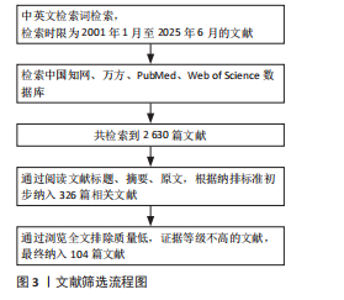
Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (24): 6316-6327.doi: 10.12307/2026.227
Previous Articles Next Articles
Traditional Chinese Medicine compound formula and effective ingredients for treating intervertebral disc degeneration: a multi-targeted and holistic regulation concept
Zhong Haotian1, Li Jianwen2, Han Weichao2, Li Songbo1, 2
- 1The First School of Clinical Medicine, Guangdong Medical University, Zhanjiang 524023, Guangdong Province, China; 2The Tenth Affiliated Hospital of Southern Medical University (Dongguan People’s Hospital), Dongguan 523069, Guangdong Province, China
-
Received:2025-06-30Revised:2025-11-06Online:2026-08-28Published:2026-02-03 -
Contact:Li Songbo, MS, Chief physician, Master’s supervisor, The First School of Clinical Medicine, Guangdong Medical University, Zhanjiang 524023, Guangdong Province, China; The Tenth Affiliated Hospital of Southern Medical University (Dongguan People’s Hospital), Dongguan 523069, Guangdong Province, China -
About author:Zhong Haotian, MS candidate, The First School of Clinical Medicine, Guangdong Medical University, Zhanjiang 524023, Guangdong Province, China -
Supported by:2021 Guangdong Basic and Applied Basic Research Fund Joint Fund, No. 2021B1515140056 (to LSB); 2022 Dongguan Social Development Science and Technology Project, No. 20221800906282 (to LJW)
CLC Number:
Cite this article
Zhong Haotian, Li Jianwen, Han Weichao, Li Songbo. Traditional Chinese Medicine compound formula and effective ingredients for treating intervertebral disc degeneration: a multi-targeted and holistic regulation concept[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6316-6327.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
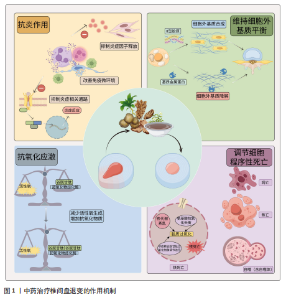
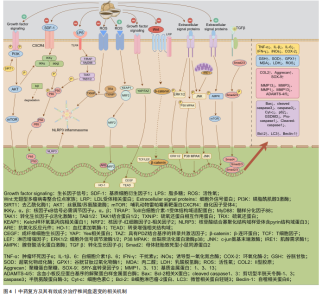
2.1 中药复方及有效成分治疗椎间盘退变的研究时间线 作者查阅并归纳相关文献,将中药复方及有效成分治疗椎间盘退变相关研究进行总结[6-17],相关研究时间线见表1。 2.2 中药复方及有效成分的抗炎作用 炎症被认为是椎间盘退变过程中的关键因素,一些炎性细胞因子如肿瘤坏死因子α、白细胞介素1及相关信号通路如核因子κB、丝裂原活化蛋白激酶被视为是潜在的治疗靶点[11],通过抑制炎症因子释放及相关通路表达可有效缓解炎症反应,从而缓解椎间盘退变的进程。其中,白细胞介素1与肿瘤坏死因子α不仅能够直接激活下游的核因子κB和丝裂原活化蛋白激酶信号通路,进而促进基质金属蛋白和含血小板反应蛋白基序的解聚蛋白样金属蛋白酶(A Disintegrin and Metalloproteinase with Throm-bospondin Motifs,ADAMTS)的表达,这些蛋白广泛作用于纤维环和软骨终板的细胞外基质降解,导致纤维环的结构松散、软骨终板厚度减少[18],还可以进一步上调其他多种炎症因子的表达,从而形成炎症级联反应[19-20]。此外,椎间盘局部的免疫微环境失衡也在椎间盘退变进展中发挥"
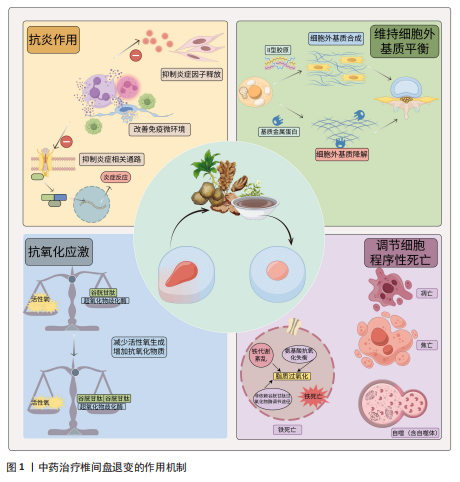

关键作用,尤其是M1型巨噬细胞、中性粒细胞、淋巴细胞等炎症细胞的浸润与活化,释放大量促炎因子,加剧局部炎症;而M2型巨噬细胞则分泌白细胞介素10、转化生长因子β等抗炎因子[21-22],有利于炎症消退与组织修复。中性粒细胞可通过释放炎症因子进一步吸引其他炎症细胞参与炎症,通过激活神经递质诱导纤维环细胞的凋亡[23]。目前大多数药物更倾向于缓解症状[24],并不能从根源上延缓椎间盘退行性变的局部炎症。近年来有越来越多的研究表明,中医药在椎间盘退变病程中具有良好的抗炎作用。 李兆勇等[25]研究发现,桂枝新加汤可降低动静力失衡法建立的颈椎间盘退变大鼠血清及椎间盘中肿瘤坏死因子α、前列腺素2水平,减轻全身与颈椎局部炎症。人参皂苷Rg1是人参中的一种四环三萜衍生物,YANG等[26]研究发现,人参皂苷Rg1能通过抑制Yes相关蛋白1/转录共激活因子1通路激活抑制椎间盘退变大鼠髓核组织中炎症因子水平,缓解炎症反应。HU等[27]研究发现,银杏素通过抑制核因子κB/核苷酸结合寡聚结构域、NLRP3炎症小体活性,抑制椎间盘退变大鼠外周血血清和白细胞介素1β诱导髓核细胞中的白细胞介素6和肿瘤坏死因子α表达,在体内体外发挥抗炎作用,明显改善椎间盘退行性病变。LIU等[28]研究独活寄生汤对基质细胞衍生因子1诱导人髓核细胞炎症的影响,发现独活寄生汤可以抑制肿瘤坏死因子α、白细胞介素1β的产生,同时通过抑制基质细胞衍生因子1/ C-X-C基序趋化因子受体4/核因子κB信号通路发挥抗炎作用,从而延缓椎间盘退变过程。三七皂苷R1是三七的主要有效成分,在骨科疾病中,三七皂苷R1对软骨损伤具有保护作用,可促进骨和软骨的形成。TANG等[29]发现三七皂苷R1在mRNA水平上逆转肿瘤坏死因子α诱导髓核细胞中促炎症因子白细胞介素1β、白细胞介素6和干扰素γ的增加,通过抑制NLRP3信号通路的激活降低白细胞介素1β蛋白水平的表达,减轻炎症反应。LIU等[30]构建了一种可注射温敏水凝胶(聚乳酸-羟基乙酸-聚乙二醇-聚乳酸-羟基乙酸)联合递送单宁酸与白藜芦醇的给药系统,其中单宁酸直接嵌入水凝胶中,白藜芦醇则由炎症响应型纳米粒携带,该系统"


植入后可在炎症刺激下定向释放,在退变髓核细胞及尾椎穿刺椎间盘退变大鼠模型中均表现出良好的抗炎效果,显著抑制肿瘤坏死因子α、白细胞介素1β 等炎症因子的表达,并通过转录组测序差异分析明确该系统主要靶向叉头盒O蛋白炎症信号通路表达的逆转。ZHU等[31]构建了香兰素修饰的明胶甲基丙烯酸酯微球,并负载转化生长因子β3,形成一种兼具抗炎与组织再生功能的多功能给药平台,该系统显著抑制髓核细胞炎症反应中白细胞介素6、肿瘤坏死因子α等炎症因子表达,并且在大鼠椎间盘退变模型中香兰素微球显著减轻椎间盘炎症反应,维持水分含量与椎间盘高度,转录组测序分析提示该系统可能通过抑制磷脂酰肌醇3-激酶-蛋白激酶B信号通路信号通路实现抗炎与组织修复的双重效应。汉黄芩素可通过激活核因子-红细胞因子2-相关因子2/抗氧化反应元件信号通路抑制丝裂原活化蛋白激酶信号通路表达,发挥相似的抗炎作用[32]。基础研究证实,使用染料木素干预髓核细胞可抑制P38丝裂原活化蛋白激酶磷酸化,从而降低炎症因子水平,进而抑制炎症发生,缓解椎间盘退 变[33]。 在调控免疫微环境改善局部炎症方面,ZHAO等[34]研究发现木兰花苷可通过抑制高迁移率族蛋白B1/髓系分化因子88信号通路/核因子κB信号通路及NLRP3炎症小体活化,阻断M1型巨噬细胞对髓核细胞的促炎作用,显著下调白细胞介素1β、肿瘤坏死因子α表达,并促进巨噬细胞M2型表型转化,从而改善椎间盘局部炎症微环境。通督活血汤通过抑制M1型巨噬细胞活性及其炎性因子表达,有效缓解了大鼠椎间盘退变模型中的局部炎症及组织结构破坏[35]。研究表明,黄芪多糖能有效调节巨噬细胞极化状态、增强调节性T细胞免疫调节功能、抑制促炎因子释放,有望成为调控椎间盘免疫微环境的潜在干预手段[36]。