Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (21): 3423-3429.doi: 10.12307/2023.457
Previous Articles Next Articles
Bioactive scaffolds in repairing osteoporotic bone defects
Hou Jianming, Li Qi
- Department of Dental Implantology, Affiliated Hospital of Inner Mongolia Medical University, Hohhot 010030, Inner Mongolia Autonomous Region, China
-
Received:2022-04-29Accepted:2022-07-25Online:2023-07-28Published:2022-11-26 -
Contact:Li Qi, Associate chief physician, Associate professor, Master’s supervisor, Department of Dental Implantology, Affiliated Hospital of Inner Mongolia Medical University, Hohhot 010030, Inner Mongolia Autonomous Region, China -
About author:Hou Jianming, Master candidate, Department of Dental Implantology, Affiliated Hospital of Inner Mongolia Medical University, Hohhot 010030, Inner Mongolia Autonomous Region, China
CLC Number:
Cite this article
Hou Jianming, Li Qi. Bioactive scaffolds in repairing osteoporotic bone defects[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(21): 3423-3429.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

2.1 理想支架的特性 制作支架通常从材料的选择开始,支架材料的特性对支架功能的发挥有着至关重要的影响,理想的支架应具备下列特性:①良好的机械性能,可以使支架承受正常的运动负荷;②合适的形态,支架的形态影响细胞与支架的相互作用,如多孔的支架可以为成骨细胞和营养物质提供更多面积的附着网络;③良好的生物相容性,支架在植入体内时不应引发免疫反应;④良好的生物活性,指支架材料可以与宿主组织相互作用和结合,支架材料具有骨传导和骨诱导特性,包含生物信号和生长因子以刺激细胞向内生长、附着和分化;⑤良好的生物降解性,支架经酶促或水解作用被生物降解,且支架的降解速率应与体内成骨速率相协调[15-17]。 2.2 支架设计 植入骨组织工程支架已经成为修复骨缺损的重要方法,根据理想支架的特性制备复合材料支架,在修复骨缺损中取得了良好的效果。但是,骨质疏松的骨改建不平衡使得骨再生变得困难,骨微结构受损使得支架常出现移位、松动。近年来,研究者对生物活性支架修复骨质疏松性骨缺损进行了研究,如何改善不平衡的骨改建和增强支架在骨缺损处的骨整合是关键[18-20]。除根据理想支架的特性设计制备支架外,在支架设计中局部递送设计具有重要作用,将支架作为缓释平台,通过局部释放生物活性物质到骨缺损处以促进骨再生。另外,围绕局部递送设计的结构设计和表面设计也受到广泛关注,通过结构设计控制材料的孔隙、比表面积和孔的连通性,以增强局部递送效率和骨整合性能,也可以在一定程度上克服材料的缺点。通过表面修饰设计对有机关能团进行修饰或经过材料之间的交联等,也可达到对生物活性物质的缓慢释放。 2.2.1 局部递送设计 负载生物活性治疗剂(离子、维生素、药物)的生物活性支架可成为良好的局部递送系统,生物治疗剂经支架递送在骨缺损处被局部缓慢释放,这样的递送方式和生物活性支架协同促进骨缺损的修复。另一方面,全身给药存在生物利用度低、疗效低等缺点,局部递送设计可以将生物治疗剂递送到局部,避免全身用药。锶是人体骨骼中必需的微量元素,但是摄入过量的锶可能会对人体器官产生细胞毒性,因此通过局部递送设计控制锶的释放速率对于骨组织工程至关重要。锶修饰的支架可以作为锶被持续释放的平台,以最大限度地减少细胞毒性。MIAO等[21]制备了由掺锶硫酸钙微球和掺锶磷酸三钙微球组成的具有多级可控降解特性的支架,体外研究表明这种复合支架显著促进了成骨细胞的增殖和分化,具有良好的成骨诱导活性。骨质疏松性骨缺损修复的实验结果表明,当支架降解率与新骨生长速度相匹配时,骨密度、骨体积/总体积比、骨小梁厚度等参数和新骨形成标志物骨钙素的表达较高,即促进了骨质疏松性骨缺损的快速修复;相反,支架降解速度慢,在一定程度上阻碍了新骨的生长。而且体内降解结果表明,通过控制掺锶硫酸三钙微球的孔隙率和相组成,可以在较大范围(6-12个月)内控制复合支架的降解时间。局部递送设计在修复骨质疏松性骨缺损的研究中被广泛应用,而如何可控、缓慢地释放生物活性物质是其中的研究热点。通过局部递送生物活性物质可以有效改善骨改建,促进骨再生。 2.2.2 材料结构设计 目前,对于修复骨质疏松性骨缺损的生物活性支架,材料设计的主要方向是改善支架材料的物理和化学性质以及生物活性[22]。LIU等[23]将硫酸钙注射到骨质疏松羊椎骨中,为期3个月的观察表明硫酸钙显著促进了骨质疏松羊的骨再生。此外,YANG等[24]将含锶的硫酸钙植入骨质疏松大鼠体内,体内结果表明含锶的硫酸钙具有良好的成骨能力,并促进了去卵巢大鼠颅骨缺损的新血管形成。然而,这些修复材料缺乏天然骨的大孔形态结构和足够的生物活性。近年来,对于支架材料的结构设计受到重视,尤其是在骨质疏松性骨缺损中,经材料结构设计控制支架材料的形态和孔隙,可以在一定程度上克服材料的缺点,并且实现对生物活性物质的缓慢释放。随着化工技术的发展,许多技术已应用于骨组织工程中,其中3D 打印技术已经得到广泛应用,3D 打印的多孔支架可以在宏观和微观结构上模仿人体骨组织的多维结构[25]。并且,通过 3D 打印技术而制备的支架是出色的运载工具,这类支架使药物和生物分子在骨缺损处持续、缓慢释放[26]。而且,传统支架在制作过程中很难完全符合骨缺损形状和获得均匀一致的孔径及孔隙率,3D打印技术可以优化传统的骨替代材料,控制支架的孔隙率和孔径连通性,有利于成骨相关细胞的增殖,也使支架易于加工成不同的物理结构,因此,3D打印技术在修复骨质疏松性骨缺损中得到广泛应用。将含骨髓间充质干细胞和骨形态发生蛋白2的双载超分子水凝胶注入3D打印的多孔钛合金支架内,经实验证明这种生物活性复合支架在骨质疏松性骨缺损中的骨整合得到增强,而且该支架使得骨形态发生蛋白2被持续释放[27]。此外,WANG等[28]通过结构设计将生物活性玻璃制作为纳米纤维结构,该结构模仿了细胞外基质的纳米纤维结构,而且该纤维具有出色的柔韧性,甚至可以进行180°弯曲;然后将纳米纤维进一步组装成3D纤维支架,并且通过均质和冻干过程将天然聚合物壳聚糖包裹和黏合于支架上,由此制得一种弹性纤维复合支架,该弹性纤维复合支架能够适当变形并适应不规则形状的骨缺损,当应用于修复骨质疏松大鼠模型中的颅骨缺损时,该复合支架显著促进了骨缺损模型的骨再生和血管形成。通过材料结构设计克服支架材料的缺点,并且与局部递送设计结合,从而使支架在骨缺损处可控地释放生物治疗剂,以此改善不平衡的骨改建和增强支架在骨缺损处的骨整合(图3)。"
