Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (17): 2731-2738.doi: 10.12307/2024.242
Previous Articles Next Articles
Application of cell derivative in periodontal regeneration
Liang Chao1, 2, Liao Li1, 2, Tian Weidong1, 2, 3
- 1National Engineering Laboratory for Oral Regenerative Medicine, Sichuan University, Chengdu 610041, Sichuan Province, China; 2National Clinical Research Center for Oral Diseases, Chengdu 610041, Sichuan Province, China; 3Department of Trauma Plastic Surgery, West China Hospital of Stomatology, Sichuan University, Chengdu 610041, Sichuan Province, China
-
Received:2023-02-14Accepted:2023-03-25Online:2024-06-18Published:2023-12-16 -
Contact:Liao Li, MD, Researcher, National Engineering Laboratory for Oral Regenerative Medicine, Sichuan University, Chengdu 610041, Sichuan Province, China; National Clinical Research Center for Oral Diseases, Chengdu 610041, Sichuan Province, China Tian Weidong, MD, Professor, National Engineering Laboratory for Oral Regenerative Medicine, Sichuan University, Chengdu 610041, Sichuan Province, China; National Clinical Research Center for Oral Diseases, Chengdu 610041, Sichuan Province, China; Department of Trauma Plastic Surgery, West China Hospital of Stomatology, Sichuan University, Chengdu 610041, Sichuan Province, China -
About author:Liang Chao, Master candidate, National Engineering Laboratory for Oral Regenerative Medicine, Sichuan University, Chengdu 610041, Sichuan Province, China; National Clinical Research Center for Oral Diseases, Chengdu 610041, Sichuan Province, China -
Supported by:National Key Research and Development Program of China No. 2022YFA1104400 (to TWD); National Natural Science Foundation of China (General Program), No. 82071092 (to LL); Key Research and Development Program of Sichuan Province, No. 2019YFS0311 (to LL)
CLC Number:
Cite this article
Liang Chao, Liao Li, Tian Weidong. Application of cell derivative in periodontal regeneration[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(17): 2731-2738.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

2.1 脱细胞基质 脱细胞基质保留了细胞外基质作为高度活跃的三维框架,可以促进组织细胞的黏附和功能。脱细胞基质由许多胶原纤维成分(如胶原蛋白、纤连蛋白和弹性蛋白)和非胶原纤维分子(如蛋白聚糖、透明质酸和糖蛋白)组成,它们通过多种受体相互作用,包括整合素、盘状结构域受体、蛋白多糖表面受体和透明质酸受体形成多组分结构网络[9]。例如,胶原蛋白、纤连蛋白和层粘连蛋白是结合整合素的受体。一些整合素还可以与细胞间黏附分子1和血管细胞黏附分子1结合,构成干细胞微环境的一部分[10]。此外,细胞外基质分子还与包括生长因子受体和Toll样受体在内的其他非传统受体相互作用并调节信号转导[11]。众所周知,不同脱细胞基质上某些受体的差异表达决定了细胞的生物学行为,并通过基质-细胞相互作用决定细胞的增殖、存活、迁移和分化命运。因此,细胞外基质分子通过调节各种细胞的表型和功能来维持组织稳态,从而与各种细胞类型的表面受体相互作用,包括间充质干细胞及成纤维细胞、成牙骨质细胞、成骨细胞和免疫细胞等[9]。 细胞外基质包括脱细胞处理后可以直接应用的固态脱细胞基质以及脱细胞后进一步溶解后具备一定流动性的脱细胞基质。固态脱细胞基质是一种经过脱细胞化并直接用作生物材料,无需进一步破坏脱细胞基质微观结构的支架。固态支架可以根据应用方式进行分类,其中包括脱细胞膜片、脱细胞组织工程的脱细胞基质模块/薄片和整个牙周组织。可溶性脱细胞基质是一种已经脱细胞的材料,然后通过额外的步骤来分解细胞外基质结构并将脱细胞基质溶解成液体形式。可溶性材料可根据重组或应用方法进行分类,包括粉末、微滴、可注射水凝胶、二维和三维水凝胶以及由可溶性脱细胞基质和其他生物材料组成的牙周再生模块。 2.1.1 固态脱细胞基质 (1)牙相关(干)细胞来源脱细胞基质(脱细胞膜片):为了提高三维细胞衍生的细胞外基质支架生成的质量和效率,人们收获细胞来源的脱细胞基质膜然后通过使用各种物理和化学技术制作成3D支架[12]。脱细胞膜片作为具有优越生物弹性和孔隙率的三维细胞衍生细胞外基质支架,可以允许更高效的营养物质交换,有利于细胞增殖、黏附和分化[13]。脱细胞膜片技术是指有效地去除膜片细胞和核成分,特别是脱氧核糖核酸和核糖核酸,同时保留了膜片的细胞外基质的基本成分、生物活性和机械完整性。由于缺乏细胞成分和主要的组织结构,在膜片细胞外基质材料中,异物反应和免疫排斥反应显著减少甚至没有;而原始的三维结构和机械完整性是高度保存的,保存了生物活性,并且具有良好生物相容性。因此,脱细胞膜片在再生医学中的应用正在上升。此外,脱细胞膜片与细胞因子或新材料结合,以获得更好的组织再生效果,已被用于许多组织再生过程,如牙科组织再生、骨再生及韧带再生等。具体而言,牙周再生常用的脱细胞膜片来源有牙周膜干细胞膜片、骨髓间充质干细胞膜片及人尿源性间充质干细胞膜片[13-15],其他诸如L929膜片来源的脱细胞膜片也可供考虑,见表1。"