除髓核外,中药复方及活性成分的抗炎效果也体现在纤维环与软骨终板上,例如,身遂痛止方能够在腰椎失稳小鼠中下调 RELA 基因及p-P65蛋白水平,从而抑制核因子κB信号通路激活,降低软骨终板中肿瘤坏死因子α、白细胞介素1β、基质金属蛋白13的表达,同时提升聚集蛋白聚糖、Ⅱ型胶原的合成水平,缓解终板结构破坏和功能退化[37]。绿原酸作为一种天然多酚类化合物,也被证实在腰椎不稳定模型小鼠中具有延缓椎间盘退变的作用,例如,GE等[38]研究发现,绿原酸可抑制核因子κB 信号通路活性,降低终板软骨细胞中白细胞介素1β、肿瘤坏死因子α和基质金属蛋白13、ADAMTS5表达,同时抑制软骨终板细胞的炎症反应。在颈椎病兔模型中,桂枝加葛根汤可显著下调纤维环细胞中白细胞介素1β、肿瘤坏死因子α表达,改善椎间盘组织形态,发挥保护作用[39]。 综上所述,大量研究表明,中医药可通过多种机制有效抑制炎症因子释放及炎症通路激活,改善椎间盘的局部炎症状态(表2),但目前对它们在不同免疫细胞介导炎症反应中的具体作用机制仍不清晰。未来研究应进一步聚焦于中药干预下炎症细胞动态变化、信号通路相互关系的调控机制,系统地揭示中药抗炎作用的关键靶点。 2.3 中药复方及有效成分的抗氧化应激作用 氧化应激是指由于线粒体功能障碍导致活性氧积累及抗氧化系统效率降低,进而引发氧化过程与抗氧化防御机制之间的失衡。椎间盘是人体中最大的无血管结构,它独特的缺氧、酸性、营养缺乏环境为氧化应激的发生提供条件[40]。在椎间盘退变的发展过程中,局部活性氧的过量产生和积累发挥着关键作用[19],积累的活性氧可引起线粒体损伤和功能障碍、降低线粒体膜电位,从而诱导纤维环细胞和软骨终板细胞发生凋亡,导致细胞功能障碍和结构退变[41-42]。因此,调控活性氧的产生、改善氧化应激,成为抑制椎间盘退变进展的关键策略。中药因多组分、多靶点且不良反应少,已显示出调控椎间盘内氧化应激和抑制椎间盘退变中的潜力[43]。 有研究报道,虎杖中的虎杖苷在减轻由去乙酰化酶1/哺乳动物雷帕毒素靶蛋白信号通路激活引起的椎间盘退变和氧化应激中扮演了关键角色,虎杖苷在对于提升抗氧化物质如还原型谷胱甘肽和超氧化物歧化酶水平以及降低丙二醛水平等抑制脂质过氧化方面均表现出积极作用[44]。GUO等[45]研究发现从小驳骨中提取出的活性成分——羽扇豆醇,可通过增强线粒体的抗氧化应激抑制高糖诱导兔髓核细胞中活性氧的升高,进而抑制氧化应激,延缓高糖诱导的髓核细胞凋亡。WANG等[46]发现染料木素能够显著改善叔丁基过氧化氢诱导的髓核细胞氧化应激,保护椎间盘免受损伤,这一作用主要归功于染料木素增强了核因子-红细胞因子2-相关因子2介导的抗氧化防御系统,有效逆转了叔丁基过氧化氢导致的髓核细胞退变,而沉默核因子-红细胞因子2-相关因子2可部分消除此保护作用,相应的,染料木素在维持椎间盘形态和促进核因子-红细胞因子2-相关因子2的表达以及改善椎间盘退变方面具有积极影响。有研究报道,硫氧还蛋白相互作用蛋白-NLRP3炎症小体信号通路与椎间盘退变相关,和厚朴酚通过抑制该信号通路下调丙二醛水平、上调超氧化物歧化酶、谷胱甘肽过氧化物酶1的表达,从而抑制线粒体中活性氧的产生,缓解氧化应激对椎间盘的破坏[47]。LI等[48]的体外实验研究发现,补肾活血方能够有效抑制核因子B/Wnt信号通路的激活,这一作用对于逆转氧化应激引起的细胞凋亡和细胞衰老至关重要,有助于维持椎间盘的力学功能和脊柱的稳定性。液相色谱-串联质谱分析显示,桃叶珊瑚苷和丹参素为补肾活血方中的有效成分,它们通过拮抗核因子κB和Wnt信号通路激动剂,有效延缓椎间盘因氧化应激而发生的退行性病变。葛根素可通过降低活性氧水平和激活Wnt/β-catenin信号通路抑制氧化应激诱导的椎间盘纤维环细胞凋亡,从而提高细胞活力,延缓椎间盘退变过程[49]。WANG等[50]研究证实,山柰酚可通过激活核因子-红细胞因子2-相关因子2信号通路及其下游抗氧化因子(如血红素加氧酶1、醌氧化还原酶1)改善过氧化氢诱导的终板软骨细胞活性氧积累和线粒体膜电位丧失,有效保护软骨终板免于氧化应激损伤。同样,SHAO等[51]研究发现,淫羊藿苷可通过激活核因子-红细胞因子2-相关因子2/血红素加氧酶1信号通路显著上调抗氧化蛋白表达,减少活性氧与丙二醛积累,提升超氧化物歧化酶活性,显著抑制软骨细胞凋亡,从而保护终板结构。 综上所述,中医药在缓解椎间盘氧化应激方面展现出较强的多靶点调控能力,尤其在提升抗氧化酶活性、稳定线粒体功能和抑制活性氧积累方面具有独特优势(表3)。然而,不同中药活性成分在不同椎间盘细胞类型中的抗氧化效应仍缺乏系统比较,并且相关信号通路之间的交互机制尚待深入探索。未来应加强多"

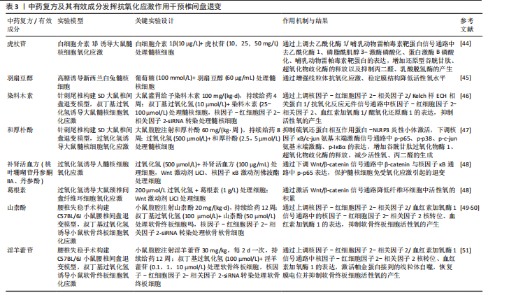
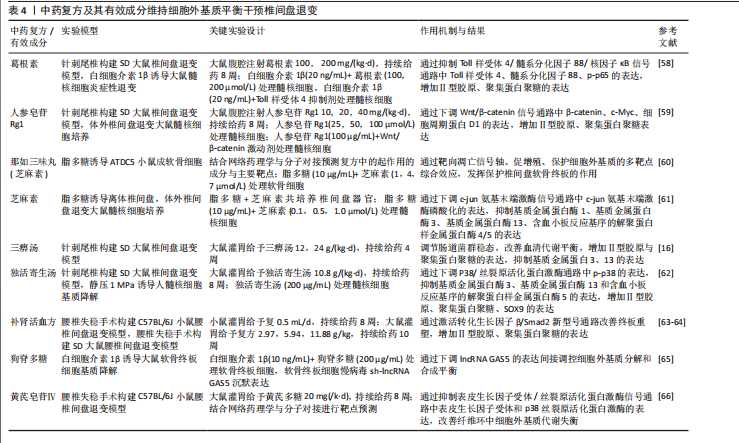
组学联合分析和体内外验证,进一步明确中药在氧化应激调控网络中的关键作用位点与干预策略。 2.4 中药复方及有效成分维持细胞外基质平衡 细胞外基质是由大约300种不同蛋白质构成的非细胞结构。椎间盘中细胞外基质的主要组成部分包括胶原蛋白、蛋白多糖以及非胶原蛋白。细胞外基质的动态平衡对于维持椎间盘的正常生理功能至关重要。髓核细胞散布在一个由多糖和胶原蛋白交织而成的复杂网络之中,这些细胞与周围的细胞外基质进行着持续的双向互动,以保持组织稳态。在健康状态下,细胞外基质的合成与降解过程保持着一种精细的动态平衡,然而当这种平衡因多种因素的干扰而受到破坏时,椎间盘退变便会发生[52]。研究表明,基质金属蛋白酶1、基质金属蛋白酶3、基质金属蛋白酶13和ADAMTS4/5过度激活,伴随合成标志物Ⅱ型胶原和聚集蛋白聚糖下调,会促发胶原纤维紊乱、弹性模量升高、钙化层增厚及渗透性下降,从而削弱纤维环的抗张强度并阻断软骨终板的营养输送链,驱动椎间盘退变进程[53-57]。因此,维持细胞外基质的平衡是改善椎间盘退变过程中的一个关键环节,对于保护和恢复椎间盘的健康状态具有重要意义。 TANG等[58]研究提到,葛根素也可通过抑制Toll样受体4/核因子κB信号通路的激活上调Ⅱ型胶原和聚集蛋白聚糖的表达,表明葛根素可能通过抑制与细胞外基质降解相关的酶的活性促进细胞外基质合成,从而具有减缓椎间盘退变进展的潜力。YU等[59]研究证明,人参皂苷Rg1可通过负调控Wnt/β-catenin信号通路促进退行性髓核细胞中细胞外基质合成,有效改善椎间盘退变。GUO等[60]研究发现,通过网络药理学预测那如三味丸的主要靶点与细胞外基质代谢相关,那如三味丸的主要成分芝麻素可显著上调ATDC5细胞中聚集蛋白聚糖与Ⅱ型胶原表达、抑制基质金属蛋白酶3表达,改善终板细胞外基质紊乱,从而延缓椎间盘退变进程。LI等[61]研究发现,芝麻素通过抑制c-jun"
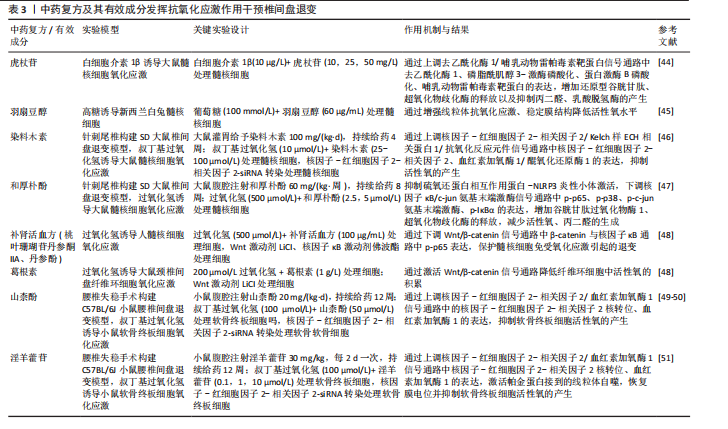
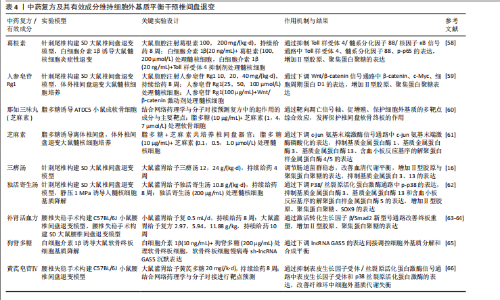
氨基末端激酶的激活来抑制细胞外基质分解代谢,维持基质代谢平衡。WANG等[16]研究发现,三痹汤能够显著降低细胞外基质中基质金属蛋白酶3、基质金属蛋白酶13和ADAMTS4的表达,同时增加聚集蛋白聚糖、Ⅱ型胶原和SOX9蛋白水平,免疫组化染色结果进一步证实了三痹汤可减少基质金属蛋白酶13表达、增加Ⅱ型胶原表达,表明三痹汤通过重塑细胞外基质的平衡恢复退行性椎间盘的结构;该研究还暗示了三痹汤的作用可能与它改善肠道菌群稳态和血清代谢平衡有关,这为中医药在椎间盘退变治疗中的应用提供了新的视角。LIU等[62]研究发现,独活寄生汤在体内外能够有效抑制P38/丝裂原活化蛋白激酶信号通路的活化,进而降低基质金属蛋白酶3、基质金属蛋白酶13和ADAMTS5的表达水平,同时提升Ⅱ型胶原、聚集蛋白聚糖、SOX9表达,延缓椎间盘的退变过程。康佳琪等[63]研究发现,补肾活血方可改善腰椎间盘退变小鼠的软骨终板结构紊乱,增加终板厚度。张若谷等[64]研究发现,补肾活血方可能通过激活转化生长因子β/SMAD家族成员2信号通路调节软骨终板的细胞外基质平衡。狗脊多糖可通过降低lncRNA GAS5表达上调聚集蛋白聚糖水平,维持终板软骨细胞的细胞外基质稳态[65]。网络药理学预测显示,黄芪皂苷Ⅳ的作用靶点主要涉及细胞外基质代谢、磷脂酰肌醇3-激酶-蛋白激酶B与核因子κB信号通路;体外实验证实,黄芪皂苷Ⅳ可上调Ⅱ型胶原与聚集蛋白聚糖表达、抑制基质金属蛋白酶3与ADAMTS5表达,维持纤维环细胞的细胞外基质稳态,从而延缓椎间盘退变进程[66]。 综上所述,现有研究已证实,中药可通过调控细胞外基质的合成与降解酶活性有效缓解椎间盘结构紊乱(表4),然而,目前对中药如何精准调控纤维环与软骨终板中不同组分的细胞外重塑及其空间异质性的理解仍不深入。未来应进一步探索不同中药成分或复方在椎间盘各区域的具体作用模式与靶向特异性,以更全面地揭示中药干预细胞外基质动态稳态的作用机制。 2.5 中药复方及有效成分参与程序性细胞死亡 椎间盘退变与髓核细胞的减少有着密切联系。在细胞死亡的主要形式中,程序性细胞死亡在椎间盘退变的发病机制中起着重要作用[67],程序性细胞死亡包括凋亡、铁死亡、焦亡、自噬等,这些过程在椎间盘退变中的作用已被广泛研究[68-69]。近年来,针对程序性细胞死亡治疗椎间盘退变的中药复方及其有效成分研究逐渐增多,为中药在椎间盘退变治疗中的应用提供了新的机制见解。 2.5.1 抑制细胞凋亡 细胞凋亡是椎间盘退变中的关键病理过程,主要通过外源性死亡受体途径、内源性线粒体途径以及内质网应激途径来启动[70-72],这些途径调节椎间盘细胞的凋亡过程。近年来,针对这些途径的治疗策略,尤其是中药复方及其有效成分的研究,已成为改善椎间盘退变的新热点[19,24]。 