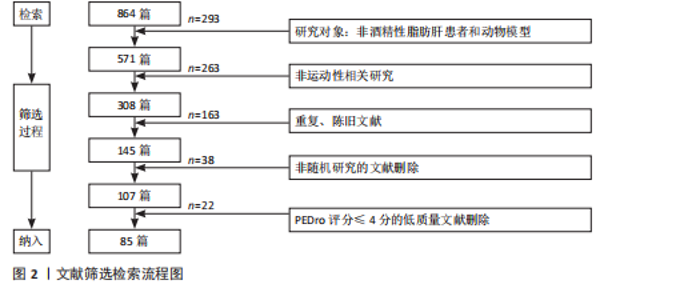
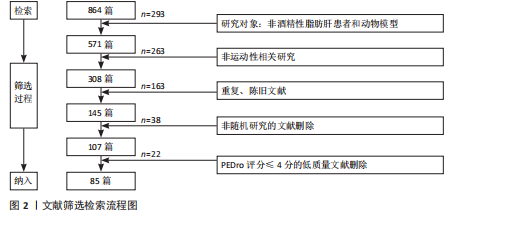
Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (24): 6328-6336.doi: 10.12307/2026.229
Previous Articles Next Articles
Mechanisms by which exercise regulates gut microbiota in the prevention and treatment of non-alcoholic fatty liver disease
Wang Wei1, Chen Jun1, Jia Shaohui2, Xue Xinxuan3, Dong Kunwei1
- 1School of Art, 2School of Sports Medicine, 3School of Sports Training, Wuhan Sports University, Wuhan 430079, Hubei Province, China
-
Received:2025-06-17Revised:2025-10-15Online:2026-08-28Published:2026-02-04 -
Contact:Dong Kunwei, Associate professor, Master’s supervisor, School of Art, Wuhan Sports University, Wuhan 430079, Hubei Province, China -
About author:Wang Wei, MS, Lecturer, School of Art, Wuhan Sports University, Wuhan 430079, Hubei Province, China -
Supported by:Youth Foundation for Scientific Research Project of Education Department of Hubei Province, No. B2019193 (to JSH)
CLC Number:
Cite this article
Wang Wei, Chen Jun, Jia Shaohui, Xue Xinxuan, Dong Kunwei. Mechanisms by which exercise regulates gut microbiota in the prevention and treatment of non-alcoholic fatty liver disease[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6328-6336.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
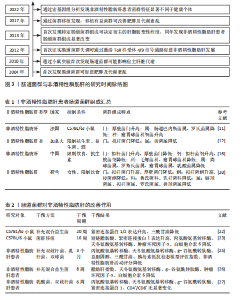
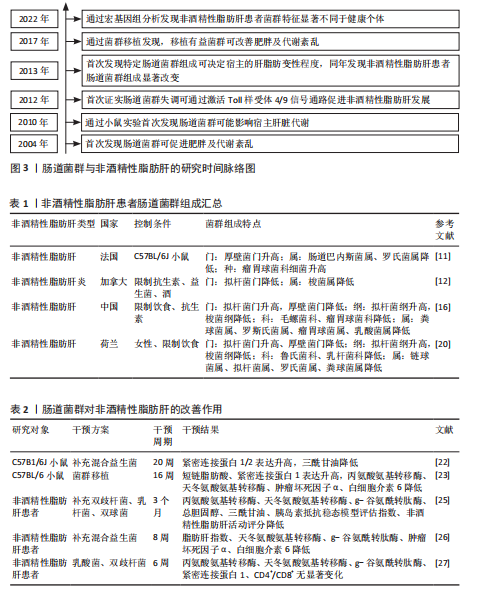
通过调控宿主的能量摄取和脂肪储存促进肥胖及相关代谢紊乱,为非酒精性脂肪肝的微生物学研究奠定了基础。2010年,RABOT等[9]在无菌小鼠研究中首次发现肠道菌群可能影响宿主的肝脏代谢,揭示了肠道微生物与代谢性疾病之间的潜在联系。2012年,HENAO-MEJIA等[10]首次证实了肠道菌群失调可通过激活Toll样受体4/9信号通路,诱导炎症反应,促进非酒精性脂肪肝的发生发展,进一步揭示了微生物-肠-肝轴的概念。2013年,LE ROY等[11]首次通过粪菌移植实验证明,特定的肠道菌群组成可决定宿主的肝脂肪变性程度,提示肠道菌群稳态在非酒精性脂肪肝防治中的重要作用。同年,MOUZAKI等[12]研究发现非酒精性脂肪肝患者的肠道菌群组成显著改变,表明特定菌群失衡可能促进非酒精性脂肪肝的发生和进展。2017年,KOOTTE等[13]的粪菌移植实验表明,通过移植健康供体的肠道菌群可改善肥胖和代谢紊乱,进一步证实了肠道菌群在代谢疾病中的关键作用。2022年,DEMIR等[14]通过宏基因组分析发现非酒精性脂肪肝患者粪便中的菌群特征显著不同于健康个体,特别是拟杆菌门和厚壁菌门的比例变化与肝纤维化程度密切相关。肠道菌群与非酒精性脂肪肝的研究历程时间脉络图,见图3。 2.2 肠道菌群组成变化与非酒精性脂肪肝的病理联系 肠道菌群是一个复杂且高度多样化的生态系统,主要由拟杆菌门、厚壁菌门、变形菌门和放线菌门组成。其中,拟杆菌门与厚壁菌门是调控宿主代谢的重要微生物类群,它们比值的变化通常被视为衡量肠道微生态稳态的关键指标[15]。尽管目前对非酒精性脂肪肝患者肠道菌群的具体组成尚未形成共识,但大量研究已证实非酒精性脂肪肝与肠道菌群紊乱密切相关。 研究表明,与健康个体相比,非酒精性脂肪肝患者的生脂多糖革兰阴性菌丰度较高,拟杆菌门的相对丰度升高约20%,而厚壁菌门的丰度下降约24%[16],提示非酒精性脂肪肝患者肠道菌群组成明显异常。肠道菌群紊乱可导致肠道通透性增加,削弱肠上皮屏障功能,使细菌及其代谢产物更易进入血液循环。例如,某些革兰阴性菌(如大肠杆菌、克雷伯氏菌和沙门菌等)的丰度增加将导致其成分脂多糖发生肠道移位,并进入循环系统,与肝细胞膜表面的Toll样受体4结合从而激活核因子κB信号通路,促进炎性因子的释放,诱发肝脏炎症反应。进一步的分析发现,非酒精性脂肪肝患者的变形菌门、埃希菌属、普雷沃菌属和链球菌属丰度升高,而粪球菌属、粪杆菌属和瘤胃球菌属丰度下降[17]。其中,变形菌门(特别是大肠杆菌和肠杆菌科)丰度的增加可能导致肠道通透性升高,进而促进脂多糖进入门静脉循环,激活炎症小体并加剧肝损伤[16];瘤胃球菌属和粪杆菌属丰度的降低可导致短链脂肪酸生成减少,而短链脂肪酸具有维持肠道稳态和发挥抗炎的作用,短链脂肪酸减少可能加剧由非酒精性脂肪肝相关病原体所诱导的肠道炎症,从而进一步促进非酒精性脂肪肝的发生发展[18]。此外,非酒精性脂肪肝患者的肠道中还富含某些具有致病潜能的细菌,如产乙醇菌(主要包括肠杆菌科)等,这些细菌可通过过量合成内源性乙醇破坏肠黏膜屏障,导致门静脉脂多糖水平升高,进而激活核因子κB信号通路,最终加重肝脏炎症[19]。有趣的是,有研究发现,与西方国家相比,中国非酒精性脂肪肝患者肠道中拟杆菌属的丰度相对更低[20],这种差异可能与不同地区的饮食结构有关,西方国家流行的高脂肪和高蛋白饮食已被证明会显著影响拟杆菌属的丰度[21]。 综上所述,非酒精性脂肪肝患者的肠道菌群组成表现为促炎菌群(如变形菌门、大肠杆菌和链球菌属)及致病菌(如肠杆菌科)的丰度增加,而具有抗炎和稳态调节功能菌群(如瘤胃球菌属和粪杆菌属)的丰度降低(表1)。这种菌群组成的不良变化可能通过增加肠道通透性促进代谢产物进入肝脏以及激活炎症通路等机制,从而推动非酒精性脂肪肝的病理进程。 2.3 肠道菌群在非酒精性脂肪肝中的改善作用 调节肠道菌群已被视为治疗非酒精性脂肪肝的一种有效策略。BRISKEY等[22]研究表明,与高脂饮食喂养的小鼠相比,接受额外益生菌补充的小鼠能够提高紧密连接蛋白的表达、维持肠道屏障功能、降低肝脏三酰甘油水平。动物实验进一步证实,健康小鼠在接受8周的粪菌移植后可调节高脂饮食引起的肠道菌群失衡,进而改善代谢相关指标,显著降低血清转氨酶水平、肝脂肪变性及小叶炎症程度[23]。此外,高脂饮食小鼠盲肠中的丁酸浓度升高,而丁酸是结肠上皮细胞的重要能量来源,这可能有助于维持肠道健康、降低肠道通透性,从而预防与非酒精性脂肪肝密切相关的代谢性内毒素血症[24]。 一项为期3个月的临床研究显示,益生菌干预组非酒精性脂肪肝患者的前谷丙转氨酶、天冬氨酸氨基转移酶、谷氨酰胺转移酶、总胆固醇、三酰甘油、胰岛素抵抗指数和粪便菌群状况显著优于安慰剂组,提示益生菌可对非酒精性脂肪肝患者的肝功能、糖脂代谢及肝脏脂肪蓄积具有一定改善作用[25]。KOBYLIAK等[26]研究发现,多益生菌联合补充(双歧杆菌属、乳杆菌属、乳球菌属和丙酸杆菌属等14种益生菌属的浓缩生物量)虽然对非酒精性脂肪肝患者的肝脏硬度无显著影响,但可明显降低非酒精性脂肪肝患者的脂肪肝指数、丙氨酸转氨酶和γ-谷氨酰转移酶水平,导致慢性全身炎症指标(肿瘤坏死因子α和白细胞介素6)水平显著下降,表明补充益生菌不仅能改善非酒精性脂肪肝患者肝酶和脂质谱,还具有一定的抗炎作用。然而,MOHAMAD NOR等[27]评估了益生菌对非酒精性脂肪肝患者免疫活性的影响,发现由6种不同乳杆菌和双歧杆菌组成的益生菌组合并未显著影响CD4+或CD8+ T细胞水平;此外,该研究还发现益生菌并未改善非酒精性脂肪肝患者的脂肪变性、纤维化、炎症评分、丙氨酸转氨酶、总胆固醇、三酰甘油或空腹血糖水平,仅观察到益生菌能够改善患者肠道环境中黏膜免疫活性并维持肠道屏障完整性。出现上述研究结果不一致的原因,可能与益生菌种类和剂量、补充持续时间以及研究对象存在较大差异有关。 