Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (2): 271-275.doi: 10.12307/2022.044
Previous Articles Next Articles
Regulatory role of Keap1/Nrf2/ARE signaling pathway in bone homeostasis
Wu Saixuan, Zhang Mi, Dong Ming, Lu Ying, Niu Weidong
- Department of Dentistry and Endodontics, School of Stomatology, Dalian Medical University, Dalian 116041, Liaoning Province, China
-
Received:2020-10-29Revised:2020-10-30Accepted:2020-12-14Online:2022-01-18Published:2021-10-27 -
Contact:Niu Weidong ,MD, Professor, Department of Dentistry and Endodontics, School of Stomatology, Dalian Medical University, Dalian 116041, Liaoning Province, China -
About author:Wu Saixuan, Master, Physician, Department of Dentistry and Endodontics, School of Stomatology, Dalian Medical University, Dalian 116041, Liaoning Province, China -
Supported by:the National Natural Science Foundation of China, No. 81700962 (to LY)
CLC Number:
Cite this article
Wu Saixuan, Zhang Mi, Dong Ming, Lu Ying, Niu Weidong. Regulatory role of Keap1/Nrf2/ARE signaling pathway in bone homeostasis[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 271-275.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
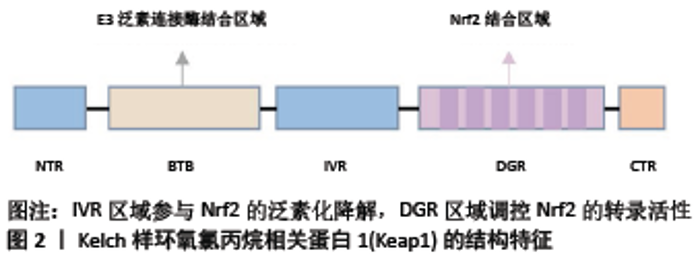
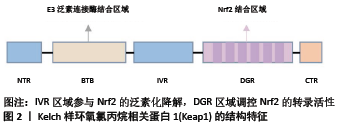
2.1 Keap1/Nrf2/ARE信号通路的组成及活化 2.1.1 Keap1 是Nrf2的特异性受体,主要存在于细胞质中。由5个结构域组成,分别为NTR、BTB、IVR、DGR和CTR区域,其中NTR、CTR为2个氨基酸末端,BTB、IVR、DGR是主要的功能区域[4],见图2。BTB区域是一个蛋白质与蛋白质相互作用的结构域,通常和其他Keap1的BTB区形成二聚体,与Nrf2以1∶1比例相结合,参与Nrf2的泛素化降解;IVR区域是氧化应激敏感区,与Nrf2的稳定性相关,在非氧化应激状态下参与Nrf2的泛素化降解;DGR区域有6个Kelch重复序列,可以识别Nrf2的ETGE和DLG两个基因序列,对Nrf2的活性具有负调控作用[5-6]。"
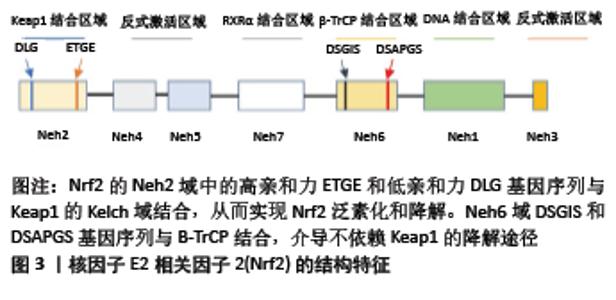
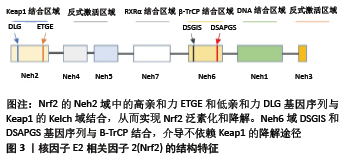
2.1.2 Nrf2 属于碱性亮氨酸拉链转录因子的Cap’n’Collar (CNC)亚家族,由核红系2组成,包括3个核红系2相关因子(NRF1、NRF2和NRF3)以及2个BTB和CNC同源蛋白(BACH1和BACH2)[5,7-8],分子质量为95-110 kDa[9]。Nrf2包含605个氨基酸和7个结构域,称为Nrf2-ECH homology(Neh)域1-7。Neh1域包含CNC/碱性亮氨酸拉链域,该域介导Nrf2与小分子肌腱纤维瘤蛋白二聚化,促进Nrf2与ARE结合[10]。Neh2域位于基因序列的最N端,促进Nrf2与Keap1结合。Neh3域位于Nrf2的最C端,通过与CHD6相互作用来辅助Nrf2反式激活,与转录活性密切相关[11]。Neh4和Neh5域通过共激活因子和共抑制因子介导转录活化或转录抑制[12-13]。Neh6域包含DSGIS和DSAPGS基因序列,提供与β-TrCP的结合位点,介导不依赖于Keap1的Nrf2降解途径[14-15]。Neh7结构域充当视黄醇受体α的结合位点,抑制依赖Nrf2/ARE的基因表达[7,16],见图3。 2.1.3 ARE 也称亲电反应元件,是一种能编码启动子区许多解毒酶和细胞保护蛋白基因的顺式反应元件。