(2)组织来源脱细胞基质:相较于细胞来源脱细胞基质,组织来源脱细胞基质可以直接从天然组织中获得,且常常具有数量上的优势。其中,同组织来源的脱细胞基质相较于非牙(颌)组织来源脱细胞基质在牙周再生的应用中更为广泛。同种异体同组织来源的脱细胞基质常难以获得,因而,作为替代的异种同类组织来源的脱细胞基质因其具有与缺损组织相适应的细胞外环境和较低的免疫反应,具有更广泛的应用,见表1。此外,异种非同类型组织来源的细胞外基质在牙周再生中也能起到一定的促进牙周再生的效果,这是因为脱细胞基质的主要成分一致或接近,故能发挥与同类型组织脱细胞基质相似的效果。目前,最常用的组织脱细胞外基质为异种脱细胞外基质,其具有来源广泛、脱细胞基质成分近似的优势,可以提供更好的模拟缺损部位微环境。 传统的脱细胞方法破坏细胞膜,除去细胞器和细胞核结构,保留了细胞外基质中的大分子胶原蛋白、纤连蛋白、糖胺聚糖和蛋白多糖等成分,因此脱细胞基质收获后常常呈现出固态结构。直接应用固态的(不可溶的)脱细胞基质是脱细胞基质衍生材料最常见的应用方式,根据脱细胞基质的来源可以分为细胞来源和组织来源两种,细胞来源的脱细胞基质是牙周再生中比较常见的脱细胞基质应用方式;因为脱细胞膜片的膜状形态与缺损部位的形态相适应。且细胞膜片培养技术的发展,使得脱细胞基质材料不受来源、种类的限制,可以提供更为广泛的细胞类型,且可以提取与原缺损组织中相同的细胞类型在体外扩增培养获得脱细胞基质,细胞脱细胞基质可以更好地支持胞内信号转导。而组织来源的脱细胞基质通常具备组织量的优势,且这个优势是数量级的。然而,正常人和其他种属的牙周组织常常无法取材或组织量小,在拔牙处理后的牙周膜经常撕裂扭曲,脱细胞处理后经常变形,因此组织脱细胞基质通常来自于其他组织来源,作为具备生物学活性的生物支架使用。通常而言,用于制备脱细胞基质材料的组织可以在取材脱细胞获得脱细胞基质后直接用于缺损组织;无需14 d或更长的体外培养时间,且同种组织来源的脱细胞基质可以为缺损部位提供正常组织结构的支架供细胞长入,为干细胞的迁移增殖分化提供了更理想更贴近正常组织微环境。 2.1.2 可溶性脱细胞基质 固态脱细胞基质支架的优点是在结构和机械上与牙周组织相似,但是同时也存在一些局限性,如需要手术植入牙周再生模块。此外,将脱细胞基质固态支架与细胞结合需要接种和体外培养,同时将脱细胞基质与其他生物材料结合较为困难,因此应用非常有限。而可溶性脱细胞外基质支架将固态的脱细胞基质制备成脱细胞基质粉末或溶解成液体,既保留了天然的组织结构,又增加其流动性[27]。可溶性脱细胞基质包含天然组织的脱细胞基质结构,但可以无需侵入性手术注射到牙周缺损区域。器官、组织的脱细胞后将脱细胞基质冻干磨成细粉,然后进行胃蛋白酶消化,进一步分解脱细胞基质,提高溶解度[10,12,28-29]。然后将溶液pH值调节以灭活胃蛋白酶,最后调整溶液的pH值和盐浓度至生理pH值,在缓冲液中洗涤,冻干保存。后续可溶解用于粉末、水凝胶微创注射疗法、光固化支架合成和3D打印的生物墨水制造等领域。 研磨和部分溶解的脱细胞基质已直接用于组织工程。颗粒状的脱细胞基质含有更多的修复成分,可以部分溶于溶液中或重悬形成悬浊液用于注射而不需要完全溶解[30-31]。此外,脱细胞骨粉作为常用的牙周组织工程材料[32-33],是脱细胞外基质粉末临床应用的另一种形式,常常与水凝胶、引导性组织再生膜联合使用促进牙周再生[26,34]。脱细胞基质衍生的水凝胶去除细胞器和DNA,保留硫酸糖胺聚糖和其他细胞外基质蛋白,如胶原蛋白,凝胶在生理温度和pH值,并具有纳米纤维结构[35]。在生理温度下,可自组装成纳米纤维水凝胶[36]。组织特异性脱细胞基质可用于涂层细胞培养皿,方法类似于其他用于培养皿铺胶的材料如胶原蛋白,或可以聚合成薄水凝胶,用于在支架上进行细胞接种。这些可溶性脱细胞基质平台已被用于模拟组织体外微环境,同时也可用于评估脱细胞基质在体外调节干细胞生物学行为和体外表型的转变的能力[37]。此外,脱细胞基质凝胶可以作为直接的人类类器官,具有稳定组织层次特征的细胞生长,用于组织工程和药物筛选。这些脱细胞基质衍生的水凝胶的发展为组织工程开辟了潜力[29,38-42],见表2。"


细胞暴露于三维水凝胶支架和环境中,与2D生长相比,具有特定空间结构的水凝胶会显著改变细胞的特性和修复潜能。挤出式生物打印依赖于结构支持的生物墨水层相互叠加,需要高黏度的解决方案来进行适当的打印和支架制造。在注射脱细胞基质时常用的浓度下,脱细胞基质预聚物由于黏度非常低,不能形成层或打印。应用溶解的脱细胞外基质作为载细胞的生物墨水通过结合甲基丙烯酸酯明胶提高浓度增加了生物墨水的可打印性[11]。另外,使用维生素b2紫外线诱导交联,也可以在没有支撑材料的情况下3D打印细胞外基质支架系统[43]。目前的研究已经试图通过更高的脱细胞基质浓度、交联修饰和与其他生物材料的结合来解决这些问题。如何提高生物墨水的可打印性还是研究者们未来研究的热点。"


2.2 细胞外囊泡 外泌体、微囊泡和凋亡小体是由细胞分泌的3种囊泡,被脂质双分子层包围,统称为细胞外囊泡[7]。囊泡可以通过其大小、生物发生和释放机制来区分。囊泡的直径分别为30-150 nm(外泌体)、100-1 000 nm(微囊泡)或1-5 μm(凋亡小体)的范围内[44-46]。一般而言,这些胞外囊泡由核酸、蛋白质、脂质和信号分子组成。根据原始细胞和囊泡类型的不同,其成分也有所不同。例如,细胞外囊泡中蛋白质的存在可能会受到该特定胞外囊泡的生物发生的影响[44-46]。 2.2.1 外泌体 外泌体生物发生的经典途径始于宿主细胞的内吞产生活性核内体,导致核内体限制膜的内陷和腔内小泡的形成,然后成熟为多泡体定向到质膜,作为外泌体被释放到细胞外环境中。微囊泡的生物发生要简单得多,在刺激下,母细胞的外膜起泡作为微囊泡与母细胞分离。外泌体几乎可以由所有类型的细胞分泌[18-20],含有特定的细胞物质:核糖核酸,游离脂肪酸,蛋白质,表面受体和细胞因子。外泌体作为局部和全身细胞间通讯介质,在各种生物学功能中起重要作用,并调节许多疾病的生理和病理过程[18,20]。 有报道表明,牙周组织可以通过从骨髓间充质干细胞中获得外泌体的移植来再生[44-46]。据报道,基于细胞外泌体的牙周炎诊治策略可以克服传统疗法缺点,具有广阔的临床应用潜力[6]。例如,间充质干细胞外泌体对中性粒细胞功能和寿命具有保护作用[47],提高机体抗感染能力;来源于牙龈干细胞和牙髓干细胞的外泌体可以促进巨噬细胞极化[48-49],抑制M1型巨噬细胞的促炎效果,激活巨噬细胞M2表型的转化;人脐血间充质干细胞的外泌体可以通过抑制树突细胞的功能,减少淋巴细胞的产生起到调节免疫的作用;牙周膜干细胞的外泌体可以通过Th17/Treg/miR-17-155p/SIRT5调控网络缓解炎症,并维持Th1/Treg平衡[50]。牙龈干细胞的外泌体miR-1260b通过靶向Wnt5a介导的RANKL途径来抑制破骨活性[51]。脂肪干细胞外泌体可通过抑制破骨细胞中的NLRP3炎症小体活化和拮抗骨细胞介导的破骨形成来减少骨吸收并修复骨缺损[6]。文章总结了外泌体在牙周再生中的应用及再生效果具体研究内容[52-54],见表3。"