总之,大多数研究表明,通过补充益生菌或进行肠道菌群移植来调控肠道菌群可有效降低非酒精性脂肪肝患者不同形式的转氨酶及慢性炎症水平(表2),提示肠道菌群可能是治疗非酒精性脂肪肝的重要靶点。 2.4 运动对肠道菌群的影响 众所周知,运动可以有效防治肥胖、糖尿病和非酒精性脂肪肝等多种代谢性疾病,而肠道菌群可能在其中发挥了关键作用。有研究显示,6?个月的有氧联合抗阻运动可有效抑制2型糖尿病患者肠道内真菌的异常增殖,减少肠道通透性增加所引发的内毒素渗漏,从而降低全身性低度炎症[28]。WANG等[29]研究发现,12周的中等强度跑台运动方案可有效逆转高脂饮食诱导的肥胖小鼠肠道菌群紊乱、增加杯状细胞数量,进而改善肠道屏障功能。MOTIANI等[30]研究显示,4?周的高强度间歇训练可显著降低胰岛素抵抗患者的厚壁菌门/拟杆菌门比值,同时降低血清肿瘤坏死因子α、C-反应蛋白和脂多糖结合蛋白水平,从而改善全身胰岛素抵抗。此外,4周的抗阻运动不仅能够提高多发性硬化症小鼠肠道菌群的多样性,还能降低厚壁菌门/拟杆菌门比值和肠黏膜通透性,调节小肠淋巴组织中的免疫炎症反应[31]。由此可见,不同运动方式均能调节肠道菌群、改善肠道屏障功能、降低炎症反应。 但需要指出的是,运动强度差异可能会对肠道菌群组成及功能产生不同的影响。例如,TORQUATI等[32]研究比较了8?周中等强度持续"
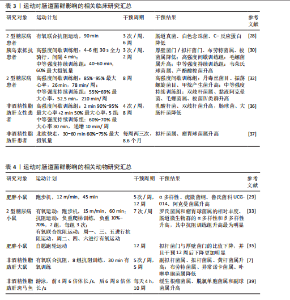
训练和高强度间歇训练对2型糖尿病患者患者肠道菌群组成和功能的影响,发现中等强度持续训练组双歧杆菌属、嗜黏蛋白阿克曼氏菌属及产丁酸菌(如毛螺菌属、肠球菌属和梭菌属)的相对丰度较高,而高强度间歇训练组瘤胃球菌属、丹毒丝菌目和甲烷产生菌的相对丰度较高;KEGG通路富集分析结果显示,丙酮酸、氨基酸及聚酮类代谢通路在中等强度持续训练组更为富集,提示不同运动强度可能通过靶向调节特定的菌群及代谢通路优化肠道微环境和宿主代谢健康。 不同运动方式、频率和持续时间亦可对肠道菌群产生不同影响。例如,韦薇等[33]研究了8周有氧运动、抗阻运动以及有氧联合抗阻运动对2型糖尿病小鼠肠道菌群的影响,结果显示3种运动方式均可改善血糖水平、调节肠道菌群的组成与结构、显著提高血清短链脂肪酸水平,其中抗阻运动组在产短链脂肪酸菌属(如罗氏菌属和瘤胃球菌属)相对丰度及肠道微生物群的α多样性和β多样性方面提升最为显著,提示抗阻运动在改善肠道菌群结构和提升短链脂肪酸含量方面可能更具优势。MCFADZEAN等[34]探讨了不同运动频率对肠道菌群的影响,结果显示,与每周运动一两次个体相比,每周运动3-5次个体在肠道菌群α多样性及厚壁菌门(尤其是普拉梭菌属)丰度方面具有更显著提升,普拉梭菌是丁酸的主要产菌,具有抗炎和增强肠道屏障功能的作用,提示适度提高运动频率有助于优化肠道微生态。此外,随着运动干预时间的延长,肠道菌群的组成可获得更为显著的优化效果,例如,EVANS等[35]在高脂饮食诱导的肥胖小鼠中观察到,相较于6周自愿跑轮运动,持续12周的自愿跑轮运动更显著地降低了拟杆菌门与厚壁菌门的比值,这可能与长期运动诱导的促炎因子减少有关,表明运动对肠道菌群结构的调节具有时间依赖性与阶段性特征。 然而,尽管近年来肠道菌群在运动防治代谢性疾病中的作用研究取得了重要进展,但当前有关运动对非酒精性脂肪肝患者肠道菌群的影响研究仍较少。王鹏等[36]以肥胖非酒精性脂肪肝女大学生为研究对象,进行8周运动联合饮食干预后发现,该干预策略能够显著减少体脂肪和内脏脂肪含量、改善脂质代谢、降低大肠杆菌和肠球菌丰度、增加有益菌乳酸杆菌及双歧杆菌丰度。类似地,CHENG等[37]针对合并糖尿病前期的非酒精性脂肪肝患者开展为期8.6个月的有氧运动干预,结果显示患者肠道菌群α多样性显著增加,拟杆菌属、瘤胃球菌属和毛螺菌科等菌群的相对丰度上升并与肝脏脂肪含量呈负相关,提示肠道菌群可能与运动改善非酒精性脂肪肝有关。动物实验表明,有氧联合抗阻运动能够抑制高脂饮食诱导的非酒精性脂肪肝大鼠体质量增长、改善代谢综合征及肝脂肪变性,同时显著提高副拟杆菌属、拟杆菌属和黄杆菌属等与健康代谢特征相关的有益菌群丰度[38]。另外,GU等[39]发现高脂饮食诱导非酒精性脂肪肝斑马鱼进行为期10周的游泳运动后,游泳运动可显著提高缓生根瘤菌属、脱氯单胞菌属和副球菌属等与代谢相关的有益菌丰度,有效降低体质量指数、肝脏相对质量、肝空泡密度、肝切片中的脂滴、三酰甘油和游离脂肪酸。进一步分析发现,肠道菌群变化影响了葡萄糖和脂肪酸代谢的分子过程,包括不饱和脂肪酸生物合成、脂肪酸生物合成、糖酵解/糖异生和胰岛素等信号通路,表明运动可能通过“肠-肝”轴来调控葡萄糖和脂肪酸代谢,从而改善非酒精性脂肪肝的病理生理状况。 综上所述,运动可调节肠道菌群组成、增加有益菌的丰度、促进关键代谢通路的激活,进而改善宿主代谢健康(表3,4)。然而,目前关于运动对非酒精性脂肪肝患者肠道菌群影响的研究仍相对有限,尤其是不同运动方式、强度及持续时间对肠道菌群及宿主代谢的影响及其作用机制尚不明确;此外,运动对非酒精性脂肪肝不同病程阶段患者肠道菌群的调节作用是否存在阶段性差异,也仍需进一步探讨。 2.5 肠道菌群介导运动防治非酒精性脂肪肝的可能机制 由于肠道和肝脏之间的解剖和功能联系,肝脏可能会持续受到肠道菌群成分及其代谢产物的影响。如前所述,肠道菌群成分及其代谢产物(如胆汁酸、短链脂肪酸和脂多糖等)在非酒精性脂肪肝的发生发展中扮演了重要角色,因此,该文在此从肠道菌群成分及其代谢产物的视角出发,探讨运动在非酒精性脂肪肝防治中的潜在机制。 2.5.1 胆汁酸 胆汁酸可分为初级胆汁酸和次级胆汁酸,初级胆汁酸(如鹅脱氧胆酸)由肝脏通过胆固醇合成,并在C24羧基上通过酰胺键与牛磺酸或甘氨酸结合,形成结合胆汁酸,进入肠道后,结合胆汁酸在含胆碱水解酶的细菌作用下被代谢转化为次级胆汁酸(如脱氧胆酸和石胆酸)[40]。临床研究表明,非酒精性脂肪肝患者的血清胆汁酸谱特征表现为胆汁酸合成明显增加、脱氧胆酸水平升高,而鹅脱氧胆酸含量降低,提示非酒精性脂肪肝患者胆汁酸代谢异常[41],其中,脱氧胆酸衍生的多种胆汁酸(如糖脱氧胆酸和7-酮脱氧胆酸和去氢胆酸)随着非酒精性脂肪肝活动度和纤维化程度的增加而升高[42]。过量的脱氧胆酸可损伤肠道屏障,诱导炎症因子释放,加剧肝损伤[43]。因此,胆汁酸代谢异常可能是非酒精性脂肪肝发展的重要病理因素之一。"
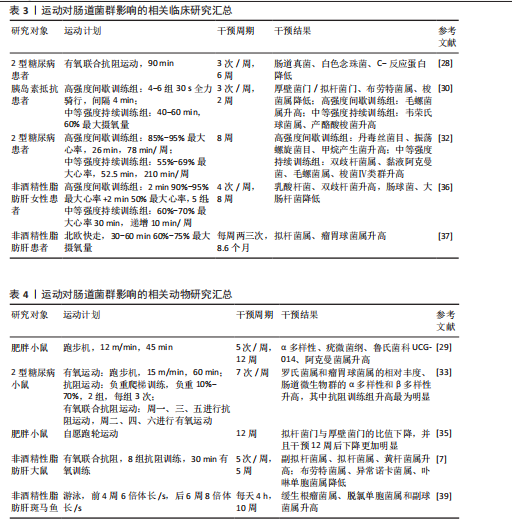
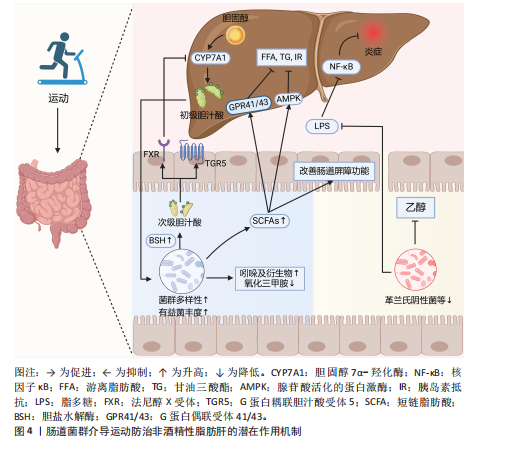
肠道菌群紊乱可导致胆汁酸代谢异常。在非酒精性脂肪肝炎患者的粪便中可观察到初级胆汁酸与次级胆汁酸的比例明显升高,这可能与其肠道中含胆碱水解酶的细菌(如乳酸杆菌属,双歧杆菌属和拟杆菌属)较少有关[44]。值得注意的是,一项针对儿童肥胖的干预研究发现,补充低聚果糖-菊粉可显著提高双歧杆菌属丰度、减少普通拟杆菌数量,从而降低粪便中初级胆汁酸浓度,减少胆汁酸积聚所致的肝损伤[45]。提示靶向调控肠道菌群或可改善胆汁酸代谢紊乱,从而防治非酒精性脂肪肝。 运动可有效逆转非酒精性脂肪肝及肥胖相关疾病引起的肠道菌群失调,提高肠道菌群的多样性,改善胆汁酸代谢紊乱,并促进胆汁酸和胆固醇的排泄[46]。SHI等[47]研究发现,8周有氧运动可显著升高非酒精性脂肪肝患者总胆汁酸和熊去氧胆酸水平、改善肝功能指标(如丙氨酸氨基转移酶和天冬氨酸氨基转移酶等)。此外,运动对非酒精性脂肪肝的保护作用可能与法尼醇X受体和G蛋白偶联胆汁酸受体5信号通路密切相关,在细胞内这些受体可与不同类型的胆汁酸结合,调节肝脏的葡萄糖、脂质及能量代谢等生理过程[48]。研究发现,6周有氧联合抗阻运动可提高非酒精性脂肪肝模型小鼠含胆碱水解酶拟杆菌属的丰度,促进初级胆汁酸水解为次级胆汁酸,从而上调法尼醇X受体表达[38]。一方面,法尼醇X受体的激活可减少胆固醇7α-羟化酶的表达,抑制胆汁酸的过度合成、维持胆汁酸稳态[49];另一方面,法尼醇X受体可通过下调固醇调节元件结合蛋白1c和脂肪酸合成酶等脂质合成相关基因表达,减少肝脏脂质蓄积、改善肝脏脂肪变性[50]。此外,法尼醇X受体还可促进肝糖原合成,并上调胰高血糖素样肽1的表达,进而增强胰岛素敏感性,改善非酒精性脂肪肝相关的胰岛素抵抗[51]。 除法尼醇X受体外,G蛋白偶联胆汁酸受体5也在胆汁酸介导的代谢调控中发挥重要作用。G蛋白偶联胆汁酸受体5可通过胰高血糖素样肽1通路影响肠道糖代谢,被认为是治疗肥胖、糖尿病和非酒精性脂肪肝等代谢性疾病的重要靶点。其中,石胆酸对G蛋白偶联胆汁酸受体5的亲和力最高,是一种有效的G蛋白偶联胆汁酸受体5激动剂。研究表明,急性有氧运动和抗阻运动均可显著提升石胆酸水平[52]。 梁家琪等[53]研究认为,运动介导肠道菌群重塑可维持胆汁酸稳态,提高石胆酸水平来激活人体G蛋白偶联胆汁酸受体5。而G蛋白偶联胆汁酸受体5的激活不仅可通过调节肝脏炎症及葡萄糖代谢,提高胰岛素敏感性[54],还可抑制巨噬细胞中核因子κB的表达,减少促炎细胞因子的释放,从而减轻肝脏炎症反应[48]。 