Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (28): 4532-4538.doi: 10.12307/2022.310
Previous Articles Next Articles
Modification of natural and composite alginate hydrogels and repair of bone defect with composite systems
Fan Yi, Liu Yadong, Cui Yutao, Liu He, Tian Yuhang, Li Shaorong, Wang Gan, Wu Dankai, Peng Chuangang
- Department of Orthopedics, Second Hospital of Jilin University, Changchun 130041, Jilin Province, China
-
Received:2021-03-23Accepted:2021-04-30Online:2022-10-08Published:2022-03-23 -
Contact:Wu Dankai, MD, Chief physician, Professor, Doctoral supervisor, Department of Orthopedics, Second Hospital of Jilin University, Changchun 130041, Jilin Province, China Peng Chuangang, MD, Associate chief physician, Associate professor, Master’s supervisor, Department of Orthopedics, Second Hospital of Jilin University, Changchun 130041, Jilin Province, China -
About author:Fan Yi, Master candidate, Physician, Department of Orthopedics, Second Hospital of Jilin University, Changchun 130041, Jilin Province, China -
Supported by:the Department of Finance Project of Jilin Province, No. 2019SCZT014 (to WDK); the Science and Technology Department Project of Jilin Province, No. 20200404140YY (to WDK); the National Natural Science Foundation of China, No. 81772456 (to LH)
CLC Number:
Cite this article
Fan Yi, Liu Yadong, Cui Yutao, Liu He, Tian Yuhang, Li Shaorong, Wang Gan, Wu Dankai, Peng Chuangang. Modification of natural and composite alginate hydrogels and repair of bone defect with composite systems[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(28): 4532-4538.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

2.1.1 天然海藻酸盐水凝胶 海藻酸盐是一种由α-L-古罗糖醛酸(G)和β-D-甘露糖醛酸(M)两种结构单元依靠1,4-糖苷键连接形成主链的嵌段天然聚多糖,主要来源于海藻类植物(褐藻)与细菌(固氮菌和假单胞菌)中,是一种高产量的、可再生的天然生物材料[16]。海藻酸盐主链由连续的M残基、连续的G残基以及交替的M和G残基组成。一般来说,海藻酸盐及其水凝胶具有良好的生物相容性,其中海藻酸的钙、钠、铵、钾盐被美国食品药品监督管理局认定为GRAS[17](Generally Recognized as Safe,一般认可为安全),在伤口愈合、药物输送和细胞移植中的应用被广泛研究。 在组织工程中,生物材料为细胞提供了三维支架,作为细胞黏附、增殖及分化的环境。细胞支架应该是多孔网络状的结构,其内的细胞在与周围环境进行细胞物质交换的同时,也被提供了必要的空间,以沉积细胞外基质。海藻酸盐形成的水凝胶的三维网络结构与人体生理状态下的细胞外基质相似[18],是潜在的骨组织工程细胞支架材料。天然海藻酸盐水凝胶自身存在的缺陷,例如其在机体内表现的缓慢的生物降解,不易被细胞识别和黏附以及传统离子交联凝胶方式下所表现的凝胶结构不稳定等,限制了其在骨组织工程中的应用。因此,为实现其骨缺损治疗的应用,需要设计海藻酸盐水凝胶体系,包括改良的生物可降解性、交联方式的选择及复合水凝胶的构建等。 2.1.2 改良的生物可降解性 理想的组织工程细胞支架材料应随着组织再生的进程逐渐生物降解,过快的降解将导致细胞支架的崩塌,而过缓的降解将阻碍组织的正常修复,因为细胞只能将矿化基质沉积到其直接的周围环境中[19]。一般生理情况下,因为缺乏降解海藻酸盐水凝胶的酶,哺乳动物体内的海藻酸盐水凝胶是难以降解的。有研究通过放射性定位实验发现,海藻酸盐进行肾脏清除的分子质量阈值约为48 kD[20],市售的海藻酸盐分子质量为3.2-400.0 kD,所以即使完成了植入部位的生物降解,大分子质量的海藻酸盐分子也将汇集在脉管系统中难以酶解或排出,影响其远期安全性。因此,增强其生物降解性以及小分子质量化海藻酸盐是其组织工程应用的重要考虑因素。 最近的研究指出,海藻酸盐的氧化可以提高其生物降解性,高碘酸盐是其常用的氧化剂[21]。在一项海藻酸钠的氧化研究中,BALAKRISHNAN等[22]通过在水-乙醇溶液和水溶液两种介质中使用高碘酸钠氧化海藻酸钠,发现在两种溶液介质中,海藻酸钠水凝胶随着氧化的进行而裂解,并且裂解产物的重均摩尔质量随着氧化体系中高碘酸钠浓度的增加而降低。在此氧化系统中,氧化和低分子质量化是一个协同的过程,是潜在的改性策略。