目前外泌体作为载体的作用也受到广泛关注,有研究人员将电穿孔用于将外源siRNA加载到外泌体中用于特定基因的敲低[55],由此可见外泌体介导的siRNA递送的治疗潜力。此外,超声处理已被用作一种主动将功能性小RNA加载到外泌体中的方法,受体细胞可以吸收并能够敲低靶向mRNA,从而导致蛋白表达降低[56]。 细胞释放的外泌体是研究最广泛的胞外囊泡。外泌体在牙周炎的诊治过程中发挥举足轻重的作用。外泌体可以被远处的细胞吸收并在胞内发挥功能[55]。间充质干细胞来源的外泌体可以调节伤口愈合,并可以调控骨髓间充质干细胞血管生成、抗炎、抗凋亡和免疫调节的功能。工程化外泌体是外泌体研究的新热点,一些研究报告了外泌体的重组可以通过修饰供体细胞来实现。另外,工程化外泌体可以通过多种治疗分子搭载发挥生物作用。例如,电穿孔可用于将外源siRNA加载到外泌体中,静脉注射后可导致特定基因的敲低,并且已观察到外泌体介导的siRNA递送的治疗潜力[56]。超声处理已被用作一种主动将功能性小RNA加载到外泌体中的方法,受体细胞可以吸收并能够敲低靶向mRNA,从而导致蛋白表达降低[57]。"


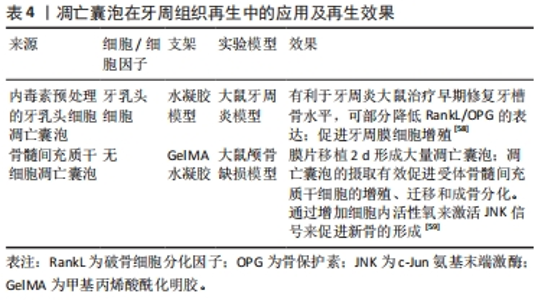
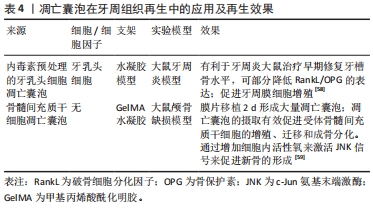
尤其在牙周再生中,骨再生是十分重要的一部分。在骨重塑过程中,破骨细胞的寿命维持2周后发生细胞凋亡并产生大量的凋亡囊泡[59]。已经表明,骨髓单核细胞受到核因子κB受体激活配体的刺激,分化为前破骨细胞和成熟破骨细胞。研究发现,源自成熟破骨细胞的凋亡囊泡可以被成骨细胞MC3T3-E1吞噬并增强后者活力。在所有破骨细胞衍生的胞外囊泡中,凋亡囊泡具有高浓度的RANK,并具有最高的成骨潜力,可以通过PI3K/Akt/mTOR/S6K信号通路诱导成骨细胞分化,这证实了凋亡囊泡的成骨能力[59]。凋亡囊泡生成减少会显著损害骨髓间充质干细胞的自我更新和分化。全身输注外源性凋亡囊泡挽救了间充质干细胞的损伤,改善了骨质疏松。此外间充质干细胞能够通过整合素αvβ3吞噬凋亡小体,并重复使用凋亡囊泡衍生的泛素连接酶来抑制Axin1,从而激活经典Wnt/β-Catenin通路促进成骨。这些研究都表明凋亡囊泡在牙周缺损骨再生治疗中的潜在用途[61]。 凋亡囊泡由凋亡细胞特异性产生,能够包封细胞凋亡过程中产生的细胞因子,可以通过细胞因子的转移促进细胞间通讯。凋亡囊泡可被巨噬细胞、树突状细胞、上皮细胞、内皮细胞和成纤维细胞识别并吞噬以进行清除,随后在溶酶体中内化、摄取和降解,被认为是细胞间通讯的关键调节因子,可以参与多种生理和病理过程,如炎症、免疫应答、凝血、肿瘤发生和宿主-微生物相互作用[62]。尽管两种胞外囊泡都被认为是十分重要的细胞间通讯介质,可以通过静脉注射的方式,调节机体的免疫微环境,但是凋亡囊泡表面的磷脂酰丝氨酸可以促进巨噬细胞募集,这是外泌体所不具备的优势。此外,凋亡囊泡作为细胞自我更新的一种手段,具备更有活力的功能性分子(RNA,DNA和蛋白质等),而外泌体作为一种载体,成分更单一。此外,凋亡囊泡密度较外泌体较高,提取更容易且提取的囊泡产量具备数量级优势[63]。"


2.3 条件培养基 条件培养基中含有细胞外基质蛋白、酶、血管生成因子、生长因子和细胞因子[64-65]。文献已经报道了一种以培养上清液的形式收集从干细胞中释放分泌因子然后移植的方法,用以取代细胞移植[62-63,66-67]。大量证据表明,间充质干细胞通过旁分泌和自分泌方式增强早期炎症期间的免疫反应,并通过产生一系列保护性生物活性因子来增强随后的组织再生[64]。这些因子广义地定义为条件培养基,是包含细胞因子、趋化因子、细胞黏附分子、脂质介质、白细胞介素、生长因子、激素、外泌体和微泡等在内的一种复合成分。 NAGATA等[64]的研究表明,从牙周干细胞中获得的条件培养基的移植可导致牙周组织的再生,牙周干细胞-条件培养基含有多种生长因子和血管生成因子,并且通过肿瘤坏死因子α的产生抑制炎症反应来促进牙周再生。牙龈干细胞和牙周膜干细胞的条件培养基移植到大鼠牙周缺损中,降低了白细胞介素1和肿瘤坏死因子α的表达水平,增强牙周再生[65]。牙周膜干细胞-条件培养基移植导致愈合的牙周组织中肿瘤坏死因子α mRNA水平降低[68],抑制了干扰素γ刺激下的单核/巨噬细胞细胞系[69]。骨髓间充质干细胞条件培养基移植的牙周组织再生也有报道。这些结果表明,间充质干细胞来源的条件培养基的移植主要通过其抗炎作用来诱导牙周再生,提示了使用干细胞来源的条件培养基进行无细胞治疗的可能性[1,70]。 干细胞在促进组织再生中的作用主要取决于它们的旁分泌功能。作为一种无细胞技术,与间充质干细胞移植相比,条件培养基移植比直接间充质干细胞移植更方便、更安全,临床转化潜力更大。它可以避免宿主免疫反应的风险,可以储存较长时间,具有成本效益。此外,间充质干细胞条件培养基具有免疫调节和组织再生潜力[72,74]。因此,使用间充质干细胞条件培养基可能是在牙周炎的炎症环境中再生牙周组织的有效方法。条件培养基具有促进骨再生[72]、牙槽骨分化[75]、血管生成的功能[76],并具有趋化活性[74],可以发挥免疫调节的作用[64]。 文章总结了条件培养基在牙周组织再生中的研究进展,见表5。"

| [1] IWASAKI K, PENG Y, KANDA R, et al. Stem cell transplantation and cell-free treatment for periodontal regeneration. Int J Mol Sci. 2022;23(3):1011. [2] SUZUKI M, KIMURA T, YOSHIDA Y et al. In vitro tissue reconstruction using decellularized pericardium cultured with cells for ligament regeneration. Polymers (Basel). 2022; 14(12):2351. [3] GOU M, HUANG YZ, HU JG, et al. Epigallocatechin-3-gallate cross-linked small intestinal submucosa for guided bone regeneration. ACS Biomater Sci Eng. 2019;5(10):5024-5035. [4] SON H, JEON M, CHOI HJ et al. Decellularized human periodontal ligament for periodontium regeneration. PLoS One. 2019;14(8):e0221236. [5] HOANG THI TT, TRAN NGUYEN DH, NGUYEN DTD, et al. Decellularized porcine epiphyseal. 2000te-derived extracellular matrix powder: synthesis and characterization. Cells Tissues Organs. 2020;209(2-3):101-109. [6] LIN H, CHEN H, ZHAO X, et al. Advances of exosomes in periodontitis treatment. J Transl Med. 2022;20(1):279. [7] BRENNAN MA, LAYROLLE P, MOONEY DJ. Biomaterials functionalized with MSC secreted extracellular vesicles and soluble factors for tissue regeneration. Adv Funct Mater. 