Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (2): 178-185.doi: 10.3969/j.issn.2095-4344.2955
Previous Articles Next Articles
Involvement of ERK5 signaling pathway in osteoporosis development in mice
Geng Bin1, 2, Xia Yayi1, 2
- 1Department of Joint Surgery, Second Hospital of Lanzhou University, Lanzhou 730000, Gansu Province, China; 2Gansu Provincial Key Laboratory of Bone and Joint Disease Research, Lanzhou 730000, Gansu Province, China
-
Received:2020-03-10Revised:2020-03-14Accepted:2020-04-15Online:2021-01-18Published:2020-11-21 -
Contact:Xia Yayi, MD, Professor, Chief physician, Doctoral supervisor, Department of Joint Surgery, Second Hospital of Lanzhou University, Lanzhou 730000, Gansu Province, China; Gansu Provincial Key Laboratory of Bone and Joint Disease Research, Lanzhou 730000, Gansu Province, China -
About author:Geng Bin, MD, Master’s supervisor, Department of Joint Surgery, Second Hospital of Lanzhou University, Lanzhou 730000, Gansu Province, China; Gansu Provincial Key Laboratory of Bone and Joint Disease Research, Lanzhou 730000, Gansu Province, China -
Supported by:the National Natural Science Foundation of China, No. 81960403; the National Natural Science Foundation of China, No. 81874017
CLC Number:
Cite this article
Geng Bin, Xia Yayi. Involvement of ERK5 signaling pathway in osteoporosis development in mice[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(2): 178-185.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
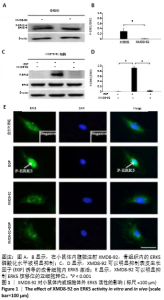
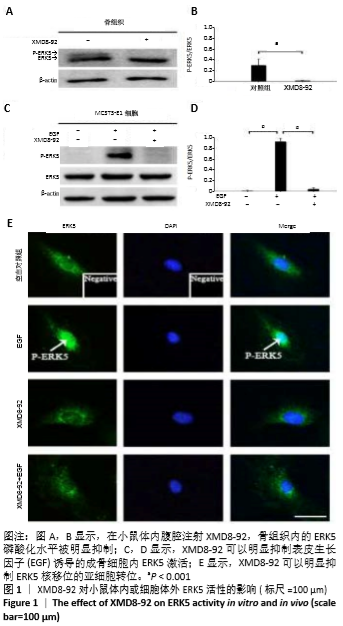
2.1 实验动物数量分析 实验选用昆明小鼠12只,分为4组,实验过程无脱失,全部进入结果分析。 2.2 小鼠体内和MC3T3-E1细胞内ERK5的活性 在小鼠体内腹腔注射XMD8-92,骨组织内的ERK5磷酸化水平被明显抑制,见图1 A,B。以往的研究证实ERK5能够被表皮生长因子所激活[30],而且以往的研究证实XMD8-92的最佳浓度为5 μmol/L,且不会影响其他MAPK信号通路[14],此次体外实验表明XMD8-92可以明显抑制表皮生长因子诱导的成骨细胞内ERK5激活,见图1 C,D。 XMD8-92作用于MC3T3-E1细胞的免疫荧光染色结果显示XMD8-92可以明显抑制ERK5核移位的亚细胞转位,见图1E。因为磷酸化的ERK5会转移至核内调控下游靶因子表达[14],XMD8-92可以阻断这一过程,进一步证实XMD8-92可以抑制成骨细胞内ERK5的活性。 "
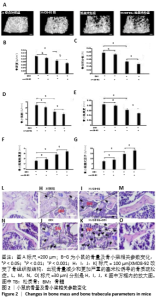
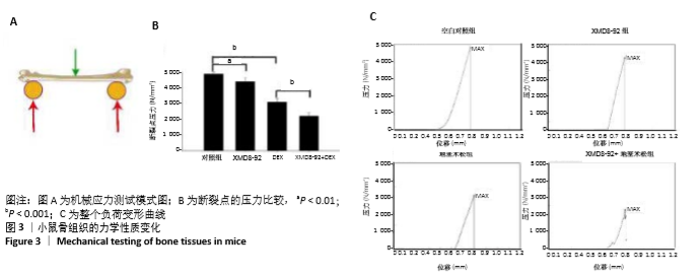
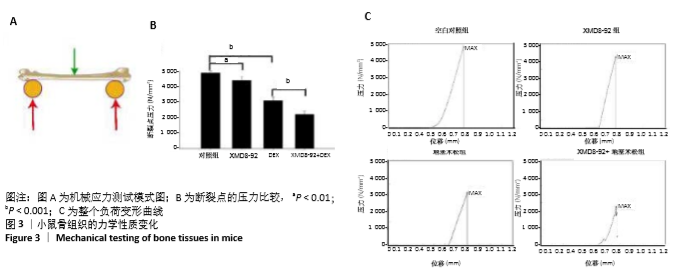
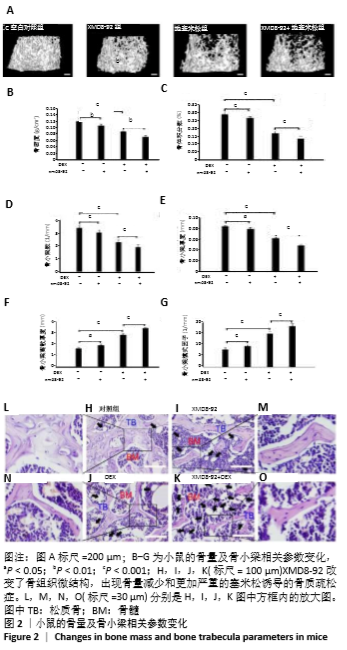
2.3 小鼠的骨量及骨小梁相关参数变化 小鼠被腹腔注射XMD8-92和/或地塞米松。μ-CT分析显示XMD8-92干预后导致小鼠股骨骨小梁的结构变稀疏,见图2。与对照组相比,骨密度定量显示XMD8-92可使股骨松质骨密度减少约11.4% (P < 0.01,见图2B)。骨体积分数、骨小梁数、骨小梁厚度也分别降低了8.0%、10.7%和5%,而骨小梁离散度和骨小梁模式因子分别增加了17.3%和20.3%(见图2 C-G)。这些结果表明,XMD8-92处理后的小鼠骨体积更小,骨小梁数目和厚度更少,且骨小梁间隙更大。 另外,腹腔注射过量的地塞米松 5周后成功建立小鼠骨质疏松症模型,结果提示XMD8-92能够显著加重塞米松诱导的骨质疏松症(见图2A)。XMD8-92+地塞米松诱导的小鼠股骨骨密度显著降低(-19.4%,P < 0.01),骨体积分数降低(-20.2%,P < 0.001),骨小梁数降低(-16.0%, P < 0.001),骨小 梁厚度(-22.6%,P < 0.001),但骨小梁离散度和骨小梁模式因子增加(+20.6 和+22.5,P < 0.001,见图2 C-G)。 同样,苏木精-伊红染色结果也显示XMD8-92处理后的小鼠、地塞米松处理后的小鼠和XMD8-92+地塞米松诱导后的小鼠其股骨均出现不同程度的骨质流失,见图2 H-O。 结果表明,抑制ERK5活性能够降低小鼠的骨量,并加重地塞米松诱导的骨质疏松症。"
