Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (34): 5421-5427.doi: 10.3969/j.issn.2095-4344.2334
Previous Articles Next Articles
Treatment of meniscus injury or degeneration: the effect and function of stem cells and artificial polymer scaffolds to form tissue engineering system
Jiang Zongrui, Zhang Zhiqi
Department of Joint Surgery, the First Affiliated Hospital of Sun Yat-sen University, Guangzhou 510080, Guangdong Province, China
-
Received:2020-03-03Revised:2020-03-07Accepted:2020-03-21Online:2020-11-08Published:2020-09-11 -
Contact:Zhang Zhiqi, MD, Associate chief physician, Department of Joint Surgery, the First Affiliated Hospital of Sun Yat-sen University, Guangzhou 510080, Guangdong Province, China -
About author:Jiang Zongrui, Master candidate, Department of Joint Surgery, the First Affiliated Hospital of Sun Yat-sen University, Guangzhou 510080, Guangdong Province, China -
Supported by:the National Natural Science Foundation of China, No. 8157211
CLC Number:
Cite this article
Jiang Zongrui, Zhang Zhiqi. Treatment of meniscus injury or degeneration: the effect and function of stem cells and artificial polymer scaffolds to form tissue engineering system[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5421-5427.
share this article
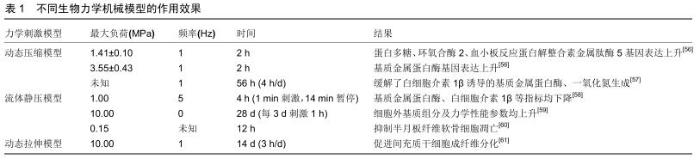
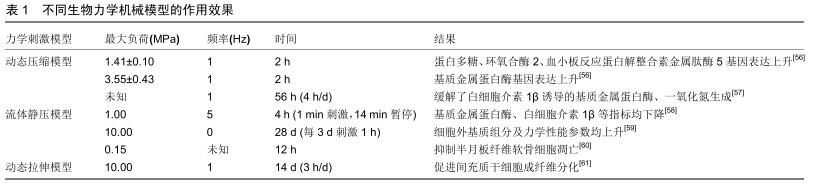
2.1 种子细胞 目前组织工程的种子细胞包括自体细胞、异体细胞、干细胞。由于自体细胞提取手段复杂且提取量稀少,同种异体细胞与异种异体细胞如若用于临床转化则存在患者免疫排斥的风险。目前最常用的种子细胞是间充质干细胞(mesenchymal stromal cell,MSCs)。 间充质干细胞具有多重分化特性,起源于人骨髓细胞,也可从成人与胎儿的其他组织获取。科学家基于以下2个原因掀起了对间充质干细胞作为组织工程学种子细胞的研究热潮:其一,间充质干细胞具有分化成多种高分化间叶组织(如骨、软骨、脂肪、韧带等)的全能性;其二,间充质干细胞可分泌多种免疫调控分子并通过旁分泌作用调节损伤组织修复。许多科学家通过注射间充质干细胞用于各种动物模型如灵长类动物[7]、新西兰兔及小鼠的半月板缺损模型[8-9],获得良好效果,为后期实现临床转化奠定了坚实基础。近年来运用间充质干细胞作为种子细胞的研究一直是组织工程学的研究热点之一。WHITEHOUSE等[10]率先将未分化的间充质干细胞种植于胶原支架上用于半月板损伤的患者,发现部分患者症状明显缓解、MRI结果未显示损伤复发且无明显的不良反应,实现初步的临床转化。 