Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (15): 2307-2314.doi: 10.12307/2024.362
Previous Articles Next Articles
Moxibustion and reduced graphene oxide/cerium dioxide nanocomposites for repairing infectious wounds
He Wei, Zhou Zheng, Wu Lingling, Wang Kai, Mu Caiyun
- College of Acupuncture and Tuina, Anhui University of Chinese Medicine, Hefei 230000, Anhui Province, China
-
Received:2023-02-09Accepted:2023-05-19Online:2024-05-28Published:2023-09-19 -
Contact:Mu Caiyun, Associate professor engaged by university, College of Acupuncture and Tuina, Anhui University of Chinese Medicine, Hefei 230000, Anhui Province, China -
About author:He Wei, Master candidate, College of Acupuncture and Tuina, Anhui University of Chinese Medicine, Hefei 230000, Anhui Province, China -
Supported by:Natural Science Foundation of Anhui Province, No. 2108085QE204 (to MCY); Natural Science Research Project of Anhui Universities, No. KJ2020A0418 (to MCY)
CLC Number:
Cite this article
He Wei, Zhou Zheng, Wu Lingling, Wang Kai, Mu Caiyun. Moxibustion and reduced graphene oxide/cerium dioxide nanocomposites for repairing infectious wounds[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(15): 2307-2314.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
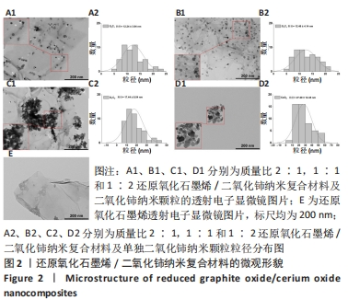
2.1 纳米复合材料的表征结果 如图2所示,通过透射电子显微镜图像观察可到还原氧化石墨烯二维纳米材料呈现铺展的片状结构(图2E),而单独合成的二氧化铈纳米颗粒显示出明显团聚,分散性很差(图2D1),这可能对其物理性质和功能发挥不利[19]。在复合纳米材料中可观察到二氧化铈纳米颗粒成功覆着于还原氧化石墨烯表面(图2A1-C1),并且随着二氧化铈比例的增加,二氧化铈在还原氧化石墨烯上的负载量也相应增加,其中G2C1和G1C1组可见二氧化铈纳米颗粒较均匀附着于还原氧化石墨烯表面,但G1C2组可观察到二氧化铈纳米颗粒出现了较明显的团聚现象,表明还原氧化石墨烯在一定程度上能够缓解二氧化铈纳米颗粒的团聚现象。"
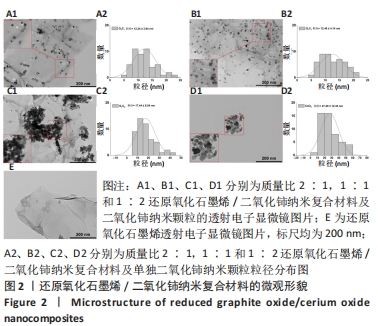
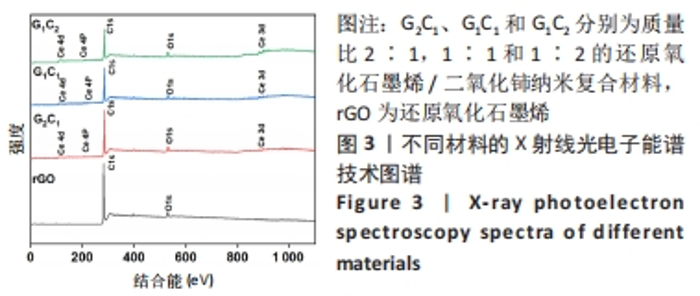
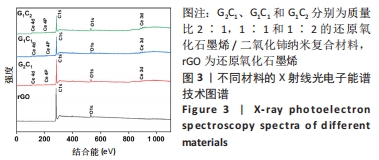