不同ARE的碱基序列是不一样的,但多项研究表明,其中都含有一个16核苷酸序列5’-TA/CAnnA/GTGAC/GnnnGCA/G-3’(n代表任一核苷酸),具有典型的功能活性。在氧化应激状态下,解离的Nrf2进入胞核内,与小分子肌腱纤维瘤蛋白形成异二聚体,进而与ARE进行特异性结合,启动下游的靶基因。Bach1是ARE的转录抑制因子,在正常的生理条件下,Bach1与肌腱纤维瘤蛋白结合形成异二聚体,从而防止Nrf2与之结合[17]。 2.1.4 Keap1/Nrf2/ARE信号通路的激活及调控机制 正常生理状态下,Keap1的DGR区域与Nrf2的Neh2域相结合,以Keap1-Nrf2复合体的形式被锚定在胞浆内肌动蛋白细胞骨架上,使Nrf2稳定在细胞质内,无法进入细胞核,从而抑制其转录。Keap1-Nrf2复合体作为Cul3/Rbx1 E3泛素连接酶的适配底物,介导Nrf2的泛素化降解,从而维持Nrf2在细胞内低表达、非活化的状态[18-19]。但在压力、氧化应激等状态下,氧化剂或亲电子化合物与Keap1的半胱氨酸残基相互作用,使其结构发生变化,从而使之与Nrf2分离[20]。Nrf2被激活后,转位进入细胞核内,通过其高度保守的碱性亮氨酸拉链结构与小分子肌腱纤维瘤蛋白形成异二聚体并且识别ARE[21],启动下游一系列保护性基因的表达,如多种抗氧化酶、Ⅱ相解毒酶,增强细胞的解毒及抗氧化能力,进而发挥保护作用[22]。 其他信号传导通路也可以影响Nrf2的转录,通过激活或抑制Cul3/Rbx1-Keap1复合物、SCF/β-TrCP复合物或HRD1来调节Nrf2蛋白水平。半胱氨酸的氧化修饰、含ETGE蛋白的竞争性结结合、蛋白质-蛋白质相互作用的抑制物、p62/SQSTM1、mTOR抑制剂和Cul3/Rbx1 E3连接酶抑制剂(MLN4924)水平的增加均可破坏Keap1与Nrf2间相互作 用[23-25]。另外,Keap1启动子的过甲基化可增加Nrf2的表达,胰岛素/生长因子或GSK3-β、CRL、PI3K-AKT-PKC、mTOR、ERK/p38-MAPK和WNT抑制剂等均能够调节SCF/β-TrCP-Nrf2间的相互作用[26-27],HRD1抑制剂(LS-102)也可用于预防内质网应激相关的Nrf2降解[28]。 2.2 Keap1/Nrf2/ARE信号通路在破骨细胞中的作用 2.2.1 破骨细胞 破骨细胞是源自造血干细胞的高度分化的多核巨噬细胞,主要功能是参与骨组织的吸收。在破骨细胞分化成熟的过程中,RANKL/RANK是调控破骨细胞分化成熟的关键信号通路[29]。RANK是RANKL的受体,是一种表达在破骨细胞前体或者成熟破骨细胞膜上的Ⅰ型跨膜糖蛋白,RANK和RANKL结合可以使破骨细胞前体分化为成熟的破骨细胞,激活核因子κB、抗酒石酸酸性磷酸酶、组织蛋白酶K和活化T细胞核因子蛋白1等与破骨细胞有关标志基因的表达[30-32]。 2.2.2 破骨细胞与Keap1/Nrf2/ARE信号通路 Keap1/Nrf2/ARE信号通路参与破骨细胞形成和活性的调节。Nrf2可以通过启动抗氧化酶的表达降低细胞内部的活性氧自由基水平[33],进而抑制MAPK通路和PI3K/Akt通路,抑制破骨细胞的分化和吸收[34],也可通过抑制核因子κB、c-Fos和NFATc1抑制破骨细胞的分化[35-36]。 在RANKL诱导的破骨细胞分化过程中,RANKL通过上调Keap1降低Nrf2/Keap1的比率,降低细胞内保护酶的表达水平,提高细胞内活性氧自由基水平,从而促进破骨细胞的分化和功能。在体外和体内模型中,Nrf2的过表达通过上调细胞保护酶血红素加氧酶1、醌氧化还原酶1和谷氨酰半胱氨酸合成酶的表达,降低细胞内的活性氧自由基水平[37],减少抗酒石酸酸性磷酸酶阳性多核细胞的数量,减少破骨细胞分化标记基因(ATP6v0d2、组织蛋白酶K、树突状细胞-特异性跨膜蛋白、基质金属蛋白酶9、破骨细胞相关受体和抗酒石酸酸性磷酸酶)的表达,并减弱骨破坏。此外,体内局部Nrf2过表达减弱了体内脂多糖介导的RANKL依赖性颅骨破坏[38]。应用Nrf2激活剂萝卜硫素和姜黄素抑制了野生型小鼠细胞中的破骨细胞分化并增加了Nrf2靶基因的表达,抑制了Nrf2缺陷型小鼠骨髓单核巨噬细胞中破骨细胞的形成[39]。说明Keap1/Nrf2信号通路通过细胞保护酶调节细胞内的活性氧自由基水平从而在破骨细胞形成中起作用。 在Nrf2缺陷型小鼠细胞中,包含3个以上核的抗酒石酸酸性磷酸酶阳性多核细胞的形成显著增强,肌动蛋白环形成能力增强,表明Nrf2可能负调控RANKL诱导的破骨细胞分化并且抑制肌动蛋白环的形成和骨吸收。敲除RAW264.7细胞中的Nrf2发现,抑制Nrf2可以增强RAW264.7细胞中的破骨细胞分化[40]。当有辐射或砷等外界刺激时,破骨细胞分化增多,功能增强。Nrf2的缺失会导致氧化应激增加,并增加对辐射或砷引起的骨丢失的敏感性[41]。 然而Nrf2在骨吸收中的作用一直集中在受活性氧自由基和RANKL影响的破骨细胞分化上,破骨细胞祖细胞和破骨细胞中Nrf2活性尚未得到很好的研究。 2.3 Keap1/Nrf2/ARE信号通路在成骨细胞中的作用 2.3.1 成骨细胞 成骨细胞起源于间充质干细胞,主要功能是参与骨组织的形成,维持骨稳态,在骨骼的生长发育以及保持骨量方面发挥关键作用,由具有多向分化潜能的间充质干细胞分化而来。