Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (10): 1497-1504.doi: 10.12307/2024.374
Previous Articles Next Articles
Various arginine configurations-modified chitosan hydrogels promote skin wound repair
Deng Jing, Li Tinghua, Zhu Hai, Yang Xiao, Cao Jun, Zhu Xiangdong
- College of Biomedical Engineering, Sichuan University, Chengdu 610064, Sichuan Province, China
-
Received:2023-05-13Accepted:2023-06-15Online:2024-04-08Published:2023-08-17 -
Contact:Zhu Xiangdong, Researcher, College of Biomedical Engineering, Sichuan University, Chengdu 610064, Sichuan Province, China -
About author:Deng Jing, Master candidate, College of Biomedical Engineering, Sichuan University, Chengdu 610064, Sichuan Province, China -
Supported by:Science and Technology Plan Project of Sichuan Province (Key Research and Development Project), No. 2020YFS0038 (to YX)
CLC Number:
Cite this article
Deng Jing, Li Tinghua, Zhu Hai, Yang Xiao, Cao Jun, Zhu Xiangdong. Various arginine configurations-modified chitosan hydrogels promote skin wound repair[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(10): 1497-1504.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
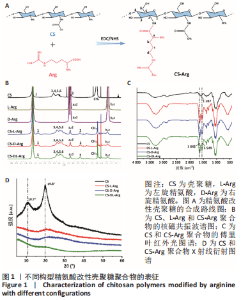
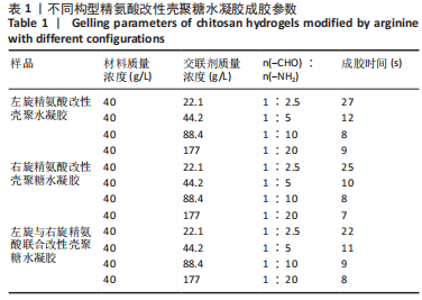
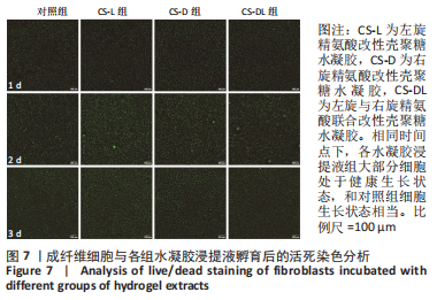
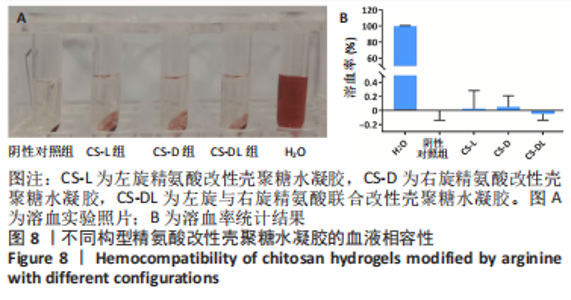
2.1 壳聚糖-精氨酸聚合物的合成及表征结果 如图1A所示,不同构型精氨酸可通过酰胺化反应键合到壳聚糖的侧链。核磁氢谱结果显示,壳聚糖-精氨酸聚合物中不仅出现了壳聚糖的特征吸收峰(δ=2.0×10-6),还在δ=1.5×10-6以及δ=3.0×10-6-3.2×10-6处均出现了精氨酸的特征吸收峰,以上结果说明精氨酸成功接枝到壳聚糖链上,见图1B。进一步,通过将精氨酸在a和d处-CH2特征吸收峰面积和壳聚糖的-CH3特征吸收峰面积进行比较,可以计算得到精氨酸的接枝率。CS-L-Arg、CS-D-Arg和CS-DL-Arg的接枝率分别为8.7%,9.2%和6.4%。 红外图谱结果如图1C所示,壳聚糖-精氨酸聚合物在 1 642 cm-1和1 549 cm-1 2个位置出现了新的吸收峰,分别是酰胺Ⅰ带C=O的伸缩振动吸收峰和酰胺Ⅱ带C-N伸缩振动和N-H变形振动的组合,说明了有新的酰胺键形成。同时,1 587 cm-1左右的N-H 弯曲振动峰被新生成的酰胺Ⅱ带覆盖,进一步证明了壳聚糖聚合物中的氨基和精氨酸分子中的羧基成功进行了酰胺化反应。 X射线衍射分析结果如图1D所示,未改性壳聚糖在衍射角为10.7°和19.8°处有2个明显的衍射峰,改性后壳聚糖的X衍射图谱有了明显改变,壳聚糖在10.7°处的衍射峰几乎完全消失,在19.8°处的衍射峰位置和强度都有变化,衍射角向大角度方向略有偏移,同时峰强度显著降低,这表明精氨酸的引入明显降低了壳聚糖的结晶度。"

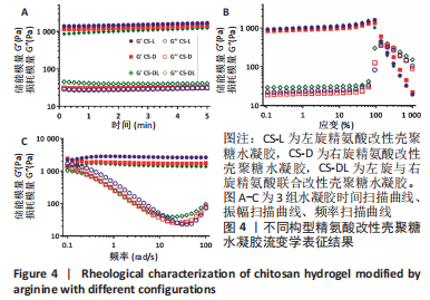
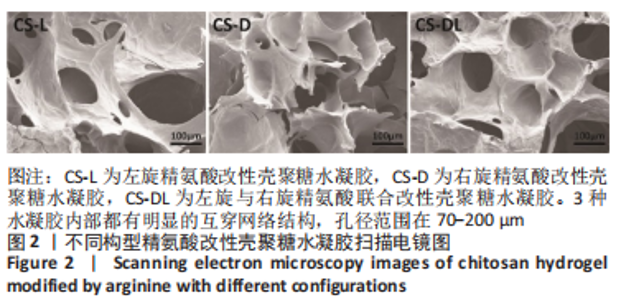
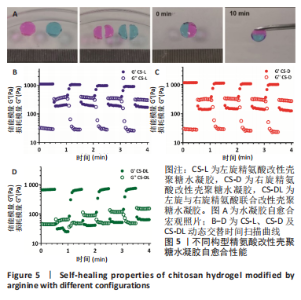
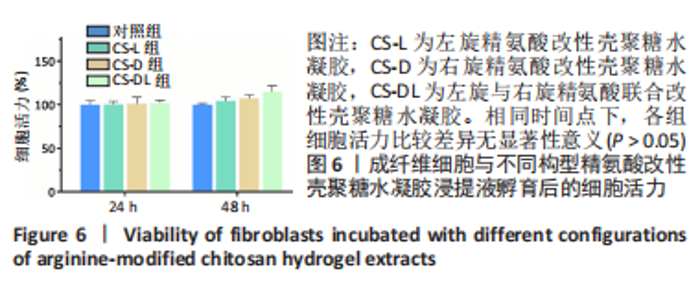
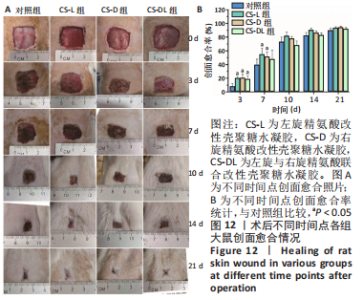
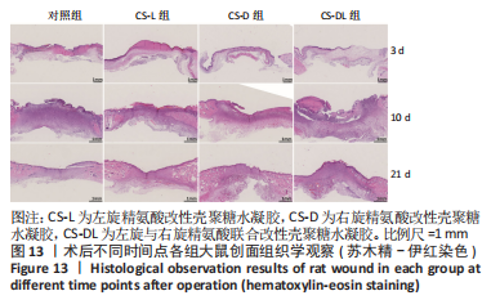
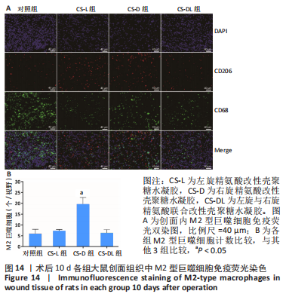
进一步考察3组水凝胶的流变学特征,结果如图4所示。由时间扫描曲线可知,3组水凝胶的储能模量(G’)均大于损耗模量(G”),表现为黏弹性固体行为特性,说明成功构建了水凝胶。同时,测量时间内3组水凝胶的储能模量和损耗模量基本保持稳定,G’约为1 000 Pa,G”为200-300 Pa。由振幅扫描曲线可知,在低应变区间(70%)以下时,3组水凝胶的G’和G”都保持稳定,此区间为水凝胶的线性黏弹区。当应变大于70%时,随着应变的增加,水凝胶的G’逐渐变小,G”逐渐变大,直至出现两条曲线相交,超过这个临界点后G’ < G”,说明水凝胶的结构已经被破坏,由黏弹性固体转变为液体状态。CS-L、CS-D、CS-DL水凝胶的转变临界值分别为158%,193%和132%。由频率扫描曲线可知,无论是在高频或者低频范围内,3组水凝胶的G’都始终大于G”,表明水凝胶形态一直维持,具有较好的稳定性。"