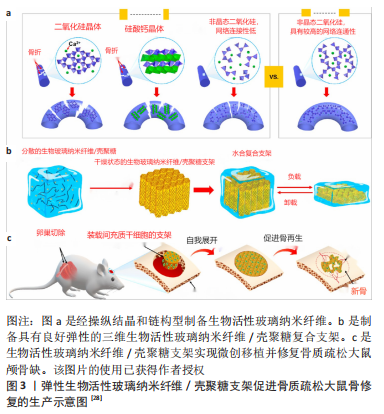
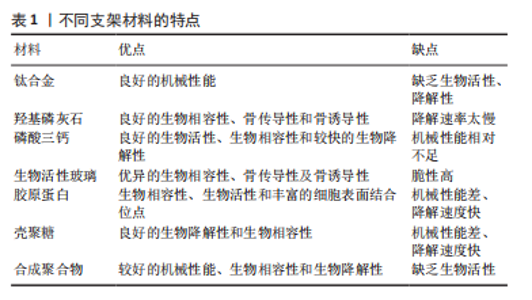
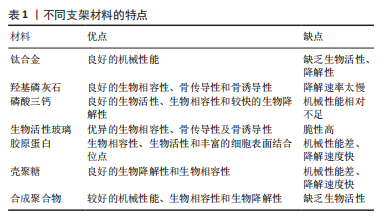
2.2.3 表面修饰设计 由于支架表面是与宿主骨组织接触的唯一区域,因此已经进行了许多尝试来改变材料表面的特性[29],例如改变支架的表面粗糙度、化学性质和通过生物活性物质涂层来修饰材料的表面,采用表面修饰以加强支架与骨骼的结合[30]。并且,通过材料结构设计,例如在一定程度上增加支架孔径可以提高表面涂层效率,同样也促进了骨质疏松性骨缺损的骨再生[31]。2,5-二羟基苯甲酸乙酯是一种促进骨形成和抑制骨吸收的药物,KWON等[32]将其掺杂于聚乳酸-乙醇酸共聚物中,并将这种复合物作为涂层涂覆于结合钛支架上,实验结果表明,这类经表面修饰的结合钛支架在骨质疏松模型中使得骨愈合加强并且植入物固定得到增强。阿仑膦酸盐是用于治疗骨质疏松症的口服药,然而,其全身给药的特点是极低的生物利用度和高毒性。WANG等[33]通过一种简单的粉末加工技术,经表面修饰制备了氨基修饰的介孔生物活性玻璃支架,并且通过局部递送设计将该复合支架作为一种新型的阿仑膦酸盐药物递送系统,体外研究证明,通过氨基修饰显著提高了阿仑膦酸盐的量和递送率,阿仑膦酸盐在该复合材料支架上被高效负载和释放,并通过体内实验证明了经表面修饰设计,该新型氨基修饰的介孔生物活性玻璃复合支架显著促进了骨质疏松性骨缺损的修复。通过对一些材料上的不同有机官能团进行修饰,或者利用某些材料的特点(材料与材料之间进行交联等),可以在一定程度上增强支架的生物功能。 总之,在修复骨质疏松性骨缺损方面,研究者通过材料设计克服支架材料的缺点,将多种材料结合,或经缓慢释放生物活性物质,从而改善不平衡的骨改建和增强支架在骨缺损处的骨整合已成为当今的热点。 2.3 生物活性支架材料 2.3.1 金属及其合金材料 钴、铬、钛及其合金等金属已被广泛用作骨支架材料,主要是因为它们具有优异的机械性能、良好的生物相容性、耐腐蚀性以及良好的骨传导性[34]。然而,金属缺乏生物降解性和生物活性,植入后需要二次手术取出支架,而且金属材料支架的高刚度会导致骨整合不良,因而不太适合用作生物活性支架材料来修复骨质疏松性骨缺损。但是通过局部递送设计,在金属支架上加载一些生物活性物质可以在改善骨改建过程中起重要作用[35]。在所有金属中,钛因其良好的机械强度和生物相容性而成为临床首选的支架材料[36]。骨形态发生蛋白2是经典的生长因子,可在体内诱导骨再生。骨保护素是一种糖蛋白,它被认为是一种抑制骨吸收的因子,主要由成骨细胞表达。研究者们设计制备了一种载有骨形态发生蛋白2和骨保护素的热敏水凝胶,然后将热敏水凝胶注射到三维多孔钛合金支架中,再将复合支架植入骨质疏松性骨缺损大鼠模型中,实验结果表明这种复合支架可以促进成骨分化并同时降低破骨细胞活化,这种复合支架与骨质疏松模型的骨整合显著加强[37]。此外,通过材料结构设计,采用3D技术打印的多孔钛合金支架可以显著降低刚度,并打印出所需的形状和更高的表面积,这种材料具有相互连接的孔隙,便于骨长入,将这种多孔钛合金支架与多糖水凝胶结合,并且封装骨髓间充质干细胞和骨形态发生蛋白2,由此制备的复合材料具有良好的生物相容性,保证了骨形态发生蛋白2的持续释放,有利于骨髓间充质干细胞的成骨分化,即诱导骨向内生长并促进微孔钛在骨质疏松性骨缺损中的骨整合[27]。根据理想支架的特性和骨质疏松性骨缺损的不平衡骨改建,通过优化金属材料支架的“结构”在一定程度上弥补材料的不足,并使其成为良好的载体,与其他材料或生物活性物质结合,既可促进骨再生,改善骨改建,又可增强支架的骨整合。 2.3.2 生物陶瓷材料 生物陶瓷是无机非金属材料,具有良好的生物活性,它们的生物活性源于它们对骨骼矿物相的模拟,因此可以为新组织的形成提供合适的表面[38-40]。用于修复骨质疏松性骨缺损的生物陶瓷材料主要有磷酸钙和生物活性玻璃等,不同类型的磷酸钙材料已一起使用或与其他材料混合使用,以弥补其缺点或突出其优点[41]。近年来,用于修复骨质疏松性骨缺损的磷酸钙支架材料主要有羟基磷灰石、磷酸三钙。 羟基磷灰石是一种天然存在的磷酸钙形式,是构成人体骨骼中最多的无机成分[42],具备理想支架中的良好的生物相容性、骨传导性,但是它的脆性高、韧性低,而且骨诱导性能不足、降解速度相对太快[43]。雷尼酸锶是一种抗骨质疏松药物,主要有效成分是锶。为提高机械性能和生物活性,ZHAO等[44]通过材料结构设计制备了一种具有三维晶须结构的掺锶羟基磷灰石生物陶瓷,这种晶须结构提高了材料的柔韧性,并与加载雷尼酸锶药物的晶须陶瓷和未进行处理的晶须陶瓷进行比较,实验结果表明掺锶的晶须陶瓷促进了去卵巢大鼠的局部骨再生和植入物骨整合,并且结果表明,未处理的晶须陶瓷组和掺锶的晶须陶瓷组的动物模型血清锶浓度较低,而载药组的血清锶浓度比正常组高 8 倍以上。 磷酸三钙是一种 Ca/P 比为 1.5 的磷酸钙,分为α相和β相,β-磷酸三钙比α-磷酸三钙具有更稳定的结构和更高的生物降解率,因此β-磷酸三钙通常用于骨再生[45]。β-磷酸三钙缺乏骨诱导性,降解速率太快,但是其纳米多孔结构使得它具有出色的生物矿化和细胞黏附性,也使得β-磷酸三钙成为生物活性物质的高效载体[46]。为此,TAO等[47]制备了搭载锶和阿司匹林的β-磷酸三钙复合支架,实验结果表明这种复合支架在体外能促进成骨细胞分化,在体内可加速去卵巢大鼠股骨缺损模型的骨再生。骨质疏松的不平衡骨改建使得骨再生能力降低,利用磷酸三钙的多孔结构,通过局部递送设计,局部给药在骨缺损修复和骨整合显示出较强的成骨能力和骨再生活性。双相磷酸钙是将更稳定的羟基磷灰石和更易溶解的磷酸三钙混合而成的材料,同时利用两者的特点,双相磷酸钙在刺激间充质干细胞的成骨分化、增加细胞黏附和增强机械性能方面的研究已得到广泛开展[48],而且在修复骨质疏松性骨缺损中也得到应用。骨化三醇是一种用于临床治疗骨质疏松症的药物,LIM等[49]将骨化三醇作为治疗剂固定在双相磷酸钙支架中,以促进卵巢切除大鼠骨髓间充质干细胞成骨能力,然后通过表面修饰设计,将羟基磷灰石/壳聚糖多层膜涂覆到支架上以提高支架的细胞相容性;体外实验表明,羟基磷灰石/壳聚糖涂层和骨化三醇的结合,不仅增强了卵巢切除大鼠的骨髓间充质干细胞在支架上的黏附和增殖,而且通过抑制钙传感受体的表达显著增强了成骨分化;在颅骨临界大小骨缺损模型中,这类复合支架显著促进了卵巢切除大鼠的骨再生。 将磷酸钙盐与一些液相混合而制成的骨水泥也得到应用,磷酸钙骨水泥有着可注射性、良好的孔隙率、机械性能和降解速率等优点[50]。硒是一种可以通过抑制破骨细胞分化而增强成骨的微量元素。LI等[51]通过局部递送设计将硒掺入磷酸钙骨水泥,然后将制得的生物陶瓷复合支架植入骨质疏松大鼠模型中,实验结果表明,这种生物陶瓷复合支架可加速修复去卵巢大鼠模型股骨缺损。 介孔生物活性玻璃有着优异的生物相容性、骨传导性及骨诱导性,它和传统生物活性玻璃有着相似的成分[52]。尽管介孔生物活性玻璃有着脆性高的缺陷,但它因高度有序的介孔结构和较大的比表面积而表现出更高的生物活性,成为了生物活性物质的高效载体[53],经表面修饰还可提高其负载效率[33],因此,其在修复骨质疏松性骨缺损中得到广泛应用。