骨形态发生蛋白-Smads、Wnt/β-catenin、Notch、Hedgehog、成纤维细胞生长因子等信号在调控成骨细胞分化及骨生成方面有重要作用[42]。Ⅰ型胶原蛋白、Runx2、骨钙蛋白、骨粘连蛋白、骨桥蛋白、骨唾液蛋白和碱性磷酸酶是成骨细胞的标志物[43]。 2.3.2 成骨细胞与Keap1/Nrf2/ARE信号通路 Nrf2在成骨细胞分化和活动中的作用仍然存在争议,可能与年龄、性别、遗传背景以及生理与病理状况等因素相关。 一些研究发现Nrf2对骨形成具有负面影响,KOOK 等[44]研究发现,辐射以剂量依赖性方式减少矿化作用,同时降低MC3T3-E1细胞中骨特异性基因骨粘连蛋白、骨桥蛋白、骨唾液蛋白和骨钙蛋白的表达。而siRNA介导的Nrf2沉默能够明显逆转辐射对细胞成骨细胞分化的负面影响,导致血红素加氧酶1减少和Runx2水平增加。类似地,PARK等[35]培养了敲除Nrf2的MC3T3-E1细胞以及颅骨来源的成骨细胞。这些体外研究发现,成骨细胞分化和钙化增强,在敲除Nrf2的成骨细胞中,碱性磷酸酶、Runx2,骨钙素的mRNA表达显著升高,Runx2蛋白的表达增加。HINOI等[45]通过在MC3T3-E1成骨细胞中过表达Nrf2,发现其会抑制成骨细胞分化的关键转录因子Runx2的表达,从而阻止成骨细胞分化。这些结果表明Nrf2负调控成骨细胞的分化和矿化。 PARK等[35]的动物实验研究表明,9周龄的野生型小鼠和敲除Nrf2的小鼠相比,Nrf2 敲除小鼠的股骨骨小梁量显著增加,结果表明Nrf2对骨量具有负面影响。为了进一步探讨Nrf2 敲除小鼠的骨量增加是由于骨形成增加还是骨吸收减少,对股骨的组织切片进行了组织形态计量学分析,结果显示,与野生型小鼠相比,Nrf2 敲除小鼠的骨形成增加,单位骨周成骨细胞数和单位骨表面成骨细胞数显著增加,而单位骨周的破骨细胞数量没有差异,单位骨表面的腐蚀表面、骨吸收活性标志物也没有差异,Nrf2 敲除小鼠的血清骨形成标记(破骨细胞)水平显著升高。这些结果表明,Nrf2 敲除小鼠的骨量增加可能是由于成骨细胞生成和骨形成增加引起的,而不是骨吸收减少所致,表明Nrf2作为骨吸收的调节剂,起到抗成骨的作用。然而PARK等[35]的研究中并未说明实验小鼠的性别,仅对9周龄的野生型小鼠和Nrf2 敲除小鼠进行对比研究,需要进一步研究来确定年龄和性别对骨骼中Nrf2调节作用的影响。 Keap1作为Nrf2的负调节剂,Keap1基因敲除小鼠可以作为Nrf2过表达的动物模型,然而Keap1-/-小鼠由于食管和前胃畸形严重过度角化而在出生后3周内死亡。为了解决这一问题,YOSHIDA等[46]通过在系统性Keap1缺乏的情况下,构建鳞状上皮特异性Nrf2缺乏(NEKO)小鼠,来纠正食管的过度角化和随后的致死性,从而建立Nrf2过表达的动物模型,发现除食管和皮肤外,在大多数组织中可观察到Nrf2的过度活化。YOSHIDA等[46]发现NEKO小鼠股骨长度、骨量和对照组小鼠相比显著减少,这些结果表明,Nrf2的过度活化导致小鼠严重骨发育不良。为了进一步分析骨发育不良的原因,将小鼠的新生颅骨进行了体外成骨细胞分化实验,观察到NEKO小鼠成骨细胞分化严重受损。因此表明Nrf2过度活化抑制了成骨细胞的分化,NEKO小鼠的骨量减少是由于成骨细胞分化减少导致的骨稳态受损。 相反,SUN等[47]研究发现,与野生型对照组小鼠相比,Nrf2-/-小鼠的骨量降低,骨骼强度降低,负载诱导的骨形成减弱。KIM等[48]发现Nrf2对于正常小鼠出生后的骨骼发育至关重要,缺乏Nrf2的小鼠表现出明显的骨骼发育受损和骨量减少。通过观察3,6,8,12和24周龄Nrf2+/+和Nrf2-/-小鼠的长骨μCT发现,对照组Nrf2+/+小鼠在8周龄时达到了峰值骨量,相比之下,Nrf2-/-小鼠在3周龄时表现出严重的骨量减少,并持续到12周龄。3,6和8周龄的Nrf2-/-小鼠和对照组相比,骨体积分数、骨小梁数量和厚度减少,而骨小梁分离增加,但是到12周龄时,Nrf2+/+和Nrf2-/-小鼠之间的所有参数都相似,结果表明Nrf2的损失会损害出生后早期的骨骼形成。进一步进行体外研究发现,早期的骨量低并不是由于成骨细胞本身存在任何缺陷。与对照组成骨细胞相比,来自Nrf2缺乏小鼠的原发颅骨成骨细胞可以正常增殖和分化。这些结果表明Nrf2-/-小鼠出生后骨骼发育受损可能是由于骨祖细胞群的增殖或存活能力衰竭所致,这导致在Nrf2-/-小鼠早期观察到的成骨细胞数量较低。KIM等[48]认为可能有其他机制可以激活独立于Nrf2信号传导的抗氧化途径,例如FoxO,从而弥补Nrf2的损失,并保证青春期生长过程中观察到的成骨细胞数量的恢复。IBANEZ等[49]有相同的观点,通过将6-9月龄的雌性Nrf2-/-小鼠和野生型小鼠随机分为卵巢切除术组和假手术组,发现Nrf2缺乏显著降低了骨小梁的体积、骨矿物质密度、骨体积分数和骨表面密度;椎骨组织形态计量学分析显示,皮质面积/总面积之比显著降低,骨小梁面积/总面积之比趋于降低。这些结果表明Nrf2是维持骨骼质量所必需的。 这与PARK等[35]的结论截然相反,KIM等[48]所用的实验动物为雌性小鼠,而PARK等[35]未说明其研究中所用动物的性别;此外,PARK等[35]仅检查了9周龄这一时间点,骨表型表现出性二态性,年龄和遗传背景等可能是造成实验结果差异的原因。 