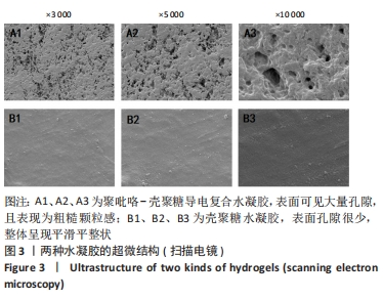
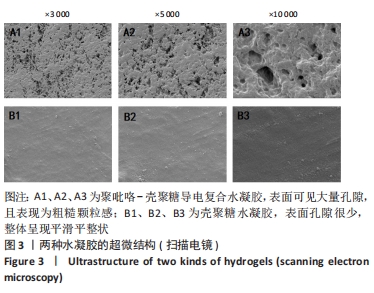
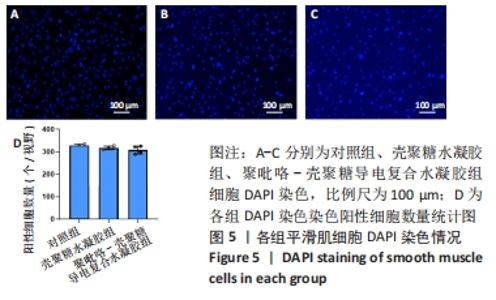
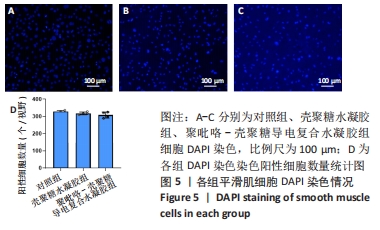
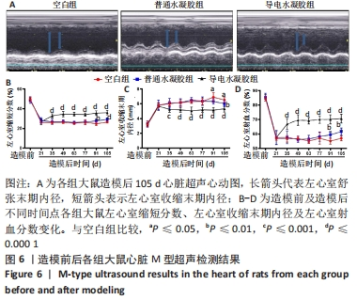
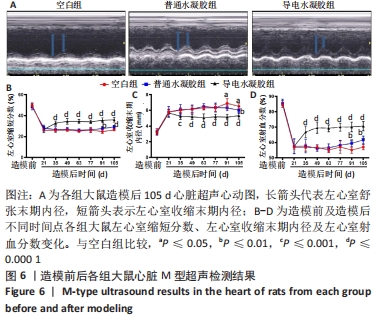
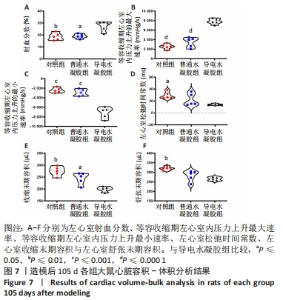
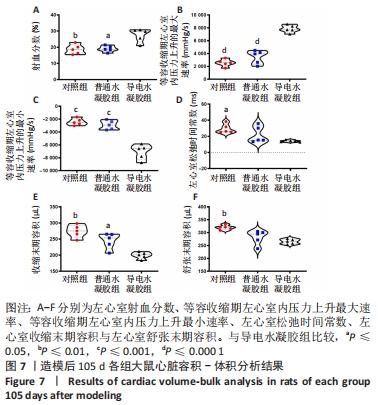
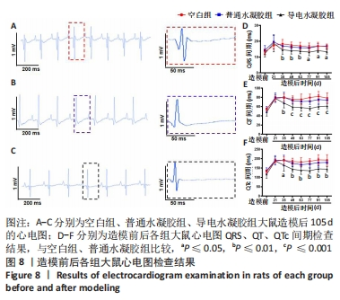
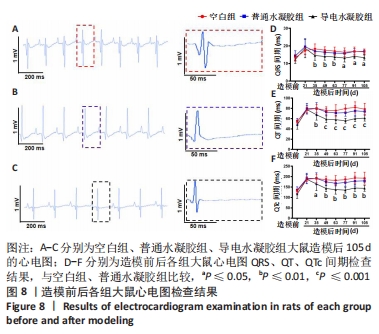
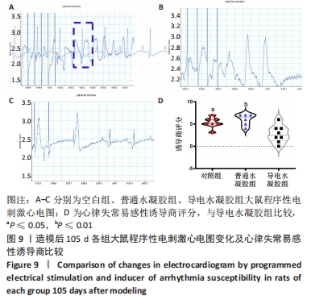
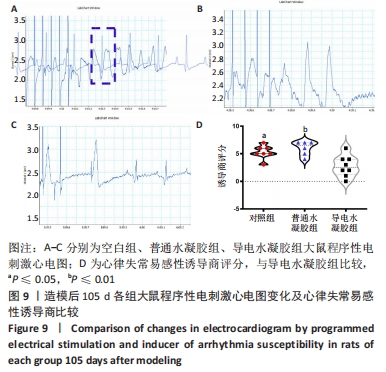
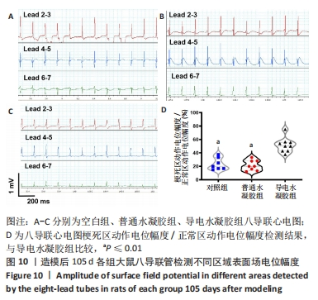
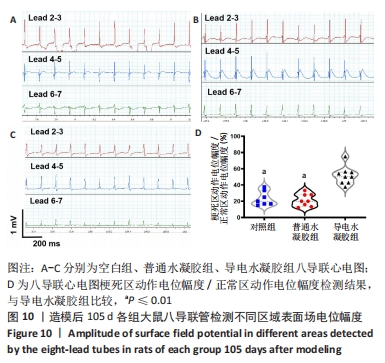
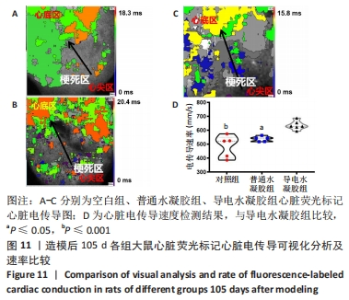
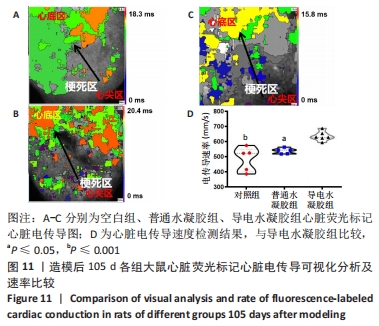
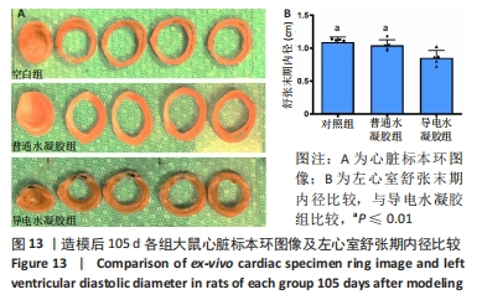
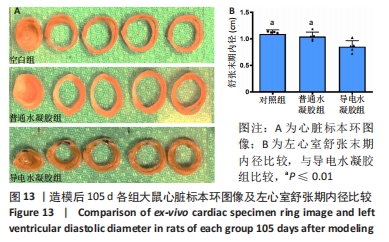
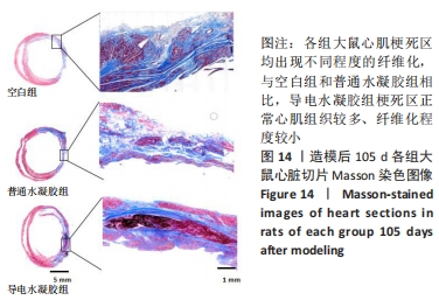
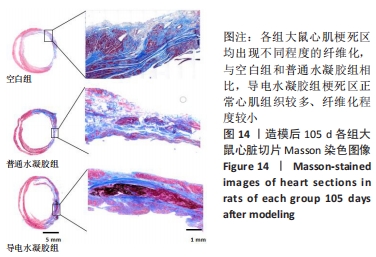
Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (15): 2315-2322.doi: 10.12307/2024.372
Previous Articles Next Articles
Polypyrrole-chitosan conductive composite hydrogel promotes recovery of cardiac function after ischemia-reperfusion injury
Wang Xinzhu1, Wang Qi1, Lang Limin2, He Sheng2
- 1Center for Regenerative Medicine, Shanxi Key Laboratory of Birth Defects and Cell Regeneration, Shanxi Medical University, Taiyuan 030001, Shanxi Province, China; 2Department of Radiology, First Hospital of Shanxi Medical University, Taiyuan 030001, Shanxi Province, China
-
Received:2023-03-02Accepted:2023-06-05Online:2024-05-28Published:2023-09-19 -
Contact:He Sheng, MD, Chief physician, Department of Radiology, First Hospital of Shanxi Medical University, Taiyuan 030001, Shanxi Province, China -
About author:Wang Xinzhu, Master, Center for Regenerative Medicine, Shanxi Key Laboratory of Birth Defects and Cell Regeneration, Shanxi Medical University, Taiyuan 030001, Shanxi Province, China -
Supported by:National Natural Science Foundation of China, No. 81900279 (to HS); China Postdoctoral Science Foundation, No. 2021M691991 (to HS)
CLC Number:
Cite this article
Wang Xinzhu, Wang Qi, Lang Limin, He Sheng. Polypyrrole-chitosan conductive composite hydrogel promotes recovery of cardiac function after ischemia-reperfusion injury[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(15): 2315-2322.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