综上所述,运动可能通过调节肠道菌群-胆汁酸轴改善胆汁酸代谢,从而介导法尼醇X受体/G蛋白偶联胆汁酸受体5信号通路来防治非酒精性脂肪肝。然而未来仍需进一步研究,以深入探讨运动调节肠道菌群-胆汁酸轴与非酒精性脂肪肝改善之间的因果关系及其潜在分子机制。 2.5.2 短链脂肪酸 短链脂肪酸是肠道菌群代谢可溶性膳食纤维和不可消化碳水化合物产生的重要产物,其中乙酸、丙酸和丁酸的含量最高,占总短链脂肪酸的90%以上。研究发现,非酒精性脂肪肝患者的产短链脂肪酸菌群(如粪杆菌属、瘤胃球菌科和优杆菌属等)丰度明显降低,同时粪便中短链脂肪酸水平也显著下降,提示短链脂肪酸可能与非酒精性脂肪肝的发生发展密切相关[55]。短链脂肪酸不仅是肠道上皮细胞的重要能量来源,还可通过肠-肝轴参与宿主脂质代谢、炎症调控及胰岛素敏感性调节,从而影响非酒精性脂肪肝的进程[56]。已有研究表明,持续4周及以上的不同运动方式干预均可有效改善代谢性疾病患者的肠道菌群组成、促进产短链脂肪酸菌群的增殖、提高短链脂肪酸的合成与吸收[57]。因此,运动可能通过增强宿主对短链脂肪酸的利用,从而发挥抗炎、改善胰岛素敏感性和调控肝脏脂质代谢的作用,最终防治非酒精性脂肪肝。具体而言,短链脂肪酸可能通过以下机制介导运动对非酒精性脂肪肝的防治作用。 运动可能通过短链脂肪酸减少肝脏脂肪积累来防治非酒精性脂肪肝:短链脂肪酸(特别是乙酸和丙酸)可通过门静脉循环进入肝脏,抑制肝脏内源性葡萄糖合成,减少糖异生,从而降低脂肪生成的底物供应,抑制肝脂肪沉 积[58]。此外,短链脂肪酸可作为G蛋白偶联受体41/43的内源性配体,调节脂肪酸氧化和脂质代谢平衡。例如,G蛋白偶联受体41/43的激活可通过钙调蛋白依赖性蛋白激酶Ⅱ/组蛋白去乙酰化酶1-环磷酸腺苷反应元件结合蛋白通路促进脂肪细胞的脂解作用,减少游离脂肪酸向肝脏的转运,从而降低肝脂肪积累[59]。同时,8周中等强度的有氧运动可通过改善肠道菌群组成来增加丁酸的生成[60]。而丁酸不仅能够增强肠道屏障功能,减少肠源性脂多糖入血,还能通过降低组蛋白去乙酰化酶活性,减少肝细胞脂肪沉积,从而延缓非酒精性脂肪肝进展[61]。 运动可能通过短链脂肪酸缓解肝脏炎症反应来防治非酒精性脂肪肝:非酒精性脂肪肝的病理进展往往伴随慢性低度炎症,而短链脂肪酸在炎症调控方面发挥着关键作用。乙酸可作为免疫调节因子,可通过抑制组蛋白去乙酰化酶活性抑制核因子κB信号通路的激活,减少促炎细胞因子(如肿瘤坏死因子α、白细胞介素6和白细胞介素1β)的释放[62]。此外,乙酸和丙酸可通过G蛋白偶联受体41/43调节免疫细胞功能,降低肝脏Kupffer细胞的活性,减少巨噬细胞极化为促炎M1型,从而减轻肝脏的炎症反应[63]。同时,短链脂肪酸还能提高跨上皮电阻来抑制脂多糖的产生,增加带状闭锁蛋白1和紧密连接蛋白的表达,从而改善肠屏障功能,这有助于阻碍毒性物质(如脂多糖和乙醇)透过肠壁进入血液循环,从源头上降低肝脏炎症的诱导因素,进而减轻非酒精性脂肪肝相关的炎症损伤[64]。 运动还可能通过短链脂肪酸降低胰岛素抵抗来防治非酒精性脂肪肝:在非酒精性脂肪肝的发生发展过程中,胰岛素抵抗既是病因也是疾病进展的结果。研究发现,运动可通过调节肠道菌群组成、增加短链脂肪酸的生成,进而促进胰岛素信号通路的激活[57]:一方面,短链脂肪酸可通过刺激肠道信号转导控制能量摄入,并以腺苷酸活化蛋白激酶依赖途径调节肝脏胰岛素敏感性和葡萄糖代谢,从而提高葡萄糖利用,降低胰岛素抵抗[65];另一方面,短链脂肪酸能够激活G蛋白偶联受体41/43,促进神经内分泌L细胞分泌多肽YY和胰高血糖素样肽1,抑制结肠上皮趋化因子和促炎因子释放,改善胰岛素抵抗引起的脂肪沉积[66]。此外,丁酸与G蛋白偶联受体结合后能够抑制组蛋白去乙酰化酶的活性,促进胰岛β细胞的增殖和功能恢复,进一步改善胰岛素敏感性[67]。 综上所述,运动可能通过调节肠道菌群组成促进短链脂肪酸的生成与吸收,从而在非酒精性脂肪肝的防治中发挥重要作用。短链脂肪酸不仅可减少肝脂肪积累、缓解肝脏炎症,还能改善胰岛素敏感性、减缓非酒精性脂肪肝进展。然而,上述关于运动通过短链脂肪酸介导非酒精性脂肪肝防治的机制尚缺乏直接证据,主要基于推测,因此,未来需进一步探讨运动对短链脂肪酸生成及代谢的影响,阐明运动在非酒精性脂肪肝防治中的具体作用机制,以提供更有力的理论支持。 2.5.3 脂多糖 脂多糖又称内毒素,是革兰阴性菌细胞壁的主要组成成分,可通过门静脉系统进入肝脏,并与肝细胞膜上广泛表达的Toll样受体4结合,从而诱导其活化。Toll样受体4的激活进一步触发库普弗细胞内核因子κB信号通路,促进炎性细胞因子的释放,如白细胞介素6、白细胞介素1β及肿瘤坏死因子α,从而引发肝脏炎症反应[63]。脂多糖还可通过Toll样受体4依赖性途径介导肌球蛋白轻链激酶的上调,并促进白细胞介素1受体相关激酶4的活化,导致肠道屏障功能受损、肠道通透性增加,进而促进脂多糖的易位和系统性炎症的发生[68]。据报道,在非酒精性脂肪肝患者和实验动物中均可观察到血清脂多糖水平升高,而降低血浆脂多糖水平可改善肝脂肪变性,提示脂多糖引发的慢性低度炎症可能是非酒精性脂肪肝进展的重要因素[69]。 运动在非酒精性脂肪肝防治中的作用可能部分通过调控肠道菌群以减少脂多糖生成与肠道移位来实现。研究表明,运动可通过改善肠道菌群组成、增加有益菌(如拟杆菌门)比例、减少脂多糖革兰阴性菌(如变形菌门)的丰度,从而降低脂多糖的生成[70]。8周有氧运动可增强非酒精性脂肪肝患者的肠道屏障功能,提高紧密连接蛋白(如闭锁蛋白和闭锁小带蛋白1)的表达,降低肠道通透性,抑制脂多糖移位进入血液循环,进而减轻肝脏炎症及其脂肪变性[71]。但值得注意的是,高强度运动可能会引起肠道菌群紊乱,损害肠道屏障功能,从而促进脂多糖易位。例如,PENG等[72]比较了12?周的高强度间隔训练和中等强度持续训练对小鼠肠道菌群和脂多糖易位的影响,发现中等强度持续训练能够增加有益菌丰度、改善肠道屏障功能、降低血液中脂多糖水平;相反,高强度间隔训练则增加了与肠道通透性升高相关的细菌丰度,导致“肠漏”现象,使血液脂多糖水平升高。该结果与前述关于高强度间隔训练对肠道菌群影响的研究不一致,可能与运动强度、频率及持续时间的差异有关,因此高强度间隔训练具体的运动方案仍需进一步探讨。 总之,适度运动可能通过重塑肠道菌群、降低含脂多糖的革兰阴性菌的丰度、改善肠道屏障功能,从而减少脂多糖的生成和肠道移位,防治非酒精性脂肪肝。 2.5.4 其他作用 除了上述机制外,运动还可能通过调控其他肠道菌群代谢物在非酒精性脂肪肝的防治中发挥重要作用。研究表明,在不饮酒的非酒精性脂肪肝炎患者中,肠道菌群组成的改变导致生态失调,从而增加血液中乙醇水平,提示肠道微生物发酵可产生内源性乙醇[73]。乙醇能够诱导细胞色素P450 2E1表达上调,促进活性氧生成,进而导致线粒体功能障碍并引发肝损伤[74]。此外,在内源性乙醇代谢过程中,还原型烟酰胺腺嘌呤二核苷酸磷酸/烟酰胺腺嘌呤二核苷酸比值升高将抑制脂肪酸β-氧化,导致肝脂肪堆积[75]。乙醇的代谢产物乙醛还可削弱肠道紧密连接蛋白的完整性,并与肠道抗菌肽表达下调相关,导致肠道屏障通透性增加,促进脂多糖易位,从而加剧肝脏炎症[76]。因此,肠道菌群来源的过量内源性乙醇参与了非酒精性脂肪肝的发生与发展。 氧化三甲胺是肠道菌群代谢胆碱、磷脂和肉碱生成的重要代谢产物。某些肠道菌(如梭菌属和脱硫弧菌属)可将膳食来源的胆碱和肉碱转化为三甲胺,后者进入肝脏后在黄素单加氧酶作用下被氧化生成氧化三甲胺。研究显示,非酒精性脂肪肝患者粪便中氧化三甲胺水平显著升高,并与疾病的严重程度呈正相关,提示氧化三甲胺可能参与了非酒精性脂肪肝的进展[77]。氧化三甲胺可抑制胆汁酸介导的肝法尼醇X受体信号通路,导致肝脂肪变性[78],并破坏肠道屏障的结构和功能,进一步激活Toll样受体4/髓分化因子88/核因子κB通路,加重肝脏炎症反应[76]。由此可见,肠道菌群代谢物氧化三甲胺可能通过损害肠道和肝脏功能来促进非酒精性脂肪肝的进展。 此外,吲哚及其衍生物(如吲哚-3-丙酸、吲哚-3-乙酸和吲哚丙酮)是肠道菌群代谢色氨酸生成的重要活性物质。研究表明,在高脂饮食诱导的非酒精性脂肪肝小鼠中,补充吲哚-3-乙酸可通过激活芳香烃受体下调肝脏脂肪生成和β-氧化相关酶的基因表达,并减少小叶炎症及促炎细胞因子的产生,从而预防脂肪变性向非酒精性脂肪性肝炎进展[79]。此外,吲哚还能通过调控胆汁酸代谢改善肠-肝轴稳态,阻碍非酒精性脂肪肝的发生[80]。 运动已被证实能够提高肥胖或非酒精性脂肪肝患者血清中吲哚衍生物的水平[81],同时降低乙醇和氧化三甲胺水平[82]。此外,4周以上的有氧运动能够促进吲哚生成菌(如Romboutsia和A. muciniphila)的生长[83],并减少乙醇生成菌(如大肠杆菌)[84]、三甲胺生成菌(如梭菌属和脱硫弧菌属)的丰度[85]。由此推测,运动可能通过调控肠道菌群组成促进吲哚及其衍生物的合成,同时抑制乙醇和氧化三甲胺的产生,从而增强肝脏代谢能力,改善肠道屏障功能,降低肝脏炎症反应,在非酒精性脂肪肝的防治中发挥积极作用。 运动调控肠道菌群防治非酒精性脂肪肝的作用机制总结,见图4。"