JIA等[54]制备并表征了含锶的介孔生物活性玻璃支架,体内实验表明这种复合支架显著促进了骨质疏松大鼠的骨再生;实验结果表明,Setd2作为一种组蛋白甲基化酶,可能成为骨质疏松症治疗选择的遗传靶点。 生物活性陶瓷类材料因其良好的生物活性,以及多种材料具有的天然多孔结构,使其可经局部递送设计而达到对生物活性物质的缓慢释放,而且通过对材料结构的设计,控制孔隙率,达到对生物活性物质的缓慢释放,这在修复骨质疏松性骨缺损方面具有重要作用。因此,多种生物陶瓷类材料在骨质疏松性骨缺损方面被广泛应用。 2.3.3 聚合物材料 用于骨植入物的聚合物材料包括天然聚合物和合成聚合物,其中天然聚合物包括胶原、丝素蛋白、藻酸盐、壳聚糖和透明质酸,人工合成聚合物包括聚乳酸、聚乙醇酸、聚己内酯、聚乙二醇及共聚物聚乳酸-乙醇酸等[55]。骨缺损修复的关键是创造一个适合新细胞增殖的环境,做到这一点的最佳方法是重建细胞正常生长的天然环境[56]。天然聚合物通常存在于人体的细胞外基质中,在修复骨质疏松性骨缺损中被广泛应用[57]。 胶原蛋白是人体内最丰富的蛋白质(占全身蛋白质含量的25%-35%),是骨骼的主要成分,具体而言,它虽然有着骨诱导能力相对不足、机械强度差等缺陷,但是它有着良好的生物相容性、生物降解性,更重要的是,它有着丰富的细胞表面结合位点,可经表面修饰设计使其成为支架修复骨质疏松性骨缺损的优良基材,其表面的生物功能分子可刺激细胞黏附、增殖和分化[58]。而且,它的一些水解产物也得到了很好的应用,金属、生物陶瓷、聚合物等材料已与胶原蛋白结合以制备复合支架,由此而得的复合材料支架在机械性能、孔隙率、结构稳定性、骨诱导性等得到改善[59]。为此,研究者们制备了经锶修饰的矿化胶原蛋白支架作为局部治疗全身性受损(例如骨质疏松)骨缺陷的潜在生物材料,通过胶原蛋白的同时原纤维化和矿化而制备了胶原蛋白-矿物纳米复合材料,体外实验证明这种复合支架使得锶被持续释放[60]。此外,阿仑膦酸盐是一种抗骨质疏松药物,ZENG等[61]将其添加到2%胶原蛋白溶液中,然后将此混合物填充到预先设计的圆形模型中,通过交联而获得工程化的胶原蛋白-阿仑膦酸盐复合支架,体外实验结果表明,这种复合支架使得阿仑膦酸被持续释放达1个月,体内实验表明这种复合支架显著促进了骨质疏松性骨缺损大鼠模型的骨再生。 明胶是一种胶原蛋白部分水解而得的聚合物,它具有良好的降解性、生物相容性和易供应性,而且它比完整的胶原蛋白具有较低的抗原性,它可以很容易使用物理和化学交联等方法进行加工和修饰[62]。LI 等[63]通过表面修饰设计,采用酪氨酸酶对明胶进行酶交联和物理折叠,通过这种顺序交联方式制备了具有药物释放控制能力的明胶纳米颗粒/丝素蛋白气凝胶,在这种复合支架上搭载雷尼酸锶,实验结果表明这类载药复合支架的空间结构优良、机械性能得到增强,体内实验证明这种载药复合支架的植入显著促进了骨质疏松骨缺损模型的骨再生。 壳聚糖是一种广泛存在于甲壳类动物外壳中的多糖,它可以提供一种与细胞外基质非常相似的机械结构[64]。除具有良好的生物相容性和生物降解性外,壳聚糖还具有抗菌、抗氧化和止血特性,而且壳聚糖中存在反应性羟基和氨基官能团,可以促进壳聚糖的化学修饰。研究人员还可以通过支架设计来控制材料的孔径及其降解速率,从而促进支架的骨整合[65]。正是由于这些原因,基于壳聚糖材料的支架在修复骨质疏松性骨缺损领域得到了越来越多的关注。CHU等[66]采用冷冻干燥技术构建了镧取代Al3+的MgAl层状双氢氧化物纳米混合材料,其中纳米混合材料在壳聚糖基质上有序排列,实验表明这种复合材料支架通过对成骨细胞和破骨细胞的双向调节而促进骨再生,并且这类复合支架能够显著促进去卵巢大鼠颅骨缺损模型骨再生。 合成聚合物具有明确的化学性质、易于加工和定制的优点,并且根据所需性能可以对聚合物进行改性。因此研究者们从这类材料的特性出发,结合其他材料的优点,设计制备了一系列聚合物材料支架[67]。近年来,聚己内酯作为合成聚合物材料成为修复骨质疏松性骨缺损的理想支架材料。 聚己内酯具有相对较好的机械性能、生物相容性和可生物降解性,而且聚己内酯的降解速率较慢,是聚酯中降解速度最慢的,因此常用来代替骨组织,然而其缺乏生物活性[68]。明胶和聚己内酯都已制备成可生物降解的电纺纳米纤维膜,而后也制备了由聚己内酯和明胶组成的混合电纺纳米可吸收引导骨再生膜,膜上负载抗氧化应激的polaprezinc药物,体内实验证明,这种复合膜在骨质疏松氧化应激的微环境中具有抗氧化应激和促进骨形成的能力[69]。此外,聚己内酯熔融状态下的热稳定性和流变性,使其具有易加工的塑料特性和充当黏合剂时良好的流变性。研究者们从这些特性出发,将介孔生物活性玻璃与ε-聚己内酯结合而制备了一种复合材料支架。在这种复合支架中,介孔生物活性玻璃为支架提供成骨特性和良好的机械性能,而ε-聚己内酯在支架制造过程中充当粘合剂并降低纯生物活性玻璃支架的脆性。这种复合支架可促进骨质疏松绵羊的骨再生,与未处理的骨质疏松绵羊相比,植入复合支架的骨质疏松绵羊在12 周后达到更大的骨化体积和骨小梁厚度[70]。 胶原蛋白有着丰富的细胞表面结合位点,明胶易通过物理和化学交联而加工修饰,壳聚糖存在反应性羟基和氨基官能团,以及聚己内酯可定制和改性,这些聚合物易于加工和修饰的特性使得其经支架设计和材料结合后,可制备成功能化的复合支架,并且在修复骨质疏松性骨缺损中具有良好的效果。不同支架材料的特点见表1。"
| [1] ASPRAY TJ, HILL TR. Osteoporosis and the Ageing Skeleton. Subcell Biochem. 2019;91:453-476. [2] ESTELL EG, ROSEN CJ. Emerging insights into the comparative effectiveness of anabolic therapies for osteoporosis. Nat Rev Endocrinol. 2021;17(1):31-46. [3] GILARSKA A, HINZ A, BZOWSKA M, et al. Addressing the Osteoporosis Problem-Multifunctional Injectable Hybrid Materials for Controlling Local Bone Tissue Remodeling. ACS Appl Mater Interfaces. 2021;13(42): 49762-49779. [4] WU Q, WANG X, JIANG F, et al. Study of Sr-Ca-Si-based scaffolds for bone regeneration in osteoporotic models. Int J Oral Sci. 2020;12(1):25. [5] RAJAN RK, CHANDRAN S, SREELATHA HV, et al. Pamidronate-Encapsulated Electrospun Polycaprolactone-Based Composite Scaffolds for Osteoporotic Bone Defect Repair. ACS Appl Bio Mater. 