2020;30(37):1909125. [8] XIN Y, XU P, WANG X, et al. Human foreskin-derived dermal stem/progenitor cell-conditioned medium combined with hyaluronic acid promotes extracellular matrix regeneration in diabetic wounds. Stem Cell Res Ther. 2021;12(1):49. [9] GU R, LIU H, ZHU Y, et al. Is extracellular matrix (ECM) a promising scaffold biomaterial for bone repair? Histol Histopathol. 2021;36(12):1219-1234. [10] KIM BS, DAS S, JANG J, et al. Decellularized extracellular matrix-based bioinks for engineering tissue- and organ-specific microenvironments. Chem Rev. 2020;120(19): 10608-10661. [11] YANG X, MA Y, WANG X, et al. A 3D-bioprinted functional module based on decellularized extracellular matrix bioink for periodontal regeneration. Adv Sci (Weinh). 2022;10(5):e2205041. [12] KIM B, VENTURA R, LEE BT. Functionalization of porous BCP scaffold by generating cell-derived extracellular matrix from rat bone marrow stem cells culture for bone tissue engineering. J Tissue Eng Regen Med. 2018;12(2):e1256-e1267. [13] XU Y, XU GY, TANG C, et al. Preparation and characterization of bone marrow mesenchymal stem cell-derived extracellular matrix scaffolds. J Biomed Mater Res B Appl Biomater. 2015;103(3):670-678. [14] FARAG A, HASHIMI SM, VAQUETTE C, et al. The effect of decellularized tissue engineered constructs on periodontal regeneration. J Clin Periodontol. 2018;45(5):586-596. [15] JIANG Y, LIU JM, HUANG JP, et al. Regeneration potential of decellularized periodontal ligament cell sheets combined with 15-Deoxy-Δ(12,14)-prostaglandin J(2)nanoparticles in a rat periodontal defect. Biomed Mater. 2021;16(4):045008. [16] ZHANG X, LI H, SUN J, et al. Cell-derived micro-environment helps dental pulp stem cells promote dental pulp regeneration. Cell Prolif. 2017;50(5):e12361. [17] NOWWAROTE N, PETIT S, FERRE FC, et al. Extracellular matrix derived from dental pulp stem cells promotes mineralization. Front Bioeng Biotechnol. 2021;9:740712. [18] HUANG JP, WU YM, LIU JM, et al. Decellularized matrix could affect the proliferation and differentiation of periodontal ligament stem cells in vitro. J Periodontal Res. 2021; 56(5):929-939. [19] YANG X, XIONG X, ZHOU W, et al. Effects of human urine-derived stem cells on the cementogenic differentiation of indirectly-cocultured periodontal ligament stem cells. Am J Transl Res. 2020;12(2):361-378. [20] WEN Y, YANG H, WU J, et al. COL4A2 in the tissue-specific extracellular matrix plays important role on osteogenic differentiation of periodontal ligament stem cells. Theranostics. 2019;9(15):4265-4286. [21] HAN X, LIAO L, ZHU T, et al. Xenogeneic native decellularized matrix carrying PPARγ activator RSG regulating macrophage polarization to promote ligament-to-bone regeneration. Mater Sci Eng C Mater Biol Appl. 2020;116:111224. [22] LEE JS, KIM SK, GRUBER R, et al. Periodontal healing by periodontal ligament fiber with or without cells: A preclinical study of the decellularized periodontal ligament in a tooth replantation model. J Periodontol. 2020;91(1):110-119. [23] NAKAMURA N, ITO A, KIMURA T, et al. Extracellular matrix induces periodontal ligament reconstruction in vivo. Int J Mol Sci. 2019;20(13):3277. [24] IMAMURA K, HAMADA Y, YOSHIDA W, et al. Investigating the effects of dehydrated human amnion-chorion membrane on periodontal healing. Biomolecules. 