YIN等[50]通过适度激活Nrf2来研究不同性别小鼠中Nrf2对小鼠骨量的影响,将Keap1+/-小鼠即Keap1杂合小鼠(Keap1 Ht)作为Nrf2适度激活小鼠模型,Keap1-/-(KO)作为Nrf2过度激活小鼠模型,Keap1野生型(WT)作为对照组。结果表明,成年雄性Keap1 Ht小鼠的骨量更高,破骨细胞的细胞数减少,血清骨吸收标志物降低,说明Nrf2适度激活负调控破骨细胞生成和骨吸收,骨的形成率更高;使用颅骨骨细胞进行的细胞培养研究进一步表明,Keap1 Ht成骨细胞中的碱性磷酸酶和Wnt5a mRNA高于野生型成骨细胞,提示Nrf2适度激活会刺激成骨细胞分化;通过破坏Keap1激活Nrf2还在Keap1 Ht小鼠的骨表型中表现出性二态性,在雄性Keap1 Ht小鼠中骨质更高,但在雌性Keap1 Ht小鼠中却没有。这些数据表明Keap1/Nrf2信号通路在成骨细胞的分化和活性中起着重要作用,适度激活Nrf2可以增加骨量,降低骨吸收,但是Nrf2激活和性类固醇受体下游信号之间的串扰需要进一步研究。 "
| [1] Bone health and nutrition: Introduction to the special issue. Mol Cell Endocrinol. 2015;410:1-2. [2] KENSLER TW, WAKABAYASHI N, BISWAL S. Cell Survival Responses to Environmental Stresses Via the Keap1-Nrf2-ARE Pathway. Annu Rev Pharmacol Toxicol. 2007;47:89-116. [3] MOTOHASHI H, YAMAMOTO M. Nrf2-Keap1 defines a physiologically important stress response mechanism. Trends Mol Med. 2004;10(11): 549-557. [4] ITOH K, WAKABAYASHI N, KATOH Y, et al. Keap1 represses nuclear activation of antioxidant responsive elements by Nrf2 through binding to the amino-terminal Neh2 domain. Genes Dev. 1999;13(1):76-86. [5] BUGNO M, DANIEL M, CHEPELEV NL, et al. Changing gears in Nrf1 research, from mechanisms of regulation to its role in disease and prevention. Biochim Biophys Acta. 2015;1849(10):1260-1276. [6] KOPACZ A, KLOSKA D, FORMAN HJ, et al. Beyond repression of Nrf2: An update on Keap1. Free Radic Biol Med. 2020;157:63-74. [7] NAM LB, KEUM YS. Binding partners of NRF2: Functions and regulatory mechanisms. Arch Biochem Biophys. 2019;678:108184. [8] MOI P, CHAN K, ASUNIS I, et al. Isolation of NF-E2-related factor 2 (Nrf2), a NF-E2-like basic leucine zipper transcriptional activator that binds to the tandem NF-E2/AP1 repeat of the beta-globin locus control region. Proc Natl Acad Sci U S A. 1994;91(21):9926-9930. [9] LAU A, TIAN W, WHITMAN SA, et al. The Predicted Molecular Weight of Nrf2: It Is What It Is Not. Antioxid Redox Signal. 2013;18(1):91-93. [10] KATSUOKA F, YAMAMOTO M. Small Maf proteins (MafF, MafG, MafK): History, structure and function. Gene. 2016;586(2):197-205. [11] HAYES JD, MCMAHON M. NRF2 and KEAP1 mutations: permanent activation of an adaptive response in cancer. Trends Biochem Sci. 2009;34(4):176-188. [12] KIM JH, YU S, CHEN JD, et al. The nuclear cofactor RAC3/AIB1/SRC-3 enhances Nrf2 signaling by interacting with transactivation domains. Oncogene. 