经测量,G2C1、G1C1、G1C2及二氧化铈纳米颗粒二氧化铈颗粒的粒径分别为(12.24±3.94),(12.46±4.14),(17.44±8.54),(27.29±12.45) nm(图2A2-D2)。这也进一步表明在还原氧化石墨烯/二氧化铈复合材料中,还原氧化石墨烯占比越多能够使二氧化铈的颗粒直径更小且更均匀。 利用X射线光电子能谱技术对不同材料表面化学组成进行分析,见图3,可见所有样品均在285 eV与530 eV出现特征峰,这分别归结于C1s和O1s[21],这与石墨烯(以sp2杂化连接的碳原子紧密堆积成单层二维蜂窝状晶格结构)和还原氧化石墨烯/二氧化铈纳米复合材料的组成元素组相符;与还原氧化石墨烯相比,还原氧化石墨烯/二氧化铈纳米复合材料表面出现了铈元素的特征峰(Ce3d、Ce4p和Ce4d),表明还原氧化石墨烯/二氧化铈纳米复合材料的成功合成。此外,表1的样品元素结果显示随着复合材料中二氧化铈质量比的增加,纳米复合材料中的铈元素含量也增加,这与实际相符。"
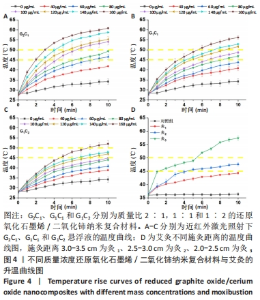
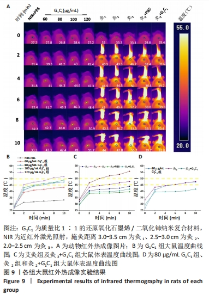
2.2 艾灸与纳米复合材料光热性能检测结果 如图4A-C所示,可以观察到在40-160 μg/mL范围内,G2C1悬浮液的温度分别为41.8,46.6,49.3,54.1,55.2,58.7,60.8 ℃,G1C1悬浮液的温度分别为40.8,43.8,46.8,48.7,51.6,52.9,56.1 ℃,G1C2悬浮液的温度分别38.8,40.8,43.9,44.9,46.9,47.8,52.0 ℃,表明纳米复合材料均具备良好的光热性能,纳米复合材料质量浓度越高,相同时间内的温度增幅越高。 艾灸作为传统疗法之一以温热刺激为显著特征,施灸时皮肤表层产生温通效应,是保证艾灸疗效的重要因素之一[22],其治疗具体操作为:将艾条点燃后对特定皮肤区域施灸,并通过控制艾条与皮肤间距离以达到特定温度,借以产生温通效应。近年研究表明10 min施灸时间能够使艾灸起效[23-24]。除此之外,艾灸疗效的发挥需使其温度达到或超过 45 ℃,因为在 40 ℃以下基本无效,而高热 (> 50 ℃ )会引起正常组织的热损伤和炎症[25-26]。图4D展示了直径为1.2 cm艾条不同施灸距离的升温曲线,可观察到对照组、灸1、灸2和灸3组在10 min时温度分别为36.4,44.3,47.6,57.4 ℃,结果表明灸2组的最终温度囊括于上述艾灸的起效温度,且不会对组织造成损害的温度区间内(45-50 ℃),因此,作者推测灸2组在动物实验中的疗效可能更佳[27]。 "

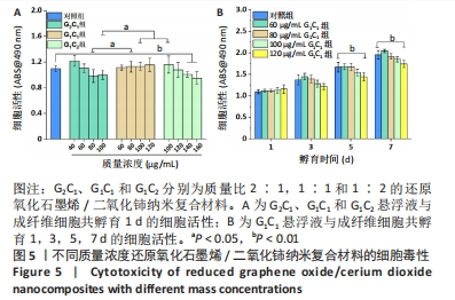
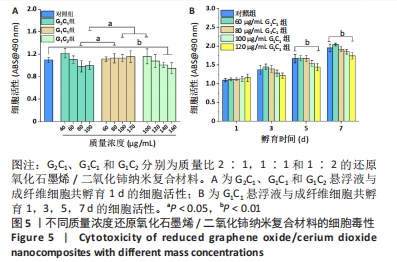
为探究45-50 ℃的温度范围是否有利于创面修复以及纳米复合材料和艾灸对感染性创面修复的积极疗效,同时使其二者疗效具有可比性,不仅选择了温度在45-50 ℃范围内的组别,还分别选择了一组小于45 ℃和一组大于 50 ℃的组别进行实验,设置如下4 组:G2C1(40,60,80,100 μg/mL)、G1C1(60,80,100,120 μg/mL)、G1C2(100,120,140,160 μg/mL)、艾灸组(灸1、灸2和灸3),其中材料组将通过细胞毒性实验和细菌悬浮液实验进一步筛选出一个配比组来进行生物膜清除实验及动物实验。 2.3 纳米复合材料的体外细胞毒性实验结果 如图5A显示,80 μg/mL G1C1组的生物相容性要优于80 μg/mL G2C1组(P < 0.05),100 μg/mL G2C1组和 100 μg/mL G1C2组生物相容性均优于100 μg/mL G2C1 组 (P < 0.05)。以上现象表明随着掺杂二氧化铈的比例增加,纳米复合材料的生物相容性升高,这与文献报道一致[28]。需要注意的是,140 μg/mL G1C2组和160 μg/mL G1C2组相较于对照组表现出轻微的毒副作用(P < 0.01),推测是源于材料质量浓度过高会降低细胞的增殖活力。 图5B中不同质量浓度G1C1组与成纤维细胞共孵育1,3 d后均未见明显毒副作用;共孵育 5,7 d 后,60,80,100 μg/mL G1C1组均表现出优良的生物相容性,直至 120 μg/mL G1C1才显示出轻微的毒副作用 (P < 0.01)。"
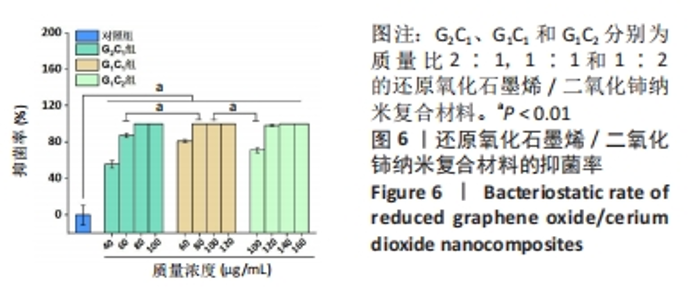