| [1] HERR DJ, SINGH T, DHAMMU T, et al. Regulation of metabolism by mitochondrial enzyme acetylation in cardiac ischemia-reperfusion injury. Biochim Biophys Acta Mol Basis Dis. 2020;1866(6):165728. [2] MARINESCU MC, LAZAR AL, MARTA MM, et al. Non-Coding RNAs: Prevention, Diagnosis, and Treatment in Myocardial Ischemia-Reperfusion Injury. Int J Mol Sci. 2022;23(5):2728. [3] BRISTOW MR, SAXON LA, BOEHMER J, et al. Cardiac-resynchronization therapy with or without an implantable defibrillator in advanced chronic heart failure. N Engl J Med. 2004;350(21):2140-2150. [4] CLELAND JG, DAUBERT JC, ERDMANN E, et al. The effect of cardiac resynchronization on morbidity and mortality in heart failure. N Engl J Med. 2005;352(15):1539-1549. [5] HE S, WU J, LI SH, et al. The conductive function of biopolymer corrects myocardial scar conduction blockage and resynchronizes contraction to prevent heart failure. Biomaterials. 2020;258:120285. [6] GABETTI S, SILEO A, MONTRONE F, et al. Versatile electrical stimulator for cardiac tissue engineering-Investigation of charge-balanced monophasic and biphasic electrical stimulations. Front Bioeng Biotechnol. 2022;10:1031183. [7] CUI Z, NI NC, WU J, et al. Polypyrrole-chitosan conductive biomaterial synchronizes cardiomyocyte contraction and improves myocardial electrical impulse propagation. Theranostics. 2018;8(10):2752-2764. [8] MASSOUMI B, ABBASIAN M, JAHANBAN-ESFAHLAN R, et al. A novel bio-inspired conductive, biocompatible, and adhesive terpolymer based on polyaniline, polydopamine, and polylactide as scaffolding biomaterial for tissue engineering application. Int J Biol Macromol. 2020;147:1174-1184. [9] LEE M, KIM MC, LEE JY. Nanomaterial-Based Electrically Conductive Hydrogels for Cardiac Tissue Repair. Int J Nanomedicine. 2022;17: 6181-6200. [10] WANJARE M, KAWAMURA M, HU C, et al. Vascularization of Engineered Spatially Patterned Myocardial Tissue Derived From Human Pluripotent Stem Cells in vivo. Front Bioeng Biotechnol. 2019;7:208. [11] BEAUCHAMP P, JACKSON CB, OZHATHIL LC, et al. 3D Co-culture of hiPSC-Derived Cardiomyocytes With Cardiac Fibroblasts Improves Tissue-Like Features of Cardiac Spheroids. Front Mol Biosci. 2020;7:14. [12] LI Y, QIU X. Bioelectricity-coupling patches for repairing impaired myocardium. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2022;14(4):e1787. [13] LI Y, WEI L, LAN L, et al. Conductive biomaterials for cardiac repair: A review. Acta Biomater. 2022;139:157-178. [14] QUE W, HAN C, ZHAO X, et al. An ECG generative model of myocardial infarction. Comput Methods Programs Biomed. 2022;225:107062. [15] FAVERE K, VAN FRAEYENHOVE J, JACOBS G, et al. Cardiac electrophysiology studies in mice via the transjugular route: a comprehensive practical guide. Am J Physiol Heart Circ Physiol. 2022;323(4):H763-h773. [16] 姜增誉,阎长平,李健丁,等.新型导电复合材料聚吡咯壳聚糖对新生大鼠心室肌细胞钙信号传导的影响及可能机制[J].