| [1] CHEN H, ZHAN Y, ZHANG J, et al. The Global, Regional, and National Burden and Trends of NAFLD in 204 Countries and Territories: An Analysis From Global Burden of Disease 2019. JMIR Public Health Surveill. 2022;8(12):e34809. [2] LEUNG PB, DAVIS AM, KUMAR S. Diagnosis and Management of Nonalcoholic Fatty Liver Disease. JAMA. 2023;330(17):1687-1688. [3] BARRETO HC, GORDO I. Intrahost evolution of the gut microbiota. Nat Rev Microbiol. 2023; 21(9):590-603. [4] JACKSON KG, WAY GW, ZHOU H. Bile acids and sphingolipids in non-alcoholic fatty liver disease. Chin Med J (Engl). 2022;135(10):1163-1171. [5] KOBAYASHI T, IWAKI M, NAKAJIMA A, et al. Current Research on the Pathogenesis of NAFLD/NASH and the Gut-Liver Axis: Gut Microbiota, Dysbiosis, and Leaky-Gut Syndrome. Int J Mol Sci. 2022;23(19):11689. [6] RALLI T, SAIFI Z, TYAGI N, et al. Deciphering the role of gut metabolites in non-alcoholic fatty liver disease. Crit Rev Microbiol. 2023;49(6):815-833. [7] VITETTA L, HENSON JD. Probiotics and synbiotics targeting the intestinal microbiome attenuate non-alcoholic fatty liver disease. Hepatobiliary Surg Nutr. 2020;9(4):526-529. [8] BÄCKHED F, DING H, WANG T, et al. The gut microbiota as an environmental factor that regulates fat storage. Proc Natl Acad Sci U S A. 2004;101(44):15718-15723. [9] RABOT S, MEMBREZ M, BRUNEAU A, et al. Germ-free C57BL/6J mice are resistant to high-fat-diet-induced insulin resistance and have altered cholesterol metabolism. FASEB J. 2010; 24(12):4948-4959. [10] HENAO-MEJIA J, ELINAV E, JIN C, et al. Inflammasome-mediated dysbiosis regulates progression of NAFLD and obesity. Nature. 2012; 482(7384):179-185. [11] LE ROY T, LLOPIS M, LEPAGE P, et al. Intestinal microbiota determines development of non-alcoholic fatty liver disease in mice. Gut. 2013; 62(12):1787-1794. [12] MOUZAKI M, COMELLI EM, ARENDT BM, et al. Intestinal microbiota in patients with nonalcoholic fatty liver disease. Hepatology. 2013;58(1):120-127. [13] KOOTTE RS, LEVIN E, SALOJÄRVI J, et al. Improvement of Insulin Sensitivity after Lean Donor Feces in Metabolic Syndrome Is Driven by Baseline Intestinal Microbiota Composition. Cell Metab. 2017;26(4):611-619.e6. [14] DEMIR M, LANG S, HARTMANN P, et al. The fecal mycobiome in non-alcoholic fatty liver disease. J Hepatol. 2022;76(4):788-799. [15] LUIS V, JEREMY DH. Probiotics and synbiotics targeting the intestinal microbiome attenuate non-alcoholic fatty liver disease. Hepatobiliary Surg Nutr. 2020;16(11):102-121. [16] CIGROVSKI BERKOVIC M, BILIC-CURCIC I, MRZLJAK A, et al. NAFLD and Physical Exercise: Ready, Steady, Go!. Front Nutr. 2021;8:734859. [17] CARBAJO-PESCADOR S, PORRAS D, GARCÍA-MEDIAVILLA MV, et al. Beneficial effects of exercise on gut microbiota functionality and barrier integrity, and gut-liver crosstalk in an in vivo model of early obesity and non-alcoholic fatty liver disease. Dis Model Mech. 2019;12(5): 314-332. [18] HSU CL, SCHNABL B. The gut-liver axis and gut microbiota in health and liver disease. Nat Rev Microbiol. 2023;21(11):719-733. [19] DUPONT AW, DUPONT HL. The intestinal microbiota and chronic disorders of the gut. Nat Rev Gastroenterol Hepatol. 2011;8(9):523-531. [20] KIMURA I, ICHIMURA A, OHUE-KITANO R, et al. Free Fatty Acid Receptors in Health and Disease. Physiol Rev. 2020;100(1):171-210. [21] XU J, LAI KKY, VERLINSKY A, et al. Synergistic steatohepatitis by moderate obesity and alcohol in mice despite increased adiponectin and p-AMPK. J Hepatol. 2011;55(3):673-682. [22] BRISKEY D, HERITAGE M, JASKOWSKI LA, et al. Probiotics modify tight-junction proteins in an animal model of nonalcoholic fatty liver disease. Therap Adv Gastroenterol. 2016;9(4): 463-472. [23] MEIJNIKMAN AS, LAPPA D, HERREMA H, et al. A systems biology approach to study non-alcoholic fatty liver (NAFL) in women with obesity. iScience. 