2.4 艾灸与纳米复合材料的体外抗菌性能评价结果 2.4.1 纳米复合材料的体外抗菌性能 图6显示,与对照组相比,不同质量浓度G2C1、G1C1和G1C2对金黄色葡萄球菌均具有明显的抗菌作用(P < 0.01),且随着复合材料质量浓度的增加抑菌率也相应提升,其中,G1C1组表现出优异的抗菌性能,当材料质量浓度为80 μg/mL时,抑菌率已达到100%。进一步观察到,100 μg/mL G1C1组和100 μg/mL G1C2组的抑菌率分别为100%和71.4%,组间比较差异有显著性意义(P < 0.01),二者在808 nm 近红外激光照射10 min时的温度分别为48.7 ℃和44.9 ℃,这表明温度是影响抗菌性能的重要因素。此外,60 μg/mL G2C1组与80 μg/mL G1C1组的抑菌率分别为87.3%和100 %,此时二者在808 nm 近红外激光照射10 min时的温度分别为46.6 ℃和46.8 ℃,显示出在温度相近的情况下,80 μg/mL G1C1组对浮游细菌的抗菌性能比60 μg/mL G2C1组更强(P < 0.01)。"
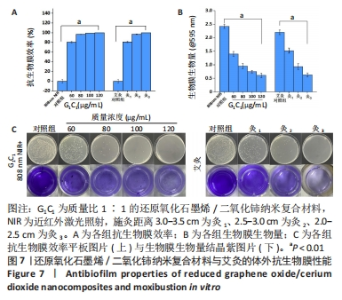
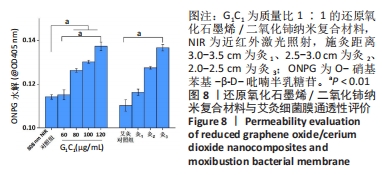
2.4.2 纳米复合材料与艾灸的生物膜清除实验 如图7A所示,相比于对照组,60-120 μg/mL G1C1 具有优良的抗生物膜效率(P < 0.01),且纳米复合材料质量浓度越高抑菌率越高,对应质量浓度纳米复合材料的抑菌率分别为80.6%,96.8%,99.2%和100%。与对照组比较,灸1、灸2和灸3组均能有效地杀死生物膜中的细菌(P < 0.01),且施灸距离越近抗生物膜效率越高,抑菌率分别为81.3%,96.9%和100%。经测量80 μg/mL G1C1组和灸2组在10 min时温度的分别为46.9 ℃和47.6 ℃,在温度相近时,80 μg/mL G1C1组和灸2组也具有十分接近的清除生物膜性能(P > 0.05)。 生物膜清除实验结果见图7B、C。实验结果显示,纳米复合材料组质量浓度越高生物膜清除能力越强,而艾灸组的施灸距离越短生物膜清除能力越强。其进一步的定量分析如图7B所示,80 μg/mL G1C1组与灸2组的吸光度值分别为0.95和0.93,组间比较差异无显著性意义(P > 0.05)。"
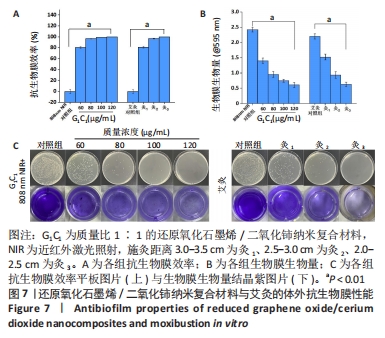
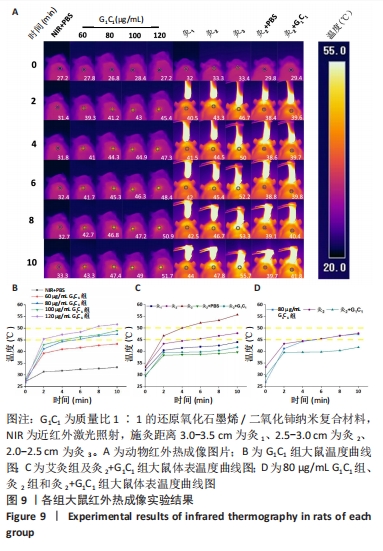
结晶紫染色与O-硝基苯基-β-D-吡喃半乳糖苷水解实验能进一步佐证,G1C1与艾灸组均通过破坏生物膜来加速细菌死亡与生物膜清除,其中80 μg/mL G1C1组和灸2组在温度设定范围内(45-50 ℃),二者对生物膜清除性能相比拟,因此后续实验将重点关注这两组在感染性创面中的修复作用。 2.5 艾灸与纳米复合材料的动物红外热成像结果 图9A为各组动物红外热成像图片。图9B为不同质量浓度G1C1组大鼠体表温度,对照组在处理10 min后温度从27.2 ℃升高到33.3 ℃,这表明近红外激光的单独照射不会明显提高温度,G1C1组温度变化是由复合纳米材料本身的光热性能引发,在G1C1组中纳米复合材料质量浓度越高,照射10 min内温度增幅越高且升温速率也越快。图9C为艾灸各组大鼠体表温度,对于艾灸组,施灸距离越近温度增幅和升温速率也越高;此外,灸2+PBS与灸2+G1C1组在10 min时温度分别为39.7 ℃(艾灸本身的温热效应)和41.8 ℃,作者推测源于艾条燃烧时辐射光谱范围宽,能够轻微的激发材料的光热效应。 值得关注的是,图9D显示,80 μg/mL G1C1组和灸2组处理2,4,10 min时的温度分别为(41.2 ℃和43.3 ℃)、(44.3 ℃和44.5 ℃)和(47.4 ℃和47.8 ℃)。这说明两者在最终温度相近的情况下,艾灸在前4 min的温度均要高于G1C1组,结合图9A推测是因为艾灸热源扩散范围更广和渗透力更强所导致。 2.6 艾灸与纳米复合材料对感染性创面的作用 "
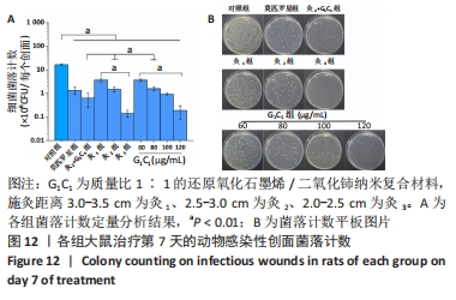
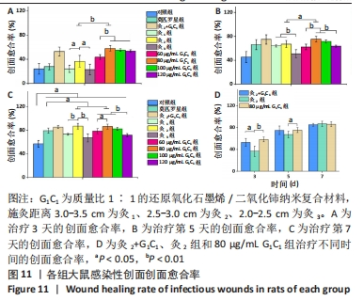
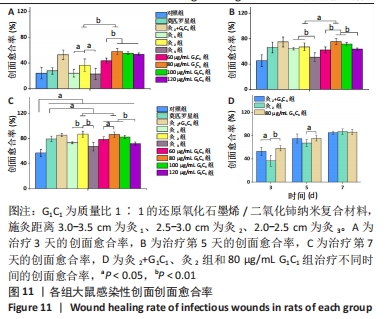
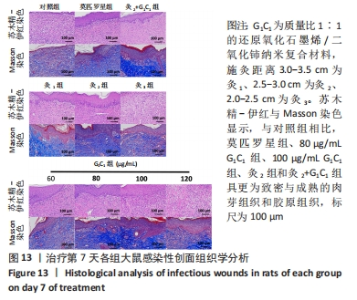
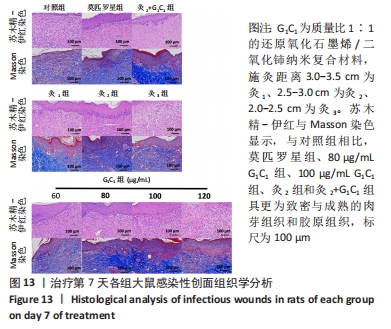