中西医结合心脑血管病杂志,2018,16(12): 1650-1655. [17] KOLETSI D, ILIADI A, TZANETAKIS GN, et al. Cardiovascular Disease and Chronic Endodontic Infection. Is There an Association? A Systematic Review and Meta-Analysis. Int J Environ Res Public Health. 2021;18(17):9111. [18] WANG Y, LI G, YANG L, et al. Development of Innovative Biomaterials and Devices for the Treatment of Cardiovascular Diseases. Adv Mater. 2022;34(46):e2201971. [19] HE M, WANG D, XU Y, et al. Nitric Oxide-Releasing Platforms for Treating Cardiovascular Disease. Pharmaceutics. 2022;14(7):1345. [20] AGGARWAL M, AGGARWAL B, RAO J. Integrative Medicine for Cardiovascular Disease and Prevention. Med Clin North Am. 2017;101(5):895-923. [21] DE GREGORIO C. Physical Training and Cardiac Rehabilitation in Heart Failure Patients. Adv Exp Med Biol. 2018;1067:161-181. [22] 郎丽敏,何生,姜增誉,等.导电复合材料在心肌梗死组织工程治疗领域的应用进展[J].中国组织工程研究,2021,25(22):3584-3590. [23] MCCARTNEY SL, PATEL C, DEL RIO JM. Long-term outcomes and management of the heart transplant recipient. Best Pract Res Clin Anaesthesiol. 2017;31(2):237-248. [24] LI Y, WEI L, LAN L, et al. Conductive biomaterials for cardiac repair: A review. Acta Biomater. 2022;139:157-178. [25] CHO S, DISCHER DE, LEONG KW, et al. Challenges and opportunities for the next generation of cardiovascular tissue engineering. Nat Methods. 2022;19(9):1064-1071. [26] MADONNA R, VAN LAAKE LW, BOTKER HE, et al. ESC Working Group on Cellular Biology of the Heart: position paper for Cardiovascular Research: tissue engineering strategies combined with cell therapies for cardiac repair in ischaemic heart disease and heart failure. Cardiovasc Res. 2019;115(3):488-500. [27] ASHTARI K, NAZARI H, KO H, et al. Electrically conductive nanomaterials for cardiac tissue engineering. Adv Drug Deliv Rev. 2019;144:162-179. [28] PHUTANE P, TELANGE D, AGRAWAL S, et al. Biofunctionalization and Applications of Polymeric Nanofibers in Tissue Engineering and Regenerative Medicine. Polymers (Basel). 2023;15(5):1202. [29] GHOVVATI M, KHARAZIHA M, ARDEHALI R, et al. Recent Advances in Designing Electroconductive Biomaterials for Cardiac Tissue Engineering. Adv Healthc Mater. 2022; 11(13):e2200055. [30] MOND HG, HELLAND JR, STOKES K, et al. The electrode-tissue interface: the revolutionary role of steroid-elution. Pacing Clin Electrophysiol. 2014;37(9):1232-1249. [31] BLACKBURN NJ, SOFRENOVIC T, KURAITIS D, et al. Timing underpins the benefits associated with injectable collagen biomaterial therapy for the treatment of myocardial infarction. Biomaterials. 