Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (1): 135-140.doi: 10.12307/2022.023
Previous Articles Next Articles
Research status and problems of stem cells and their derived exosomes for prevention and treatment of vascular restenosis
Shuai Zhiqin1, Chen Jiameng1, Liu Taotao1, Hu Anling1, Li Lisheng1, Yu Limei2, Xu Shangfu1, 2
- 1Key Laboratory of Basic Pharmacology of Ministry of Education and Joint International Research Laboratory of Ethnocentric of Ministry of Education, Zunyi Medical University, Zunyi 563003, Guizhou Province, China; 2Key Laboratory of Cell Engineering in Guizhou Province, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China
-
Received:2020-12-11Revised:2021-01-05Accepted:2021-02-07Online:2022-01-08Published:2021-10-25 -
Contact:Xu Shangfu, PhD, Associate professor, Key Laboratory of Basic Pharmacology of Ministry of Education and Joint International Research Laboratory of Ethnocentric of Ministry of Education, Zunyi Medical University, Zunyi 563003, Guizhou Province, China; Key Laboratory of Cell Engineering in Guizhou Province, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China -
About author:Shuai Zhiqin, Master candidate, Key Laboratory of Basic Pharmacology of Ministry of Education and Joint International Research Laboratory of Ethnocentric of Ministry of Education, Zunyi Medical University, Zunyi 563003, Guizhou Province, China -
Supported by:the National Natural Science Foundation of China, No. 81560592 (to XSF), No. 82060817 (to XSF); Academic Seedling Program of Zunyi Medical University, (to XSF)
CLC Number:
Cite this article
Shuai Zhiqin, Chen Jiameng, Liu Taotao, Hu Anling, Li Lisheng, Yu Limei, Xu Shangfu. Research status and problems of stem cells and their derived exosomes for prevention and treatment of vascular restenosis[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 135-140.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