G1C1组组内,80,100 μg/mL是更有利于感染性创面修复的质量浓度。治疗第3天,60 μg/mL组创面愈合率低于其他质量浓度组(P < 0.01)。第5天,60,120 μg/mL组大鼠创面愈合率低于其他质量浓度组(P < 0.01)。第7天,120 μg/mL组大鼠创面愈合率低于其他质量浓度组(P < 0.01),60 μg/mL组大鼠创面愈合率低于80 μg/mL组(P < 0.05),80 μg/mL组和100 μg/mL组大鼠创面愈合率未见明显差异(P > 0.05)。艾灸组组内,治疗第3天,灸2组大鼠创面愈合率高于灸1组、灸3组(P < 0.05);第5天,灸3组大鼠创面愈合率低于灸1组、灸2组(P < 0.01);第7天,灸2组大鼠创面愈合率高于灸1组、灸3组(P < 0.01)。 80 μg/mL G1C1组第3,5天的创面愈合率高于灸2组(P < 0.01,P < 0.05)。灸2+G1C1组大鼠治疗第3天的创面愈合率高于灸2组(P < 0.05),并且与80 μg/mL G1C1组效果接近;第5天,创面愈合率由高到低依次为80 μg/mL G1C1组、灸2+G1C1和灸2组;第7天,3组创面愈合率的组间比较差异无显著性意义(P > 0.05)。 2.6.2 各组大鼠创面菌落计数 如图12所示,与对照组相比,莫匹罗星组、各质量浓度G1C1组、各艾灸组和灸2+G1C1组均能有效清除创面细菌残留(P < 0.01),并且G1C1质量浓度越高细菌残留量越少,艾灸组施灸距离越近细菌残留量越少。除此之外还观察到,材料与艾灸联合后对创面细菌清除能力要优于单独使用(P < 0.01)。"

| [1] 唐娅妮,崔艺敏,何轶帆,等.艾灸热、光、烟作用机制研究进展[J].中国中医药信息志,2022,29(11):148-151. [2] 陈日新.热敏灸:中医灸法的传承、创新与发展[J].中国针灸,2023,43(4):483-488. [3] 黄河,欧阳里知,胡明岸,等.基于生物传热学的艾灸疗法物理学剂量研究[J].世界中医药,2019,14(3):521-526. [4] WU MC, DEOKAR AR, LIAO JH, et al. Graphene-based photothermal agent for rapid and effective killing of bacteria. ACS Nano. 2013;7(2):1281-1290. [5] XIA MY, XIE Y, YU CH, et al. Graphene-based nanomaterials: the promising active agents for antibiotics-independent antibacterial applications. J Controll Release. 2019;307:16-31. [6] TABISH TA, ZHANG S, WINYARD PG. Developing the next generation of graphene-based platforms for cancer therapeutics: The potential role of reactive oxygen species. Redox Biol. 2018;15:34-40. [7] MATTEINI P, TATINI F, CAVIGLI L, et al. Graphene as a photothermal switch for controlled drug release. Nanoscale. 2014;6(14):7947-7953. [8] HUI L, AULETTA JT, HUANG Z, et al. Surface disinfection enabled by a layer-by-layer thin film of polyelectrolyte-stabilized reduced graphene oxide upon solar near-infrared irradiation. ACS Appl Mater Interfaces. 