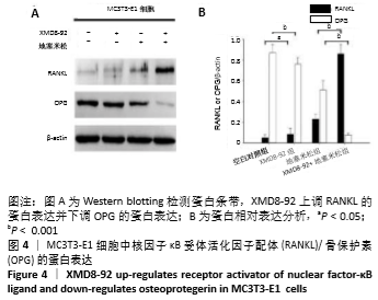
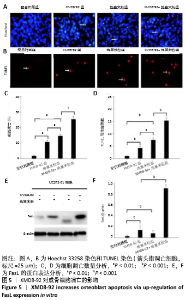
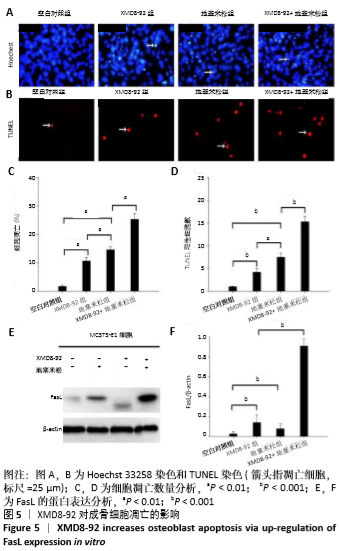
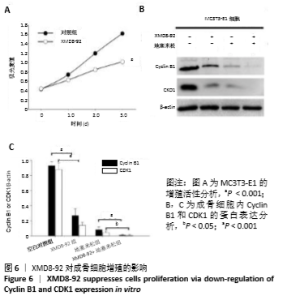
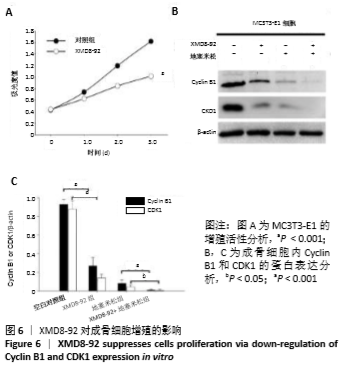
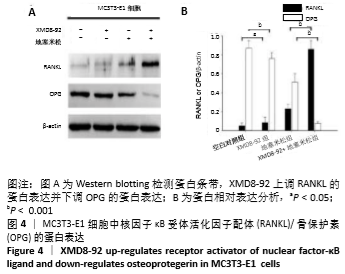
2.5 MC3T3-E1细胞中RANKL/OPG的蛋白表达 RANKL和骨保护素在骨重塑过程中起着重要作用,由成骨细胞合成并分泌,其表达量的变化能够反映出成骨细胞或破骨细胞的活性,间接显示骨形成和骨吸收的动态平衡关系,是检测骨质疏松症常用的生物学指标[1-3]。Western blotting结果显示XMD8-92和地塞米松显著提高了MC3T3-E1细胞中RANKL的蛋白水平(P < 0.05和P < 0.001,见图4 A和B),而且XMD8-92能够进一步促进地塞米松诱导的RANKL上调(P < 0.001,见图4 A和B)。相反,XMD8-92和地塞米松明显下调了骨保护素蛋白的表达,且XMD8-92能够进一步促进地塞米松对骨保护素的下调作用 (P < 0.001,见图4 A和B)。结果提示,ERK5活性的抑制能够明显上调RANKL并下调骨保护素的蛋白表达,XMD8-92可以通过调控RANKL/OPG的蛋白表达来调节骨量的变化。"
| [1] MARUOTTI N, CORRADO A,ROTONDO C, et al. Janus kinase inhibitors role in bone remodeling. J Cell Physiol. 2020;235(3): 915-1920. [2] TOOSI S,BEHRAVAN J. Osteogenesis and bone remodeling: A focus on growth factors and bioactive peptides. Biofactors. 2019;3(9): e10213. [3] KATSIMBRI P. The biology of normal bone remodelling. Eur J Cancer Care (Engl). 2017;26(6). doi: 10.1111/ecc.12740. [4] KHOSLA S, MONROE DG. Regulation of Bone Metabolism by Sex Steroids. Cold Spring Harb Perspect Med. 2018;8(1): pii: a031211. [5] FRENKEL B, WHITE W, TUCKERMANN J. Glucocorticoid-induced osteoporosis. Adv Exp Med Biol. 2015;872:179-215. [6] BUCKLEY L, HUMPHREY MB. Glucocorticoid-Induced Osteoporosis. N Engl J Med. 2018;379(26): 2547-2556. [7] WEINSTEIN RS, JILKA RL, PARFITT AM, et al. Inhibition of osteoblastogenesis and promotion of apoptosis of osteoblasts and osteocytes by glucocorticoids. Potential mechanisms of their deleterious effects on bone. J Clin Invest. 1998;102(2): 274-282. [8] JIA D, O’BRIEN CA, STEWART SA, et al. Glucocorticoids act directly on osteoclasts to increase their life span and reduce bone density. Endocrinology. 2006;147(12): 5592-5599. [9] WEINSTEIN RS.Glucocorticoid-induced osteoporosis and osteonecrosis. Endocrinol Metab Clin North Am. 2012;41(3):595-611. [10] BRIOT K. Bone and glucocorticoids. Ann Endocrinol (Paris). 