另外,γ射线照射也能够降低海藻酸盐分子质量,提高其在机体内的降解速率[23]。以上2种提高海藻酸盐水凝胶生物降解性的方法都伴随着海藻酸盐主链的缩短,实现了小分子质量化,有利于海藻酸盐材料在机体内生物降解后的代谢排出,提升了其远期生物安全性。 2.1.3 交联方式 离子交联和共价交联是海藻酸盐水凝胶主要的两种凝胶方式。一般情况下,离子交联凝胶方式能在安全温和的条件下成胶。海藻酸盐(主要是海藻酸钠)与Ca2+交联形成离子交联型水凝胶,其机制在于Ca2+与海藻酸盐中的4个G残基进行配位,形成典型的“蛋盒”结构[24]。除Ca2+以外,Zn2+、Ba2+、Sr2+和Cu2+等二价金属离子也可以与海藻酸盐反应形成水凝胶[25-27]。在体内液体环境下,离子交联型水凝胶容易因为二价金属阳离子的浸出从而导致水凝胶机械强度降低甚至水凝胶的解体[28-29],并且离子交联的凝胶方式容易因为快速地凝胶化而导致不均质水凝胶的产生,所产生凝胶的机械性能也难以控制[30-31],而有研究表明,凝胶不同的机械性能会导致不同的细胞谱系分化行为[32]。一般来说,随着离子浓度的增加,离子交联型水凝胶的机械强度也会增加[33-35],但需要注意的是,已有证据表明,过高的离子浓度会表现出细胞毒性[36]。为解决以上问题,不同的解决方案被提出。DIAZ-RODRIGUEZ等[37]制备了Ca2+交联水凝胶,在海藻酸钙水凝胶中加入了CaCO3晶体,CaCO3的存在可以改善海藻酸盐水凝胶的长期稳定性,因为碳酸钙晶体的部分溶解会导致Ca2+离子的释放,从而促进海藻酸盐凝胶的形成。SUN等[38]通过原位缓慢释放D-葡萄糖酸-d-内酯水解产生的二价离子,成功交联了多巴胺修饰的海藻酸钠复合水凝胶,缓释的二价阳离子能使海藻酸盐形成均质的水凝胶,其中的羟基磷灰石可以帮助稳定水凝胶网络。基于相同的原理,其他能够提供缓释阳离子的成分,如磷酸钙等,也用于海藻酸盐复合水凝胶的添加材料以保持凝胶结构的稳定性。 共价交联型海藻酸盐水凝胶是海藻酸盐分子通过共价键结合形成的水凝胶,相比于离子交联型水凝胶,能够减少因离子浸出而导致的凝胶结构问题。共价交联型水凝胶同样存在着局限性,例如在制备共价交联型水凝胶的过程中,应该选择合适的交联剂和交联体系,以避免或减少因为有毒交联剂(如四官能团聚酰胺胺[39])的使用而造成的不良反应。基于仿海底贻贝黏附的设计,LEE等[40]通过共价交联制备形成了海藻酸盐-邻苯二酚水凝胶,其细胞毒性小,可忽略,在流变测试下显示出均一的力学性能;相比于海藻酸钙水凝胶,海藻酸盐-邻苯二酚水凝胶在水溶液条件下具有更高的稳定性,并且在此凝胶中,通过调节凝胶中的海藻酸盐-邻苯二酚的质量分数,可实现对机械强度的调节。 因此,离子交联与共价交联的凝胶方式各有其优势和局限性,所以在设计构建水凝胶体系时需要结合所需功能进行综合考量。 2.1.4 海藻酸盐复合水凝胶 人体骨组织由约70%的无机物和约30%的有机成分组成,从材料的角度考虑,骨为复合材料[41]。羟基磷灰石是骨组织主要的无机物成分,基于仿生设计,羟基磷灰石常被加入水凝胶材料中以构建复合水凝胶体系,在骨缺损修复的初期,骨组织就能从修复材料中获得所需的钙源。在此体系中,羟基磷灰石浸出的Ca2+可以帮助海藻酸盐水凝胶保持结构的稳定,还可以作为其他二价阳离子的载体,例如具有促进细胞成骨活性的Sr2+[42]。另外,其他组分也被选择性地加入构建海藻酸盐复合水凝胶,以实现其特定的功能,如透明质酸作为细胞识别的位点,可以参与CD44介导的细胞识别与黏附[43],弥补天然海藻酸盐水凝胶在细胞识别和黏附中的不足,刺激结缔组织的修复;明胶来源于胶原蛋白,可以用来模拟正常骨组织细胞外有机成分[44];壳聚糖凝胶带有正电荷,可以构建通过静电相互作用组装分层凝胶[29],也可以提供抗菌性能等。 2.2 海藻酸盐水凝胶修复骨缺损 在临床工作中,骨缺损的修复是一个复杂的问题,例如缺损的大小各异,形状不同,缺损所在的骨的生理功能也不一样。不同的骨缺损修复,需要修复体系具有不同的功能,如细胞、生物活性因子、药物的递送和承重等。"

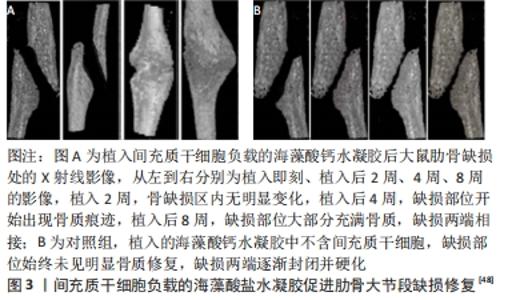
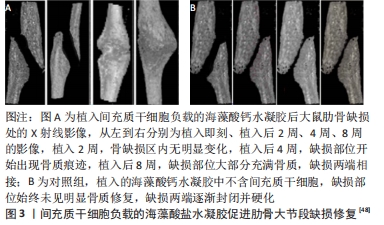
2.2.1 细胞、生物活性因子、药物的控制性递送 在一般情况下,骨组织固有的自我修复能力,使得骨组织能够在一定情况下完成缺损的自身修复[45-46]。但在某些特殊情况下,例如临界性骨缺损时,骨组织难以完成自身修复。骨组织的再生修复是细胞与细胞基质、生物活性因子、药物等环境因素共同作用的结果,控制性地向骨缺损部位输送细胞和药物等生物活性物质以协助骨缺损修复是现阶段骨组织工程的常用策略。运用海藻酸盐水凝胶及其复合水凝胶温和、低机械强度的特点,在植入时能够避免对周围组织的侵入,同时完成目标细胞、生物活性因子以及药物的递送。 以骨髓间充质干细胞为代表的间充质干细胞是骨缺损治疗中常用的、能够进行成骨分化的细胞[47],同时,也被认为是骨缺损细胞疗法的金标准。有研究证据表明,利用简单的海藻酸钙水凝胶作为细胞包裹基质,向骨缺损部位输送间充质干细胞是一种有效的骨缺损修复促进途径[48],见图3。"

海藻酸盐/透明质酸复合水凝胶在细胞输送中以促进骨组织修复过程中体现出与简单海藻酸钙水凝胶类似的性能[43]。有研究利用海藻酸盐甲基丙烯酸甲酯、多巴胺、RGD短肽和羟基磷灰石合成了光交联的海藻酸基黏合剂水凝胶[49],并用其向免疫缺陷小鼠体内输送牙龈间充质干细胞,在异位成骨实验中形成了与天然骨相似的矿化结节。因此,基于细胞输送的治疗是骨缺损修复的有效途径。 目前,已经有多种生物活性因子,如骨形成蛋白、血管内皮生长因子、血小板衍生生长因子和转化生长因子β等,在研究中被用于促进骨组织再生。骨形成蛋白能促进间充质细胞定向分化为成骨细胞,是体内诱导骨和软骨形成的细胞因子,其中在骨缺损修复中最常用的是骨形成蛋白2。在一项大鼠股骨临界性骨缺损的修复实验中,KOLAMBKA等[50]用纳米纤维网状管和海藻酸钠水凝胶构建了一种复合生长因子输送系统,纳米纤维网状管单独或在没有重组人骨形成蛋白2的海藻酸盐水凝胶存在下,不足以桥接大鼠股骨8 mm的节段性缺损,而在海藻酸盐水凝胶中输送重组人骨形成蛋白2显著增加了骨形成,形成了完全性的骨桥接。另外,CHEN等[51]的研究表明骨形成蛋白7能够促进人真皮成纤维细胞的成骨作用,并证实海藻酸盐水凝胶可以用于负载表达骨形成蛋白7的腺病毒,实现基因治疗。 氧、微量营养物质等在组织中扩散的距离有限,因此,血管化在成功构建工程组织中起着关键性作用。