LI等[73]研究发现,白藜芦醇可清除过量的活性氧,显著抑制硝普钠诱导髓核细胞经内源性线粒体途径的凋亡、降低椎间盘中矢状面Tunel阳性细胞百分比,在抗凋亡方面具有潜力。实验发现,柚皮苷可通过下调Caspase-3和Bax的表达、增加抗凋亡分子Bcl-2的表达,从而减弱叔丁基过氧化氢诱导的椎间盘退变效果[74]。LUO 等[75]研究揭示了小檗碱在过氧化氢介导髓核细胞凋亡中的保护作用,可能与抑制肌醇需求酶1/c-jun氨基末端激酶信号通路激活调节内质网应激和自噬过程、诱导细胞内钙离子失衡有关。GAO等[76]研究结果表明,补肾活血方可恢复线粒体膜电位,从而恢复线粒体膜通透性转换孔功能,减少活性氧的积累,保护线粒体功能,抑制细胞凋亡,发挥抗椎间盘退变活性。实验证明,独活寄生汤能够下调miR-494表达、上调去乙酰化酶3表达,通过调节miR494/去乙酰化酶3/线粒体自噬信号通路抑制白细胞介素1β诱导的髓核细胞凋亡和线粒体功能障碍,促进Bcl-2蛋白分泌、降低Bax蛋白含量,进而阻断椎间盘退变进展[77]。WAN等[78]研究发现,姜黄素在体外白细胞介素1β诱导的大鼠终板软骨细胞模型中表现出明显的抗凋亡作用,姜黄素干预可显著提高细胞活力,并通过上调Bcl-2表达、下调Bax表达抑制线粒体通路介导的细胞凋亡,从而保护终板结构、延缓椎间盘退变进程。 2.5.2 抑制细胞焦亡 焦亡是近10年来备受关注的新型细胞死亡,它的特征是炎症因子分泌和细胞破坏,与椎间盘退变有关[79]。近几年内也有不少研究表明,中医药通过抑制细胞焦亡改善椎间盘退变。GONG等[80]研究发现麦芽酚可抑制髓核细胞的焦亡,通过使用白细胞介素1β建立细胞焦亡模型,分别给予梯度浓度的麦芽酚干预,结果显示与对照组相比,各治疗组中NLRP3、Pro-Caspase1、Cleaved-Caspase-1、Cleaved-白细胞介素1β的表达量呈浓度依赖性形式降低。WANG等[81]在脂多糖诱导髓核"

细胞焦亡的实验中发现,强筋壮骨祛风合剂可通过抑制NLRP3、白细胞介素1β的表达降低细胞焦亡相关蛋白NLRP3、Caspase-1、Gasdermin D(GSDMD)表达,减少脂多糖诱导的髓核细胞焦亡。GE等[82]实验证实,增加六味地黄汤中山茱萸含量能够抑制椎间盘退变小鼠模型中白细胞介素1β、Caspase-1和GSDMD表达,抑制髓核细胞焦亡,同时上调Ⅱ型胶原表达水平,增强腰椎的整体功能,其中5倍量山茱萸的改良六味地黄汤组表现出卓越的功效。GUO等[83]在动物椎间盘退变模型中发现,独活寄生汤可通过抑制基质细胞衍生因子1/C-X-C基序趋化因子受体4-核因子κB-NLRP3轴抑制椎间盘促炎因子产生和减轻髓核细胞焦亡,进而发挥延缓、防治椎间盘退变的作用。YAO等[84]在腰椎失稳手术构建的腰椎间盘退变小鼠模型中发现,骨痹汤同样能通过抑制核因子κB信号通路活化,显著降低软骨终板细胞中 NLRP3、Caspase-1、GSDMD等焦亡相关因子的表达,从而改善软骨终板的多孔结构,提升椎间盘结构的完整性与基质代谢,延缓椎间盘退变进程。 2.5.3 抑制铁死亡 铁死亡在2012年首先被提出[85],铁死亡的特点是细胞内活性氧在铁离子作用下使细胞膜多不饱和脂肪酸过氧化并出现膜损伤,导致铁代谢、脂质代谢、氨基酸代谢失衡,引起细胞死亡[86-87]。近年来随着铁死亡研究的深入,多种退行性疾病已被发现与铁死亡相关。椎间盘退变是一种代表性且常见的退行性疾病,也被证明与铁死亡显著相关[88-91],而中医药因多靶点的优势在改善铁死亡方面有着巨大潜力。 ZHANG等[92]在肿瘤坏死因子α诱导的髓核细胞铁死亡与大鼠椎间盘退变模型中发现,仙鹤草素可增加髓核细胞和退变椎间盘中核因子-红细胞因子2-相关因子2、谷胱甘肽过氧化物酶4 的表达,抑制髓核细胞中Fe2+和丙二醛的积累,减轻肿瘤坏死因子α诱导的细胞铁死亡,从而抑制椎间盘退变的进展。通过肿瘤坏死因子α和叔丁基过氧化氢诱导的终板软骨细胞炎症和氧化应激模型发现,淫羊藿苷可增加谷胱甘肽过氧化物酶4、铁重链蛋白1、溶质载体家族7成员11的蛋白水平和超氧化物歧化酶活性,并且敲低核因子-红细胞因子2-相关因子2表达阻断了淫羊藿苷的上述作用,表明淫羊藿苷可能通过激活核因子-红细胞因子2-相关因子2/血红素加氧酶1信号通路抑制肿瘤坏死因子α和过氧化氢诱导的软骨细胞铁死亡,最终在椎间盘退变中发挥保护作用[51]。实验证实,橙皮苷通过增强核因子-红细胞因子2-相关因子2表达和抑制核因子κB信号通路,上调谷胱甘肽过氧化物酶4、铁重链蛋白1的表达,下调长链酰基辅酶a合成酶4、前列腺素内过氧化物合酶2的表达,抑制氧化应激依赖的铁死亡,从而保护人髓核细胞[93]。CUI等[94]研究发现,葛根素通过LINC01535途径显著增强人类髓核间充质干细胞中Ⅱ型胶原和聚集蛋白聚糖的表达,下调基质金属蛋白酶3、基质金属蛋白酶13和ADAMTS5的mRNA表达。 2.5.4 调节细胞自噬 细胞自噬是细胞适应环境变化、维持体内平衡的过程[95],主要通过自噬泡与溶酶体相结合进而降解细胞内衰老和受损的细胞器,清除代谢产物及有毒物质,从而在饥饿、低氧等应激状态下保护细胞[96]。近年来在延缓椎间盘退变方面,自噬作为研究热点受到广泛关注和深入探索。 WANG等[97]研究发现,经槲皮素治疗后退变髓核细胞中微管相关蛋白轻链3-Ⅱ/微管相关蛋白轻链3-Ⅰ的比率以剂量依赖性方式增加,p62表达水平则以剂量依赖性方式下降,并在后续研究发现槲皮素通过去乙酰化酶1介导的自噬保护髓核细胞免于凋亡,减轻椎间盘退变的进展。芹菜素是一种广泛存在于中药中的黄酮类成分,XIE等[98]发现芹菜素可以恢复叔丁基过氧化氢诱导的髓核细胞自噬通量阻滞,逆转叔丁基过氧化氢引发的转录因子EB核转移减少,促进转录因子EB的核表达水平,并在进一步研究发现,芹菜素可能通过激活腺苷酸活化蛋白激酶/哺乳动物雷帕毒素靶蛋白通路靶向恢复转录因子EB水平的自噬流量,发挥治疗椎间盘退变的作用。DAI等[12]基于动物实验和细胞实验提出,益气活血汤可以调控腺苷酸活化蛋白激酶/液泡蛋白分选34通路提高自噬水平、促进Beclin1-液泡蛋白分选34复合物形成,通过激活上游蛋白腺苷酸活化蛋白激酶和上调去泛素化酶13来激活自噬,达到延缓椎间盘退变的作用。ZHANG等[99]研究通过对脂多糖诱导与芝麻素诱导的ATDC5细胞转录组测序分析发现,BECN2是调控该细胞的关键靶点,并在后续研究中证明芝麻素可通过上调BECN2抑制自噬相关基因14、液泡蛋白分选34、生长与分化因子相关血清蛋白1等自噬关键蛋白的表达,从而减轻软骨终板的退变。柚皮苷通过激活去乙酰化酶3/FOXO3a/帕金蛋白信号通路促进线粒体自噬,有效减少软骨终板钙化,保护椎间盘终板结构[100]。CHEN等[17]构建了一种基于基质金属蛋白酶13响应的时序递释系统,将银杏素与雷帕霉素联合递送,通过靶向调控自噬显著缓解椎间盘退变的进展,该系统利用外泌体递送银杏素早期抑制白细胞介素1β诱导的炎症反应,雷帕霉素则于中后期由介孔二氧化硅纳米粒释放,进一步增强微管相关蛋白轻链3-Ⅱ表达、减少p62积聚、促进溶酶体相关膜蛋白1共定位,从而恢复受炎症阻断的自噬流;体内研究表明该中药递送系统可显著改善椎间盘水分含量、基质合成能力及组织结构完整性。这一研究不仅验证了银杏素作为中药单体成分在调控自噬方面的显著效应,也表明中药结合智能材料系统的策略为精准干预椎间盘退变提供了新方向。 综上所述,凋亡、焦亡、铁死亡及自噬等多种程序性死亡形式共同参与了椎间盘退变的发生发展(表5),它们之间并非孤立存在,而是通过复杂的信号网络相互影响。由于这些程序性死亡途径彼此交织,单一靶向一种细胞死亡的干预可能不足以全面阻止退变进程,甚至抑制某一路径后可能出现其他死亡途径的补偿性增强,因此,利用中药多靶点、整体调节的优势有望同时调控多个程序性死亡过程,从多层面保护椎间盘细胞,延缓退变。然而,目前针对椎间盘退变的中药研究大多按不同程序性死亡类型分别展开,对于这些细胞死亡形式间相互作用机制及中药综合调控作用的认识仍显不足。未来有必要加强对不同程序性死亡途径机制的研究,阐明中药如何协同影响这些途径,并优化中药复方的组分配比,以实现对椎间盘退变更有效的整体干预;此外,将上述实验结果向临床转化、评估长期应用的安全性和有效性,也是中药介入椎间盘退变领域亟待解决的挑战和空白。"