间充质干细胞存在缺陷如易感染性及增殖能力弱,而移植前培养具有突变或表观遗传改变的自体间充质干细胞后发生肿瘤样异常的发展是目前科学家最为担忧的风险[11];因此,如何排除这些缺陷并将间充质干细胞真正实现临床转化成为当前研究的热点之一。 2.2 支架 当今支架为种子细胞提供增殖、分化的模具与环境,使其形成符合相应解剖部位形态的结构,从而起到替代损伤退变组织的作用。目前认为组织工程学中运用的支架需要满足以下3个条件:其一,满足原有组织结构的生物机械负荷;其二,满足原有组织的生物学功能如增殖、分化等;其三,满足实行组织工程操作如提取、制作、消毒以及移植的可行性。目前常见的支架主要有:人工聚合物支架、水凝胶支架、细胞外基质组分支架、组织源性支架。 2.2.1 人工聚合物支架 人工聚合物支架如聚氨酯(polyurethane, PU)、聚乙酸丙酯(polycaprolactone, PCL)、聚乳酸(polylactic acid, PLA)、聚乙醇酸(polyglycolic acid,PGA)、聚乳酸-羟基乙酸共聚物(polylactic co-glycolic acid, PLGA)等均为人工合成,在各种动物模型中均表现出良好的生物机械性能。此外在研究中人工聚合物支架也展现了其他功能,KOCH等[12]发现聚氨酯可促进血管形成;KWAK等[13]发现运用富血小板血浆预处理的聚乳酸-羟基乙酸共聚物支架有更强的细胞黏着能力促进半月板再生进程。 聚乙酸丙酯支架是近年来研究最多的一种新型支架,由乙酸、异丙醇在硫酸催化下酯化合成,具有优异的生物机械性能、可加工性,而且其在生物体内降解速度较其他支架慢,给予种子细胞更多时间生长发育。ZHANG等[14]系统性研究种子细胞、支架与刺激物组合,通过3D打印合成聚乙酸丙酯支架并种植间充质干细胞于其中,结果显示间充质干细胞/聚乙酸丙酯支架组合相较单纯支架组分有更强的生物机械性能与软骨保护作用,原因可能与分泌更多细胞外基质组分有关。通过组织转化生长因子β3以及动态压缩负荷刺激成功恢复了半月板的各向异性,即恢复半月板内区、外区各自不同的细胞类型、组织形态、生化组分与生理功能[15],提示未来组织工程的研究趋向。然而其缺陷在于其聚合链上缺乏生物官能团从而导致细胞难以黏附于其表面形成聚合物,科学家通过将聚乙酸丙酯支架与疏水性和亲水性聚合物如水凝胶结合来加强细胞黏附及细胞外基质合成[16];或者通过填充碳纳米材料增加其导电功能促进黏附[17]。 目前有多种技术应用于半月板人工聚合物支架来增强其生物机械负荷需求,如静电技术以及纤维纺织技术[18-20],然而在仿生性与生物活性方面的研究仍然缺乏,且其生物相容性与细胞毒性有待进一步考察。 2.2.2 水凝胶支架 水凝胶支架可人工合成,也可天然提取。天然水凝胶如海藻酸、壳聚糖与细菌纤维素的主要成分以多糖为主。FOLLIN等[21]将脂肪干细胞用海藻酸包裹,培养1周后脂肪干细胞细胞活性>93%,沉积的细胞恢复了原有细胞表型,并抑制了淋巴细胞增殖能力从而降低免疫排斥反应,体现出良好的生物相容性。MORADI等[22]合成交联壳聚糖/聚乙烯醇合成的支架具备良好的生物可降解性与机械性能,接种软骨细胞可促进半月板再生。BODIN等[23]评估细菌纤维素杨氏模量、压缩模量及拉伸模量,发现其对比胶原支架显示出更好的生物机械性能,而在高负荷环境下力学负荷参数却不及猪半月板;此外,细菌纤维素体外培养时可缠绕形成具有纳米结构的膜,促进骨髓干细胞与其黏附[24]。 明胶由动物体内的胶原蛋白制备而成,高度模拟细胞外基质,因此常用于组织工程领域。NARITA等[25]用明胶包裹成纤维生长因子2植入新西兰白兔半月板缺损模型可刺激半月板细胞增殖且抑制其凋亡。TANAKA 等[26]利用明胶水凝胶包裹辛伐他汀注射于骨关节炎小鼠膝关节腔可缓解软骨退变加重。近年来对于明胶的研究更多聚焦于其与甲基丙烯酸酐通过光反应制备而得的新型水凝胶——甲基丙烯酸酐化明胶(Gelatin methacrylate,GelMA),除外明胶原本具有的特性如仿生性,甲基丙烯酸酐化明胶容易成型。