血管内皮生长因子是一种强有力的血管生成因子,通过刺激内皮细胞的迁移和增殖,在血管网络的形成中起促进作用。KAIGLER等[52]研究发现,海藻酸钙水凝胶负载血管内皮生长因子的应用促进了早期的血管生成,而在较晚的时间点,它促进了骨的再生,并且这种结果可能来自于通过血管内皮生长因子的促进血管生成的作用,间接影响骨的愈合和成熟。 骨组织的再生涉及到多种生物活性因子的作用,这提示了适当递送多种生物活性因子可能是有效组织工程策略的前提。然而,在组织重建过程中,协调的生物“信号”并不是同时进行两种或更多种的活性因子同时递送那么简单[53-54]。例如,骨形成蛋白的作用存在于骨再生的各个阶段中,而血小板衍生生长因子主要作用在骨再生的早期,Bayer等[55]设计了海藻酸盐水凝胶/磷酸钙复合支架以控制性释放骨形成蛋白2和血小板衍生生长因子BB,证实,早期的血小板衍生生长因子BB递送能够有效地刺激支架的细胞浸润,且并不对延后释放的骨形成蛋白2的促进成骨细胞分化的能力产生抑制,证明了顺序释放的血小板衍生生长因子-BB和骨形成蛋白2有可能协同促进骨再生过程。 另外,海藻酸盐水凝胶可以作为生物活性物质和细胞相互作用的体系,用于生物活性物质与细胞的反应,并且可以实现生物活性物质与细胞的一起递送。在一项体外实验中,SEVARI等[56]在海藻酸盐水凝胶中加入了生物活性玻璃微粒和基质胶,结果表明,生物活性玻璃微粒和基质胶的浓度越高,牙髓干细胞成骨基因骨钙蛋白、矮小相关转录因子2和碱性磷酸酶的表达水平越高,矿化程度越高。GE等[57]在海藻酸盐水凝胶中加入了基底膜蛋白、人胎盘Ⅳ型胶原、层粘连蛋白以及硫酸软骨素来模拟细胞外基质,发现,在含有骨形成蛋白2和基底膜蛋白的水凝胶微珠中发现了矿物质结节,且基底膜蛋白装载的微珠中的矿化结节更多,而在小鼠颅顶的临界性骨缺损植入中,含有骨形成蛋白2和基底膜蛋白的水凝胶微珠负载人间充质干细胞都有新生类骨组织,但无骨形成蛋白2和基底膜蛋白的水凝胶微珠组则未见。 因此,海藻酸盐水凝胶在骨组织修复中的应用是因为其能够用作选择性地递送细胞、生物活性因子、药物等的载体。在递送过程中,海藻酸盐水凝胶保持了递送物质的生物活性,并且能够实现细胞、生物活性物质、药物的组合递送,时间顺序缓释等功能,见表1。"


2.2.2 海藻酸盐水凝胶联合高强度支架修复骨缺损 为周围组织提供机械支撑及承重是骨的重要生理功能。在骨缺损修复中,一个稳定的机械环境是必要的[64],并且承重骨缺损所需的早期承重也是必须考虑的问题。 海藻酸盐水凝胶的机械强度是凝胶成分、凝胶浓度及交联方式等条件综合的结果,但无论采用哪种途径,目前的海藻酸盐水凝胶都难以满足承重骨缺损修复所需要的机械强度。在修复体系中引入具有高机械强度的支架,例如金属支架、骨水泥、高强度聚合物等,使复合的海藻酸盐水凝胶体系能够表现更优的机械性能,对于大节段骨缺损(由其是承重骨缺损)的修复是必需的。 金属支架:目前运用于体内的金属植入物主要为钛合金,其与机体能够较好的相容,但是骨整合能力欠缺,可能出现无菌性松动[65]。裸钛(Ti)材料表现出一定的生物惰性,这阻止了生物分子的粘附,基于此问题,MUDERRISOGLU等[66]开发了一种新型的由藻酸盐水凝胶,多孔CaCO3生物陶瓷和活性酶碱性磷酸酶组成的钛表面纳米结构生物活性涂层,对钛(Ti)表面进行改性,以构建骨刺激界面。在此涂层中,释放的碱性磷酸酶和Ca2+增强了成骨细胞MC3T3的分化和增殖,并使其保持了良好的成骨活性,这表明海藻酸盐水凝胶良好的生物活性分子负载特性是促进界面骨整合的潜在途径。 虽然金属支架能够提供机械支撑,但骨再生不足时,骨缺损也难以愈合,基于此,生物活性分子、细胞等被引入钛合金支架中,以促进骨再生和骨长入。KUMAR等[58]在钛合金(Ti6Al4V)支架中引入负载了前成骨细胞(MC3T3-E1)的海藻酸盐/明胶/羟基磷灰石凝胶,在该体系中,细胞具有良好的存活率,并且表现出相比于对照组(孔板表面)更高的碱性磷酸酶活性。因此,钛合金以及海藻酸盐水凝胶复合体系在提供良好机械支撑的同时,也是良好的细胞输送载体。 骨水泥:是骨科临床工作中常用的植入物,具有较高的机械强度,广泛用于骨缺损填充、感染控制和骨科移植物锚定。磷酸钙基骨水泥可以模制和原位注射形成支架,具有良好的骨传导性和生物可降解性[67-68]。但是其存在着一些缺陷,例如不耐冲洗和较低的生物活性。XU等[69]的研究数据表明,在海藻酸盐水凝胶/磷酸钙骨水泥复合支架中,骨水泥体内降解过程中释放的Ca2+,为海藻酸盐凝胶结构的保持提供了二价金属阳离子,并且海藻酸盐凝胶的加入提高了骨水泥的耐冲洗性能,在体外细胞相容性实验中,骨水泥/海藻酸盐水凝胶组表现出了更高的人骨髓间充质干细胞增殖活性。 在复合体系中,海藻酸盐水凝胶也可以用于细胞、药物的递送。QIU等[61]合成了壳聚糖/磷酸钙骨水泥/多西环素支架,在体外实验中表现出超过3周的抗生素释放,并且呈现出随剂量增长的对牙龈卟啉单胞菌和金黄色葡萄球菌的抑菌性能。掺入复合支架的海藻酸盐水凝胶微珠负载了人牙周膜干细胞,表现出成骨谱系分化,有望应用于颅面、骨科中的感染性骨缺损的修复。 因此,在大节段骨缺损时,尤其承重骨缺损时,需要具有高机械强度的植入物以提供机械支撑。海藻酸盐水凝胶体系的加入可以提供生物活性因子、细胞等,并且增强了植入体骨整合及促进骨再生的能力,可以帮助加速骨的功能性重建。"

| [1] HO-SHUI-LING A, BOLANDER J, RUSTOM LE, et al. Bone regeneration strategies: engineered scaffolds, bioactive molecules and stem cells current stage and future perspectives. Biomaterials. 