2020;3(4): 1924-1933. [6] ZHAO R, SHANG T, YUAN B, et al. Osteoporotic bone recovery by a bamboo-structured bioceramic with controlled release of hydroxyapatite nanoparticles. Bioact Mater. 2022;17:379-393. [7] YANG YS, XIE J, CHAUGULE S, et al. Bone-Targeting AAV-Mediated Gene Silencing in Osteoclasts for Osteoporosis Therapy. Mol Ther Methods Clin Dev. 2020;17:922-935. [8] DEININGER C, WAGNER A, HEIMEL P, et al. Enhanced BMP-2-Mediated Bone Repair Using an Anisotropic Silk Fibroin Scaffold Coated with Bone-like Apatite. Int J Mol Sci. 2021;23(1):283. [9] TROMBETTA RP, KNAPP EK, Awad HA. A Mouse Femoral Ostectomy Model to Assess Bone Graft Substitutes. Methods Mol Biol. 2021;2230: 75-89. [10] ZHAO Y, LI Z, JIANG Y, et al. Bioinspired mineral hydrogels as nanocomposite scaffolds for the promotion of osteogenic marker expression and the induction of bone regeneration in osteoporosis. Acta Biomater. 2020;113:614-626. [11] 李祖浩,王辰宇,王中汉,等.骨质疏松性骨缺损的治疗进展:支架植入与局部药物递送[J].中国组织工程研究,2018,22(18):2939-2945. [12] MARYCZ K, ŚMIESZEK A, KORNICKA-GARBOWSKA K, et al. Novel Nanohydroxyapatite (nHAp)-Based Scaffold Doped with Iron Oxide Nanoparticles (IO), Functionalized with Small Non-Coding RNA (miR-21/124) Modulates Expression of Runt-Related Transcriptional Factor 2 and Osteopontin, Promoting Regeneration of Osteoporotic Bone in Bilateral Cranial Defects in a Senescence-Accelerated Mouse Model (SAM/P6). PART 2. Int J Nanomedicine. 2021;16:6049-6065. [13] SHUAI C, YU L, FENG P, et al. Interfacial reinforcement in bioceramic/biopolymer composite bone scaffold: The role of coupling agent. Colloids Surf B Biointerfaces. 2020;193:111083. [14] Zhang B, Li J, He L, et al. Bio-surface coated titanium scaffolds with cancellous bone-like biomimetic structure for enhanced bone tissue regeneration. Acta Biomater. 2020;114:431-448. [15] HE J, CHEN G, LIU M, et al. Scaffold strategies for modulating immune microenvironment during bone regeneration. Mater Sci Eng C Mater Biol Appl. 2020;108:110411. [16] 汪雕雕,孙雨阳,田壮,等.不同骨组织工程支架设计与骨传导性、骨诱导性及生物降解性变化的关系[J].中国组织工程研究,2022, 26(21):3435-3444. [17] TURNBULL G, CLARKE J, PICARD F, et al. 3D bioactive composite scaffolds for bone tissue engineering. Bioact Mater. 2017;3(3):278-314. [18] GARCÍA-GARCÍA P, REYES R, PÉREZ-HERRERO E, et al. Alginate-hydrogel versus alginate-solid system. Efficacy in bone regeneration in osteoporosis. Mater Sci Eng C Mater Biol Appl. 2020;115:111009. [19] RUMIAN Ł, WOLF-BRANDSTETTER C, RÖßLER S, et al. Sodium alendronate loaded poly(l-lactide- co-glycolide) microparticles immobilized on ceramic scaffolds for local treatment of bone defects. Regen Biomater. 2020;7(3):293-302. [20] Tang Q, Hu Z, Jin H, et al. Microporous polysaccharide multilayer coated BCP composite scaffolds with immobilised calcitriol promote osteoporotic bone regeneration both in vitro and in vivo.Theranostics. 2019;9(4):1125-1143. [21] MIAO Q, JIANG N, YANG Q, et al. Multi-stage controllable degradation of strontium-doped calcium sulfate hemihydrate-tricalcium phosphate microsphere composite as a substitute for osteoporotic bone defect repairing: degradation behavior and bone response. Biomed Mater. 2021;17(1):10.1088/1748-605X/ac4323. [22] MAI X, KANG Z, WANG N, et al. Oxygen Plasma Technology-Assisted Preparation of Three-Dimensional Reduced Graphene Oxide/Polypyrrole/Strontium Composite Scaffold for Repair of Bone Defects Caused by Osteoporosis. Molecules. 