BAHCECIOGLU等[27]设计的一种模仿半月板结构的聚乙酸丙酯/水凝胶构建体经过培养发现甲基丙烯酸酐化明胶组分水凝胶表现出高水平的Ⅰ型胶原、Ⅱ型胶原与糖胺聚糖表达,同时表现出半月板软骨样内区和纤维软骨样外区的结构特点。此外,他们还发现离体情况下甲基丙烯酸酐化明胶比聚乙酸丙酯支架更有利于半月板再生[16]。 水凝胶支架优越的仿生性,为细胞增殖与组织再生提供与高度模仿正常生理的微环境,常用于体外3D培养观察细胞增殖、迁移、分化以及与细胞外基质之间的联系[28];此外,水凝胶生物可降解且具有原位可成形性,体内外均可有效均匀地包裹细胞或者药物,形成可注射性水凝胶支架,可避免骨关节炎患者采取开放性手术治疗,从而减轻患者痛苦[29]。FENG等[30]通过合成一种细胞可渗透及可注射性(cell-infiltratable and injectable,Ci-I)的水凝胶支架包裹骨髓干细胞与淫羊藿黄素,其在激素诱导性股骨头坏死动物模型中促进原位骨再生及防止骨矿物密度的减少,原理在于通过弱而高度动态的主客体络合而物理交联,并进一步通过有限的化学交联来增强稳定性,从而实现高度动态及可逆转化的结构网络以实现细胞渗透,创造间充质干细胞成骨分化的有利微环境。因此水凝胶支架非常适合运用于组织工程,但其生物机械性能仍不及正常生理半月板,仍需要更有效的方法加工各种水凝胶支架以增强其负荷能力。 2.2.3 细胞外基质组分支架 细胞外基质组分支架较人工聚合物支架与水凝胶支架更具仿生性,人软骨与半月板中均含有大量胶原与透明质酸,因此两者合成的支架免疫排斥性非常低。胶原支架通过酸性溶液溶解Ⅰ型胶原制得,兼具合适的支架孔径与再生组织的自我再塑功能[31],ODA等[32]移植Ⅰ型胶原支架与髌下脂肪垫至已行半月板缺损新西兰白兔中发现其能有效修复半月板,基质金属蛋白酶表达下调且白细胞介素1β表达无明显上调,大体、组织学观察缺损半月板修复情况较对照组更满意;此外,鉴于表皮生长因子受体信号通路在半月板损伤与动物骨关节炎模型中有上调倾向,结合表皮生长因子小分子抑制剂吉菲替尼与胶原支架可延缓小鼠早期骨关节炎的进展;透明质酸为体内普遍的糖胺聚糖,可与胶原等生物大分子作用合成细胞外基质,也可通过胞膜表面受体与细胞结合,从而激活相关下游通路并产生相应的生物学功能[33]。透明质酸支架用于半月板的研究较少,但干燥状态下通过调节透明质酸中的葡萄糖醛酸制得衍生物透明质酸苄酯(hyaluronan benzyl ester,HYAFF-11)结构较稳定且生物机械性能评估较好[34]。CHIARI等[35]比较胶原组分支架与透明质酸苄酯组分支架发现两者糖胺聚糖以及Ⅰ型胶原合成量差异无统计学意义,以上研究均表明透明质酸具备满足半月板组织工程支架需求的条件可成为深入研究的支架选择。 丝蛋白也是一种细胞外基质支架,来源于昆虫,主要由蛋白质及少量磷酸酯多糖与脂质构成,丝素蛋白可通过介导钙磷合成从而影响骨生长与发育[36]。GRUCHENBURG等[37]将一种新型的丝蛋白组分支架植入羊部分半月板切除模型,发现其拉伸与压缩参数与正常生理半月板相似,且植入后无明显炎性反应与软骨退变,但因固定技术不成熟,导致植入物无法与缺损部位贴合。WARNECKE等[38]通过动态挤压试验、μCT以及生物相容性测试等方式评估丝蛋白组分支架的生物机械性能、结构以及生物学特点,初步认为其可作为早期半月板切除术后的取代物。而用于缝合时天然丝素纤维中与丝素蛋白共存的丝胶往往会引发抗原-抗体反 应[39],这一缺陷通过物理、化学修饰或者添加其他聚合物或矿物质去除丝胶得以解决[36]。合适的缝合术式、合并使用的有效材料调节细胞外基质组分支架降解动力学仍有待科学家探索与研究。 2.2.4 组织衍生支架 来源于发育完全组织的组织衍生支架,如小肠黏膜下层、脱细胞半月板支架、脱细胞基质,具备良好的仿生性,为细胞提供合适的增殖、分化、迁移及细胞外基质合成的内环境。小肠黏膜下层源于猪小肠的细胞外基质,主要由胶原、蛋白聚糖以及猪小肠分泌的生长因子构成。猪小肠黏膜下层可支持宿主细胞的种植生长、激活并维持细胞的增殖与分化、不会诱发免疫排斥且使用后可生物降解[40]。猪小肠黏膜下层已在各种犬类动物半月板缺损或者切除模型中证实能促进半月板再生[41-43],COOK等[44]发现通过移植猪小肠黏膜下层可以促进无血管区域的类半月板组织再生,起到软骨保护作用;在长期预后方面,他们又发现猪小肠黏膜下层植入半月板缺损模型中后具有长期稳定的软骨保护作用且下肢运动功能康复更好[40]。