2018;180:143-162. [2] SHAKYA AK, KANDALAM U. Three-dimensional macroporous materials for tissue engineering of craniofacial bone. Br J Oral Maxillofac Surg. 2017;55(9):875-891. [3] TOURNIER P, GUICHEUX J, PARE A, et al. A partially demineralized allogeneic bone graft: in vitro osteogenic potential and preclinical evaluation in two different intramembranous bone healing models. Sci Rep. 2021;11(1):4907. [4] DIMITRIOU R, MATALIOTAKIS GI, ANGOULES AG, et al. Complications following autologous bone graft harvesting from the iliac crest and using the RIA:A systematic review. Injury-Int J Care Inj. 2011;42:S3-S15. [5] 谢程欣,余城墙,王维,等.骨形态发生蛋白与自体骨移植治疗四肢长骨骨不连的Meta分析[J].中国组织工程研究,2020,24(5): 803-810. [6] YUAN HP, FERNANDES H, HABIBOVIC P, et al. Osteoinductive ceramics as a synthetic alternative to autologous bone grafting. Proc Natl Acad Sci U S A. 2010;107(31):13614-13619. [7] HAUGEN HJ, LYNGSTADAAS SP, ROSSI F, et al. Bone grafts:which is the ideal biomaterial? J Clin Periodontol. 2019;46:92-102. [8] ZHENG Y, WANG J, CHANG B, et al. Clinical study on repair of metacarpal bone defects using titanium alloy implantation and autologous bone grafting. Exp Ther Med. 2020;20(6):233. [9] AGARWAL R, GARCIA AJ. Biomaterial strategies for engineering implants for enhanced osseointegration and bone repair. Adv Drug Deliv Rev. 2015;94:53-62. [10] BOSE S, ROY M, BANDYOPADHYAY A. Recent advances in bone tissue engineering scaffolds. Trends Biotechnol. 2012;30(10):546-554. [11] REAKASAME S, BOCCACCINI AR. Oxidized alginate-based hydrogels for tissue engineering applications: a review. Biomacromolecules. 2018;19(1):3-21. [12] RANA D, KUMAR TSS, RAMALINGAM M. Cell-laden hydrogels for tissue engineering. J Biomater Tissue Eng. 2014;4(7):507-535. [13] SPICER C D. Hydrogel scaffolds for tissue engineering:the importance of polymer choice. Polym Chem. 2020;11(2):184-219. [14] GONG T, XIE J, LIAO JF, et al. Nanomaterials and bone regeneration. Bone Res. 2015;3:15029. [15] MAO A S, MOONEY DJ. Regenerative medicine: current therapies and future directions. Proc Natl Acad Sci U S A. 2015;112(47):14452-14459. [16] LALZAWMLIANA V, ANAND A, MUKHERJEE P, et al. Marine organisms as a source of natural matrix for bone tissue engineering. Ceram Int. 2019;45(2):1469-1481. [17] LIU J, YANG SQ, LI XT, et al. Alginate oligosaccharides:production, biological activities, and potential applications. Compr Rev Food Sci Food Saf. 2019;18(6):1859-1881. [18] TIBBITT MW, ANSETH KS. Hydrogels as extracellular matrix mimics for 3D cell culture. Biotechnol Bioeng. 2009;103(4):655-663. [19] XIANG G, LIPPENS E, HAFEEZ S, et al. Oxidized alginate beads for tunable release of osteogenically potent mesenchymal stromal cells. Mater Sci Eng C-Mater Biol Appl. 2019;104:8. [20] AL-SHAMKHANI A, DUNCAN R. Radioiodination of alginate via covalently-bound tyrosinamide allows monitoring of its fate in vivo. J Bioact Compat Polym. 1995;10(1):4-13. [21] LIANG Y, LIU WS, HAN BQ, et al. An in situ formed biodegradable hydrogel for reconstruction of the corneal endothelium. Colloid Surf B-Biointerfaces. 