| [1] MOHD ISA IL, TEOH SL, MOHD NOR NH, et al. Discogenic Low Back Pain: Anatomy, Pathophysiology and Treatments of Intervertebral Disc Degeneration. Int J Mol Sci. 2022;24(1):208. [2] BINCH ALA, FITZGERALD JC, GROWNEY EA, et al. Cell-based strategies for IVD repair: clinical progress and translational obstacles. Nat Rev Rheumatol. 2021;17(3):158-175. [3] KNEZEVIC NN, CANDIDO KD, VLAEYEN JWS, et al. Low back pain. Lancet. 2021;398(10294):78-92. [4] ZHU S, WANG J, SUO M, et al. Can extracellular vesicles be considered as a potential frontier in the treatment of intervertebral disc disease? Ageing Res Rev. 2023;92:102094. [5] REN JL, YANG L, QIU S, et al. Efficacy evaluation, active ingredients, and multitarget exploration of herbal medicine. Trends Endocrinol Metab. 2023;34(3):146-157. [6] 金天翔,袁军,高俊,等.从《黄帝内经》论椎间盘及椎间盘突出症的中医病因病机与治疗方法[J].中医正骨,2024,36(7):59-62,68. [7] 林垂聪,吴春雷,叶澄宇,等.丹参注射液对退变颈椎间盘影响的实验研究[J].中国中医基础医学杂志,2003(8):22-25. [8] HAYASHI AM, MATERA JM, DA SILVA TS, et al. Electro-acupuncture and Chinese herbs for treatment of cervical intervertebral disk disease in a dog. J Vet Sci. 2007;8(1):95-98. [9] CHOW DH, LAI A, TANG FH, et al. Effects of Panax ginseng-containing herbal plasters on compressed intervertebral discs in an in vivo rat tail model. Chin Med. 2013;8(1):4. [10] YU PF, JIANG H, LIU JT, et al. Traditional Chinese medicine treatment for ruptured lumbar disc herniation: clinical observations in 102 cases. Orthop Surg. 2014;6(3):229-235. [11] ZHANG B, XU H, WANG J, et al. A narrative review of non-operative treatment, especially traditional Chinese medicine therapy, for lumbar intervertebral disc herniation. Biosci Trends. 2017; 11(4):406-417. [12] DAI F, YU P, YU Z, et al. Yiqi Huoxue Recipe Delayed Intervertebral Disc Degeneration by Activating Autophagy. Front Pharmacol. 2021;12:705747. [13] SUN C, SUN K, WANG S, et al. Effect of Baimai ointment on lumbar disc herniation: A multicentre, prospective, randomised, double-blind, placebo-controlled trial. Phytomedicine. 2024;122:155138. [14] QIN X, SUN K, XU W, et al. An evidence-based guideline on treating lumbar disc herniation with traditional Chinese medicine. J Evid Based Med. 2024;17(1):187-206. [15] 秦晓宽,孙凯,徐卫国,等.腰椎间盘突出症中医循证实践指南[J].西部中医药,2024, 37(5):1-15. [16] WANG N, CHEN S, XIE Y, et al. The Sanbi Decoction alleviates intervertebral disc degeneration in rats through intestinal flora and serum metabolic homeostasis modulation. Phytomedicine. 2024; 127:155480. [17] CHEN J, GAN X, SU S, et al. Conditional sequential delivery of ginkgetin and rapamycin orchestrates inflammation and autophagy to alleviate intervertebral disc degeneration. J Control Release. 2025;381:113556. [18] KIM H, HONG JY, LEE J, et al. IL-1β promotes disc degeneration and inflammation through direct injection of intervertebral disc in a rat lumbar disc herniation model. Spine J. 2021;21(6):1031-1041. [19] 郭佳霖,何志伟,杨学军.天然小分子治疗椎间盘退变的作用机制研究进展[J].中国脊柱脊髓杂志,2022,32(10):945-953. [20] 王伯阳,刘瑞,王跃文.椎间盘退行性变相关分子机制相关研究进展[J].内蒙古医科大学学报,2019, 41(5):555-557,560. [21] YAMAGISHI A, NAKAJIMA H, KOKUBO Y, et al. Polarization of infiltrating macrophages in the outer annulus fibrosus layer associated with the process of intervertebral disc degeneration and neural ingrowth in the human cervical spine. Spine J. 2022;22(5):877-886. [22] LI L, WEI K, DING Y, et al. M2a Macrophage-Secreted CHI3L1 Promotes Extracellular Matrix Metabolic Imbalances via Activation of IL-13Rα2/MAPK Pathway in Rat Intervertebral Disc Degeneration. Front Immunol. 2021;12:666361. [23] ZHANG Y, ZHANG J, SUN Z, et al. MAPK8 and CAPN1 as potential biomarkers of intervertebral disc degeneration overlapping immune infiltration, autophagy, and ceRNA. Front Immunol. 2023;14: 1188774. [24] RIDER SM, MIZUNO S, KANG JD. Molecular Mechanisms of Intervertebral Disc Degeneration. Spine Surg Relat Res. 2019;3(1):1-11. [25] 李兆勇,杨雷,郭彦涛,等.桂枝新加汤对颈椎间盘退变大鼠模型椎间盘TNF-α及血清中炎症因子的影响[J].辽宁中医杂志,2024,51(12): 184-188,238-239. [26] YANG YH, GU XP, HU H, et al. Ginsenoside Rg1 inhibits nucleus pulposus cell apoptosis, inflammation and extracellular matrix degradation via the YAP1/TAZ pathway in rats with intervertebral disc degeneration. J Orthop Surg Res. 2022;17(1):555. [27] HU B, LIN S, LIN S, et al. Ginkgetin Alleviates Intervertebral Disc Degeneration by Inhibiting Apoptosis, Inflammation, and Disturbance of Extracellular Matrix Synthesis and Catabolism via Inactivation of NLRP3 Inflammasome. Immunol Invest. 2023;52(5):546-560. [28] LIU ZC, WANG ZL, HUANG CY, et al. Duhuo Jisheng Decoction inhibits SDF-1-induced inflammation and matrix degradation in human degenerative nucleus pulposus cells in vitro through the CXCR4/NF-κB pathway. Acta Pharmacol Sin. 2018; 39(6):912-922. [29] TANG K, SU W, HUANG C, et al. Notoginsenoside R1 suppresses inflammatory response and the pyroptosis of nucleus pulposus cells via inactivating NF-κB/NLRP3 pathways. Int Immunopharmacol. 