2021;26(15):4451. [23] LIU D, WU ZX, ZHANG Y, et al. Local Treatment of Osteoporotic Sheep Vertebral Body With Calcium Sulfate for Decreasing the Potential Fracture Risk: Microstructural and Biomechanical Evaluations. Clin Spine Surg. 2016;29(7):E358-E364. [24] YANG S, WANG L, FENG S, et al. Enhanced bone formation by strontium modified calcium sulfate hemihydrate in ovariectomized rat critical-size calvarial defects. Biomed Mater. 2017;12(3):035004. [25] LI K, ZHANG F, WANG D, et al. Silkworm-inspired electrohydrodynamic jet 3D printing of composite scaffold with ordered cell scale fibers for bone tissue engineering. Int J Biol Macromol. 2021;172:124-132. [26] MISHRA A, SRIVASTAVA V. Biomaterials and 3D printing techniques used in the medical field. J Med Eng Technol. 2021;45(4):290-302. [27] BAI H, ZHAO Y, WANG C, et al. Enhanced osseointegration of three-dimensional supramolecular bioactive interface through osteoporotic microenvironment regulation. Theranostics. 2020;10(11):4779-4794. [28] WANG L, QIU Y, GUO Y, et al. Smart, Elastic, and Nanofiber-Based 3D Scaffolds with Self-Deploying Capability for Osteoporotic Bone Regeneration. Nano Lett. 2019;19(12):9112-9120. [29] KUMARI S, TIYYAGURA HR, POTTATHARA YB, et al. Surface functionalization of chitosan as a coating material for orthopaedic applications: A comprehensive review. Carbohydr Polym. 2021;255: 117487. [30] ARCOS D, VALLET-REGÍ M. Substituted hydroxyapatite coatings of bone implants. J Mater Chem B. 2020;8(9):1781-1800. [31] KRIEGHOFF J, PICKE AK, SALBACH-HIRSCH J, et al. Increased pore size of scaffolds improves coating efficiency with sulfated hyaluronan and mineralization capacity of osteoblasts. Biomater Res. 2019;23:26. [32] KWON BJ, SEON GM, LEE MH, et al. Locally delivered ethyl-2,5-dihydroxybenzoate using 3D printed bone implant for promotion of bone regeneration in a osteoporotic animal model. Eur Cell Mater. 2018;35:1-12. [33] WANG X, ZENG D, WENG W, et al. Alendronate delivery on amino modified mesoporous bioactive glass scaffolds to enhance bone regeneration in osteoporosis rats. Artif Cells Nanomed Biotechnol. 2018;46(sup2):171-181. [34] CALABRESE G, FRANCO D, PETRALIA S, et al. Dual-Functional Nano-Functionalized Titanium Scaffolds to Inhibit Bacterial Growth and Enhance Osteointegration. Nanomaterials (Basel). 2021;11(10): 2634. [35] LI J, YUAN H, CHANDRAKAR A, et al. 3D porous Ti6Al4V-beta-tricalcium phosphate scaffolds directly fabricated by additive manufacturing. Acta Biomater. 2021;126:496-510. [36] KAUR M, SINGH K. Review on titanium and titanium based alloys as biomaterials for orthopaedic applications. Mater Sci Eng C Mater Biol Appl. 2019;102:844-862. [37] WANG X, LI Z, WANG Z, et al. Incorporation of Bone Morphogenetic Protein-2 and Osteoprotegerin in 3D-Printed Ti6Al4V Scaffolds Enhances Osseointegration Under Osteoporotic Conditions. Front Bioeng Biotechnol. 