2022;25(8):104828. [24] YATSUNENKO T, REY FE, MANARY MJ, et al. Human gut microbiome viewed across age and geography. Nature. 2012;486(7402):222-227. [25] ZHOU D, PAN Q, SHEN F, et al. Total fecal microbiota transplantation alleviates high-fat diet-induced steatohepatitis in mice via beneficial regulation of gut microbiota. Sci Rep. 2017;7(1):1529. [26] KOBYLIAK N, ABENAVOLI L, MYKHALCHYSHYN G, et al. A Multi-strain Probiotic Reduces the Fatty Liver Index, Cytokines and Aminotransferase levels in NAFLD Patients: Evidence from a Randomized Clinical Trial. J Gastrointestin Liver Dis. 2018;27(1):41-49. [27] MOHAMAD NOR MH, AYOB N, MOKHTAR NM, et al. The Effect of Probiotics (MCP(®) BCMC(®) Strains) on Hepatic Steatosis, Small Intestinal Mucosal Immune Function, and Intestinal Barrier in Patients with Non-Alcoholic Fatty Liver Disease. Nutrients. 2021;13(9):3192. [28] PASINI E, CORSETTI G, ASSANELLI D, et al. Effects of chronic exercise on gut microbiota and intestinal barrier in human with type 2 diabetes. Minerva Med. 2019;110(1):3-11. [29] WANG J, ZHANG Q, XIA J, et al. Moderate Treadmill Exercise Modulates Gut Microbiota and Improves Intestinal Barrier in High-Fat-Diet-Induced Obese Mice via the AMPK/CDX2 Signaling Pathway. Diabetes Metab Syndr Obes, 2022;15:209-223. [30] MOTIANI KK, COLLADO MC, ESKELINEN JJ, et al. Exercise Training Modulates Gut Microbiota Profile and Improves Endotoxemia. Med Sci Sports Exerc. 2020;52(1):94-104. [31] CHEN H, SHEN L, LIU Y, et al. Strength Exercise Confers Protection in Central Nervous System Autoimmunity by Altering the Gut Microbiota. Front Immunol. 2021;12:628629. [32] TORQUATI L, GAJANAND T, COX ER, et al. Effects of exercise intensity on gut microbiome composition and function in people with type 2 diabetes. Eur J Sport Sci. 2023;23(4):530-541. [33] 韦薇,张秋,黄燕凤,等.不同运动方式对2型糖尿病小鼠肠道菌群及短链脂肪酸的影响[J].广西医科大学学报,2022,39(4):643-648. [34] MCFADZEAN R. Exercise can help modulate human gut microbiota. University of Colorado, 2014. [35] EVANS CC, LEPARD KJ, KWAK JW, et al. Exercise prevents weight gain and alters the gut microbiota in a mouse model of high fat diet-induced obesity. PLoS One. 2014;9(3):e92193. [36] 王鹏,刘宝亮,刘岩,等.运动结合饮食干预对肥胖非酒精性脂肪肝女大学生身体成分和脂代谢及肠道菌群的影响[J].中国学校卫生, 2023,44(8):1169-1173. [37] CHENG R, WANG L, LE S, et al. A randomized controlled trial for response of microbiome network to exercise and diet intervention in patients with nonalcoholic fatty liver disease. Nat Commun. 2022;13(1):2555. [38] GREENBERG NA, GASSULL MA, MEIER R. Short-chain fatty acids: ready for prime time? Nutr Clin Pract. 2006;21(6):639-640. [39] GU X, YUAN L, GAN L, et al. Understanding the Role of Exercise and Probiotic Interventions on Non-Alcoholic Fatty Liver Disease Alleviation in Zebrafish: Dialogue Between the Gut and Liver. Int J Mol Sci. 2025;26(3):1360. [40] CAI GS, SU H, ZHANG J. Protective effect of probiotics in patients with non-alcoholic fatty liver disease. Medicine (Baltimore). 2020;99(32): e21464. [41] RIMAL B, COLLINS SL, TANES CE, et al. Bile salt hydrolase catalyses formation of amine-conjugated bile acids. Nature. 2024;626(8000):859-863. [42] JIAO N, BAKER SS, CHAPA-RODRIGUEZ A, et al. Suppressed hepatic bile acid signalling despite elevated production of primary and secondary bile acids in NAFLD. Gut. 2018;67(10): 1881-1891. [43] LI Z, YUAN H, CHU H, et al. The Crosstalk between Gut Microbiota and Bile Acids Promotes the Development of Non-Alcoholic Fatty Liver Disease. Microorganisms. 2023;11(8):2059. [44] ADAMS LA, WANG Z, LIDDLE C, et al. Bile acids associate with specific gut microbiota, low-level alcohol consumption and liver fibrosis in patients with non-alcoholic fatty liver disease. Liver Int. 2020;40(6):1356-1365. [45] NICOLUCCI AC, HUME MP, MARTÍNEZ I, et al. Prebiotics Reduce Body Fat and Alter Intestinal Microbiota in Children Who Are Overweight or With Obesity. Gastroenterology. 