2021;101(Pt B):107866. [30] LIU Y, GUO C, WANG Y, et al. Application of an Injectable Thermosensitive Hydrogel Drug Delivery System for Degenerated Intervertebral Disc Regeneration. Biomacromolecules. 2025;26(1): 209-221. [31] ZHU Z, YU Q, LI H, et al. Vanillin-based functionalization strategy to construct multifunctional microspheres for treating inflammation and regenerating intervertebral disc. Bioact Mater. 2023;28:167-182. [32] FANG W, ZHOU X, WANG J, et al. Wogonin mitigates intervertebral disc degeneration through the Nrf2/ARE and MAPK signaling pathways. Int Immunopharmacol. 2018;65:539-549. [33] GE J, ZHOU Q, CHENG X, et al. The protein tyrosine kinase inhibitor, Genistein, delays intervertebral disc degeneration in rats by inhibiting the p38 pathway-mediated inflammatory response. Aging (Albany NY). 2020;12(3):2246-2260. [34] ZHAO F, GUO Z, HOU F, et al. Magnoflorine Alleviates “M1” Polarized Macrophage-Induced Intervertebral Disc Degeneration Through Repressing the HMGB1/Myd88/NF-κB Pathway and NLRP3 Inflammasome. Front Pharmacol. 2021;12:701087. [35] 鄢来军,葛海雅,汪正明,等.通督活血汤抑制巨噬细胞炎症延缓大鼠椎间盘退变的机制[J].中国组织工程研究,2025,29(32):6851-6857. [36] LI CX, LIU Y, ZHANG YZ, et al. Astragalus polysaccharide: a review of its immunomodulatory effect. Arch Pharm Res. 2022;45(6):367-389. [37] WANG X, ZENG Q, GE Q, et al. Protective effects of Shensuitongzhi formula on intervertebral disc degeneration via downregulation of NF-κB signaling pathway and inflammatory response. J Orthop Surg Res. 2024;19(1):80. [38] GE Q, YING J, SHI Z, et al. Chlorogenic Acid retards cartilaginous endplate degeneration and ameliorates intervertebral disc degeneration via suppressing NF-κB signaling. Life Sci. 2021;274: 119324. [39] 李扬,张志文,黎邦直,等.桂枝加葛根汤对颈椎病模型兔纤维环细胞凋亡和炎性因子的影响[J].湖北中医药大学学报,2021,23(5):13-16. [40] XIA Q, ZHAO Y, DONG H, et al. Progress in the study of molecular mechanisms of intervertebral disc degeneration. Biomed Pharmacother. 2024; 174:116593. [41] NI B, SHEN H, WANG W, et al. TGF-β1 reduces the oxidative stress-induced autophagy and apoptosis in rat annulus fibrosus cells through the ERK signaling pathway. J Orthop Surg Res. 2019;14(1):241. [42] KANG L, LIU S, LI J, et al. Parkin and Nrf2 prevent oxidative stress-induced apoptosis in intervertebral endplate chondrocytes via inducing mitophagy and anti-oxidant defenses. Life Sci. 2020;243:117244. [43] 杨正汉,赵继荣,马俊飞,等.基于氧化应激的中医药干预椎间盘退变的研究进展[J].中国现代应用药学,2024,41(1):138-144. [44] 左斌,夏晓枫,车彪,等.虎杖苷对小鼠腰椎间盘退变髓核细胞凋亡及SIRT1/mTOR通路的影响[J].中国组织工程研究,2021,25(35): 5619-5625. [45] GUO MB, WANG DC, LIU HF, et al. Lupeol against high-glucose-induced apoptosis via enhancing the anti-oxidative stress in rabbit nucleus pulposus cells. Eur Spine J. 2018;27(10):2609-2620. [46] WANG K, HU S, WANG B, et al. Genistein protects intervertebral discs from degeneration via Nrf2-mediated antioxidant defense system: An in vitro and in vivo study. J Cell Physiol. 2019; 234(9):16348-16356. [47] TANG P, GU J M, XIE Z A, et al. Honokiol alleviates the degeneration of intervertebral disc via suppressing the activation of TXNIP-NLRP3 inflammasome signal pathway. Free Radic Biol Med. 2018;120:368-379. [48] LI L, WEI X, LI K, et al. Traditional Chinese Medicine formula Bu-Shen-Huo-Xue-Fang (BSHXF) protects nucleus pulposus cells against the inflammatory and oxidative stress-induced degenerative changes. J Pharm Biomed Anal. 2023;236:115656. [49] 任彬彬,宋晓磊,刘永涛,等.葛根素对H2O2诱导的颈椎间盘纤维环细胞凋亡的影响及机制[J].中国老年学杂志,2019,39(6):1424-1427. [50] WANG H, LIU X, YANG H, et al. Activation of the Nrf-2 pathway by pinocembrin safeguards vertebral endplate chondrocytes against apoptosis and degeneration caused by oxidative stress. Life Sci. 2023;333:122162. [51] SHAO Y, SUN L, YANG G, et al. Icariin protects vertebral endplate chondrocytes against apoptosis and degeneration via activating Nrf-2/HO-1 pathway. Front Pharmacol. 