然而因猪小肠黏膜下层主要在犬类动物模型中运用而未运用于其他动物模型,需进一步结合多种实验动物模型进行评估。 脱细胞技术的出现解决了同种异体组织移植的缺陷如感染、缓慢的组织重塑与结合以及免疫排斥反应,脱细胞半月板支架与脱细胞基质均通过脱细胞技术合成[45]。 YAMASAKI等[46]通过液氮冻融3次将使半月板脱细胞,然后种植间充质干细胞于该脱细胞支架上,观察4周后发现细胞外基质、蛋白聚糖、Ⅹ型胶原表达升高,各项力学机械模数近似于正常半月板的坚固度。SANDMANN等[47]提取人半月板并运用去垢剂十二烷基硫酸钠脱细胞并体外评估其机械力学特性也出现类似结果。脱细胞基质也采用了脱细胞技术,YUAN等[48]使用胃蛋白酶及乙酸处理猪半月板并冷藏于4 ℃实现脱细胞合成的支架可促进细胞外基质组分合成,然而只能通过将牛软化松质骨(demineralized cancellous bone,DCB)浸入方可增强其机械性能,并实现半月板再生;且脱细胞基质/软化松质骨支架的关节软骨完整性、膝关节全器官磁共振成像评分(Whole-Organ Magnetic Resonance Imaging Scores,WORMS)均优于单纯脱细胞基质组。以上均表明以及许多组织衍生支架缺乏正常半月板的生理机械负荷功能,且这一缺陷在体内实验中可能影响细胞外基质原有功能;同时组织衍生材料提取手段常涉及手术切除天然组织,故其供应缺乏及成本高昂是需要进一步解决的问题;此外,脱细胞处理等组织衍生材料的加工程序很可能破坏了组织结构的完整性[49]。 2.3 刺激物 生物化学刺激物对于半月板及其他膝关节组织发育至关重要,以生长因子作用最为突出。以往研究说明生长因子如表皮上生长因子、转化生长因子、血小板源性生长因子、纤维母细胞生长因子、胰岛素样生长因子、肝细胞生长因子对半月板细胞增殖、迁移、分化等一系列生理活动有调节作用[50]、也具有促进半月板细胞合成细胞外基质的作用[51];其中以转化生长因子β3在半月板组织工程中运用最广泛[52-53]。近来趋势逐渐倾向将生长因子、种子细胞、支架整合作为一个体系研究,LEE等[54]通过运用组织转化生长因子、转化生长因子β3刺激种植于人半月板脱细胞支架上的间充质干细胞,观察发现这种干预措施可促使其分化成为区域特异性细胞与细胞外基质,即半月板外区合成分泌Ⅰ型胶原,内区合成分泌Ⅱ型胶原,同时也恢复了正常半月板内外区的生物机械负荷性能。这项研究一方面彰显了生长因子对半月板细胞的增殖、分化的作用,另一方面提示将具有诱导增殖、分化以及内环境调节的生长因子、全能性种子细胞以及仿生性强且满足正常组织生物功能的支架作为一个体系研究最有可能实现组织工程的最终目的并且实现临床转化。 适当生物力学刺激对半月板组织再生修复有积极作用。科学家一直在从压力大小、持续时间及刺激频率层面寻求符合要求的动态力学负荷刺激模型,相关刺激方法包括动态压缩试验、循环静水压模型以及拉伸负荷(表1)。经研究,三者的作用原理均以调节细胞外基质合成有关且均需于动态环境下进行,因为研究指出静态负荷可能会加速半月板的退变,其机制与下调细胞外基质基因表达有 关[55]。相反,一定范围内的动态负荷可以促进半月板再生修复,然而超出正常生理动态负荷会促进炎性递质如白细胞介素1β及基质金属蛋白酶如基质金属蛋白酶1、基质金属蛋白酶3、基质金属蛋白酶13等表达,加速膝关节各组织的退化[56]。因此探索合适压力大小、持续时间、刺激频率的动态压力负荷刺激模型对于正常生理半月板及组织工程复合物十分重要。此外,ZHANG等[14]将合适的动态压缩模型运用于已用组织转化生长因子、转化生长因子β3刺激已种植间充质干细胞的聚乙酸丙酯支架后可高度还原正常半月板的结构,提示系统性结合合理生物力学模型对未来半月板组织工程领域的必要性。 "
|
[1] SALATA MJ, GIBBS AE, SEKIYA JK. A systematic review of clinical outcomes in patients undergoing meniscectomy.Am J Sports Med. 2010; 38(9):1907-1916.
