Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (15): 2320-2325.doi: 10.3969/j.issn.2095-4344.2014.15.005
Previous Articles Next Articles
Bony ingrowth induced by treadmill exercise in rats with femoral shaft bone defects
Zhang Jie1, Zhang Wen2, Chen Xi2, Luo Zong-ping2, Yang Hui-lin1
- 1 First Affiliated Hospital of Soochow University, Suzhou 215006, Jiangsu Province, China; 2 Orthopaedic Institute of Soochow University, Suzhou 215006, Jiangsu Province, China
-
Online:2014-04-09Published:2014-04-09 -
Contact:Luo Zong-ping, Professor, Doctoral supervisor, Orthopaedic Institute of Soochow University, Suzhou 215006, Jiangsu Province, China -
About author:Zhang Jie, Studying for master’s degree, First Affiliated Hospital of Soochow University, Suzhou 215006, Jiangsu Province, China -
Supported by:the National Natural Science Foundation of China, No. 11072165
CLC Number:
Cite this article
Zhang Jie, Zhang Wen, Chen Xi, Luo Zong-ping, Yang Hui-lin. Bony ingrowth induced by treadmill exercise in rats with femoral shaft bone defects[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(15): 2320-2325.
share this article
2.1 实验动物数量分析 实验纳入12只SD大鼠,全部进行动物造模,实验组给予跑步锻炼,对照组的活动限制在盒子中,12只大鼠没有出现死亡等异常现象,全部进入结果分析。 2.2 micro-CT获得的骨形态学参数 术后1周时,已经有少量的新骨生成。随时间推移,两组的新骨生成量都在逐渐地增加。术后2,3,4周实验组的骨形成分数(BV/TV)均比对照组高,且差异有显著性意义,在第4周时,实验组的骨形成分数比对照组高出11.91%(图1A)。表明给予跑步锻炼之后,成骨效应有明显的提高。 两组新生骨的骨密度都时间推移逐步地升高,说明骨组织在不断地成熟。虽然实验组的骨密度在后3个时间点(术后2,3,4周)均高于对照组,但差异无显著性意义(图1B)。"
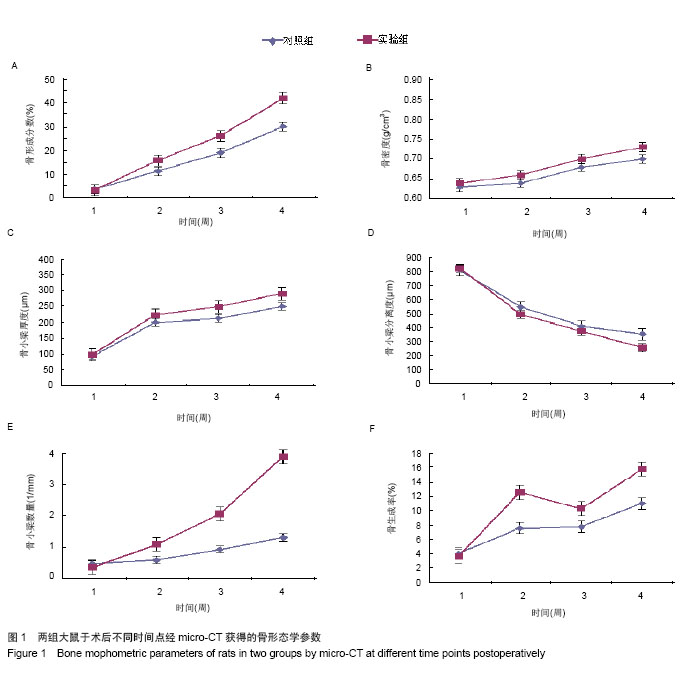
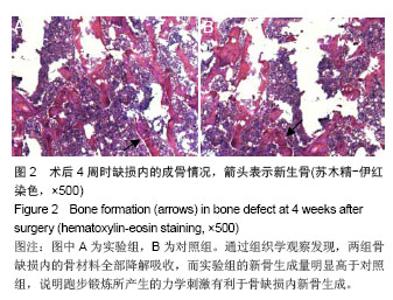
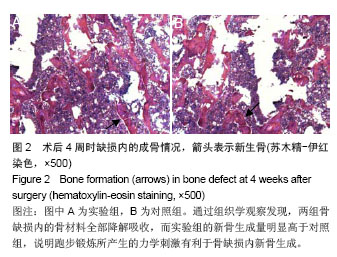
两组骨小梁厚度(Tb.Th)随时间推移逐渐地增加,实验组骨小梁厚度增加的程度要高于对照组,并在后3个时间点(术后2,3,4周)差异有显著性意义(图1C)。 相反,两组骨小梁分离度(Tb.Sp)随时间推移逐渐地降低,但是实验组骨小梁分离度的下降程度要大于对照组,且在第二和最后一个时间点(术后2,4周),差异有显著性意义(图1D)。 两组骨小梁数量(Tb.N)随时间推移逐渐增多,实验组骨小梁数量快速的上升,且在后3个时间(术后2,3,4周),实验组骨小梁数量都要显著高于对照组,差异有显著性意义,表明适度跑步锻炼有利于骨小梁数量的增加(图1E)。 2.3 动态骨形态学变化 不管对于实验组或对照组,骨形成分数(BV/TV)都在不断地增加,说明这样一个事实:缺损处新骨生成速度总是快于骨质吸收速度。通过分析骨生成率(BFR),发现给予跑步锻炼后,实验组骨生成率迅速大幅度上升,而对照组上升幅度明显低于实验组,说明给予跑步锻炼之后,成骨效率是明显增加的。虽然,在此之后实验组骨生成率有所下降,但仍比对照组要高。并且,在最后一个时间点,实验组骨生成率再次出现快速地上升,与对照组相比,差异有显著性意义(图1F)。 2.4 组织学变化 通过组织学观察发现,两组骨缺损内的骨材料全部降解吸收,而实验组的新骨生成量明显高于对照组,说明跑步锻炼所产生的力学刺激有利于骨缺损内新骨生成(图2)。"
| [1]Arrington ED, Smith WJ, Chambers HG,et al. Complications of iliac crest bone graft harvesting.Clin Orthop Relat Res. 1996;(329):300-309. [2]Grauer JN, Beiner JM, Kwon B,et al.The evolution of allograft bone for spinal applications.Orthopedics. 2005;28(6): 573-579. [3]Porter JR, Ruckh TT, Popat KC.Bone tissue engineering: a review in bone biomimetics and drug delivery strategies. Biotechnol Prog. 2009;25(6):1539-1560. [4]Forwood MR, Owan I, Takano Y,et al. Increased bone formation in rat tibiae after a single short period of dynamic loading in vivo.Am J Physiol. 