2013;32(4):514-527. [13] ZENKOV NK, KOZHIN PM, CHECHUSHKOV AV, et al. Mazes of Nrf2 Regulation. Biochemistry (Mosc). 2017;82(5):556-564. [14] RADA P, ROJO AI, CHOWDHRY S, et al. SCF/{beta}-TrCP promotes glycogen synthase kinase 3-dependent degradation of the Nrf2 transcription factor in a Keap1-independent manner. Mol Cell Biol. 2011;31(6):1121-1133. [15] CHOWDHRY S, ZHANG Y, MCMAHON M, et al. Nrf2 is controlled by two distinct β-TrCP recognition motifs in its Neh6 domain, one of which can be modulated by GSK-3 activity. Oncogene. 2013;32(32):3765-3781. [16] KUMAR H, KIM IS, MORE SV, et al. Natural product-derived pharmacological modulators of Nrf2/ARE pathway for chronic diseases. Nat Prod Rep. 2014;31(1):109-139. [17] SCHMIDLIN CJ, DODSON MB, MADHAVAN L, et al. Redox regulation by NRF2 in aging and disease. Free Radic Biol Med. 2019;134:702-707. [18] DINKOVA-KOSTOVA AT, HOLTZCLAW WD, COLE RN, et al. Direct evidence that sulfhydryl groups of Keap1 are the sensors regulating induction of phase 2 enzymes that protect against carcinogens and oxidants. Proc Natl Acad Sci U S A. 2002;99(18):11908-11913. [19] NEZU M, SUZUKI N, YAMAMOTO M. Targeting the KEAP1-NRF2 System to Prevent Kidney Disease Progression. Am J Nephrol. 2017;45(6): 473-483. [20] KEUM YS, CHOI BY. Molecular and chemical regulation of the Keap1-Nrf2 signaling pathway. Molecules. 2014;19(7):10074-10089. [21] KIMURA M, YAMAMOTO T, ZHANG J, et al. Molecular basis distinguishing the DNA binding profile of Nrf2-Maf heterodimer from that of Maf homodimer. J Biol Chem. 2015;290(17):10644. [22] GAN L, JOHNSON JA. Oxidative damage and the Nrf2-ARE pathway in neurodegenerative diseases. Biochim Biophys Acta. 2014;1842(8): 1208-1218. [23] KAGEYAMA S, SAITO T, OBATA M, et al. Negative Regulation of the Keap1-Nrf2 Pathway by a p62/Sqstm1 Splicing Variant. Mol Cell Biol. 2018;38(7):e00642-e00617. [24] ANDERICA C, HERNANDEZ J, VAZQUEZ I, et al. The MLN4924 inhibitor exerts a neuroprotective effect against oxidative stress injury via Nrf2 protein accumulation. Redox Biol. 2016;8:341-347. [25] WANG R, YU Z, SUNCHU B, et al. Rapamycin inhibits the secretory phenotype of senescent cells by a Nrf2-independent mechanism. Aging Cell. 2017;16(3):564-574. [26] TIAN D, SHI Y, CHEN D, et al. The Wnt inhibitor LGK-974 enhances radiosensitivity of HepG2 cells by modulating Nrf2 signaling. Int J Oncol. 