2022;12(6):857. [25] BIANCHI S, BERNARDI S, SIMEONE D, et al. Proliferation and morphological assessment of human periodontal ligament fibroblast towards bovine pericardium membranes: an in vitro study. Materials (Basel). 2022;15(23):8284. [26] SUZUKI M, KIMURA T, NAKANO Y, et al. Preparation of mineralized pericardium by alternative soaking for soft-hard interregional tissue application. J Biomed Mater Res A. 2023;111(2):198-208. [27] CHOI JS, YANG HJ, KIM BS, et al. Human extracellular matrix (ECM) powders for injectable cell delivery and adipose tissue engineering. J Control Release. 2009;139(1):2-7. [28] HUA Y, HUO Y, BAI B, et al. Fabrication of biphasic cartilage-bone integrated scaffolds based on tissue-specific photo-crosslinkable acellular matrix hydrogels. Materials Today Bio. 2022;17:100489. [29] GIOBBE GG, CROWLEY C, LUNI C, et al. Extracellular matrix hydrogel derived from decellularized tissues enables endodermal organoid culture. Nat Commun. 2019;10(1): 5658. [30] PETERSON B, WHANG PG, IGLESIAS R, et al. Osteoinductivity of commercially available demineralized bone matrix. Preparations in a spine fusion model. J Bone Joint Surg Am. 2004;86(10):2243-2250. [31] TABUCHI M, NEGISHI J, YAMASHITA A, et al. Effect of decellularized tissue powders on a rat model of acute myocardial infarction. Mater Sci Eng C Mater Biol Appl. 2015;56:494-500. [32] NOBAKHT S, MILNE TJ, DUNCAN WJ, et al. Expression of the pleiotrophin-midkine axis in a sheep tooth socket model of bone healing. J Periodontal Res. 2023;58(1):109-121. [33] LIM J, JUN SH, TALLARICO M, et al. A randomized controlled trial of guided bone regeneration for peri-implant dehiscence defects with two anorganic bovine bone materials covered by titanium meshes. Materials (Basel). 2022;15(15):5294. [34] GAO C, SOW WT, WANG Y, et al. Hydrogel composite scaffolds with an attenuated immunogenicity component for bone tissue engineering applications. J Mater Chem B. 2021;9(8):2033-2041. [35] UNGERLEIDER JL, JOHNSON TD, RAO N, et al. Fabrication and characterization of injectable hydrogels derived from decellularized skeletal and cardiac muscle. Methods. 2015;84:53-59. [36] SINGELYN JM, DEQUACH JA, SEIF-NARAGHI SB, et al. Naturally derived myocardial matrix as an injectable scaffold for cardiac tissue engineering. Biomaterials. 2009;30(29): 5409-5416. [37] FARNEBO S, WOON CY, SCHMITT T, et al. Design and characterization of an injectable tendon hydrogel: a novel scaffold for guided tissue regeneration in the musculoskeletal system. Tissue Eng Part A. 2014;20(9-10):1550-1561. [38] SPANG MT, CHRISTMAN KL. Extracellular matrix hydrogel therapies: in vivo applications and development. Acta Biomater. 2018;68:1-14. [39] NOGUEIRA DMB, FIGADOLI ALF, ALCANTARA PL, et al. Biological behavior of xenogenic scaffolds in alcohol-induced rats: histomorphometric and picrosirius red staining analysis. Polymers (Basel). 2022;14(3):584. [40] LINK PA, RITCHIE AM, COTMAN GM, et al. Electrosprayed extracellular matrix nanoparticles induce a pro-regenerative cell response. J Tissue Eng Regen Med. 2018; 12(12):2331-2336. [41] AGARWAL T, NARAYAN R, MAJI S, et al. Decellularized caprine liver extracellular matrix as a 2D substrate coating and 3D hydrogel platform for vascularized liver tissue engineering. J Tissue Eng Regen Med. 2018;12(3):e1678-e1690. [42] SHEN X, LI S, ZHAO X, et al. Dual-crosslinked regenerative hydrogel for sutureless long-term repair of corneal defect. Bioact Mater. 