1996;270(3 Pt 1):E419-423. [5]Turner CH, Forwood MR, Otter MW.Mechanotransduction in bone: do bone cells act as sensors of fluid flow. FASEB J. 1994;8(11):875-878. [6]Tanaka SM, Sun HB, Roeder RK,et al.Osteoblast responses one hour after load-induced fluid flow in a three-dimensional porous matrix.Calcif Tissue Int. 2005;76(4):261-271. [7]Zhao F, Chella R, Ma T.Effects of shear stress on 3-D human mesenchymal stem cell construct development in a perfusion bioreactor system: Experiments and hydrodynamic modeling. Biotechnol Bioeng. 2007;96(3):584-595. [8]Song G, Ju Y, Shen X,et al.Mechanical stretch promotes proliferation of rat bone marrow mesenchymal stem cells. Colloids Surf B Biointerfaces. 2007;58(2):271-277. [9]Sikavitsas VI, Bancroft GN, Lemoine JJ,et al.Flow perfusion enhances the calcified matrix deposition of marrow stromal cells in biodegradable nonwoven fiber mesh scaffolds.Ann Biomed Eng. 2005;33(1):63-70. [10]Nordström A, Olsson T, Nordström P.Sustained benefits from previous physical activity on bone mineral density in males.J Clin Endocrinol Metab. 2006;91(7):2600-2644. [11]Yeh JK, Aloia JF, Tierney JM,et al. Effect of treadmill exercise on vertebral and tibial bone mineral content and bone mineral density in the aged adult rat: determined by dual energy X-ray absorptiometry.Calcif Tissue Int. 1993; 52(3):234-238. [12]Bourrin S, Palle S, Pupier R,et al.Effect of physical training on bone adaptation in three zones of the rat tibia.J Bone Miner Res. 1995;10(11):1745-1752. [13]Bouxsein ML, Boyd SK, Christiansen BA,et al.Guidelines for assessment of bone microstructure in rodents using micro-computed tomography.J Bone Miner Res. 2010;25(7): 1468-1486. [14]Meinel L, Karageorgiou V, Fajardo R,et al.Bone tissue engineering using human mesenchymal stem cells: effects of scaffold material and medium flow.Ann Biomed Eng. 2004; 32(1):112-122. [15]Pioletti DP, Montjovent MO, Zambelli PY,et al. Bone tissue engineering using foetal cell therapy.Swiss Med Wkly. 2006; 136(35-36):557-560. [16]Murakami N, Saito N, Horiuchi H,et al. Repair of segmental defects in rabbit humeri with titanium fiber mesh cylinders containing recombinant human bone morphogenetic protein-2 (rhBMP-2) and a synthetic polymer.J Biomed Mater Res. 2002; 62(2):169-174. [17]Richardson TP, Peters MC, Ennett AB,et al. Polymeric system for dual growth factor delivery.Nat Biotechnol. 2001;19(11): 1029-1034. [18]Frost HM.Skeletal structural adaptations to mechanical usage (SATMU): 2. Redefining Wolff's law: the remodeling problem. Anat Rec. 1990;226(4):414-422. [19]Turner CH, Pavalko FM.Mechanotransduction and functional response of the skeleton to physical stress: the mechanisms and mechanics of bone adaptation.J Orthop Sci. 1998;3(6): 346-355. [20]Richardson RS, Wagner H, Mudaliar SR,et al. Human VEGF gene expression in skeletal muscle: effect of acute normoxic and hypoxic exercise.Am J Physiol. 1999;277(6 Pt 2): H2247-2252. [21]Hiscock N, Fischer CP, Pilegaard H,et al.Vascular endothelial growth factor mRNA expression and arteriovenous balance in response to prolonged, submaximal exercise in humans.Am J Physiol Heart Circ Physiol. 2003;285(4):H1759-1763. [22]Kohno S, Kaku M, Tsutsui K,et al.Expression of vascular endothelial growth factor and the effects on bone remodeling during experimental tooth movement. 