2011;82(1):1-7. [22] BALAKRISHNAN B, LESIEUR S, LABARRE D, et al. Periodate oxidation of sodium alginate in water and in ethanol-water mixture:a comparative study. Carbohydr Res. 2005;340(7):1425-1429. [23] ALSBERG E, KONG HJ, HIRANO Y, et al. Regulating bone formation via controlled scaffold degradation. J Dent Res. 2003;82(11):903-908. [24] PLAZINSKI W. Molecular basis of calcium binding by polyguluronate chains. revising the egg-box model. J Comput Chem. 2011;32(14): 2988-2995. [25] LIU YG, TONG YS, WANG SB, et al. Influence of different divalent metal ions on the properties of alginate microcapsules and microencapsulated cells. J Sol-Gel Sci Technol. 2013;67(1):66-76. [26] ISKANDAR L, ROJO L, DI SILVIO L, et al. The effect of chelation of sodium alginate with osteogenic ions, calcium, zinc, and strontium. J Biomater Appl. 2019;34(4):573-584. [27] CATTALINI JP, HOPPE A, PISHBIN F, et al. Novel nanocomposite biomaterials with controlled copper/calcium release capability for bone tissue engineering multifunctional scaffolds. J R Soc Interface. 2015;12(110):13. [28] LUECKGEN A, GARSKE DS, ELLINGHAUS A, et al. Hydrolytically-degradable click-crosslinked alginate hydrogels. Biomaterials. 2018; 181:189-198. [29] YAN HQ, CHEN XQ, FENG MX, et al. Layer-by-layer assembly of 3D alginate-chitosan-gelatin composite scaffold incorporating bacterial cellulose nanocrystals for bone tissue engineering. Mater Lett. 2017; 209:492-496. [30] JEON O, BOUHADIR KH, MANSOUR JM, et al. Photocrosslinked alginate hydrogels with tunable biodegradation rates and mechanical properties. Biomaterials. 2009;30(14):2724-2734. [31] JEON O, POWELL C, AHMED SM, et al. Biodegradable, photocrosslinked alginate hydrogels with independently tailorable physical properties and cell adhesivity. Tissue Eng Part A. 2010;16(9):2915-2925. [32] HUEBSCH N, ARANY PR, MAO AS, et al. Harnessing traction-mediated manipulation of the cell/matrix interface to control stem-cell fate. Nat Mater. 2010;9(6):518-526. [33] 赵德路,铁朝荣,王新,等.复合锶离子光交联海藻酸盐水凝胶支架的机械和生物学性能[J].中国组织工程研究,2019,23(18):2880-2887. [34] KAYGUSUZ H, EVINGUR GA, PEKCAN O, et al. Surfactant and metal ion effects on the mechanical properties of alginate hydrogels. Int J Biol Macromol. 2016;92:220-224. [35] 张漫,李昊,徐婷,等.含锶交联藻酸钠凝胶对前成骨细胞黏附和增殖的影响[J].上海口腔医学,2019,28(2):123-127. [36] AMIN AK, HUNTLEY JS, BUSH PG, et al. Chondrocyte death in mechanically injured articular cartilage-the influence of extracellular calcium. J Orthop Res. 2009;27(6):778-784. [37] DIAZ-RODRIGUEZ P, GARCIA-TRINANES P, LOPEZ MME, et al. Mineralized alginate hydrogels using marine carbonates for bone tissue engineering applications. Carbohydr Polym. 