2023;20:434-448. [43] HWANG SH, KIM J, HEO C, et al. 3D printed multi-growth factor delivery patches fabricated using dual-crosslinked decellularized extracellular matrix-based hybrid inks to promote cerebral angiogenesis. Acta Biomater. 2023;157:137-148. [44] WU J, CHEN L, WANG R, et al. Exosomes secreted by stem cells from human exfoliated deciduous teeth promote alveolar bone defect repair through the regulation of angiogenesis and osteogenesis. ACS Biomater Sci Eng. 2019;5(7):3561-3571. [45] CHEW JRJ, CHUAH SJ, TEO KYW, et al. Mesenchymal stem cell exosomes enhance periodontal ligament cell functions and promote periodontal regeneration. Acta Biomater. 2019;89:252-264. [46] MOHAMMED E, KHALIL E, SABRY D. Effect of adipose-derived stem cells and their exo as adjunctive therapy to nonsurgical periodontal treatment: a histologic and histomorphometric study in rats. Biomolecules. 2018;8(4):167. [47] TAGHAVI-FARAHABADI M, MAHMOUDI M, REZAEI N, et al. Wharton’s Jelly mesenchymal stem cells exosomes and conditioned media increased neutrophil lifespan and phagocytosis capacity. Immunol Invest. 2021;50(8):1042-1057. [48] WANG R, JI Q, MENG C, et al. Role of gingival mesenchymal stem cell exosomes in macrophage polarization under inflammatory conditions. Int Immunopharmacol. 2020; 81:106030. [49] SHEN Z, KUANG S, ZHANG Y, et al. Chitosan hydrogel incorporated with dental pulp stem cell-derived exosomes alleviates periodontitis in mice via a macrophage-dependent mechanism. Bioact Mater. 2020;5(4):1113-1126. [50] ZHENG Y, DONG C, YANG J, et al. Exosomal microRNA-155-5p from PDLSCs regulated Th17/Treg balance by targeting sirtuin-1 in chronic periodontitis. J Cell Physiol. 2019; 234(11):20662-20674. [51] NAKAO Y, FUKUDA T, ZHANG Q, et al. Exosomes from TNF-α-treated human gingiva-derived MSCs enhance M2 macrophage polarization and inhibit periodontal bone loss. Acta Biomater. 2021;122:306-324. [52] HUANG CC, KANG M, Lu Y, et al. Functionally engineered extracellular vesicles improve bone regeneration. Acta Biomater. 2020;109:182-194. [53] KANG M, HUANG CC, LU Y, et al. Bone regeneration is mediated by macrophage extracellular vesicles. Bone. 2020;141:115627. [54] HUANG CC, KANG M, SHIRAZI S, et al. 3D Encapsulation and tethering of functionally engineered extracellular vesicles to hydrogels. Acta Biomater. 2021;126:199-210. [55] NIK MOHAMED KAMAL NNS, SHAHIDAN WNS. Salivary exosomes: from waste to promising periodontitis treatment. Front Physiol. 2021;12:798682. [56] LAMICHHANE TN, JEYARAM A, Patel DB, et al. Oncogene knockdown via active loading of small rnas into extracellular vesicles by sonication. Cell Mol Bioeng. 2004;9(3):315-324. [57] DOU G, TIAN R, LIU X, et al. Chimeric apoptotic bodies functionalized with natural membrane and modular delivery system for inflammation modulation. Sci Adv. 2020; 6(30):eaba2987. [58] KAKARLA R, HUR J, KIM YJ, et al. Apoptotic cell-derived exosomes: messages from dying cells. Exp Mol Med. 2020;52(1):1-6. [59] MA Q, LIANG M, WU Y, et al. Mature osteoclast-derived apoptotic bodies promote osteogenic differentiation via RANKL-mediated reverse signaling. J Biol Chem. 2019; 294(29):11240-11247. [60] LIU D, KOU X, CHEN C, et al. Circulating