2017;153(3): 711-722. [46] ZHANG M, XIAO B, CHEN X, et al. Physical exercise plays a role in rebalancing the bile acids of enterohepatic axis in non-alcoholic fatty liver disease. Acta Physiol (Oxf). 2024;240(1): e14065. [47] SHI J, CUI J, ZHENG T, et al. Comparative effects of aerobic and resistance exercise on bile acid profiles and liver function in patients with non-alcoholic fatty liver disease. BMC Gastroenterol. 2025;25(1):239. [48] 田盟,严俊,李汛.胆汁酸受体在非酒精性脂肪性肝病中的作用[J].中国生物化学与分子生物学报,2022,38(5):587-594. [49] WEI M, TU W, HUANG G. Regulating bile acids signaling for NAFLD: molecular insights and novel therapeutic interventions. Front Microbiol. 2024;15:1341938. [50] JADHAV K, XU Y, XU Y, et al. Reversal of metabolic disorders by pharmacological activation of bile acid receptors TGR5 and FXR. Mol Metab. 2018;9: 131-140. [51] OSCAR CT, ANNE T, PHILIPPE L, et al. Bile Acid Control of Metabolism and Inflammation in Obesity, Type 2 Diabetes, Dyslipidemia, and Nonalcoholic Fatty Liver Disease. Gastroenterology. 2017;52(7):1679-1694. [52] MORVILLE T, SAHL RE, TRAMMELL SA, et al. Divergent effects of resistance and endurance exercise on plasma bile acids, FGF19, and FGF21 in humans. JCI Insight. 2018;3(15):e122737. [53] 梁家琪,刘恒旭,阳金鑫,等.运动与肠道菌健康效益的关系[J].中国组织工程研究,2023, 27(8):1292-1299. [54] FINN PD, RODRIGUEZ D, KOHLER J, et al. Intestinal TGR5 agonism improves hepatic steatosis and insulin sensitivity in Western diet-fed mice. Am J Physiol Gastrointest Liver Physiol. 2019;316(3):G412-G424. [55] ZHANG S, ZHAO J, XIE F, et al. Dietary fiber-derived short-chain fatty acids: A potential therapeutic target to alleviate obesity-related nonalcoholic fatty liver disease. Obes Rev. 2021;22(11):e13316. [56] LI X, HE M, YI X, et al. Short-chain fatty acids in nonalcoholic fatty liver disease: New prospects for short-chain fatty acids as therapeutic targets. Heliyon. 2024;10(5):e26991. [57] 刘伊依,邱俊强.运动与肠道菌群代谢产物:短链脂肪酸在2型糖尿病中的代谢调控作用[J].中国运动医学杂志,2023,42(10):818-824. [58] YANG X, ZHANG M, LIU Y, et al. Inulin-enriched Megamonas funiformis ameliorates metabolic dysfunction-associated fatty liver disease by producing propionic acid. NPJ Biofilms Microbiomes. 2023;9(1):84. [59] ZHENG M, YANG X, WU Q, et al. Butyrate Attenuates Hepatic Steatosis Induced by a High-Fat and Fiber-Deficient Diet via the Hepatic GPR41/43-CaMKII/HDAC1-CREB Pathway. Mol Nutr Food Res. 2023;67(1):e2200597. [60] HAN Y, QUAN H, JI W, et al. Moderate-intensity continuous training and high-intensity interval training alleviate glycolipid metabolism through modulation of gut microbiota and their metabolite SCFAs in diabetic rats. Biochem Biophys Res Commun. 2024;735:150831. [61] AMIRI P, AREFHOSSEINI S, BAKHSHIMOGHADDAM F, et al. Mechanistic insights into the pleiotropic effects of butyrate as a potential therapeutic agent on NAFLD management: A systematic review. Front Nutr. 2022;9:1037696. [62] DU Y, HE C, AN Y, et al. The Role of Short Chain Fatty Acids in Inflammation and Body Health. Int J Mol Sci. 2024;25(13):7379. [63] 李永强,唐文娟,周永健.肠道菌群及其代谢产物在非酒精性脂肪性肝病发生发展及治疗中的作用[J]. 临床肝胆病杂志,2023,39(8):1805-1810. [64] LI Y, LIU T, YAN C, et al. Diammonium Glycyrrhizinate Protects against Nonalcoholic Fatty Liver Disease in Mice through Modulation of Gut Microbiota and Restoration of Intestinal Barrier. Mol Pharm. 2018;15(9):3860-3870. [65] CAO F, DING Q, ZHUGE H, et al. Lactobacillus plantarum ZJUIDS14 alleviates non-alcoholic fatty liver disease in mice in association with modulation in the gut microbiota. Front Nutr. 2022;9:1071284. [66] 陶永彪,汪龙德,李正菊,等.肠道菌群代谢物短链脂肪酸改善非酒精性脂肪肝病的作用研究进展[J]. 中国药理学与毒理学杂志,2023, 37(1):47-53. [67] KOBAYASHI M, MIKAMI D, KIMURA H, et al. Short-chain fatty acids, GPR41 and GPR43 ligands, inhibit TNF-α-induced MCP-1 expression by modulating p38 and JNK signaling pathways in human renal cortical epithelial cells. Biochem Biophys Res Commun. 