2015;7(19):10511-10517. [9] LIAO C, LI Y, TJONG SC. Graphene nanomaterials: Synthesis, biocompatibility, and cytotoxicity. Int J Mol Sci. 2018;19(11):3564. [10] VUPPALADADIUM SSR, AGARWAL T, KULANTHAIVEL S, et al. Silanization improves biocompatibility of graphene oxide. Mater Sci Eng C Mater Biol Appl. 2020;110:110647. [11] WANG SD, MA Q, WANG K, et al. Improving antibacterial activity and biocompatibility of bioinspired electrospinning silk fibroin nanofibers modified by graphene oxide. ACS Omega. 2018;3(1):406-413. [12] MA S, LU Y, ZHU X, et al. Efficient Modulation of Electron Pathways by Constructing a MnO2–x@ CeO2 Interface toward Advanced Lithium–Oxygen Batteries. ACS Appl Mater Interfaces. 2022;14(19):22104-22113. [13] CHENG X, ZHANG X, SU D, et al. NO reduction by CO over copper catalyst supported on mixed CeO2 and Fe2O3: Catalyst design and activity test. Appl Catal B-Environ. 2018;239:485-501. [14] EMA T, CHOI PG, TAKAMI S, et al. Facet-Controlled Synthesis of CeO2 Nanoparticles for High-Performance CeO2 Nanoparticle/SnO2 Nanosheet Hybrid Gas Sensors. ACS Appl Mater Interfaces. 2022;14(51):56998-57007. [15] WANG Y, HUANG Y, FU Y, et al. Reductive damage induced autophagy inhibition for tumor therapy. Nano Res. 2023;16(4):5226-5236. [16] ZHENG H, WANG S, CHENG F, et al. Bioactive anti-inflammatory, antibacterial, conductive multifunctional scaffold based on MXene@ CeO2 nanocomposites for infection-impaired skin multimodal therapy. Chem EngJ. 2021;424:130148. [17] ALSHARIF NB, SAMU GF, SÁRINGER S, et al. Antioxidant colloids via heteroaggregation of cerium oxide nanoparticles and latex beads. Colloids Surf B Biointerfaces. 2022;216: 112531. [18] SHIN SR, AGHAEI-GHAREH-BOLAGH B, DANG TT, et al. Cell-laden microengineered and mechanically tunable hybrid hydrogels of gelatin and graphene oxide. Adv Mater. 2013; 25(44):6385-6391. [19] SRIVASTAVA M, DAS AK, KHANRA P, et al. Characterizations of in situ grown ceria nanoparticles on reduced graphene oxide as a catalyst for the electrooxidation of hydrazine. J Mater Chem A Mater. 