apoptotic bodies maintain mesenchymal stem cell homeostasis and ameliorate osteopenia via transferring multiple cellular factors. Cell Res. 2018;28(9):918-933. [61] LI M, XING X, HUANG H, et al. BMSC-derived apoevs promote craniofacial bone repair via ROS/JNK signaling. J Dent Res. 2022;101(6):714-723. [62] LI X, LIU Y, LIU X, et al. Advances in the therapeutic effects of apoptotic bodies on systemic diseases. Int J Mol Sci. 2022;23(15):8202. [63] HUA S, BARTOLD PM, GULATI K, et al. Periodontal and dental pulp cell-derived small extracellular vesicles: a review of the current status. Nanomaterials (Basel). 2021;11(7):1858. [64] NAGATA M, IWASAKI K, AKAZAWA K, et al. Conditioned medium from periodontal ligament stem cells enhances periodontal regeneration. Tissue Eng Part A. 2017;23(9-10): 367-377. [65] QIU J, WANG X, ZHOU H, et al. Enhancement of periodontal tissue regeneration by conditioned media from gingiva-derived or periodontal ligament-derived mesenchymal stem cells: a comparative study in rats. Stem Cell Res Ther. 2020;11(1):42. [66] LAI H, LI J, KOU X, et al. Extracellular vesicles for dental pulp and periodontal regeneration. Pharmaceutics. 2023;15(1):282. [67] LIU C, LI Y, HAN G, Advances of mesenchymal stem cells released extracellular vesicles in periodontal bone remodeling. DNA Cell Biol. 2022;41(11):935-950. [68] YANG F, ZHANG R, XU J, et al. Comparative effects of concentrated growth factors on the biological characteristics of periodontal ligament cells and stem cells from apical papilla. J Endod. 2022;48(8):1029-1037. [69] LIU J, WANG H, ZHANG L, et al. Periodontal ligament stem cells promote polarization of M2 macrophages. J Leukoc Biol. 2022;111(6):1185-1197. [70] NOVELLO S, TRICOT-DOLEUX S, NOVELLA A, et al. Influence of periodontal ligament stem cell-derived conditioned medium on osteoblasts. Pharmaceutics. 2022;14(4):729. [71] OGISU K, FUJIO M, TSUCHIYA S, et al. Conditioned media from mesenchymal stromal cells and periodontal ligament fibroblasts under cyclic stretch stimulation promote bone healing in mouse calvarial defects. Cytotherapy. 2020;22(10):543-551. [72] JIN Z, FENG Y, LIU H: Conditioned media from differentiating craniofacial bone marrow stromal cells influence mineralization and proliferation in periodontal ligament stem cells. Hum Cell. 2016;29(4):162-175. [73] KATAGIRI W, OSUGI M, KAWAI T, et al. First-in-human study and clinical case reports of the alveolar bone regeneration with the secretome from human mesenchymal stem cells. Head Face Med. 2016;12:5. [74] INUKAI T, KATAGIRI W, YOSHIMI R, et al. Novel application of stem cell-derived factors for periodontal regeneration. Biochem Biophys Res Commun. 2013;430(2):763-768. [75] LIU N, GU B, LIU N, et al. Wnt/β-catenin pathway regulates cementogenic differentiation of adipose tissue-deprived stem cells in dental follicle cell-conditioned medium. PLoS One. 2014;9(5):e93364. [76] KATAGIRI W, KAWAI T, OSUGI M, et al. Angiogenesis in newly regenerated bone by secretomes of human mesenchymal stem cells. Maxillofac Plast Reconstr Surg. 2017; 39(1):8. |
| [1] | Liu Hanfeng, Wang Jingjing, Yu Yunsheng. Artificial exosomes in treatment of myocardial infarction: current status and prospects [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1118-1123. |