| [1] KOEHLER J, BRANDL FP, GOEPFERICH AM. Hydrogel wound dressings for bioactive treatment of acute and chronic wounds. Eur Polym J. 2018;100:1-11. [2] WINTER GD. Formation of the scab and the rate of epithelization of superficial wounds in the skin of the young domestic pig. Nature. 1962;193:293-294. [3] DONG RN, GUO BL. Smart wound dressings for wound healing. Nano Today. 2021;41:22. [4] GHOMI ER, KHALILI S, KHORASANI SN, et al. Wound dressings: Current advances and future directions. J Appl Polym Sci. 2019;136(27):12. [5] FARAHANI M, SHAFIEE A. Wound Healing: From Passive to Smart Dressings. Adv Healthc Mater. 2021;10(16):32. [6] GALLASTEGUI A, SPESIA MB, DELL’ERBA IE, et al. Controlled release of antibiotics from photopolymerized hydrogels: Kinetics and microbiological studies. Mater Sci Eng C-Mater Biol Appl. 2019;102:896-905. [7] SU JJ, LI JK, LIANG JH, et al. Hydrogel Preparation Methods and Biomaterials for Wound Dressing. Life-Basel. 2021;11(10):22. [8] LIANG YP, HE JH, GUO BL. Functional Hydrogels as Wound Dressing to Enhance Wound Healing. ACS Nano. 2021;15(8):12687-12722. [9] CHEN SJ, TIAN HR, MAO JL, et al. Preparation and application of chitosan-based medical electrospun nanofibers. Int J Biol Macromol. 2023;226:410-422. [10] TORKAMAN S, RAHMANI H, ASHORI A, et al. Modification of chitosan using amino acids for wound healing purposes: A review. Carbohydr Polym. 2021;258:8. [11] QU J, ZHAO X, LIANG YP, et al. Antibacterial adhesive injectable hydrogels with rapid self-healing, extensibility and compressibility as wound dressing for joints skin wound healing. Biomaterials. 2018;183:185-199. [12] LI H, ZHOU XJ, LUO L, et al. Bio-orthogonally crosslinked catechol-chitosan hydrogel for effective hemostasis and wound healing. Carbohydr Polym. 2022;281:11. [13] HRISTINA K, LANGERHOLC T, TRAPECAR M. Novel metabolic roles of L-arginine in body energy metabolism and possible clinical applications. J Nutr Health Aging. 2014;18(2):213-218. [14] HUSSEIN Y, EL-FAKHARANY EM, KAMOUN EA, et al. Electrospun PVA/hyaluronic acid/L-arginine nanofibers for wound healing applications: Nanofibers optimization and in vitro bioevaluation. Int J Biol Macromol. 2020;164:667-676. [15] YU J, ZHANG RL, CHEN BH, et al. Injectable Reactive Oxygen Species-Responsive Hydrogel Dressing with Sustained Nitric Oxide Release for Bacterial Ablation and Wound Healing. Adv Funct Mater. 