2013;1(34):9792-9801. [20] ZHAO H, HUANG J, LI Y, et al. ROS-scavenging hydrogel to promote healing of bacteria infected diabetic wounds. Biomaterials. 2020;258:120286. [21] TAO B, SHEN X, YUAN Z, et al. N-halamine-based multilayers on titanium substrates for antibacterial application. Colloids Surf B Biointerfaces. 2018;170:382-392. [22] 成鸿群,刘宜成,徐金鹏,等.温和灸和雀啄灸温度场分布特点的实验研究[J].中国针灸,2021,41(10):1113-1117. [23] 李松梅,赵彦春,赵娟,等.不同艾灸灸距和时长对糖尿病周围神经病变的疗效与安全性研究[J].北京中医药,2023,42(2):143-145. [24] 毛珍,江润,段骏,等.不同灸量热敏灸治疗膝骨关节炎临床研究[J].针灸临床杂志,2022,38(10):52-56. [25] 朱兵 . 关于灸材和灸温的思考 [J]. 针刺研究 ,2018,43(2):63-67. [26] SONG Y, WANG Y, ZHU Y, et al. Biomodal tumor‐targeted and redox‐responsive Bi2Se3 hollow nanocubes for MSOT/CT imaging guided synergistic low‐temperature photothermal radiotherapy. Adv Healthc Mater. 2019;8(16):1900250. [27] 刘磊,王敏君,吴立斌,等.不同艾条直径和施灸距离对大鼠穴区皮肤表面温度的影响[J].针刺研究,2020,45(5):396-401. [28] LIAO C, LI Y, TJONG SC. Graphene nanomaterials: Synthesis, biocompatibility, and cytotoxicity. Int J Mol Sci. 2018;19(11): 3564. [29] 王永灵,蒉纲.艾灸疗法治疗外科创面的研究进展[J].湖北中医药大学学报,2012, 14(1):73-74. [30] 张雪琳,高崚,陈新旺,等.艾烟的化学成分及药理活性研究进展[J].中华中医药杂志,2022,37(8):4560-4564. [31] 张帆,吴立斌,刘磊,等.艾灸烟热效应促进大鼠外伤创面愈合及组织修复的研究[J].针刺研究,2021,46(7):575-579+585. [32] XU C, PU K. Second near-infrared photothermal materials for combinational nanotheranostics. Chem Soc Rev. 2021;50(2):1111-1137. [33] MEI L, ZHU S, YIN W, et al. Two-dimensional nanomaterials beyond graphene for antibacterial applications: current progress and future perspectives. Theranostics. 2020;10(2):757. [34] WANG Y, LI J, LI X, et al. Graphene-based nanomaterials for cancer therapy and anti-infections. Bioact Mater. 2022;14:335-349. [35] XIA MY, XIE Y, YU CH, et al. Graphene-based nanomaterials: the promising active agents for antibiotics-independent antibacterial applications. J Controll Release. 2019;307:16-31. [36] LUO M, SHAITAN K, QU X, et al. Bioactive rare earth-based inorganic-organic hybrid biomaterials for wound healing and repair. Appl Mater Today. 2022;26:101304. |