[2] HUNTER DJ, ZHANG YQ, NIU JB, et al. The association of meniscal pathologic changes with cartilage loss in symptomatic knee osteoarthritis.Arthritis Rheum.2006; 54(3):795-801.
[3] BERTHIAUME MJ, RAYNAULD JP, MARTEL-PELLETIER J, et al. Meniscal tear and extrusion are strongly associated with progression of symptomatic knee osteoarthritis as assessed by quantitative magnetic resonance imaging.Ann Rheum Dis.2005; 64(4):556-563.
[4] LAAVOLA M, LEPPANEN T, HAMALAINEN M, et al. IL-6 in Osteoarthritis: Effects of Pine Stilbenoids. Molecules. 2018;24(1). pii: E109.
[5] CHAUFFIER K, LAIGUILLON MC, BOUGAULT C, et al. Induction of the chemokine IL-8/Kc by the articular cartilage: possible influence on osteoarthritis.Joint Bone Spine.2012;79(6):604-609.
[6] KAPOOR M, MARTEL-PELLETIER J, LAJEUNESSE D, et al. Role of proinflammatory cytokines in the pathophysiology of osteoarthritis.Nat Rev Rheumatol.2011;7(1):33-42.
[7] KONDO S, MUNETA T, NAKAGAWA Y, et al. Transplantation of autologous synovial mesenchymal stem cells promotes meniscus regeneration in aged primates.J Orthop Res.2017;35(6): 1274-1282.
[8] SAULNIER N, VIGUIER E, PERRIER-GROULT E, et al. Intra-articular administration of xenogeneic neonatal Mesenchymal Stromal Cells early after meniscal injury down-regulates metalloproteinase gene expression in synovium and prevents cartilage degradation in a rabbit model of osteoarthritis. Osteoarthritis Cartilage.2015; 23(1):122-133.
[9] HATSUSHIKA D, MUNETA T, HORIE M, et al. Intraarticular injection of synovial stem cells promotes meniscal regeneration in a rabbit massive meniscal defect model.J Orthop Res.2013;31(9): 1354-1359.
[10] WHITEHOUSE MR, HOWELLS NR, PARRY MC, et al. Repair of Torn Avascular Meniscal Cartilage Using Undifferentiated Autologous Mesenchymal Stem Cells: From In Vitro Optimization to a First-in-Human Study.Stem Cells Transl Med.2017;6(4): 1237-1248.
[11] MUSCHLER GF, NAKAMOTO C, GRIFFITH LG. Engineering principles of clinical cell-based tissue engineering.J Bone Joint Surg Am.2004; 86(7):1541-1558.
[12] KOCH M, ACHATZ FP, LANG S, et al. Tissue Engineering of Large Full-Size Meniscus Defects by a Polyurethane Scaffold: Accelerated Regeneration by Mesenchymal Stromal Cells.Stem Cells Int.2018; 2018(8207071.
[13] KWAK HS, NAM J, LEE JH, et al. Meniscal repair in vivo using human chondrocyte-seeded PLGA mesh scaffold pretreated with platelet-rich plasma.J Tissue Eng Regen Med.2017;11(2): 471-480.
[14] ZHANG ZZ, WANG SJ, ZHANG JY, et al. 3D-Printed Poly(epsilon-caprolactone) Scaffold Augmented With Mesenchymal Stem Cells for Total Meniscal Substitution: A 12- and 24-Week Animal Study in a Rabbit Model.Am J Sports Med. 2017; 45(7):1497-1511.
[15] ZHANG ZZ, CHEN YR, WANG SJ, et al. Orchestrated biomechanical, structural, and biochemical stimuli for engineering anisotropic meniscus.Sci Transl Med. 2019;11(487). pii: eaao0750.