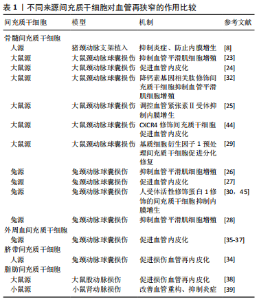
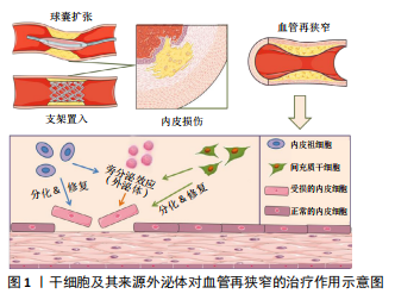
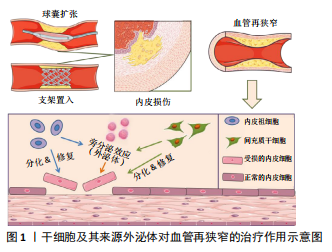
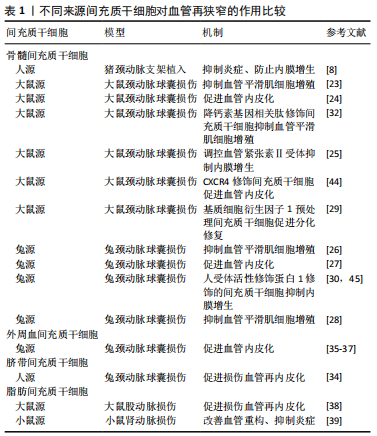
2.1 血管再狭窄形成 经皮冠状动脉血管腔内成形术所致的血管内皮细胞损伤是再狭窄发生的始动因素,早期血管弹性回缩,血小板聚集和附壁血栓形成,中晚期血管平滑肌细胞在多种反应性生长因子,如血小板衍生生长因子、碱性成纤维细胞生长因子、白细胞介素10、血管紧张素Ⅱ等的推动下,从血管中层向内膜下迁移,由收缩型向分泌型转化,并分泌细胞外基质从而形成新生内膜引起血管再狭窄[5-7]。血管内皮细胞是血管平滑肌细胞和循环中反应性生长刺激因子之间的屏障,可产生一氧化氮等多种活性因子。因此,血管内皮细胞损伤和血管平滑肌细胞表型转化是再狭窄形成的关键环节,而干细胞可有效抑制血管平滑肌细胞表型转化和异常增殖,也是防治再狭窄的重要策略。 2.2 干细胞移植防治血管再狭窄 干细胞具有自我复制能力和多潜能分化能力,可条件性分化为多种细胞。目前成功用于临床治疗的疾病包括恶性血液系统疾病,如白血病、淋巴瘤等;神经系统疾病,如脑损伤、阿尔茨海默病、帕金森综合征等;免疫系统疾病,如类风湿性关节炎、系统红斑狼疮等。在心血管系统,造血干细胞和间充质干细胞也都表现出其应用潜力。干细胞对器官和组织的功能维持起到重要作用。研究发现,外周血干/祖细胞参与血管形成、修复与重塑,并且可分化为多种与血管形成有关的细胞亚型,促进血管生 成[7-8]。研究认为参与血管损伤修复的干细胞主要为间充质干细胞,而内皮祖细胞对血管再狭窄也具有较好的预防和治疗作用。 2.2.1 内皮祖细胞防治血管再狭窄的研究 内皮祖细胞是1997年由日本学者ASAHARA等[9]首次从人外周血单个核细胞中成功分离出并能够表达血管内皮细胞特异性抗原的单核细胞。内皮祖细胞可以分化为成熟的血管内皮细胞,内皮祖细胞治疗为促进再内皮化提供了新的途径。骨髓来源内皮祖细胞移植已被证明具有明确的定植、分化为血管内皮细胞的作用,也有研究认为内皮祖细胞的分泌作用在血管损伤修复过程中具有重要作用[10]。骨髓来源内皮祖细胞分为早期 (3-6 d)、晚期(9-14 d)和极晚期(17-21 d),多数研究认为晚期内皮祖细胞移植促进内膜形成的作用最为显著,而且部分表达血小板-内皮细胞黏附分子的细胞可直接代替损伤的血管内皮细胞,移植后降低损伤血管中肿瘤坏死因子α、白细胞介素6、单核细胞趋化蛋白1、血管细胞黏附因子1的表达和线粒体成熟,促进再内皮化形成,但机制还有待进一步研究[11-12]。轴突导向蛋白netrin-1可改善内皮祖细胞功能,诱导内皮祖细胞归巢,经netrin-1预处理的内皮祖细胞移植通过NO/cGMP/p38 MAPK途径阻断血管平滑肌细胞的迁移和增殖[13]。还有研究发现,内皮祖细胞移植能减少动脉粥样硬化再狭窄模型兔血管内膜的炎症浸润,其调控作用与降低血小板释放的血小板衍生生长因子与血管内皮细胞生长因子的表达有关[14];携带人血栓调节蛋白基因的内皮祖细胞局部移植可有效预防动脉成形术后再狭窄,并在血管损伤部位得到Fe2O3磁性纳米颗粒标记的外源性内皮祖细胞定植的证据[15]。此外,内皮祖细胞动员和捕获内皮祖细胞支架也是血管再狭窄防治的研究热点[16-19],但有研究统计捕获内皮祖细胞支架仍存在不良事件发生[20]。 2.2.2 间充质干细胞防治血管再狭窄的研究 间充质干细胞是具有自我更新和多向分化能力的多能干细胞,其来源广泛。骨髓来源间充质干细胞的研究最早、最多,此外脐带、胎盘、羊膜、脂肪等也是间充质干细胞的重要来源[21]。