2003;82(3):177-182. [23]Fong KD, Nacamuli RP, Loboa EG,et al.Equibiaxial tensile strain affects calvarial osteoblast biology.J Craniofac Surg. 2003;14(3):348-355. [24]Hiltunen MO, Ruuskanen M, Huuskonen J,et al. Adenovirus-mediated VEGF-A gene transfer induces bone formation in vivo.FASEB J. 2003;17(9):1147-1149. [25]Yao Z, Lafage-Proust MH, Plouët J,et al. Increase of both angiogenesis and bone mass in response to exercise depends on VEGF.J Bone Miner Res. 2004;19(9):1471-1480. [26]Hausman MR, Schaffler MB, Majeska RJ.Prevention of fracture healing in rats by an inhibitor of angiogenesis.Bone. 2001;29(6):560-564. [27]Kasten TP, Collin-Osdoby P, Patel N,et al. Potentiation of osteoclast bone-resorption activity by inhibition of nitric oxide synthase.Proc Natl Acad Sci U S A. 1994;91(9):3569-3573. [28]MacIntyre I, Zaidi M, Alam AS,et al.Osteoclastic inhibition: an action of nitric oxide not mediated by cyclic GMP.Proc Natl Acad Sci U S A. 1991;88(7):2936-2940. [29]Riancho JA, Salas E, Zarrabeitia MT, et al.Expression and functional role of nitric oxide synthase in osteoblast-like cells. J Bone Miner Res. 1995;10(3):439-446. [30]Riancho JA, Zarrabeitia MT, Fernandez-Luna JL,et al. Mechanisms controlling nitric oxide synthesis in osteoblasts. Mol Cell Endocrinol. 1995;107(1):87-92. [31]Johnson DL, McAllister TN, Frangos JA.Fluid flow stimulates rapid and continuous release of nitric oxide in osteoblasts.Am J Physiol. 1996;271(1 Pt 1):E205-208. [32]Hung CT, Pollack SR, Reilly TM, et al. Real-time calcium response of cultured bone cells to fluid flow.Clin Orthop Relat Res. 1995;(313):256-269. [33]Dillaman RM.Movement of ferritin in the 2-day-old chick femur. Anat Rec. 1984;209(4):445-453. [34]Otter MW, Palmieri VR, Cochran GV.Transcortical streaming potentials are generated by circulatory pressure gradients in living canine tibia.J Orthop Res. 1990;8(1):119-126. [35]Seliger WG.Tissue fluid movement in compact bone.Anat Rec. 1970;166(2):247-255.http://www.ncbi.nlm.nih.gov/pubmed/5414695 [36]Fox SW, Chambers TJ, Chow JW.Nitric oxide is an early mediator of the increase in bone formation by mechanical stimulation.Am J Physiol. 1996;270(6 Pt 1):E955-960. [37]Turner CH, Takano Y, Owan I,et al.Nitric oxide inhibitor L-NAME suppresses mechanically induced bone formation in rats.Am J Physiol. 1996;270(4 Pt 1):E634-639. [38]Tan SD, de Vries TJ, Kuijpers-Jagtman AM,et al.Osteocytes subjected to fluid flow inhibit osteoclast formation and bone resorption.Bone. 2007;41(5):745-751. [39]Robling AG, Bellido T, Turner CH.Mechanical stimulation in vivo reduces osteocyte expression of sclerostin.J Musculoskelet Neuronal Interact. 