2022;13:937502. [52] LIANG H, LUO R, LI G, et al. The Proteolysis of ECM in Intervertebral Disc Degeneration. Int J Mol Sci. 2022;23(3):1715. [53] ARIPAKA SS, BECH-AZEDDINE R, JØRGENSEN L M, et al. The expression of metalloproteinases in the lumbar disc correlates strongly with Pfirrmann MRI grades in lumbar spinal fusion patient. Brain Spine. 2022;2:100872. [54] KIBBLE MJ, DOMINGOS M, HOYLAND JA, et al. Importance of Matrix Cues on Intervertebral Disc Development, Degeneration, and Regeneration. Int J Mol Sci. 2022;23(13):6915. [55] WANG WJ, YU XH, WANG C, et al. MMPs and ADAMTSs in intervertebral disc degeneration. Clin Chim Acta. 2015;448:238-246. [56] LIU MH, SUN C, YAO Y, et al. Matrix stiffness promotes cartilage endplate chondrocyte calcification in disc degeneration via miR-20a targeting ANKH expression. Sci Rep. 2016;6: 25401. [57] PENG B, HOU S, SHI Q, et al. The relationship between cartilage end-plate calcification and disc degeneration: an experimental study. Chin Med J (Engl). 2001;114(3):308-312. [58] TANG H, ZHANG S, LU X, et al. Effects of puerarin on the intervertebral disc degeneration and biological characteristics of nucleus pulposus cells. Pharm Biol. 2023;61(1):12-22. [59] YU L, HAO Y, PENG C, et al. Effect of Ginsenoside Rg1 on the intervertebral disc degeneration rats and the degenerative pulposus cells and its mechanism. Biomed Pharmacother. 2020;123:109738. [60] GUO J, XUE J, HE Z, et al. The mechanism by which Naru 3 pill protects against intervertebral disc cartilage endplate degeneration based on network pharmacology and experimental verification. J Orthop Surg Res. 2023;18(1):552. [61] LI K, LI Y, XU B, et al. Sesamin inhibits lipopolysaccharide-induced inflammation and extracellular matrix catabolism in rat intervertebral disc. Connect Tissue Res. 2016;57(5):347-359. [62] LIU W, JIN S, HUANG M, et al. Duhuo jisheng decoction suppresses matrix degradation and apoptosis in human nucleus pulposus cells and ameliorates disc degeneration in a rat model. J Ethnopharmacol. 2020;250:112494. [63] 康佳琪,郭宵飞,苏红梅,等.补肾活血方对大鼠肾虚血瘀型腰椎间盘退变的干预作用[J].中国药物警戒,2025,22(3):249-257. [64] 张若谷,葛钦文,邹凯奥,等.补肾活血方对腰椎间盘退变小鼠腰椎终板软骨的影响及其作用机制[J]. 中医正骨,2025,37(3):5-13. [65] 付长龙,梅阳阳,金灵璐,等.狗脊多糖调控lncRNA GAS5维持软骨细胞外基质稳态的机制[J].中华中医药杂志,2022,37(12):7379-7383. [66] CHEN D, FAN T, SUN K, et al. Network pharmacology and experimental validation to reveal the pharmacological mechanisms of Astragaloside Ⅳ in treating intervertebral disc degeneration. Eur J Pharmacol. 2024;982:176951. [67] YUAN J, OFENGEIM D. A guide to cell death pathways. Nat Rev Mol Cell Biol. 2024;25(5):379-395. [68] ZHAO CQ, JIANG LS, DAI LY. Programmed cell death in intervertebral disc degeneration. Apoptosis. 2006;11(12):2079-2088. [69] 裴彩霞,贾楠,刘俊伶,等.细胞死亡的多种方式及其机制研究进展[J/OL].解放军医学杂志,1-17[2025-08-04].https://link.cnki.net/urlid/11.1056.R.20250327.1520.006. [70] VOSS AK, STRASSER A. The essentials of developmental apoptosis. F1000Res. 2020;9:F1000 Faculty Rev-148. doi: 10.12688/f1000research.21571.1. eCollection 2020. [71] WU Y, CHEN M, JIANG J. Mitochondrial dysfunction in neurodegenerative diseases and drug targets via apoptotic signaling. Mitochondrion. 2019;49:35-45. [72] REDZA-DUTORDOIR M, AVERILL-BATES DA. Activation of apoptosis signalling pathways by reactive oxygen species. Biochim Biophys Acta. 2016;1863(12):2977-2992. [73] LI K, LI Y, MI J, et al. Resveratrol protects against sodium nitroprusside induced nucleus pulposus cell apoptosis by scavenging ROS. Int J Mol Med. 2018;41(5):2485-2492. [74] ZHANG Z, WANG C, LIN J, et al. Therapeutic Potential of Naringin for Intervertebral Disc Degeneration: Involvement of Autophagy Against Oxidative Stress-Induced Apoptosis in Nucleus Pulposus Cells. Am J Chin Med. 2018: 1-20.doi:10.1142/s0192415x18500805 [75] LUO R, LIAO Z, SONG Y, et al. Berberine ameliorates oxidative stress-induced apoptosis by modulating ER stress and autophagy in human nucleus pulposus cells. Life Sci. 2019;228: 85-97. [76] GAO S, WANG C, QI L, et al. Bushen Huoxue Formula Inhibits IL-1β-Induced Apoptosis and Extracellular Matrix Degradation in the Nucleus Pulposus Cells and Improves Intervertebral Disc Degeneration in Rats. J Inflamm Res. 2024; 17:121-136. [77] LIU W, ZHAO X, WU X. Duhuo Jisheng Decoction suppresses apoptosis and mitochondrial dysfunction in human nucleus pulposus cells by miR-494/SIRT3/mitophagy signal axis. J Orthop Surg Res. 2023;18(1):177. [78] WAN C, LIU S, ZHAO L, et al. Curcumin protects rat endplate chondrocytes against IL-1β-induced apoptosis via Bcl-2/Bax regulation. J Mol Histol. 