研究已明确间充质干细胞可诱导分化为血管内皮细胞,在体外可形成血管移植物用于临床治疗缺血性疾病、血管组织工程及再生医学等领域[21-22]。研究发现,大鼠骨髓间充质干细胞移植治疗颈总动脉球囊损伤模型大鼠,可抑制血管平滑肌细胞增殖,促进损伤血管内皮化,调控血管紧张素Ⅱ受体而抑制再狭 窄[23-25]。兔腹主动脉球囊损伤后导致血管平滑肌细胞增殖及动脉管腔狭窄,经血管局部移植骨髓间充质干细胞可抑制血管平滑肌细胞增殖,减轻动脉粥样硬化及狭窄程度,降低平滑肌肌动蛋白、增殖细胞核抗原在血管内膜上的表达[26]。骨髓间充质干细胞可促进颈动脉球囊损伤后血管早期再内皮化和血管平滑肌细胞表型转化,抑制血管新生内膜增生,减轻血管再狭窄[27-28]。移植术后1周,损伤血管的内膜有骨髓间充质干细胞归巢,术后2周骨髓间充质干细胞移植组血管内膜有连续性CD31的表达,术后4周颈动脉造影结果显示:骨髓间充质干细胞移植组较对照组血管再狭窄率降低[27]。在猪颈动脉支架手术模型中,移植人骨髓间充质干细胞可抑制血管损伤引起的炎症,防止再狭窄的发生[8]。还有研究发现骨髓间充质干细胞经过基因修饰对血管再狭窄的治疗作用增强。基质细胞衍生因子预处理的骨髓间充质干细胞移植可归巢至损伤处并诱导细胞分化,抑制中膜血管平滑肌细胞增殖来减轻血管狭窄程度,显著提高损伤内膜CD31以及血管内皮生长因子水平,并抑制增殖细胞核抗原的表达[29]。兔颈动脉粥样硬化并球囊成形术后,采用人受体活性修饰蛋白1基因修饰的骨髓间充质干细胞移植治疗,发现归巢的干细胞增多,凋亡的血管平滑肌细胞增多,抑制血管平滑肌细胞增殖作用增强。人受体活性修饰蛋白1基因修饰的骨髓间充质干细胞更有效地抑制内膜增生,从而降低血管成形术后再狭窄的发生[30]。降钙素基因相关肽修饰的间充质干细胞移植能明显抑制体外培养血管平滑肌细胞的表型转化和增殖,这可能与上调降钙素基因相关肽受体表达,增强降钙素基因相关肽的产生有关[31-32]。研究发现,在猪支架植入模型中,人脐带间充质干细胞移植或联合血管内皮生长因子治疗,可以抑制支架引起的炎症反应,预防新生内膜增生,减少心脏支架植入术后再狭窄的发生[33-34]。兔颈动脉损伤模型中移植人脐带间充质干细胞,也可促进损伤血管再内皮化[34]。在兔颈动脉球囊损伤模型中移植兔外周血来源间充质干细胞,可促进损伤血管再内皮化,抑制再狭窄发生[35-37]。在大鼠股动脉损伤模型和小鼠肾动脉损伤模型中,移植大鼠或小鼠来源脂肪间充质干细胞,可改善血管重构,抑制炎症,促进损伤血管再内皮化而防止损伤血管发生再狭窄[38-39]。但也有研究表明损伤血管内皮分泌的转化生长因子β1通过Smad和基质金属蛋白酶14通路招募骨髓间充质干细胞集聚损伤处有关,并诱导骨髓间充质干细胞向内膜细胞分化,也可分化为血管平滑肌细胞,参与新生内膜的形成和血管重塑[40-43]。对于间充质干细胞治疗再狭窄的作用,部分研究认为是间充质干细胞直接分化为内皮细胞,促进血管再内皮化而抑制血管再狭窄;也有研究认为,治疗作用与间充质干细胞分泌功能有 关[23]。以上不同来源间充质干细胞对血管再狭窄的作用汇总见表1[8,23-30,32,34-39,44-45]。"

2.3 干细胞来源外泌体防治血管再狭窄的研究 研究认为内皮祖细胞、血管内皮细胞与血管平滑肌细胞之间存在信息传递,信息传递的介质主要是以Ca2+、IP3为代表的第二信使[46],以及内皮祖细胞、血管内皮细胞分泌释放的多种生物效应因子,包括血管内皮生长因子、血管紧张素Ⅱ、内皮素等。血管内皮细胞损伤也可引起血小板衍生生长因子的释放,炎症细胞释放炎症因子作用于血管平滑肌细胞,引起血管平滑肌细胞发生表型转化和异常增殖[47]。近年来的研究发现,外泌体是心血管系统细胞间信息传递的重要方式,其内包含多种蛋白质、脂质及核酸,介导细胞间的物质传递与信号转导,参与机体的生理功能和病理过程[48-49],是新的细胞间通讯系统——细胞外囊泡的一种。内容物不同的外泌体可作用于邻近或远距离靶细胞并介导细胞间信号传递,是心血管疾病诊断和治疗的新策略,也是相关研究的新靶标。干细胞来源外泌体是其发挥作用的重要机制之一。研究表明,干细胞来源外泌体通过运输释放各种特有的生物活性分子,如特有的蛋白质、mRNA和microRNA等改变靶细胞基因调控网络发挥生物学功能。