2018;79(3): 115-118. [11] DING N, GENG B, LI Z, et al. Fluid shear stress promotes osteoblast proliferation through the NFATc1-ERK5 pathway. Connect Tissue Res. 2018;60(2): 107-116. [12] BO Z,BIN G, JING W, et al. Fluid shear stress promotes osteoblast proliferation via the Gαq-ERK5 signaling pathway. Connect Tissue Res. 2016;57(4): 299-306. [13] JIANG J, ZHAO LG, TENG YJ, et al. ERK5 signalling pathway is essential for fluid shear stress-induced COX-2 gene expression in MC3T3-E1 osteoblast. Mol Cell Biochem. 2015;406(1-2): 237-243. [14] BIN G, CUIFANG W, BO Z, et al. Fluid shear stress inhibits TNF-alpha-induced osteoblast apoptosis via ERK5 signaling pathway. Biochem Biophys Res Commun. 2015;466(1):117-123. [15] ZHOU G, BAO ZQ, DIXON JE. Components of a new human protein kinase signal transduction pathway. J Biol Chem. 1995;270(21): 12665-12669. [16] LEE JD, ULEVITCH RJ, HAN J. Primary structure of bmk1: A new mammalian map kinase. Biochem Biophys Res Commun. 1995;213(2): 715-724. [17] ABE J, KUSUHARA M, ULEVITCH RJ, et al. Big mitogen-activated protein kinase 1 (BMK1) is a redox-sensitive kinase. J Biol Chem. 1996;271(28): 16586-16590. [18] KATO Y, KRAVCHENKO VV, TAPPING RI, et al. BMK1/ERK5 regulates serum-induced early gene expression through transcription factor MEF2C. EMBO J. 1997;16 (23):7054-7066. [19] FUKUHARA S, MARINISSEN MJ, CHIARIELLO M, et al. Signaling from G protein-coupled receptors to ERK5/Big MAPK 1 involves Galpha q and Galpha 12/13 families of heterotrimeric G proteins. Evidence for the existence of a novel Ras AND Rho-independent pathway. J Biol Chem. 2000;275 (28): 21730-21736. [20] KIM EK, CHOI EJ. Pathological roles of MAPK signaling pathways in human diseases. Biochim Biophys Acta. 2010;1802 (4): 396-405. [21] REGAN CP, LI W, BOUCHER DM, et al. Erk5 null mice display multiple extraembryonic vascular and embryonic cardiovascular defects. Proc Natl Acad Sci U S A. 2002;99(14): 9248-9253. [22] WANG X, MERRITT AJ, SEYFRIED J, et al. Targeted deletion of mek5 causes early embryonic death and defects in the extracellular signal-regulated kinase 5/myocyte enhancer factor 2 cell survival pathway. Mol Cell Biol. 2005;25(1): 336-345. [23] YAN L, CARR J, ASHBY PR, et al. Knockout of ERK5 causes multiple defects in placental and embryonic development. BMC Dev Biol. 2003;3 (11). [24] HAYASHI M, KIM SW, IMANAKA-YOSHIDA K, et al. Targeted deletion of BMK1/ERK5 in adult mice perturbs vascular integrity and leads to endothelial failure. J Clin Invest. 