[16] BAHCECIOGLU G, HASIRCI N, BILGEN B, et al. Hydrogels of agarose, and methacrylated gelatin and hyaluronic acid are more supportive for in vitro meniscus regeneration than three dimensional printed polycaprolactone scaffolds.Int J Biol Macromol. 2019;122(1152-1162.
[17] GOPINATHAN J, PILLAI MM, SAHANAND KS, et al. Synergistic effect of electrical conductivity and biomolecules on human meniscal cell attachment, growth, and proliferation in poly-epsilon-caprolactone nanocomposite scaffolds.Biomed Mater.2017;12(6):065001.
[18] PATEL JM, GHODBANE SA, BRZEZINSKI A, et al. Tissue-Engineered Total Meniscus Replacement With a Fiber-Reinforced Scaffold in a 2-Year Ovine Model.Am J Sports Med.2018; 46(8):1844-1856.
[19] PATEL JM, MERRIAM AR, CULP BM, et al. One-Year Outcomes of Total Meniscus Reconstruction Using a Novel Fiber-Reinforced Scaffold in an Ovine Model.Am J Sports Med.2016; 44(4):898-907.
[20] MERRIAM AR, PATEL JM, CULP BM, et al. Successful Total Meniscus Reconstruction Using a Novel Fiber-Reinforced Scaffold: A 16- and 32-Week Study in an Ovine Model.Am J Sports Med.2015;43(10):2528-2537.
[21] FOLLIN B, JUHL M, COHEN S, et al. Human adipose-derived stromal cells in a clinically applicable injectable alginate hydrogel: Phenotypic and immunomodulatory evaluation.Cytotherapy.2015; 17(8):1104-1118.
[22] MORADI L, VASEI M, DEHGHAN MM, et al. Regeneration of meniscus tissue using adipose mesenchymal stem cells-chondrocytes co-culture on a hybrid scaffold: In vivo study. Biomaterials.2017;126:18-30.
[23] BODIN A, CONCARO S, BRITTBERG M, et al. Bacterial cellulose as a potential meniscus implant.J Tissue Eng Regen Med.2007; 1(5):406-408.
[24] SILVA MA, LEITE YKC, DE CARVALHO CES, et al. Behavior and biocompatibility of rabbit bone marrow mesenchymal stem cells with bacterial cellulose membrane.Peer J.2018;6:e4656.
[25] NARITA A, TAKAHARA M, SATO D, et al. Biodegradable gelatin hydrogels incorporating fibroblast growth factor 2 promote healing of horizontal tears in rabbit meniscus.Arthroscopy.2012; 28(2): 255-263.
[26] TANAKA T, MATSUSHITA T, NISHIDA K, et al. Attenuation of osteoarthritis progression in mice following intra-articular administration of simvastatin-conjugated gelatin hydrogel.J Tissue Eng Regen Med.2019; 13(3):423-432.
[27] BAHCECIOGLU G, BILGEN B, HASIRCI N, et al. Anatomical meniscus construct with zone specific biochemical composition and structural organization.Biomaterials.2019; 218(119361.
[28] YUE K, TRUJILLO-DE SANTIAGO G, ALVAREZ MM, et al. Synthesis, properties, and biomedical applications of gelatin methacryloyl (GelMA) hydrogels.Biomaterials. 2015;73:254-271.
[29] LI Y, RODRIGUES J, TOMAS H. Injectable and biodegradable hydrogels: gelation, biodegradation and biomedical applications. Chem Soc Rev.2012;41(6):2193-2221.
[30] FENG Q, XU J, ZHANG K, et al. Dynamic and Cell-Infiltratable Hydrogels as Injectable Carrier of Therapeutic Cells and Drugs for Treating Challenging Bone Defects.ACS Cent Sci.2019;5(3): 440-450.
[31] CHEN Y, CHEN J, ZHANG Z, et al. Current advances in the development of natural meniscus scaffolds: innovative approaches to decellularization and recellularization. Cell Tissue Res. 2017;370(1):41-52.
[32] ODA S, OTSUKI S, KUROKAWA Y, et al. A new method for meniscus repair using type I collagen scaffold and infrapatellar fat pad.J Biomater Appl.2015;29(10):1439-1448.
[33] ROONEY P, KUMAR S. Inverse relationship between hyaluronan and collagens in development and angiogenesis.Differentiation. 1993;54(1):1-9.