外泌体在干细胞和受损细胞间的信息传递是双向性的,受损细胞分泌含有细胞特异性蛋白、microRNA的外泌体,被干细胞内化后诱导其重组分化特征性表型,再由干细胞分泌外泌体,调控受损细胞相关基因表达,促进受损细胞再生和修复。 2.3.1 内皮祖细胞来源外泌体及其对血管再狭窄的作用 随着内皮祖细胞研究不断地丰富和深入,内皮祖细胞来源外泌体现也备受关注,被认为是内皮祖细胞治疗疾病的重要机制之一。近年来,内皮祖细胞来源外泌体在多种疾病模型中表现出积极的内皮修复功能,如血管内皮细胞缺氧性损伤[50]、皮肤创伤性损伤[51]、脑损伤[52]、急性肺损伤等[53]。在血管损伤模型中,内皮祖细胞来源外泌体可减弱内皮细胞缺氧性损伤,其机制主要通过改善其增殖、迁移能力从而减少内皮细胞的凋亡[50]。在缺血再灌注损伤等疾病中,内皮祖细胞来源外泌体中的microRNA分子起到促进血管再生作用,降低缺血缺氧微环境带来的损伤风险并促进组织再生修复[50]。在大鼠皮肤创伤性损伤模型中,内皮祖细胞来源外泌体对内皮细胞具有促进修复的功能,其作用机制与促进缺损皮肤愈合、增强皮肤组织胶原成熟以及上调血管内皮生长因子A表达水平有关[51],并且还具有抗炎、抗氧化、抗凋亡和保护血管内膜作用。脑损伤模型中,通过尾静脉向创伤小鼠注射内皮祖细胞来源外泌体,发现脑创伤灶局部血管新生增强,脑组织灌流量增加,神经修复加快[52],从而减轻脑组织水肿程度,减少神经细胞凋亡;内皮祖细胞来源外泌体促进血管新生的机制可能是其分泌血管内皮生长因子,促进基质金属蛋白酶9表达,抑制抑癌基因PTEN表达[54]。在急性肺损伤模型中,由于各种非心源性因素引起肺组织结构发生特征性病理改变,包括肺毛细血管内皮细胞和肺泡上皮细胞损伤所造成的弥漫性肺间质及肺泡水肿导致的低氧性呼吸功能不全或衰竭,其主要标志为微循环血管内皮损伤[53]。内皮祖细胞来源外泌体可促进新生血管生成从而减缓器官的功能障碍,同时促血管生成相关蛋白表达增加,包括内皮型一氧化氮合酶、血管内皮生长因子和血管内皮生长因子受体2 [55]。用脂多糖诱导大鼠急性肺损伤模型后,通过静脉注射途径将内皮祖细胞来源外泌体移植于大鼠体内,结果证明内皮祖细胞来源外泌体可有效改善大鼠肺损伤,其机制可能是内皮祖细胞来源外泌体将miR-126转移至靶血管内皮细胞后,导致SPRED1表达下调从而激活RAF/ERK信号通路,减轻肺血管的渗透性,减少肺含水量,最终改善急性肺损伤[56]。由此可见,内皮祖细胞来源外泌体在血管生成、皮肤创伤、肺损伤及脑损伤方面均具有较好的作用,其机制均与其促进血管内皮修复,改善损伤微环境有关。除此之外,外泌体内含物也在这些相关模型治疗中起到重要作用。研究发现,内皮祖细胞来源外泌体分泌的miR-218 能够促进肾微血管损伤修复;内皮祖细胞来源外泌体转染miR-210后能更好地解决缺氧诱导的血管内皮细胞存活率下降和血管生成功能障碍的问题;内皮祖细胞来源外泌体中miR-126部分增强RAF/ERK信号通路,从而促进细胞增殖、迁移和血管生成。此外,内皮祖细胞来源外泌体分泌的miR-21-5p、miR-221等也参与血管生成、血管内膜修复[50-53]。 在血管再狭窄损伤模型中,研究发现内皮祖细胞本身和分化成熟的血管内皮细胞来源外泌体均可被血管平滑肌细胞内化并影响其表型和功能。血管内皮细胞衍生的囊泡和外泌体含有血管平滑肌细胞表型调控的miRNA和蛋白[57-58],血管内皮细胞来源外泌体明显抑制颈动脉内皮损伤后的血管平滑肌细胞表型转化和血管增生内膜形成,其机制与CD137信号通路调控血管内皮细胞来源外泌体中的平滑肌表型调节蛋白TET2向血管平滑肌细胞转移有关[59]。高糖刺激的血管内皮细胞来源外泌体富含Notch3蛋白,被血管平滑肌细胞内化,并通过mTOR信号通路增加骨钙素和P21表达,从而促进血管平滑肌细胞钙化/老化[60]。骨髓源性内皮祖细胞对血管平滑肌细胞的表型转化和迁移具有抑制作用,其机制与增加共培养体系中一氧化氮和血管内皮生长因子有关[61-62]。内皮祖细胞来源外泌体可增强血管内皮细胞的分泌功能,并且促进球囊损伤大鼠的血管修复,其主要机制是内皮祖细胞来源外泌体促进内皮损伤后早期的再内皮化[63-65]。