2022;32(33):11. [16] PELUFFO RD. L-Arginine currents in rat cardiac ventricular myocytes. J Physiol-London. 2007; 580(3):925-936. [17] LIM HK, RAHIM AB, LEO VI, et al. Polyamine Regulator AMD1 Promotes Cell Migration in Epidermal Wound Healing. J Invest Dermatol. 2018;138(12):2653-2665. [18] ITO D, ITO H, IDETA T, et al. Systemic and topical administration of spermidine accelerates skin wound healing. Cell Commun Signal. 2021;19(1):12. [19] LING ZX, DENG J, ZHANG ZR, et al. Spatiotemporal manipulation of L-arginine release from bioactive hydrogels initiates rapid skin wound healing accompanied with repressed scar formation. Appl Mater Today. 2021;24:16. [20] MARIANI E, LISIGNOLI G, BORZI RM, et al. Biomaterials: Foreign Bodies or Tuners for the Immune Response? Int J Mol Sci. 2019;20(3):42. [21] VISHWAKARMA A, BHISE NS, EVANGELISTA MB, et al. Engineering Immunomodulatory Biomaterials To Tune the Inflammatory Response. Trends Biotechnol. 2016;34(6): 470-482. [22] JULIER Z, PARK AJ, BRIQUEZ PS, et al. Promoting tissue regeneration by modulating the immune system. Acta Biomater. 2017;53:13-28. [23] LYNCH RI, LAVELLE EC. Immuno-modulatory biomaterials as anti-inflammatory therapeutics. Biochem Pharmacol. 2022;197:17. [24] WYNN TA, VANNELLA KM. Macrophages in Tissue Repair, Regeneration, and Fibrosis. Immunity. 2016;44(3):450-462. [25] QIAN YN, ZHENG YJ, JIN J, et al. Immunoregulation in Diabetic Wound Repair with a Photoenhanced Glycyrrhizic Acid Hydrogel Scaffold. Adv Mater. 2022;34(29):15. [26] GREEN DW, LEE JM, KIM EJ, et al. Chiral Biomaterials: From Molecular Design to Regenerative Medicine. Adv Mater Interfaces. 2016;3(6):13. [27] DUAN YY, ZHENG HH, LI ZH, et al. Unsaturated polyurethane films grafted with enantiomeric polylysine promotes macrophage polarization to a M2 phenotype through PI3K/Akt1/mTOR axis. Biomaterials. 2020;246:14. [28] GRIFFIN DR, ARCHANG MM, KUAN CH, et al. Activating an adaptive immune response from a hydrogel scaffold imparts regenerative wound healing. Nat Mater. 2021;20(4):560-569. [29] ZHU JW, TIAN J, YANG C, et al. L-Arg-Rich Amphiphilic Dendritic Peptide as a Versatile NO Donor for NO/Photodynamic Synergistic Treatment of Bacterial Infections and Promoting Wound Healing. Small. 2021;17(32):18. [30] WILKINSON HN, HARDMAN MJ. Wound healing: cellular mechanisms and pathological outcomes. Open Biol. 2020;10(9):14. [31] RAZIYEVA K, KIM Y, ZHARKINBEKOV Z, et al. Immunology of Acute and Chronic Wound Healing. Biomolecules. 