2004;113 (8): 1138-1148. [25] SOHN SJ, SARVIS BK, CADO D, et al. ERK5 MAPK regulates embryonic angiogenesis and acts as a hypoxia-sensitive repressor of vascular endothelial growth factor expression. J. Biol. Chem. 2002;277(45): 43344-43351. [26] YANG Q, DENG X, LU B, et al. Pharmacological inhibition of BMK1 suppresses tumor growth through promyelocytic leukemia protein. Cancer Cell. 2010;18(3): 258-267. [27] YANG Q, LEE JD. Targeting the BMK1 MAP kinase pathway in cancer therapy. Clin Cancer Res. 2011;17(11): 3527-3532. [28] SUREBAN SM,MAY R,WEYGANT N,et al.XMD8-92 inhibits pancreatic tumor xenograft growth via a DCLK1-dependent mechanism. Cancer Lett. 2014;351(1): 151-161. [29] ROVIDA E, DI MAIRA G, TUSA I, et al. The mitogen-activated protein kinase ERK5 regulates the development and growth of hepatocellular carcinoma. Gut. 2015;64(9): 1454-1465. [30] KATO Y, TAPPING RI, HUANG S, et al. Bmk1/Erk5 is required for cell proliferation induced by epidermal growth factor. Nature. 1998;395(6703): 713-716. [31] NAKAMURA H, AOKI K, MASUDA W, et al. Disruption of NF-kappaB1 prevents bone loss caused by mechanical unloading. J Bone Miner Res. 2013;28(6): 1457-1467. [32] NAKAMURA K, JOHNSON GL. Activity assays for extracellular signal-regulated kinase 5. Methods Mol Biol 2010; 661:91-106. [33] PELLETIER JP, FERNANDES JC, JOVANOVIC DV, et al. Chondrocyte death in experimental osteoarthritis is mediated by MEK 1/2 and p38 pathways: role of cyclooxygenase-2 and inducible nitric oxide synthase. J Rheumatol. 2001;28(11): 2509-2519. [34] PAWLIK TM, KEYOMARSI K. Role of cell cycle in mediating sensitivity to radiotherapy. Int J Radiat Oncol Biol Phys. 2004;59(4): 928-942. [35] THORNTON TM, RINCON M. Non-classical p38 map kinase functions: Cell cycle checkpoints and survival. Int J Biol Sci. 2009;5(1): 44-51. [36] PELLETIER JP, MINEAU F, BOILEAU C, et al. Diacerein reduces the level of cartilage chondrocyte DNA fragmentation and death in experimental dog osteoarthritic cartilage at the same time that it inhibits caspase-3 and inducible nitric oxide synthase. Clin Exp Rheumatol. 2003;21(2): 171-177. [37] LOHBERGER B, LEITHNER A, STUENDL N, et al. Diacerein retards cell growth of chondrosarcoma cells at the G2/M cell cycle checkpoint via cyclin B1/CDK1 and CDK2 downregulation. BMC Cancer. 2015; 15:891. [38] KANESHIRO S, OTSUKI D, YOSHIDA K, et al. MEK5 suppresses osteoblastic differentiation. Biochem Biophys Res Commun. 