还有研究显示内皮祖细胞来源外泌体在体外能促进血管平滑肌细胞的增殖和迁移[66],分析其原因可能与内皮祖细胞所处的分化状态有关。 2.3.2 间充质干细胞来源外泌体及其对血管再狭窄的作用 间充质干细胞来源外泌体是研究最多的干细胞来源外泌体,具有与间充质干细胞相似的组织损伤修复和再生功能,并且具有重要的免疫调节作用。间充质干细胞来源外泌体涉及伤口愈合和皮肤再生、肿瘤微环境、神经损伤、肝肾损伤、心血管系统疾病、肠炎、软骨损伤等研究领域。越来越多的证据表明,间充质干细胞的免疫抑制主要归因于间充质干细胞衍生的细胞外囊泡及外泌体的作用,尤其是外泌体。研究表明,间充质干细胞来源外泌体局部和全身给药均能有效抑制炎症组织的有害免疫反应,促进损伤实质细胞的存活和再生。间充质干细胞来源外泌体的抗炎作用依赖于在炎症免疫细胞(M1巨噬细胞、树突状细胞、CD4+的Th1和Th17细胞)中传递免疫调节miRNA和免疫调节蛋白,使其表型转化为免疫抑制的M2巨噬细胞、耐受性树突状细胞和调节性T细胞[67]。此外,间充质干细胞来源外泌体通过调控mRNA和miRNA,在损伤的肝细胞、神经元、视网膜细胞、肺、肠和肾上皮细胞中激活自噬和/或抑制凋亡、坏死和氧化应激,促进其存活和再生[67]。脑出血模型大鼠给予脂肪间充质干细胞来源外泌体治疗后,大脑功能明显改善,表现出具有更多的轴突,高表达的少突胶质细胞形成标记物和更多的髓鞘染色[68]。人骨髓间充质干细胞来源外泌体对小鼠结肠炎模型具有显著改善作用,骨髓间充质干细胞来源外泌体通过下调促炎细胞因子水平、抑制NF-κBp65信号转导途径、调节抗氧化/氧化平衡和抑制细胞凋亡的发生而实现对结肠炎的保护作用[69-70]。 研究表明,骨髓间充质干细胞来源外泌体在体外具有软骨保护和抗炎作用,在体内对小鼠骨关节炎有保护作用[71]。研究者采用白细胞介素1β对人脐带间充质干细胞进行处理后发现miR-146a的表达显著增加,其外泌体能有效促进败血症小鼠肝和肺组织中的巨噬细胞从M1型向抗炎M2型极化,明显改善小鼠败血症的症状,从而降低小鼠死亡率[72]。 间充质干细胞来源外泌体主要通过调节炎症免疫、微环境等机制对急性心肌梗死、脑卒中、肺动脉高压和败血性心肌病等都具有治疗作用[73]。脂肪间充质干细胞来源外泌体通过激活S1P/SK1/S1PR1信号转导途径改善缺氧条件下诱导的H9c2细胞凋亡、转化生长因子β1诱导的心肌成纤维细胞纤维化以及巨噬细胞M1极化导致的心脏损害,同时促进巨噬细胞M2极化,进而改善心肌梗死后出现的心脏纤维化及炎症反应[74];在大鼠急性心肌梗死模型局部梗死区注射骨髓间充质干细胞来源外泌体后,能明显降低梗死区心肌的炎症细胞浸润,促进新生血管形成,恢复血流,从而改善心功能[75];大鼠结扎左冠状动脉前降支建立大鼠心肌缺血再灌注模型,骨髓间充质干细胞来源外泌体可减轻大鼠缺血再灌注心肌组织的病理损伤,并且发现携带miR-125b的骨髓间充质干细胞来源外泌体可通过下调长寿蛋白家族(Sirtuins)成员SIRT7保护心肌缺血再灌注损伤[76]。研究也发现人脂肪间充质干细胞来源外泌体通过携带高表达的miR-125a抑制δ样蛋白4的表达,同时Dil标记的外泌体可以转移到人脐静脉内皮细胞的胞质中,促进血管生成[77]。 最新研究发现间充质干细胞来源外泌体对再狭窄有防治作用。在体外研究中,间充质干细胞来源外泌体减轻了低氧低糖诱导的内皮细胞活力下降,减少细胞凋亡、炎症因子水平,抑制炎症通路的激活,从而修复内皮细胞损伤[78];此外也有研究发现,间充质干细胞来源外泌体在体外被血管平滑肌细胞内化并抑制其增殖和迁移[79]。体内实验进一步分析显示miR-125b在间充质干细胞来源外泌体中富集,并通过靶向其3’非翻译区抑制肌球蛋白1E的表达。此外,间充质干细胞来源外泌体释放的miR-125b在体内抑制Myo1e表达,抑制血管平滑肌细胞增殖、迁移和内膜增生[79]。骨髓间充质干细胞来源外泌体抑制大鼠颈动脉球囊损伤后内膜增生,其潜在机制可能与长链非编码RNA XR602080、XR591017通过糖原合成酶激酶3β、周期素依赖性激酶抑制因子2B、细胞周期素A2等分子调控平滑肌细胞周期变化有关[80]。此外,也有研究显示间充质干细胞来源外泌体激活ERK1/2信号通路,加速再内皮化抑制颈动脉损伤后内膜增生[81],也为介入治疗后再狭窄疾病提供了一种新的无细胞治疗方法。间充质干细胞、内皮祖细胞及其外泌体移植防治血管再狭窄的作用及机制汇总见表2及图1。"