2006;6(4):354. [40]Santos A, Bakker AD, Zandieh-Doulabi B,et al.Pulsating fluid flow modulates gene expression of proteins involved in Wnt signaling pathways in osteocytes.J Orthop Res. 2009;27(10): 1280-1287. [41]You L, Temiyasathit S, Lee P,et al.Osteocytes as mechanosensors in the inhibition of bone resorption due to mechanical loading.Bone. 2008;42(1):172-179. [42]Gardner MJ, van der Meulen MC, Demetrakopoulos D,et al.In vivo cyclic axial compression affects bone healing in the mouse tibia.J Orthop Res. 2006;24(8):1679-1686. [43]Roshan-Ghias A, Terrier A, Bourban PE,et al. In vivo cyclic loading as a potent stimulatory signal for bone formation inside tissue engineering scaffold.Eur Cell Mater. 2010; 19:41-49. [44]Claes L, Eckert-Hübner K, Augat P.The effect of mechanical stability on local vascularization and tissue differentiation in callus healing.J Orthop Res. 2002;20(5):1099-1105. [45]Goodship AE, Cunningham JL, Kenwright J.Strain rate and timing of stimulation in mechanical modulation of fracture healing.Clin Orthop Relat Res. 1998;(355 Suppl):S105-115. [46]Hente R, Cordey J, Rahn BA,et al.Fracture healing of the sheep tibia treated using a unilateral external fixator. Comparison of static and dynamic fixation.Injury. 1999;30 Suppl 1:A44-51. [47]Roshan-Ghias A, Lambers FM, Gholam-Rezaee M,et al.In vivo loading increases mechanical properties of scaffold by affecting bone formation and bone resorption rates.Bone. 2011;49(6):1357-1364. [48]Claes L, Eckert-Hübner K, Augat P.The effect of mechanical stability on local vascularization and tissue differentiation in callus healing.J Orthop Res. 2002;20(5):1099-1105. [49]Wallace AL, Draper ER, Strachan RK,et al.The vascular response to fracture micromovement.Clin Orthop Relat Res. 1994;(301):281-290. [50]Schulte FA, Lambers FM, Kuhn G,et al. In vivo micro-computed tomography allows direct three-dimensional quantification of both bone formation and bone resorption parameters using time-lapsed imaging.Bone. 2011;48(3):433-442. [51]Hillam RA, Skerry TM.Inhibition of bone resorption and stimulation of formation by mechanical loading of the modeling rat ulna in vivo.J Bone Miner Res. 1995;10(5):683-689. [52]Yeh JK, Aloia JF, Chen MM,et al. Influence of exercise on cancellous bone of the aged female rat.J Bone Miner Res. 1993;8(9):1117-1125. [53]Mow VC, Huiskers R. Basic orthopaedic biomechanics and mechano-biology[M]. Lippincott Williams & Wilkins,2005 [54]Wolf S, Janousek A, Pfeil J,et al.The effects of external mechanical stimulation on the healing of diaphyseal osteotomies fixed by flexible external fixation.Clin Biomech (Bristol, Avon). 1998;13(4-5):359-364. |
| [1] | Hua Haotian, Zhao Wenyu, Zhang Lei, Bai Wenbo, Wang Xinwei. Meta-analysis of clinical efficacy and safety of antibiotic artificial bone in the treatment of chronic osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 970-976. |
| [2] | Chen Junyi, Wang Ning, Peng Chengfei, Zhu Lunjing, Duan Jiangtao, Wang Ye, Bei Chaoyong. Decalcified bone matrix and lentivirus-mediated silencing of P75 neurotrophin receptor transfected bone marrow mesenchymal stem cells to construct tissue-engineered bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 510-515. |