| [1] | Li Wenlan, Wang Wenyuan, Ren Wenxiu, Zhang Yupei, Yang Xiaoyan, Wang Zhigang, Xia Jizhu. Preparation of a near-infrared photoresponsive biomimetic nanoprobe and its application in photothermal detection and treatment of breast cancer [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 669-675. |
| [2] | Zhang Ya, Mu Qiuju, Wang Zilin, Liu Hongjie, Zhu Lili. Hydrogel loaded with platelet-rich plasma promotes wound healing in diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 690-696. |
| [3] | Li Chun, Zhang Yanlin, Liu Di, Wang Minglei, Wang Duo, Liu Junwei, Wu Yongli. Effect of warm-needling moxibustion on anterior cruciate ligament injury repair and related growth factors in rabbits with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(23): 3621-3626. |
| [4] | Zhu Yongzhao, Fang Chao, Zhao Fang, Zhang Qing, Zhao Dan. Mechanism by which lycium barbarum polysaccharides inhibit keratinocyte apoptosis in burn wounds via autophagy [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(23): 3686-3691. |
| [5] | Shi Jiao, Li Xingjie, Liu Qiqi, Liu Jun, Yuan Xu, Chen Shangjie. Effect of electronic moxibustion on the volume of hippocampal subregion in patients with amnestic mild cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(20): 3176-3181. |
| [6] | Xiao Ziteng, Wang Tingyu, Zhang Wenwen, Tan Fengyi, Su Haiwei, Li Siting, Wu Yahui, Zhou Yanfang, Peng Xinsheng. Exosomes and skin wound healing [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(19): 3104-3110. |
| [7] | Wang Jinlei, Li Ke, Zhao Liang. Platelet-camouflaged silver nanoparticle hydrogel accelerates wound healing in type 1 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(17): 2659-2666. |