| [2] | Ma Shuwei, He Sheng, Han Bing, Zhang Liaoyun. Exosomes derived from mesenchymal stem cells in treatment of animals with acute liver failure: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1137-1142. |
| [3] | Shen Ziqing, Xia Tian, Shan Yibo, Zhu Ruijun, Wan Haoxin, Ding Hao, Pan Shu, Zhao Jun. Vascularized tracheal substitutes constructed by exosome-load hydrogel-modified 3D printed scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 697-705. |
| [4] | Yin Tong, Yang Jilei, Li Yourui, Liu Zhuoran, Jiang Ming. Application of core-shell structured nanofibers in oral tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 766-770. |
| [5] | Dai Jing, Liu Shasha, Shen Mingjing. Exosome-loaded injectable hydrogel for repairing bone defects around implants [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 347-354. |
| [6] | He Bo, He Zhijun, Liu Tao, Ma Suilu, Wei Xiaotao, Wang Weiwei. Action mechanism and advantages of mesenchymal stem cells for treating flap ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(25): 4065-4071. |
| [7] | Yang Mengxiao, Fu Changxi. Mechanism by which strength training improves bone injury in ovariectomized rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(20): 3150-3156. |
| [8] | Zhu Shijie, Yang Yiting, Cao Yuting, Zheng Liangdong, Lin Kaili, Zhu Rui. Cartilage protective effect of swimming exercise in aged mice with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(20): 3170-3175. |
| [9] | Ge Ruiyang, Ni Can, Yang Kun, Yan Fuhua. The role of macrophage polarization in the pathogenesis and treatment of periodontitis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(20): 3246-3251. |
| [10] | Long Yi, Yang Jiaming, Ye Hua, Zhong Yanbiao, Wang Maoyuan. Extracellular vesicles in sarcopenic obesity: roles and mechanisms [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 315-320. |
| [11] | Long Zhisheng, Gong Feipeng, Wen Jiabin, Min Huan, Shu Yang, Lai Zhuoxi, Chen Gang. Bone marrow mesenchymal stem cell exosomes combined with epigallocatechin-3-gallate in treatment of spinal cord ischemia/reperfusion injury in rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(19): 2953-2959. |
| [12] | Gu Chengxu, Zhang Naili, Meng Yongchun, Liu Qing, Guo Qixuan, Fu Li, Zhang Luping, Huang Fei. Adipose-derived mesenchymal stem cell-derived exosomes alleviate hydrogen peroxide-induced PC12 cell apoptosis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(19): 2988-2995. |
| [13] | Xiao Ziteng, Wang Tingyu, Zhang Wenwen, Tan Fengyi, Su Haiwei, Li Siting, Wu Yahui, Zhou Yanfang, Peng Xinsheng. Exosomes and skin wound healing [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(19): 3104-3110. |
| [14] | Yang Yunyun, Chen Qiqing, Zhao Jirong, Zhu Bao, Ma Dong, Huang Junkai, An Dehao, Zou Jipeng, Liu Weihang. Research status of traditional Chinese medicine monomer mediating related signaling pathways in treatment of intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(18): 2918-2924. |
| [15] | Zhao Shasha, He Qing, Li Jia, Wu Ying. Effects of recombinant human collagen supplementation on extracellular matrix remodeling in mouse skeletal muscle after eccentric exercise [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(16): 2542-2549. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||