2017;486(2):499-505. [68] ZHOU M, LV J, CHEN X, et al. From gut to liver: Exploring the crosstalk between gut-liver axis and oxidative stress in metabolic dysfunction-associated steatotic liver disease. Ann Hepatol. 2025;30(1):101777. [69] CARPINO G, DEL BEN M, PASTORI D, et al. Increased Liver Localization of Lipopolysaccharides in Human and Experimental NAFLD. Hepatology. 2020;72(2):470-485. [70] CHEN J, JIA S, XUE X, et al. Gut microbiota: a novel target for exercise-mediated regulation of NLRP3 inflammasome activation. Front Microbiol. 2024;15:1476908. [71] GAO LL, MA JM, FAN YN, et al. Lycium barbarum polysaccharide combined with aerobic exercise ameliorated nonalcoholic fatty liver disease through restoring gut microbiota, intestinal barrier and inhibiting hepatic inflammation. Int J Biol Macromol. 2021;183:1379-1392. [72] PENG M, ZOU R, YAO S, et al. High-intensity interval training and medium-intensity continuous training may affect cognitive function through regulation of intestinal microbial composition and its metabolite LPS by the gut-brain axis. Life Sci. 2024;352:122871. [73] ZHU L, BAKER SS, GILL C, et al. Characterization of gut microbiomes in nonalcoholic steatohepatitis (NASH) patients: a connection between endogenous alcohol and NASH. Hepatology. 2013;57(2):601-609. [74] CHO YE, KIM DK, SEO W, et al. Fructose Promotes Leaky Gut, Endotoxemia, and Liver Fibrosis Through Ethanol-Inducible Cytochrome P450-2E1-Mediated Oxidative and Nitrative Stress. Hepatology. 2021;73(6):2180-2195. [75] LU Y, GEORGE J. Interaction between fatty acid oxidation and ethanol metabolism in liver. Am J Physiol Gastrointest Liver Physiol. 2024;326(5): G483-G494. [76] MIR H, MEENA AS, CHAUDHRY KK, et al. Occludin deficiency promotes ethanol-induced disruption of colonic epithelial junctions, gut barrier dysfunction and liver damage in mice. Biochim Biophys Acta. 2016;1860(4):765-774. [77] NIAN F, CHEN Y, XIA Q, et al. Gut microbiota metabolite trimethylamine N-oxide promoted NAFLD progression by exacerbating intestinal barrier disruption and intrahepatic cellular imbalance. Int Immunopharmacol. 2024;142(Pt B): 113173. [78] TAN X, LIU Y, LONG J, et al. Trimethylamine N-Oxide Aggravates Liver Steatosis through Modulation of Bile Acid Metabolism and Inhibition of Farnesoid X Receptor Signaling in Nonalcoholic Fatty Liver Disease. Mol Nutr Food Res. 2019;63(17):e1900257. [79] DING Y, YANAGI K, YANG F, et al. Oral supplementation of gut microbial metabolite indole-3-acetate alleviates diet-induced steatosis and inflammation in mice. Elife. 2024;12:RP87458. [80] NIU B, PAN T, XIAO Y, et al. The therapeutic potential of dietary intervention: based on the mechanism of a tryptophan derivative-indole propionic acid on metabolic disorders. Crit Rev Food Sci Nutr. 2025;65(9):1729-1748. [81] KASPEREK MC, MAILING L, PICCOLO BD, et al. Exercise training modifies xenometabolites in gut and circulation of lean and obese adults. Physiol Rep. 2023;11(6):e15638. [82] BRANDAO CFC, KREMPF M, GIOLO DE CARVALHO F, et al. Sphingolipid and Trimethylamine-N-Oxide (TMAO) Levels in Women with Obesity after Combined Physical Training. Metabolites. 2024; 14(8):398. [83] VAZQUEZ-MEDINA A, RODRIGUEZ-TRUJILLO N, AYUSO-RODRIGUEZ K, et al. Exploring the interplay between running exercises, microbial diversity, and tryptophan metabolism along the microbiota-gut-brain axis. Front Microbiol. 2024;15:1326584. [84] SHI J, YU D, YANG Y, et al. Association Between Long-Term Regular Exercise and Gut Microbiota Among Middle-Aged and Older Urban Chinese. Int J Sport Nutr Exerc Metab. 2022;32(3):144-152. [85] AHRENS A P, CULPEPPER T, SALDIVAR B, et al. A Six-Day, Lifestyle-Based Immersion Program Mitigates Cardiovascular Risk Factors and Induces Shifts in Gut Microbiota, Specifically Lachnospiraceae, Ruminococcaceae , Faecalibacterium prausnitzii : A Pilot Study. Nutrients. 2021;13(10):3459. |