2015;39:182-192. [32] CUPA J, STREBEL I, BADERTSCHER P, et al. Diagnostic and prognostic value of QRS duration and QTc interval in patients with suspected myocardial infarction. Cardiol J. 2018;25(5):601-610. [33] JASTRZĘBSKI M, MOSKAL P, HUYBRECHTS W, et al. Left bundle branch-optimized cardiac resynchronization therapy (LOT-CRT): Results from an international LBBAP collaborative study group. Heart Rhythm. 2022;19(1):13-21. [34] THEUNS D, VAN BOVEN N, SCHAER BA, et al. Predicting Early Mortality Among Implantable Defibrillator Patients Treated With Cardiac Resynchronization Therapy. J Card Fail. 2019; 25(10):812-818. [35] BEHAR JM, JACKSON T, HYDE E, et al. Optimized Left Ventricular Endocardial Stimulation Is Superior to Optimized Epicardial Stimulation in Ischemic Patients With Poor Response to Cardiac Resynchronization Therapy: A Combined Magnetic Resonance Imaging, Electroanatomic Contact Mapping, and Hemodynamic Study to Target Endocardial Lead Placement. JACC Clin Electrophysiol. 2016;2(7):799-809. [36] MORSINK M, SEVERINO P, LUNA-CERON E, et al. Effects of electrically conductive nano-biomaterials on regulating cardiomyocyte behavior for cardiac repair and regeneration. Acta Biomater. 2022;139:141-156. [37] MOUSAVI A, VAHDAT S, BAHEIRAEI N, et al. Multifunctional Conductive Biomaterials as Promising Platforms for Cardiac Tissue Engineering. ACS Biomater Sci Eng. 2021;7(1):55-82. [38] UL HAQ A, CAROTENUTO F, DE MATTEIS F, et al. Intrinsically Conductive Polymers for Striated Cardiac Muscle Repair. Int J Mol Sci. 2021;22(16):8550. [39] UL HAQ A, CAROTENUTO F, DI NARDO P, et al. Extrinsically Conductive Nanomaterials for Cardiac Tissue Engineering Applications. Micromachines (Basel). 2021;12(8):914. [40] WANG L, LIU Y, YE G, et al. Injectable and conductive cardiac patches repair infarcted myocardium in rats and minipigs. Nat Biomed Eng. 2021;5(10):1157-1173. |
| [1] | Yang Yifeng, Ye Nan, Wang Lin, Guo Shuaicheng, Huang Jian. Signaling pathway of dexmedetomidine against ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1464-1469. |
| [2] | Lou Guo, Zhang Yan, Fu Changxi. Role of endothelial nitric oxide synthase in exercise preconditioning-induced improvement of myocardial ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1283-1288. |
| [3] | Yue Yun, Wang Peipei, Yuan Zhaohe, He Shengcun, Jia Xusheng, Liu Qian, Li Zhantao, Fu Huiling, Song Fei, Jia Menghui. Effects of croton cream on JNK/p38 MAPK signaling pathway and neuronal apoptosis in cerebral ischemia-reperfusion injury rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1186-1192. |