2021;11(5):25. [32] HE YX, LI Y, SUN YD, et al. A double-network polysaccharide-based composite hydrogel for skin wound healing. Carbohydr Polym. 2021;261:11. [33] ZHANG M, YANG M, WOO MW, et al. High-mechanical strength carboxymethyl chitosan-based hydrogel film for antibacterial wound dressing. Carbohydr Polym. 2021;256:9. [34] ZOU FX, WANG YS, ZHENG YD, et al. A novel bioactive polyurethane with controlled degradation and L-Arg release used as strong adhesive tissue patch for hemostasis and promoting wound healing. Bioact Mater. 2022;17:471-487. [35] AHMED R, AUGUSTINE R, CHAUDHRY M, et al. Nitric oxide-releasing biomaterials for promoting wound healing in impaired diabetic wounds: State of the art and recent trends. Biomed Pharmacother. 2022;149:12707. [36] FENG ZJ, SU Q, ZHANG CN, et al. Bioinspired Nanofibrous Glycopeptide Hydrogel Dressing for Accelerating Wound Healing: A Cytokine-Free, M2-Type Macrophage Polarization Approach. Adv Funct Mater. 2020;30(52):13. [37] EMING SA, KRIEG T, DAVIDSON JM. Inflammation in wound repair: Molecular and cellular mechanisms. J Invest Dermatol. 2007;127(3):514-525. [38] MAO JY, CHEN L, CAI ZW, et al. Advanced Biomaterials for Regulating Polarization of Macrophages in Wound Healing. Adv Funct Mater. 2022;32(12):25. [39] LIU GW, MA HX, QIU L, et al. Phenotypic and functional switch of macrophages induced by regulatory CD4(+)CD25(+) T cells in mice. Immunol Cell Biol. 2011;89(1):130-142. [40] ZHANG W, XIA SZ, WENG TT, et al. Antibacterial coaxial hydro-membranes accelerate diabetic wound healing by tuning surface immunomodulatory functions. Mater Today Bio. 2022;16:18. [41] YUAN Y, FAN DD, SHEN SH, et al. An M2 macrophage-polarized anti-inflammatory hydrogel combined with mild heat stimulation for regulating chronic inflammation and impaired angiogenesis of diabetic wounds. Chem Eng J. 2022;433:18. |
| [1] | Wang Weiqing, Zhou Yue. Chronic inflammation regulates adipose tissue fibrosis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1307-1312. |
| [2] | Zeng Fanzhuo, Li Yuxin, Sun Jiachen, Gu Xinyang, Wen Shan, Tian He, Mei Xifan. Efficient strategies for microglia replacement in spinal cord injury models [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1007-1014. |
| [3] | Chen Xiaofang, Zheng Guoshuang, Li Maoyuan, Yu Weiting. Preparation and application of injectable sodium alginate hydrogels [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 789-794. |