2017;51(2):545-554. [27] MOBASHER A, GONZALEZ A, SANTAMARIA B, et al. Protein tyrosine phosphatase 1B modulates GSK3β/Nrf2 and IGFIR signaling pathways in acetaminophen-induced hepatotoxicity. Cell Death Dis. 2013;4(5): e626. [28] DODSON M, DE LA VEGA MR, CHOLANIANS AB, et al. Modulating NRF2 in Disease: Timing Is Everything. Annu Rev Pharmacol Toxicol. 2019;59:555-575. [29] LERNER UH. Osteoclasts in Health and Disease. Pediatr Endocrinol Rev. 2019;17(2):84-99. [30] AMIN N, BOCCARDI V, TAGHIZADEH M, et al. Probiotics and bone disorders: the role of RANKL/RANK/OPG pathway. Aging Clin Exp Res. 2020;32(3):363-371. [31] 谢冰洁,冯捷,韩向龙.破骨细胞生物学特征的研究与进展[J].中国组织工程研究,2017,21(11):1770-1775. [32] SUN X, ZHANG B, PAN X, et al. Octyl itaconate inhibits osteoclastogenesis by suppressing Hrd1 and activating Nrf2 signaling. Faseb J. 2019;33: 12929-12940. [33] KIM HJ, PARK C, KIM GY, et al. Sargassum serratifolium attenuates RANKL-induced osteoclast differentiation and oxidative stress through inhibition of NF-κB and activation of the Nrf2/HO-1 signaling pathway. Biosci Trends. 2018;12(3):257-265. [34] MA Q, LIANG M, TANG X, et al. Vitamin B5 inhibit RANKL induced osteoclastogenesis and ovariectomy induced osteoporosis by scavenging ROS generation. Am J Transl Res. 2019;11:5008-5018. [35] PARK CK, LEE Y, KIM KH, et al. Nrf2 is a novel regulator of bone acquisition. Bone. 2014;63:36-46. [36] CHUNG S, KIM S, SON M, et al. Inhibition of p300/CBP-Associated Factor Attenuates Renal Tubulointerstitial Fibrosis through Modulation of NF-kB and Nrf2. Int J Mol Sci. 2019;20(7):1554. [37] LI Z, CHEN C, ZHU X, et al. Glycyrrhizin Suppresses RANKL-Induced Osteoclastogenesis and Oxidative Stress Through Inhibiting NF-κB and MAPK and Activating AMPK/Nrf2. Calcif Tissue Int. 2018;103(3): 324-337. [38] KANZAKI H, SHINOHARA F, KAJIYA M, et al. The Keap1/Nrf2 protein axis plays a role in osteoclast differentiation by regulating intracellular reactive oxygen species signaling. J Biol Chem. 2013;288(32): 23009-23020. [39] HYEON S, LEE