[34] FENG Z, FAN Y, GUO J, et al. Research progress of scaffold materials for tissue engineered meniscus. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi.2019;33(8):1019-1028.
[35] CHIARI C, KOLLER U, KAPELLER B, et al. Different behavior of meniscal cells in collagen II/I,III and Hyaff-11 scaffolds in vitro. Tissue Eng Part A.2008;14(8):1295-1304.
[36] BHATTACHARJEE P, KUNDU B, NASKAR D, et al. Silk scaffolds in bone tissue engineering: An overview. Acta Biomater.2017; 63:1-17.
[37] GRUCHENBERG K, IGNATIUS A, FRIEMERT B, et al. In vivo performance of a novel silk fibroin scaffold for partial meniscal replacement in a sheep model.Knee Surg Sports Traumatol Arthrosc.2015;23(8):2218-2229.
[38] WARNECKE D, STEIN S, HAFFNER-LUNTZER M, et al. Biomechanical, structural and biological characterisation of a new silk fibroin scaffold for meniscal repair. J Mech Behav Biomed Mater. 2018;86:314-324.
[39] MEINEL L, HOFMANN S, KARAGEORGIOU V, et al. The inflammatory responses to silk films in vitro and in vivo. Biomaterials. 2005;26(2):147-155.
[40] MOSALA NEZHAD Z, PONCELET A, DE KERCHOVE L, et al. Small intestinal submucosa extracellular matrix (CorMatrix(R)) in cardiovascular surgery: a systematic review. Interact Cardiovasc Thorac Surg. 2016; 22(6):839-850.
[41] COOK JL, FOX DB, MALAVIYA P, et al. Evaluation of small intestinal submucosa grafts for meniscal regeneration in a clinically relevant posterior meniscectomy model in dogs.J Knee Surg.2006;19(3):159-167.
[42] WELCH JA, MONTGOMERY RD, LENZ SD, et al. Evaluation of small-intestinal submucosa implants for repair of meniscal defects in dogs.Am J Vet Res.2002;63(3):427-431.
[43] COOK JL, TOMLINSON JL, KREEGER JM, et al. Induction of meniscal regeneration in dogs using a novel biomaterial.Am J Sports Med.1999;27(5):658-665. [44] COOK JL, TOMLINSON JL, ARNOCZKY SP, et al. Kinetic study of the replacement of porcine small intestinal submucosa grafts and the regeneration of meniscal-like tissue in large avascular meniscal defects in dogs. Tissue Eng. 2001; 7(3):321-334.
[45] KHOURY MA, GOLDBERG VM, STEVENSON S. Demonstration of HLA and ABH antigens in fresh and frozen human menisci by immunohistochemistry.J Orthop Res.1994;12(6):751-757.
[46] YAMASAKI T, DEIE M, SHINOMIYA R, et al. Meniscal regeneration using tissue engineering with a scaffold derived from a rat meniscus and mesenchymal stromal cells derived from rat bone marrow.J Biomed Mater Res A.2005; 75(1):23-30.
[47] SANDMANN GH, EICHHORN S, VOGT S, et al. Generation and characterization of a human acellular meniscus scaffold for tissue engineering.J Biomed Mater Res A.2009; 91(2):567-574.
[48] YUAN Z, LIU S, HAO C, et al. AMECM/DCB scaffold prompts successful total meniscus reconstruction in a rabbit total meniscectomy model. Biomaterials. 2016; 111:13-26.
[49] MAKRIS EA, HADIDI P, ATHANASIOU KA. The knee meniscus: structure-function, pathophysiology, current repair techniques, and prospects for regeneration.Biomaterials.2011; 32(30): 7411-7431.
[50] BHARGAVA MM, ATTIA ET, MURRELL GA, et al. The effect of cytokines on the proliferation and migration of bovine meniscal cells.Am J Sports Med.1999; 27(5):636-643.
[51] PANGBORN CA, ATHANASIOU KA. Effects of growth factors on meniscal fibrochondrocytes. Tissue Eng. 2005;11(7-8):1141-1148.
[52] FREYMANN U, ENDRES M, GOLDMANN U, et al. Toward scaffold-based meniscus repair: effect of human serum, hyaluronic acid and TGF-ss3 on cell recruitment and re-differentiation. Osteoarthritis Cartilage. 2013; 21(5):773-781.