2021;9:754205. [38] BATTAFARANO G, ROSSI M, DE MARTINO V, et al. Strategies for Bone Regeneration: From Graft to Tissue Engineering. Int J Mol Sci. 2021; 22(3):1128. [39] CODREA CI, CROITORU AM, BACIU CC, et al. Advances in Osteoporotic Bone Tissue Engineering. J Clin Med. 2021;10(2):253. [40] LIAO HT, TSAI MJ, BRAHMAYYA M, et al. Bone Regeneration Using Adipose-Derived Stem Cells in Injectable Thermo-Gelling Hydrogel Scaffold Containing Platelet-Rich Plasma and Biphasic Calcium Phosphate. Int J Mol Sci. 2018;19(9):2537. [41] WANG J, WANG M, CHEN F, et al. Nano-Hydroxyapatite Coating Promotes Porous Calcium Phosphate Ceramic-Induced Osteogenesis Via BMP/Smad Signaling Pathway. Int J Nanomedicine. 2019;14:7987-8000. [42] JI X, YUAN X, MA L, et al. Mesenchymal stem cell-loaded thermosensitive hydroxypropyl chitin hydrogel combined with a three-dimensional-printed poly(ε-caprolactone) /nano-hydroxyapatite scaffold to repair bone defects via osteogenesis, angiogenesis and immunomodulation. Theranostics. 2020;10(2):725-740. [43] QAYOOM I, TEOTIA AK, KUMAR A. Nanohydroxyapatite Based Ceramic Carrier Promotes Bone Formation in a Femoral Neck Canal Defect in Osteoporotic Rats. Biomacromolecules. 2020;21(2):328-337. [44] ZHAO R, CHEN S, ZHAO W, et al. A bioceramic scaffold composed of strontium-doped three-dimensional hydroxyapatite whiskers for enhanced bone regeneration in osteoporotic defects. Theranostics. 2020;10(4):1572-1589. [45] JEONG J, KIM JH, SHIM JH, et al. Bioactive calcium phosphate materials and applications in bone regeneration. Biomater Res. 2019;23:4. [46] KANG HJ, MAKKAR P, PADALHIN AR, et al. Comparative study on biodegradation and biocompatibility of multichannel calcium phosphate based bone substitutes. Mater Sci Eng C Mater Biol Appl. 2020;110:110694. [47] TAO ZS, ZHOU WS, XU HG, et al. Aspirin modified strontium-doped β-tricalcium phosphate can accelerate the healing of femoral metaphyseal defects in ovariectomized rats. Biomed Pharmacother. 2020;132:110911. [48] CHEN Y, LIU Z, JIANG T, et al. Strontium-substituted biphasic calcium phosphate microspheres promoted degradation performance and enhanced bone regeneration. J Biomed Mater Res A. 2020;108(4):895-905. [49] LIM HK, KWON IJ, ON SW, et al. Enhanced Bone Regeneration in Variable-Type Biphasic Ceramic Phosphate Scaffolds Using rhBMP-2. Int J Mol Sci. 2021;22(21):11485. [50] TAN S, WANG Y, DU Y, et al. Injectable bone cement with magnesium-containing microspheres enhances osteogenesis via anti-inflammatory immunoregulation. Bioact Mater. 2021;6(10):3411-3423. [51] LI TL, TAO ZS, WU XJ, et al. Selenium-modified calcium phosphate cement can accelerate bone regeneration of osteoporotic bone defect. J Bone Miner Metab. 2021;39(6):934-943. [52] SHEN X, YU P, CHEN H, et al. Icariin controlled release on a silk fibroin/mesoporous bioactive glass nanoparticles scaffold for promoting stem cell osteogenic differentiation. RSC Adv. 2020;10(20):12105-12112. [53] GUDURIC V, BELTON N, RICHTER RF, et al. Tailorable Zinc-Substituted Mesoporous Bioactive Glass/Alginate-Methylcellulose Composite Bioinks. Materials (Basel). 2021;14(5):1225. [54] JIA X, LONG Q, MIRON RJ, et al. Setd2 is associated with strontium-induced bone regeneration. Acta Biomater. 