H, YANG Y, et al. Nrf2 deficiency induces oxidative stress and promotes RANKL-induced osteoclast differentiation. Free Radic Biol Med. 2013;65:789-799. [40] LI W, SUN Y. Nrf2 is required for suppressing osteoclast RANKL-induced differentiation in RAW 264.7 cells via inactivating cannabinoid receptor type 2 with AM630. Regen Ther. 2020;14:191-195. [41] LIU Z, HOU Y, LI L, et al. Nrf2 deficiency aggravates the increase in osteoclastogenesis and bone loss induced by inorganic arsenic. Toxicol Appl Pharmacol. 2019;367:62-70. [42] MAEDA K, KOBAYASHI Y, KOIDE M, et al. The Regulation of Bone Metabolism and Disorders by Wnt Signaling. Int J Mol Sci. 2019;20(22): 5525. [43] TRESGUERRES FGF, TORRES J, LÓPEZ-QUILES J, et al. The osteocyte: A multifunctional cell within the bone. Ann Anat. 2020;227:151422. [44] KOOK SH, KIM KA, JI H, et al. Irradiation inhibits the maturation and mineralization of osteoblasts via the activation of Nrf2/HO-1 pathway. Mol Cell Biochem. 2015;410(1-2):255-266. [45] HINOI E, FUJIMORI S, WANG L, et al. Nrf2 negatively regulates osteoblast differentiation via interfering with Runx2-dependent transcriptional activation. J Biol Chem. 2006;281(26):18015-18024. [46] YOSHIDA E, SUZUKI T, MORITA M, et al. Hyperactivation of Nrf2 leads to hypoplasia of bone in vivo. Genes Cells. 2018;23(5):386-392. [47] SUN YX, LI L, CORRY KA, et al. Deletion of Nrf2 reduces skeletal mechanical properties and decreases load-driven bone formation. Bone. 2015;74:1-9. [48] KIM JH, SINGHAL V, BISWAL S, et al. Nrf2 is required for normal postnatal bone acquisition in mice. Bone Res. 2014;2:14033. [49] IBANEZ L, FERRANDIZ ML, BRINES R, et al. Effects of Nrf2 deficiency on bone microarchitecture in an experimental model of osteoporosis. Oxid Med Cell Longev. 2014;2014:726590. [50] YIN Y, CORRY KA, LOUGHRAN JP, et al. Moderate Nrf2 Activation by Genetic Disruption of Keap1 Has Sex-Specific Effects on Bone Mass in Mice. Sci Rep. 2020;10(1):348. |
| [1] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhou Qian, Zhang Qiang, Chen Qiu. Human salivary components and osteoporosis/osteopenia [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1439-1444. |