| [4] | Wang Wu, Fan Xiaolei, Xie Jie, Hu Yihe, Zeng Min. Hydroxyapatite-polyvinyl alcohol/collagen-chitosan-gelatin composite hydrogel for repairing rabbit osteochondral defect [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 682-689. |
| [5] | Zhang Ya, Mu Qiuju, Wang Zilin, Liu Hongjie, Zhu Lili. Hydrogel loaded with platelet-rich plasma promotes wound healing in diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 690-696. |
| [6] | Shen Ziqing, Xia Tian, Shan Yibo, Zhu Ruijun, Wan Haoxin, Ding Hao, Pan Shu, Zhao Jun. Vascularized tracheal substitutes constructed by exosome-load hydrogel-modified 3D printed scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 697-705. |
| [7] | Zhu Liwei, Wang Jiangyue, Bai Ding. Application value of nanocomposite gelatin methacryloyl hydrogels in different bone defect environments [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 753-758. |
| [8] | Yang Yuqing, Chen Zhiyu. Role and application of early transient presence of M1 macrophages in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 594-601. |
| [9] | Xing Hao, Meng Qingfeng, Chang Zhengqi. Mechanism of negative pressure wound therapy in the auxiliary treatment of bone and soft tissue infection [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 621-626. |
| [10] | Dai Jing, Liu Shasha, Shen Mingjing. Exosome-loaded injectable hydrogel for repairing bone defects around implants [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 347-354. |
| [11] | Gu Mingxi, Wang Changcheng, Tian Fengde, An Ning, Hao Ruihu, Guo Lin. Preparation and in vitro evaluation of a three-dimensional porous cartilage scaffold made of silk fibroin/gelatin/chitosan [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 366-372. |
| [12] | Cao Sheng, Kong Lingwei, Xu Kun, Sun Zhijie. Effect of gelatin methacryloyl hydrogel loaded with salvianolic acid B on intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 380-386. |
| [13] | Bi Yujie, Ma Dujun, Peng Liping, Zhou Ziqiong, Zhao Jing, Zhu Houjun, Zhong Qiuhui, Yang Yuxin. Strategy and significance of Chinese medicine combined with medical hydrogel for disease treatment [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 419-425. |
| [14] | Chen Pinrui, Pei Xibo, Xue Yiyuan. Function and advantages of magnetically responsive hydrogel in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 452-457. |
| [15] | Long Zhirui, Huang Lei, Xiao Fang, Wang Lin, Wang Xiaobei. Characteristics of hydrogel microspheres in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 472-478. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||