2018;195:235-242. [38] SUN XJ, LI ZY, CUI ZD, et al. Preparation and physicochemical properties of an injectable alginate-based hydrogel by the regulated release of divalent ions via the hydrolysis of d-glucono-delta-lactone. J Biomater Appl. 2020;34(7):891-901. [39] PATIL SS, NUNE KC, MISRA RDK. Alginate/poly(amidoamine) injectable hybrid hydrogel for cell delivery. J Biomater Appl. 2018;33(2):295-314. [40] LEE C, SHIN J, LEE JS, et al. Bioinspired, calcium-free alginate hydrogels with tunable physical and mechanical properties and improved biocompatibility. Biomacromolecules. 2013;14(6):2004-2013. [41] SALGADO AJ, COUTINHO OP, REIS RL. Bone tissue engineering: state of the art and future trends. Macromol Biosci. 2004;4(8):743-765. [42] LOURENCO AH, NEVES N, RIBEIRO-MACHADO C, et al. Injectable hybrid system for strontium local delivery promotes bone regeneration in a rat criticalsized defect model. Sci Rep. 2017;7(1):15. [43] INGAVLE GC, GIONET-GONZALES M, VORWALD CE, et al. Injectable mineralized microsphere-loaded composite hydrogels for bone repair in a sheep bone defect model. Biomaterials. 2019;197:119-128. [44] CECOLTAN S, STANCU IC, DRAGUSIN DM, et al. Nanocomposite particles with improved microstructure for 3D culture systems and bone regeneration. J Mater Sci-Mater Med. 2017;28(10):153. [45] CUI YT, ZHU TT, LI D, et al. Bisphosphonate-functionalized scaffolds for enhanced bone regeneration. Adv Healthc Mater. 2019;8(23): e1901073. [46] LOI F, CORDOVA LA, PAJARINEN J, et al. Inflammation, fracture and bone repair. Bone. 2016;86:119-130. [47] ZHOU WY, LIN JX, ZHAO K, et al. Single-cell profiles and clinically useful properties of human mesenchymal stem cells of adipose and bone marrow origin. Am J Sports Med. 2019;47(7):1722-1733. [48] 王俊钢,李聪聪,毛广显,等.骨生物材料复合骨髓间充质干细胞异位成骨修复肋骨大段缺损[J].中国组织工程研究,2017,21(2): 182-186. [49] HASANI-SADRABADI MM, SARRION P, POURAGHAEI S, et al. An engineered cell-laden adhesive hydrogel promotes craniofacial bone tissue regeneration in rats. Sci Transl Med. 2020;12(534):eaay6853. [50] KOLAMBKAR YM, DUPONT KM, BOERCKEL JD, et al. An alginate-based hybrid system for growth factor delivery in the functional repair of large bone defects. Biomaterials. 2011;32(1):65-74. [51] CHEN F, BI D, CAO G, et al. Bone morphogenetic protein 7-transduced human dermal-derived fibroblast cells differentiate into osteoblasts and form bone in vivo. Connecti Tissue Res. 2018;59(3):223-232. [52] KAIGLER D, SILVA EA, MOONEY DJ. Guided bone regeneration using injectable vascular endothelial growth factor delivery gel. J Periodontol. 2013;84(2):230-238. [53] SUBBIAH R, CHENG A, RUEHLE MA, et al. Effects of controlled dual growth factor delivery on bone regeneration following composite bone-muscle injury. Acta Biomaterialia. 