2025;56(2):111. [79] LUO J, YANG Y, WANG X, et al. Role of Pyroptosis in Intervertebral Disc Degeneration and Its Therapeutic Implications. Biomolecules. 2022; 12(12):1804. [80] GONG Y, QIU J, JIANG T, et al. Maltol ameliorates intervertebral disc degeneration through inhibiting PI3K/AKT/NF-κB pathway and regulating NLRP3 inflammasome-mediated pyroptosis. Inflammopharmacology. 2023;31(1):369-384. [81] WANG D, ZHANG L, HE D, et al. Systemic pharmacology reveal the mechanism by which the Qiangjin Zhuanggu Qufeng mixture inhibits LPS-induced pyroptosis of rat nucleus pulposus cells. Phytomedicine. 2023;119:154998. [82] GE Y, XIE Y, CHAI J, et al. Augmented Cornus officinalis Levels in Liuwei Dihuang Decoction Inhibits Nucleus Pulposus Cell Pyroptosis to Enhance Therapeutic Efficacy Against Intervertebral Disc Degeneration. J Inflamm Res. 2024;17:4453-4465. [83] GUO D, CHENG K, SONG C, et al. Mechanisms of inhibition of nucleus pulposus cells pyroptosis through SDF1/CXCR4-NFkB-NLRP3 axis in the treatment of intervertebral disc degeneration by Duhuo Jisheng Decoction. Int Immunopharmacol. 2023;124(Pt A):110844. [84] YAO S, LI Y, RUAN H, et al. Gubi Decoction Ameliorates Porous Cartilage Endplate in an Intervertebral Disc Degeneration Model Mouse Through Inhibition of NF-κB Activity and Pyroptosis. J Inflamm Res. 2025;18:5293-5309. [85] DIXON SJ, LEMBERG KM, LAMPRECHT MR, et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell. 2012;149(5): 1060-1072. [86] JIANG X, STOCKWELL BR, CONRAD M. Ferroptosis: mechanisms, biology and role in disease. Nat Rev Mol Cell Biol. 2021;22(4):266-282. [87] FANG Y, CHEN X, TAN Q, et al. Inhibiting Ferroptosis through Disrupting the NCOA4-FTH1 Interaction: A New Mechanism of Action. ACS Cent Sci. 2021;7(6):980-989. [88] CHEN J, YANG X, FENG Y, et al. Targeting Ferroptosis Holds Potential for Intervertebral Disc Degeneration Therapy. Cells. 2022;11(21):3508. [89] RU Q, LI Y, XIE W, et al. Fighting age-related orthopedic diseases: focusing on ferroptosis. Bone Res. 2023;11(1):12. [90] 武豪,吴世祥,尹恒,等.铁死亡与椎间盘退变的研究进展[J].中国医学前沿杂志(电子版), 2023,15(11):103-109. [91] 赵继荣,马俊飞,薛旭,等.铁死亡与椎间盘退变关系的研究进展[J].中国脊柱脊髓杂志, 2024,34(5):548-554. [92] ZHANG P, RONG K, GUO J, et al. Cynarin alleviates intervertebral disc degeneration via protecting nucleus pulposus cells from ferroptosis. Biomed Pharmacother. 2023;165:115252. [93] ZHU J, SUN R, YAN C, et al. Hesperidin mitigates oxidative stress-induced ferroptosis in nucleus pulposus cells via Nrf2/NF-κB axis to protect intervertebral disc from degeneration. Cell Cycle. 2023;22(10):1196-1214. [94] CUI P, SHENG Y, WU C, et al. Puerarin modulates proliferation, inflammation and ECM metabolism in human nucleus pulposus mesenchymal stem cells via the lncRNA LINC01535. Heliyon. 2024; 10(12):e33083. [95] DEBNATH J, GAMMOH N, RYAN KM. Autophagy and autophagy-related pathways in cancer. Nat Rev Mol Cell Biol. 2023;24(8):560-575. [96] YURUBE T, ITO M, KAKIUCHI Y, et al. Autophagy and mTOR signaling during intervertebral disc aging and degeneration. JOR Spine. 2020;3(1):e1082. [97] WANG D, HE X, WANG D, et al. Quercetin Suppresses Apoptosis and Attenuates Intervertebral Disc Degeneration via the SIRT1-Autophagy Pathway. Front Cell Dev Biol. 2020;8: 613006. [98] XIE C, SHI Y, CHEN Z, et al. Apigenin Alleviates Intervertebral Disc Degeneration via Restoring Autophagy Flux in Nucleus Pulposus Cells. Front Cell Dev Biol. 2021;9:787278. [99] ZHANG B, HE Z, GUO J, et al. Sesamin-mediated high expression of BECN2 ameliorates cartilage endplate degeneration by reducing autophagy and inflammation. Aging (Albany NY). 2024;16(2): 1145-1160. [100] WANG J, JING X, LIU X, et al. Naringin safeguards vertebral endplate chondrocytes from apoptosis and NLRP3 inflammasome activation through SIRT3-mediated mitophagy. Int Immunopharmacol. 