2015; 463(3): 241-247. [39] ZHANG B, AN L, GENG B, et al. ERK5 negatively regulates Kruppel-like factor 4 and promotes osteogenic lineage cell proliferation in response to MEK5 overexpression or fluid shear stress. Connect Tissue Res. 2019;21: 1-12. [40] MORIMOTO H, KONDOH K, NISHIMOTO S, et al. Activation of a C-terminal transcriptional activation domain of ERK5 by autophosphorylation. J Biol Chem. 2007;282(49): 35449-35456. [41] THOUVEREY C, CAVERZASIO J. Ablation of p38alpha MAPK signaling in osteoblast lineage cells protects mice from bone loss induced by estrogen deficiency. Endocrinology. 2015;156(12): 4377-4387. [42] BOYLE DL, HAMMAKER D, EDGAR M, et al. Differential roles of MAPK kinases MKK3 and MKK6 in osteoclastogenesis and bone loss. PLoS ONE. 2014;9(1): e84818. [43] EL KHASSAWNA T, BOCKER W, BRODSKY K, et al. Impaired extracellular matrix structure resulting from malnutrition in ovariectomized mature rats. Histochem Cell Biol. 2015;144(5): 491-507. [44] ZHANG P, DAI Q, OUYANG N, et al. Mechanical Strain Promotes Osteogenesis of BMSCs from Ovariectomized Rats via the ERK1/2 but not p38 or JNK-MAPK Signaling Pathways. Curr Mol Med. 2015:15(8): 780-789. [45] KRUM SA, CHANG J, MIRANDA-CARBONI G, et al.Novel functions for NFkappaB: inhibition of bone formation. Nat Rev Rheumatol. 2010:6(10): 607-611. |
| [1] | Li Xiaoqun, Xu Kaihang, Ji Fang. Corylin inhibits osteoclastogenesis and attenuates postmenopausal osteoporosis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(2): 186-190. |
| [2] | Xu Hui, Kang Bingxin, Zhong Sheng, Gao Chenxin, Zhao Chi, Qiu Guowei, Sun Songtao, Xie Jun, Xiao Lianbo, Shi Qi. Pressing local acupoints plus adjustion of the knee joint in a sitting position for treating knee osteoarthritis: a randomized controlled trial [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(2): 216-221. |
| [3] | Wang Yue, Wang Xinjun, Yuan Yinpeng, Wang Yuze. Mechanism of DAIa2GIP inhibiting mitochondrial apoptosis in chondrocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(11): 1652-1657. |
| [4] | Wang Qian, Li Lu, Shu Jingyuan, Dong Zhiheng, Jin Youshi, Wang Qingshan. Micro-morphology and phase of zirconia-based nano-hydroxyapatite functional gradient biomaterials [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1517-1521. |
| [5] | Li Rui, Wang Chen, Zhang Wenyi, Ma Shiqing, Sun Yingchun. Various surface treatment methods and resin cement type on the influence of bonding strength of zirconia ceramics [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1528-1532. |
| [6] | Xu Changkui, Pu Xiaobing, Lu Yao, Chen Jiarong, Pan Lei. Safety and antibacterial properties of gentamicin-loaded silk fibroin in meniscus repair [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1545-1549. |