2017;53:495-505. [55] BHARADWAZ A, JAYASURIYA AC. Recent trends in the application of widely used natural and synthetic polymer nanocomposites in bone tissue regeneration. Mater Sci Eng C Mater Biol Appl. 2020;110:110698. [56] KIM YS, MAJID M, MELCHIORRI AJ, et al. Applications of decellularized extracellular matrix in bone and cartilage tissue engineering. Bioeng Transl Med. 2018;4(1):83-95. [57] LYNCH CR, KONDIAH PPD, CHOONARA YE. Advanced Strategies for Tissue Engineering in Regenerative Medicine: A Biofabrication and Biopolymer Perspective. Molecules. 2021;26(9):2518. [58] ZHAO C, XIAO Y, LING S, et al. Structure of Collagen. Methods Mol Biol. 2021;2347:17-25. [59] MAILLARD S, SICARD L, ANDRIQUE C, et al. Combining sclerostin neutralization with tissue engineering: An improved strategy for craniofacial bone repair. Acta Biomater. 2022;140:178-189. [60] QUADE M, SCHUMACHER M, BERNHARDT A, et al. Strontium-modification of porous scaffolds from mineralized collagen for potential use in bone defect therapy. Mater Sci Eng C Mater Biol Appl. 2018;84: 159-167. [61] ZENG Y, ZHOU M, MOU S, et al. Sustained delivery of alendronate by engineered collagen scaffold for the repair of osteoporotic bone defects and resistance to bone loss. J Biomed Mater Res A. 2020;108(12): 2460-2472. [62] PUROHIT SD, BHASKAR R, SINGH H, et al. Development of a nanocomposite scaffold of gelatin-alginate-graphene oxide for bone tissue engineering. Int J Biol Macromol. 2019;133:592-602. [63] LI D, CHEN K, DUAN L, et al. Strontium Ranelate Incorporated Enzyme-Cross-Linked Gelatin Nanoparticle/Silk Fibroin Aerogel for Osteogenesis in OVX-Induced Osteoporosis. ACS Biomater Sci Eng. 2019;5(3):1440-1451. [64] ISLAM MM, SHAHRUZZAMAN M, BISWAS S, et al. Chitosan based bioactive materials in tissue engineering applications-A review. Bioact Mater. 2020;5(1):164-183. [65] HE J, HU X, CAO J, et al. Chitosan-coated hydroxyapatite and drug-loaded polytrimethylene carbonate/polylactic acid scaffold for enhancing bone regeneration. Carbohydr Polym. 2021;253:117198. [66] CHU M, SUN Z, FAN Z, et al. Bi-directional regulation functions of lanthanum-substituted layered double hydroxide nanohybrid scaffolds via activating osteogenesis and inhibiting osteoclastogenesis for osteoporotic bone regeneration. Theranostics. 2021;11(14):6717-6734. [67] LEI B, GUO B, RAMBHIA KJ, et al. Hybrid polymer biomaterials for bone tissue regeneration. Front Med. 2019;13(2):189-201. [68] DWIVEDI R, KUMAR S, PANDEY R, et al. Polycaprolactone as biomaterial for bone scaffolds: Review of literature. J Oral Biol Craniofac Res. 2020; 10(1):381-388. [69] GAO X, AL-BAADANI MA, WU M, et al. Study on the Local Anti-Osteoporosis Effect of Polaprezinc-Loaded Antioxidant Electrospun Membrane. Int J Nanomedicine. 2022;17:17-29. [70] GÓMEZ-CEREZO N, CASARRUBIOS L, SAIZ-PARDO M, et al. Mesoporous bioactive glass/ɛ-polycaprolactone scaffolds promote bone regeneration in osteoporotic sheep. Acta Biomater. 2019;90:393-402. |
| [1] | Li Xiaomin, Tian Xiangdong, Tan Yetong, Zhu Guangyu, Wang Rongtian, Wang Jian, Xue Zhipeng, Ma Sheng, Hu Yuanyi, Huang Ye, Ding Tiansong. Changes of lower limb force line and knee function after high tibial osteotomy in osteoporotic medial ventricular knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1325-1329. |