| [3] | Ma Zhijie, Li Jingyu, Cao Fang, Liu Rong, Zhao Dewei. Influencing factors and biological property of novel biomedical materials: porous silicon carbide coated with bioactive tantalum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 558-563. |
| [4] | Shi Xiaoxiu, Mao Shilong, Liu Yang, Ma Xingshuang, Luo Yanfeng. Comparison of tantalum and titanium (alloy) as orthopedic materials: physical and chemical indexes, antibacterial and osteogenic ability [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 593-599. |
| [5] | Yang Junhui, Luo Jinli, Yuan Xiaoping. Effects of human growth hormone on proliferation and osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3956-3961. |
| [6] | Li Xinping, Cui Qiuju, Zeng Shuguang, Ran Gaoying, Zhang Zhaoqiang, Liu Xianwen, Fang Wei, Xu Shuaimei. Effect of modification of β-tricalcium phosphate/chitosan hydrogel on growth and mineralization of dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3493-3499. |
| [7] | Zhou Anqi, Tang Yufei, Wu Bingfeng, Xiang Lin. Designing of periosteum tissue engineering: combination of generality and individuality [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3551-3557. |
| [8] | Chen Song, He Yuanli, Xie Wenjia, Zhong Linna, Wang Jian. Advantages of calcium phosphate nanoparticles for drug delivery in bone tissue engineering research and application [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3565-3570. |
| [9] | Ailimaierdan·Ainiwaer, Wang Ling, Gu Li, Dilidaer•Taxifulati, Wang Shan, Yin Hongbin. Effect of transforming growth factor-beta3 on the proliferation and osteogenic capability of osteoblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(17): 2664-2669. |
| [10] | Huang Na, Liu Jiayue, Huang Yingjie, Wen Junmao, Wang Haibin, Zhang Qingwen, Zhou Chi . Bibliometric and visualized analysis of research on osteonecrosis of the femoral head from the Web of Science in the last 5 years [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(17): 2711-2718. |
| [11] | Zhang Xianjun, Zhao Xijiang. In vivo osteogenic properties of silicon-incorporated titanium dioxide nanotubes on titanium screw surface [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2461-2465. |
| [12] | Chen Jufang, Tian Yulou, Hao Xin. Role and potential of adipose-derived stem cells in cranio-maxillofacial bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2087-2096. |
| [13] | Zhuang Chuanji, Chen Wenzhao, Jiang Xinmin. Osteogenesis in vitro and bone defect repair in vivo of bone morphogenetic protein 9 composite collagen based bone repair material [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1489-1494. |
| [14] | Wu Ming, Zhang Yan. Related factors regulating osteogenic differentiation of bone marrow mesenchymal stem cells through Wnt/β-catenin signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(1): 116-122. |
| [15] | Liu Zhendong, Qin Sihe. Four-dimensional space events of fracture healing [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(6): 903-910. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||