| [4] | Yang Yifeng, Huang Jian, Ye Nan, Wang Lin. Ischemia-reperfusion injury in total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 955-960. |
| [5] | Wang Wu, Fan Xiaolei, Xie Jie, Hu Yihe, Zeng Min. Hydroxyapatite-polyvinyl alcohol/collagen-chitosan-gelatin composite hydrogel for repairing rabbit osteochondral defect [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 682-689. |
| [6] | Zhang Ya, Mu Qiuju, Wang Zilin, Liu Hongjie, Zhu Lili. Hydrogel loaded with platelet-rich plasma promotes wound healing in diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 690-696. |
| [7] | Shen Ziqing, Xia Tian, Shan Yibo, Zhu Ruijun, Wan Haoxin, Ding Hao, Pan Shu, Zhao Jun. Vascularized tracheal substitutes constructed by exosome-load hydrogel-modified 3D printed scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 697-705. |
| [8] | Zhu Liwei, Wang Jiangyue, Bai Ding. Application value of nanocomposite gelatin methacryloyl hydrogels in different bone defect environments [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 753-758. |
| [9] | Chen Xiaofang, Zheng Guoshuang, Li Maoyuan, Yu Weiting. Preparation and application of injectable sodium alginate hydrogels [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 789-794. |
| [10] | Dai Jing, Liu Shasha, Shen Mingjing. Exosome-loaded injectable hydrogel for repairing bone defects around implants [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 347-354. |
| [11] | Gu Mingxi, Wang Changcheng, Tian Fengde, An Ning, Hao Ruihu, Guo Lin. Preparation and in vitro evaluation of a three-dimensional porous cartilage scaffold made of silk fibroin/gelatin/chitosan [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 366-372. |
| [12] | Cao Sheng, Kong Lingwei, Xu Kun, Sun Zhijie. Effect of gelatin methacryloyl hydrogel loaded with salvianolic acid B on intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 380-386. |
| [13] | Bi Yujie, Ma Dujun, Peng Liping, Zhou Ziqiong, Zhao Jing, Zhu Houjun, Zhong Qiuhui, Yang Yuxin. Strategy and significance of Chinese medicine combined with medical hydrogel for disease treatment [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 419-425. |
| [14] | Chen Pinrui, Pei Xibo, Xue Yiyuan. Function and advantages of magnetically responsive hydrogel in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 452-457. |
| [15] | Long Zhirui, Huang Lei, Xiao Fang, Wang Lin, Wang Xiaobei. Characteristics of hydrogel microspheres in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 472-478. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||