2024;140: 112801. [101] WANG K, YIN J, CHEN J, et al. Inhibition of inflammation by berberine: Molecular mechanism and network pharmacology analysis. Phytomedicine. 2024;128:155258. [102] 黄巧波,刘明平.基于网络药理学与分子对接技术探讨大黄肝毒性的作用机制[J].现代药物与临床,2023,38(12):2965-2970. [103] POIVRE M, DUEZ P. Biological activity and toxicity of the Chinese herb Magnolia officinalis Rehder & E. Wilson (Houpo) and its constituents . J Zhejiang Univ Sci B. 2017;18(3):194-214. [104] 魏巍,张明发,沈雅琴.厚朴酚及和厚朴酚的胃肠道药理作用及其机制的研究进展[J].药物评价研究,2022,45(9):1914-1921. |
| [1] | Zhu Xiaolong, Zhang Wei, Yang Yang. Visualization analysis of research hotspots and cutting-edge information in the field of intervertebral disc regeneration and repair [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2391-2402. |
| [2] | Cai Ziming, Yu Qinghe, Ma Pengfei, Zhang Xin, Zhou Longqian, Zhang Chongyang, Lin Wenping. Heme oxygenase-1 alleviates lipopolysaccharide-induced inflammatory response in nucleus pulposus mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1624-1631. |
| [3] | Wang Peng, Li Zhijun, Zhang Shaojie, Wu Yimin. Intervertebral disc rehydration after posterior lumbar dynamic internal fixation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(3): 711-720. |
| [4] | Li Huaying, Li Hao, Peng Wuxun, Dong Wentao. Mechanism of cuproptosis in the diagnosis and treatment of orthopedic-related diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6275-6281. |
| [5] | Wang Zhengye, Liu Wanlin, Zhao Zhenqun. Mechanisms of miRNAs involved in cartilage development: new strategies and targets [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6289-6296. |
| [6] | Kan Weiyi, Wang Linrong, Cheng Leping. The interaction and balance between cellular senescence and tissue repair [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6297-6305. |
| [7] | Wang Wei, Chen Jun, Jia Shaohui, Xue Xinxuan, Dong Kunwei. Mechanisms by which exercise regulates gut microbiota in the prevention and treatment of non-alcoholic fatty liver disease [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6328-6336. |
| [8] | Li Zijing, Chen Xuwu, Ouyang Xinye, Wang Maoyuan. Mitophagy impairment mediated muscular atrophy: insights from the Drosophila model [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 5897-5905. |
| [9] | Jiang Chao, Che Yanjun. Biological mechanisms and future research trends of cartilaginous endplate degeneration [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 5915-5924. |
| [10] | Zhou Tianle, Wang Wei, Zhang Zhiwen, Liu Ximing. Application and progress of transcriptomics and proteomics techniques in the study of intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 5925-5933. |
| [11] | Xie Ziying, Li Songbo, Li Jianwen, Yin Yuchao, Zheng Baichuan, Hu Chengshang. Animal experimental study on the treatment of lumbar intervertebral disc degeneration with Chinese herbal compound: species selection, modeling method and drug administration [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 5934-5942. |
| [12] | Ji Kaizhong, Kong Yihao, Zhi Yiqing, Jin Yingying, Chen Jianquan. Effects and mechanisms of palmitoyl acyltransferase ZDHHC5 in tissue homeostasis and diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(17): 4430-4445. |
| [13] | Mao Sujie, Gao Jie, Pan Zhuangli. Immune cells synergistically regulate inflammatory response, muscle regeneration and metabolic homeostasis in training-induced stress responses [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(10): 2671-2680. |
| [14] | Qian Kun, Li Ziqing, Sun Shui . Endoplasmic reticulum stress in the occurrence and development of common degenerative bone diseases [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(6): 1285-1295. |
| [15] | Xiang Pan, Che Yanjun, Luo Zongping. Compressive stress induces degeneration of cartilaginous endplate cells through the SOST/Wnt/beta-catenin pathway [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(5): 951-957. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||