2020;114:63-75. [54] SIMMONS CA, ALSBERG E, HSIONG S, et al. Dual growth factor delivery and controlled scaffold degradation enhance in vivo bone formation by transplanted bone marrow stromal cells. Bone. 2004;35(2):562-569. [55] BAYER EA, JORDAN J, ROY A, et al. Programmed platelet-derived growth factor-bb and bone morphogenetic protein-2 delivery from a hybrid calcium phosphate/alginate scaffold. Tissue Eng Part A. 2017;23(23-24): 1382-1393. [56] SEVARI SP, SHAHNAZI F, CHEN C, et al. Bioactive glass-containing hydrogel delivery system for osteogenic differentiation of human dental pulp stem cells. J Biomed Mater Res Part A. 2020;108(3): 557-564. [57] GE Q, GREEN DW, LEE DJ, et al. Mineralized polysaccharide transplantation modules supporting human msc conversion into osteogenic cells and osteoid tissue in a non-union defect. Mol Cells. 2018;41(12):1016-1023. [58] KUMAR A, NUNE KC, MISRA R DK. Design and biological functionality of a novel hybrid Ti-6Al-4V/hydrogel system for reconstruction of bone defects. J Tissue Eng Regen Med. 2018;12(4):1133-1144. [59] CIPITRIA A, BOETTCHER K, SCHOENHALS S, et al. In-situ tissue regeneration through SDF-1 alpha driven cell recruitment and stiffness-mediated bone regeneration in a critical-sized segmental femoral defect. Acta Biomateri. 2017;60:50-63. [60] 郑旺,高丽娜,李西成,等.胶原蛋白膜包裹藻酸钙凝胶/骨髓基质细胞/BMP-2复合体修复兔桡骨缺损[J].中国矫形外科杂志,2012, 20(4):363-366. [61] QIU G, HUANG M, LIU J, et al. Antibacterial calcium phosphate cement with human periodontal ligament stem cell-microbeads to enhance bone regeneration and combat infection. J Tissue Eng Regen Med. 2021;15(3):232-243. [62] SANGEETHA K, GIRIJA EK. Tailor made alginate hydrogel for local infection prophylaxis in orthopedic applications. Mater Sci Eng C-Mater Biol Appl. 2017;78:1046-1053. [63] MA FB, LI SJ, RUIZ-ORTEGA LI, et al. Effects of alginate/chondroitin sulfate-based hydrogels on bone defects healing. Mater Sci Eng C-Mater Biol Appl. 2020;116:11212. [64] DIMITRIOU R, JONES E, MCGONAGLE D, et al. Bone regeneration: current concepts and future directions. BMC Med. 2011;9:66. [65] RAPHEL J, KARLSSON J, GALLI S, et al. Engineered protein coatings to improve the osseointegration of dental and orthopaedic implants. Biomaterials. 2016;83:269-282. [66] MUDERRISOGLU C, SAVELEVA M, ABALYMOV A, et al. Nanostructured biointerfaces based on bioceramic calcium carbonate/hydrogel coatings on titanium with an active enzyme for stimulating osteoblasts growth. Adv Mater Interfaces. 2018;5(19):1800452. [67] ZHAO LA, WEIR MD, XU HHK. An injectable calcium phosphate-alginate hydrogel-umbilical cord mesenchymal stem cell paste for bone tissue engineering. Biomaterials. 2010;31(25):6502-6510. [68] WEIR MD, XU HHK, SIMON CG. Strong calcium phosphate cement-chitosan-mesh construct containing cell-encapsulating hydrogel beads for bone tissue engineering. J Biomed Mater Res Part A. 2006; 77A(3):487-496. [69] XU C, WANG XY, ZHOU J, et al. Bioactive tricalcium silicate/alginate composite bone cements with enhanced physicochemical properties. J Biomed Mater Res Part B. 2018;106(1):237-244. |
