 2019;133(5):709-722. [30] KEPKA M, SZWEJSER E, PIJANOWSKI L, et al. A role for melatonin in maintaining the pro- and anti-inflammatory balance by influencing leukocyte migration and apoptosis in carp. Dev Comp Immunol. 2015;53(1):179-190.
[31] LUCHETTI F, CANONICO B, BARTOLINI D, et al. Melatonin regulates mesenchymal stem cell differentiation: a review. J Pineal Res. 2014;56(4):382-397.
[32] MORTEZAEE K, PASBAKHSH P, RAGERDI KASHANI I, et al. Melatonin Pretreatment Enhances the Homing of Bone Marrow-derived Mesenchymal Stem Cells Following Transplantation in a Rat Model of Liver Fibrosis. Iran Biomed J. 2016;20(4):207-216.
[33] CEREZO AB, HORNEDO-ORTEGA R, ÁLVAREZ-FERNÁNDEZ MA, et al. Inhibition of VEGF-Induced VEGFR-2 Activation and HUVEC Migration by Melatonin and Other Bioactive Indolic Compounds. Nutrients. 2017;9(3):249.
[34] LIU Z, ZOU D, YANG X, et al. Melatonin inhibits colon cancer RKO cell migration by downregulating Rho‑associated protein kinase expression via the p38/MAPK signaling pathway. Mol Med Rep. 2017;16(6):9383-9392.
[35] YE X, QIU Y, GAO Y, et al. A Subtle Network Mediating Axon Guidance: Intrinsic Dynamic Structure of Growth Cone, Attractive and Repulsive Molecular Cues, and the Intermediate Role of Signaling Pathways. Neural Plast. 2019;2019:1719829.
[36] LV J, SUN X, MA J, et al. Netrin-1 induces the migration of Schwann cells via p38 MAPK and PI3K-Akt signaling pathway mediated by the UNC5B receptor. Biochem Biophys Res Commun. 2015;464(1):263-268.
[37] GUO WL, QI ZP, YU L, et al. Melatonin combined with chondroitin sulfate ABC promotes nerve regeneration after root-avulsion brachial plexus injury. Neural Regen Res. 2019; 14(2):328-338.
[38] LI X, LI Z, WANG J, et al. Wnt4 signaling mediates protective effects of melatonin on new bone formation in an inflammatory environment. FASEB J. 2019;33(9): 10126-10139.
[39] LIU L, WANG T, YANG X, et al. MTNR1B loss promotes chordoma recurrence by abrogating melatonin-mediated β-catenin signaling repression. J Pineal Res. 2019;67(2): e12588.
[40] HAN B, ZHAO J, WANG W, et al. Cdc42 Promotes Schwann Cell Proliferation and Migration Through Wnt/β-Catenin and p38 MAPK Signaling Pathway After Sciatic Nerve Injury. Neurochem Res. 2017;42(5):1317-1324.
[41] WHITEHEAD MJ, MCGONIGAL R, WILLISON HJ, et al. Heparanase attenuates axon degeneration following sciatic nerve transection. Sci Rep. 2018;8(1):5219.
[42] TAWK M, MAKOUKJI J, BELLE M, et al. Wnt/beta-catenin signaling is an essential and direct driver of myelin gene expression and myelinogenesis. J Neurosci. 2011;31(10): 3729-3742.
[43] KILIC U, YILMAZ B, UGUR M, et al. Evidence that membrane-bound G protein-coupled melatonin receptors MT1 and MT2 are not involved in the neuroprotective effects of melatonin in focal cerebral ischemia. J Pineal Res. 2012; 52(2):228-235. |
| [1] | Hu Guang, Guan Zhiyu, Zhang Kaiwei . Mechanism underlying the interventional effect of Panax Notoginsenosides on ovariectomized osteoporotic fracture rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(2): 172-177. |
| [2] | Geng Bin, Xia Yayi. Involvement of ERK5 signaling pathway in osteoporosis development in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(2): 178-185. |
| [3] | Li Xiaoqun, Xu Kaihang, Ji Fang. Corylin inhibits osteoclastogenesis and attenuates postmenopausal osteoporosis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(2): 186-190. |
| [4] | Hua Kunchi, Hu Yongcheng. Defatting effect of gradient alcohol treatment on cancellous bone allograft and the biomechanical properties of defatted cancellous bone allograft [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5502-5507. |
| [5] | Yuan Yiming, Wang Yan, Chen Chengcheng, Zhao Mingyue, Pei Fei. Efficacy of exosomes in peripheral nerve injury [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 5079-5084. |
| [6] | Chen Jia, Yang Yiqiang, Hu Chen, Chen Qi, Zhao Tian, Yong Min, Ma Dongyang, Ren Liling. Fabrication of prevascularized osteogenic differentiated cell sheet based on human bone marrow mesenchymal stem cells and human umbilical vein endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 4934-4940. |
| [7] | Liao Jian, Huang Xiaolin, Zhou Qian, Huo Hua, Qi Yuhan, Wu Chao, Shi Qianhui, Yang Tongjing, Liao Yunmao, Liang Xing. Calcined bone/chitosan composite promotes osteogenic differentiation of bone marrow mesenchymal stem cells in Sprague-Dawley rats [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 4941-4947. |
| [8] | Wang Guoliang, Li Yanlin, Xiang Yaoyu, Jia Di, Li Canzhang, He Lu. MicroRNA expression profiles of chondrocytes in osteoarthritis induced by stromal cell derived factor 1 [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 4948-4953. |
| [9] | Li Yuanqi, Lin Hai, Luo Hongrong, Zhang Xingdong. Relationship between mitochondrial autophagy and chondrogenesis of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 4954-4960. |
| [10] | Ling Xuejian, Yang Qingchun, Zhang Yanding, Ma Gang. Effect of Noggin overexpression in hair follicle dermal sheath cells on the growth of skin and hair follicles [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 5013-5017. |
| [11] | Cai Bozhi, Chen Xiancai, Ni Na, Lin Weishen, Yuan Yanping, Lin Changmin, Huang Keng. Wnt5a expression in the regeneration of the inferior segment of rat hair follicle at embryonic and postnatal stages [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 5018-5022. |
| [12] | Yin Jing, Wang Dan, Wang Peng, Zhang Hong, Jiang Haijun. Comparison of immune responses of acellular vascular materials prepared by the two methods in animals [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(28): 4555-4561. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||