| [2] | Jin Tao, Liu Lin, Zhu Xiaoyan, Shi Yucong, Niu Jianxiong, Zhang Tongtong, Wu Shujin, Yang Qingshan. Osteoarthritis and mitochondrial abnormalities [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1452-1458. |
| [3] | Zhang Lichuang, Xu Hao, Ma Yinghui, Xiong Mengting, Han Haihui, Bao Jiamin, Zhai Weitao, Liang Qianqian. Mechanism and prospects of regulating lymphatic reflux function in the treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1459-1466. |
| [4] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [5] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [6] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhang Qiang, Liu Jing, Shao Ming. Acupuncture for Parkinson’s disease: an insight into the action mechanism in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1272-1277. |
| [7] | Hui Xiaoshan, Bai Jing, Zhou Siyuan, Wang Jie, Zhang Jinsheng, He Qingyong, Meng Peipei. Theoretical mechanism of traditional Chinese medicine theory on stem cell induced differentiation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1125-1129. |
| [8] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [9] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1137-1142. |
| [10] | Guo Jia, Ding Qionghua, Liu Ze, Lü Siyi, Zhou Quancheng, Gao Yuhua, Bai Chunyu. Biological characteristics and immunoregulation of exosomes derived from mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1093-1101. |
| [11] | Wu Weiyue, Guo Xiaodong, Bao Chongyun. Application of engineered exosomes in bone repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1102-1106. |
| [12] | Zhou Hongqin, Wu Dandan, Yang Kun, Liu Qi. Exosomes that deliver specific miRNAs can regulate osteogenesis and promote angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1107-1112. |
| [13] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [14] | Huang Chenwei, Fei Yankang, Zhu Mengmei, Li Penghao, Yu Bing. Important role of glutathione in stemness and regulation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1119-1124. |
| [15] | Hu Weifan, Zheng Li, Li Dadi, Sun Yang, Zhao Fengchao. Overexpression of miR-25 downregulates titanium particle-induced osteoclast differentiation through the NFATc1 signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 682-687. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||