| [1] | Xue Yadong, Zhou Xinshe, Pei Lijia, Meng Fanyu, Li Jian, Wang Jinzi . Reconstruction of Paprosky III type acetabular defect by autogenous iliac bone block combined with titanium plate: providing a strong initial fixation for the prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1424-1428. |
| [2] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhou Qian, Zhang Qiang, Chen Qiu. Human salivary components and osteoporosis/osteopenia [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1439-1444. |
| [3] | Jin Tao, Liu Lin, Zhu Xiaoyan, Shi Yucong, Niu Jianxiong, Zhang Tongtong, Wu Shujin, Yang Qingshan. Osteoarthritis and mitochondrial abnormalities [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1452-1458. |
| [4] | Zhang Lichuang, Xu Hao, Ma Yinghui, Xiong Mengting, Han Haihui, Bao Jiamin, Zhai Weitao, Liang Qianqian. Mechanism and prospects of regulating lymphatic reflux function in the treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1459-1466. |
| [5] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [6] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [7] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhang Qiang, Liu Jing, Shao Ming. Acupuncture for Parkinson’s disease: an insight into the action mechanism in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1272-1277. |
| [8] | Gao Yujin, Peng Shuanglin, Ma Zhichao, Lu Shi, Cao Huayue, Wang Lang, Xiao Jingang. Osteogenic ability of adipose stem cells in diabetic osteoporosis mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 999-1004. |
| [9] | Hui Xiaoshan, Bai Jing, Zhou Siyuan, Wang Jie, Zhang Jinsheng, He Qingyong, Meng Peipei. Theoretical mechanism of traditional Chinese medicine theory on stem cell induced differentiation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1125-1129. |
| [10] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [11] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1137-1142. |
| [12] | Guo Jia, Ding Qionghua, Liu Ze, Lü Siyi, Zhou Quancheng, Gao Yuhua, Bai Chunyu. Biological characteristics and immunoregulation of exosomes derived from mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1093-1101. |
| [13] | Wu Weiyue, Guo Xiaodong, Bao Chongyun. Application of engineered exosomes in bone repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1102-1106. |
| [14] | Zhou Hongqin, Wu Dandan, Yang Kun, Liu Qi. Exosomes that deliver specific miRNAs can regulate osteogenesis and promote angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1107-1112. |
| [15] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||