| [1] | Zhang Qingtong, Chen Leqin, Liu Chang, Chen Yuting, Guo Ruiwu. Neuromechanism of the endocannabinoid system in regulating exercise motivation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(在线): 1-11. |
| [2] | Liu Jinlong, Abuduwupuer·Haibier, Bai Zhen, Su Danyang, Miao Xin, Li Fei, Yang Xiaopeng. Efficacy of different nonsurgical treatments for adolescent idiopathic scoliosis: a systematic review and network meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2370-2379. |
| [3] | Cao Yong, Teng Hongliang, Tai Pengfei, Li Junda, Zhu Tengqi, Li Zhaojin. Interactions between cytokines and satellite cells in muscle regeneration [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1808-1817. |
| [4] | Cai Ziming, Yu Qinghe, Ma Pengfei, Zhang Xin, Zhou Longqian, Zhang Chongyang, Lin Wenping. Heme oxygenase-1 alleviates lipopolysaccharide-induced inflammatory response in nucleus pulposus mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1624-1631. |
| [5] | Zou Yulian, Chen Chaopei, Huang Haixia, Lan Yuyan, Liu Min, Huang Ting. Resveratrol promotes osteogenic differentiation of bone marrow mesenchymal stem cells in an inflammatory microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1669-1678. |
| [6] | Xia Linfeng, Wang Lu, Long Qianfa, Tang Rongwu, Luo Haodong, Tang Yi, Zhong Jun, Liu Yang. Human umbilical cord mesenchymal stem cell-derived exosomes alleviate blood-brain barrier damage in mice with septic encephalopathy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1711-1719. |
| [7] | Pan Dong, Yang Jialing, Tian Wei, Wang Dongji, Zhu Zheng, Ma Wenchao, Liu Na, Fu Changxi. Resistance exercise activates skeletal muscle satellite cells in aged rats: role of adiponectin receptor 1 pathway [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1736-1746. |
| [8] | Hou Chaowen, Li Zhaojin, Kong Jianda, Zhang Shuli. Main physiological changes in skeletal muscle aging and the multimechanism regulatory role of exercise [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1464-1475. |
| [9] | Sun Yaotian, Xu Kai, Wang Peiyun. Potential mechanisms by which exercise regulates iron metabolism in immune inflammatory diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1486-1498. |
| [10] | Liu Yu, Lei Senlin, Zhou Jintao, Liu Hui, Li Xianhui. Mechanisms by which aerobic and resistance exercises improve obesity-related cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1171-1183. |
| [11] | Wen Fan, Xiang Yang, Zhu Huan, Tuo Yanfang, Li Feng. Exercise improves microvascular function in patients with type 2 diabetes [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1225-1235. |
| [12] | Chen Qiang, Wu Wenjuan, Jiang Shuhua, Huang Da. Physical exercise improves physical function in burn patients: a systematic review and meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1269-1281. |
| [13] | Jiang Yang, Peng Hao, Song Yanping, Yao Na, Song Yueyu, Yin Xingxiao, Li Yanqi, Chen Qigang. Isometric exercise reduces resting blood pressure: a meta-analysis of moderating factors and dose effects [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 975-986. |
| [14] | Li Hanyue, Li Yini, Xiang Linmei, Li Sen. Effects of resistance exercise therapy on pain and function in patients with cervical spondylotic radiculopathy: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 987-996. |
| [15] | Sun Jiahe, Shi Jipeng, Zhu Tianrui, Quan Helong, Xu Hongqi. Effect of exercise intervention in elderly individuals with sarcopenia and its comorbidities: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 997-1007. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||