| [7] | Jia Wei, Zhang Mandong, Chen Weiyi, Wang Chenyan, Guo Yuan. Effects of femoral prosthetic materials on artificial knee arthroplasty performance [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1477-1481. |
| [8] | Fan Haixia, Tan Qingkun, Wang Hong, Cheng Huanzhi, Liu Xue, Ching-chang Ko, Geng Haixia. Rabbit skull defects repaired by the hydroxyapatite/geltin scaffold combined with bone marrow mesenchymal stem cells and umbilical vein endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1495-1499. |
| [9] | Liao Jian, Huang Xiaolin, Huo Hua, Zhou Qian, Cheng Yuting, Qi Yuhan, Wu Chao, Yang Tongjing, Liao Yunmao, Liang Xing. Effects of calcined bone/chitosan composite materials on proliferation and adhesion in osteoblasts [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5447-5453. |
| [10] | Dong Wenjie, Zhang Shiyang, Zhao Lei, Wang Yukun. Icariin deproteinized inorganic bovine bone composite and deproteinized inorganic bovine bone material in repairing mandibular defects [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5467-5472. |
| [11] | Chen Shaobo, Shuang Feng, Hu Wei, Li Hao, Shao Yinchu. Injectable bone graft for the repair of nonunion of midshaft clavicular fracture after internal fixation [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5473-5477. |
| [12] | Li Yongtao, Song Wenhui, Liu Changwen, Liang Kaiheng, Zhang Kaixi, Liu Changfeng. Clinical efficacy of calcium sulphate/demineralized bone matrix versus bone allograft in anterior cervical discectomy and fusion [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5478-5485. |
| [13] | Hua Kunchi, Hu Yongcheng. Defatting effect of gradient alcohol treatment on cancellous bone allograft and the biomechanical properties of defatted cancellous bone allograft [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5502-5507. |
| [14] | Jiang Zongrui, Zhang Zhiqi. Treatment of meniscus injury or degeneration: the effect and function of stem cells and artificial polymer scaffolds to form tissue engineering system [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5421-5427. |
| [15] | Zhang Bin, Sun Lihua, Zhang Junhua, Liu Tongbin, Liu Yusan, Cui Caiyun, Li Jun. Short-term effect comparison of a modified socket shield technique and conventional flapless immediate implant and immediate restoration in maxillary aesthetic area [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5514-5519. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||