[53] MANDAL BB, PARK SH, GIL ES, et al. Stem cell-based meniscus tissue engineering. Tissue Eng Part A.2011; 17(21-22): 2749-2761.
[54] LEE CH, RODEO SA, FORTIER LA, et al. Protein-releasing polymeric scaffolds induce fibrochondrocytic differentiation of endogenous cells for knee meniscus regeneration in sheep.Sci Transl Med.2014; 6(266):266ra171.
[55] UPTON ML, CHEN J, GUILAK F, et al. Differential effects of static and dynamic compression on meniscal cell gene expression.J Orthop Res.2003;21(6):963-969.
[56] ZIELINSKA B, KILLIAN M, KADMIEL M, et al. Meniscal tissue explants response depends on level of dynamic compressive strain. Osteoarthritis Cartilage.2009; 17(6):754-760.
[57] MCNULTY AL, ESTES BT, WILUSZ RE, et al. Dynamic loading enhances integrative meniscal repair in the presence of interleukin-1. Osteoarthritis Cartilage. 2010;18(6):830-838.
[58] NATSU-UME T, MAJIMA T, RENO C, et al.Menisci of the rabbit knee require mechanical loading to maintain homeostasis: cyclic hydrostatic compression in vitro prevents derepression of catabolic genes. J Orthop Sci.2005;10(4):396-405.
[59] GUNJA NJ, UTHAMANTHIL RK, ATHANASIOU KA. Effects of TGF-beta1 and hydrostatic pressure on meniscus cell-seeded scaffolds.Biomaterials.2009;30(4):565-573.
[60] ZHANG Y, WANG F, BAO L, et al. Cyclic hydrostatic compress force regulates apoptosis of meniscus fibrochondrocytes via integrin alpha5beta1.Physiol Res.2019;68(4):639-649.
[61] BAKER BM, SHAH RP, HUANG AH, et al. Dynamic tensile loading improves the functional properties of mesenchymal stem cell-laden nanofiber-based fibrocartilage.Tissue Eng Part A.2011; 17(9-10):1445-1455. |
| [1] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [2] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [3] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [4] | Huang Dengcheng, Wang Zhike, Cao Xuewei. Intravenous, topical tranexamic acid alone or their combination in total knee arthroplasty: a meta-analysis of randomized controlled trials [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 948-956. |
| [5] | Zhan Fangbiao, Cheng Jun, Zou Xinsen, Long Jie, Xie Lizhong, Deng Qianrong. Intraoperative intravenous application of tranexamic acid reduces perioperative bleeding in multilevel posterior spinal surgery: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 977-984. |
| [6] | Yang Xin, Jin Zhe, Feng Xu, Lu Bing. The current situation of knowledge and attitudes towards organ, eye tissue, body donation of residents in Shenyang [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 779-784. |
| [7] | Liu Bo, Chen Xianghe, Yang Kang, Yu Huilin, Lu Pengcheng. Mechanism of DNA methylation in exercise intervention for osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 791-797. |
| [8] | Zhang Guomei, Zhu Jun, Hu Yang, Jiao Hongwei. Stress of three-dimensional finite element models of E-MAX porcelain inlay [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 537-541. |
| [9] | Cheng Jun, Tan Jun, Zhao Yun, Cheng Fangdong, Shi Guojia. Effect of thrombin concentration on the prevention of postoperative cerebrospinal leakage by fibrin glue [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 570-575. |
| [10] | Li Li, Ma Li. Immobilization of lactase on magnetic chitosan microspheres and its effect on enzymatic properties [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 576-581. |
| [11] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [12] | Ye Haimin, Ding Linghua, Kong Weihao, Huang Zutai, Xiong Long. Role and mechanism of hierarchical microchanneled bone scaffolds in promoting osteogenesis and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 621-625. |
| [13] | Yu Langbo, Qing Mingsong, Zhao Chuntao, Peng Jiachen. Hot issues in clinical application of dynamic contrast-enhanced magnetic resonance imaging in orthopedics [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 449-455. |
| [14] | Li Yanle, Yue Xiaohua, Wang Pei, Nie Weizhi, Zhang Junwei, Tan Yonghai, Jiang Hongjiang. Intramedullary nail fixation versus plate fixation in the treatment of displaced midshaft clavicular fractures in adults: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 471-476. |
| [15] | Wang Xinting, Xu Dandi, Zhang Junxia, Su Hailong Wang Qi. Stability of load-bearing cross barrier of different arch structures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3838-3843. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||