| [2] | Wen Xinghua, Ding Huanwen, Cheng Kai, Yan Xiaonan, Peng Yuanhao, Wang Yuning, Liu Kang, Zhang Huiwu. Three-dimensional finite element model analysis of intramedullary nailing fixation design for large femoral defects in Beagle dogs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1371-1376. |
| [3] | Jiang Xiaocheng, Shi Lu, Wang Yinbin, Li Qiujiang, Xi Chuangzhen, Ma Zefeng, Cai Lijun. Systematical evaluation of bone fusion rate after interbody fusion in patients with osteoporosis and lumbar degenerative disease treated with teriparatide [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1427-1433. |
| [4] | Sun Jiajia, Zhu Haidi, Lu Yun, Zhang Kai. Comparison of bone metabolism markers between type 2 diabetes mellitus and non-type 2 diabetes mellitus patients with hip fracture [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1156-1160. |
| [5] | Yang Yitian, Wang Lu, Yao Wei, Zhao Bin. Application of the interaction between biological scaffolds and macrophages in bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1071-1079. |
| [6] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [7] | Lu Di, Zhang Cheng, Duan Rongquan, Liu Zongxiang. Osteoinductive properties of calcium phosphate ceramic bone repair materials [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1103-1109. |
| [8] | Tang Haotian, Liao Rongdong, Tian Jing. Application and design of piezoelectric materials for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1117-1125. |
| [9] | Xu Yan, Li Ping, Lai Chunhua, Zhu Peijun, Yang Shuo, Xu Shulan. Piezoelectric materials for vascularized bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1126-1132. |
| [10] | Long Yanming, Xie Mengsheng, Huang Jiajie, Xue Wenli, Rong Hui, Li Xiaojie. Casein kinase 2-interaction protein-1 regulates the osteogenic ability of bone marrow mesenchymal stem cells in osteoporosis rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 878-882. |
| [11] | Qin Yuxing, Ren Qiangui, Li Zilong, Quan Jiaxing, Shen Peifeng, Sun Tao, Wang Haoyu. Action mechanism and prospect of bone microvascular endothelial cells for treating femoral head necrosis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 955-961. |
| [12] | Wang Jinling, Huang Xiarong, Qu Mengjian, Huang Fujin, Yin Lingwei, Zhong Peirui, Liu Jin, Sun Guanghua, Liao Yang, Zhou Jun. Effects of exercise training on bone mass and bone microstructure in aged osteoporotic rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 676-682. |
| [13] | Zhang Min, Zhang Xiaoming, Liu Tongbin. Application potential of naringin in bone tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 787-792. |
| [14] | Cheng Yunzhong, Liu Yuzeng, Hai Yong, Guan Li, Pan Aixing, Zhang Xinuo, Tao Luming, Li Yue. Bibliometric and visual analysis of the research status and development trend of cortical bone trajectory screws [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 513-519. |
| [15] | Liu Hao, Yang Hongsheng, Zeng Zhimou, Wang Liping, Yang Kunhai, Hu Yongrong, Qu Bo. Lumbar MRI vertebral bone quality score to evaluate the severity of osteoporosis in postmenopausal women [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 606-611. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||