| [8] | Tang Ziyan, Gu Shunqiang, Chen Xiaoling, Wang Lei, Ma Chengbang, Zhou Mei, Chen Tianbao, Du Lina, Jin Yiguang. Recombinant expression and in vitro activity identification of a bioactive peptide QUB2984 from skin secretion of Agalychnis callidryas [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(17): 2675-2681. |
| [9] | Gao Li, Liu Liu, Ren Wenyan, Liu Xue, Wang Yiyu. Influential mechanism of graphene and its derivatives on angiogenesis and vascularized bone [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(17): 2716-2722. |
| [10] | Wang Xindong, Liang Chengzhi, Zhang Yongxian. Functional characteristics and clinical applications of MXene nanoparticles in wound healing [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(17): 2739-2746. |
| [11] | Bian Zhihong, Zhang Yuntao, Li Zeming, Hou Yudong. Potential of shikonin and its derivatives in oral soft and hard tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(17): 2747-2752. |
| [12] | Jiang Jie, Liu Juan, Yu Yanyan, Yang Yue, Wang Qianhui. Mechanism by which moxibustion pretreatment attenuates oxidative stress injury in a rat model of cerebral ischemia-reperfusion [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(16): 2488-2493. |
| [13] | Zhai Yunhao, Qian Yun. Research and application of carbon nanomaterials in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(15): 2423-2429. |
| [14] | Dong Hongfei, Huang Xi, Li Xianhui, Zhang Yanbiao, Wang Xuyang, Wang Bing, Sun Hongyu. Placenta-derived mesenchymal stem cells in promoting acute skin wound healing in rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(13): 2047-2053. |
| [15] | Zhao Lixin, Tang Qingxi, Zhang Kezhong. Influence of adipose tissue and its derivative on wound repair and vascularization [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(13): 2120-2125. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||