| [1] 马丽媛,吴亚哲,王文,等.《中国心血管病报告2017》要点解读[J].中国心血管杂志,2018,23(1):3-6. [2] SIMARD T, HIBBERT B, RAMIREZ FD, et al. The evolution of coronary stents: a brief review. Can J Cardiol. 2014;30(1):35-45. [3] 谢秀峰,张越,曲泽.药物洗脱球囊和药物洗脱支架对支架内再狭窄的疗效比较[J].中国组织工程研究,2020,24(4):555-560. [4] 姚雯,赵金河.冠心病介入治疗术后再狭窄防治的研究进展[J].中西医结合心脑血管病杂志,2018,16(23):3456-3458. [5] ZHANG J, GAO F, NI T, et al. Linc-POU3F3 is overexpressed in in-stent restenosis patients and induces VSMC phenotypic transformation via POU3F3/miR-449a/KLF4 signaling pathway. Am J Transl Res. 2019; 11(7):4481-4490. [6] GAO Y, ZHU P, XU SF, et al. Ginsenoside Re inhibits PDGF-BB-induced VSMC proliferation via the eNOS/NO/cGMP pathway. Biomed Pharmacother. 2019;115:108934. [7] 罗靖莹,郭阳,黄玮玮,等.间充质干细胞传递线粒体对肺微血管内皮细胞的修复作用[J].医学研究生学报,2019,32(9):931-936. [8] NAKAZAKI M, OKA S, SASAKI M, et al. Prevention of neointimal hyperplasia induced by an endovascular stent via intravenous infusion of mesenchymal stem cells. J Neurosurg. 2019;133(6):1-13. [9] ASAHARA T, MUROHARA T, SULLIVAN A, et al. Isolation of putative progenitor endothelial cells for angiogenesis. Science. 1997;275(5302): 964-967. [10] BRIASOULIS A, TOUSOULIS D, ANTONIADES C, et al. The role of endothelial progenitor cells in vascular repair after arterial injury and atherosclerotic plaque development. Cardiovasc Ther. 2011;29(2): 125-139. [11] IKUTOMI M, SAHARA M, NAKAJIMA T, et al. Diverse contribution of bone marrow-derived late-outgrowth endothelial progenitor cells to vascular repair under pulmonary arterial hypertension and arterial neointimal formation. J Mol Cell Cardiol. 2015;86:121-135. [12] XU RW, ZHANG WJ, ZHANG JB, et al. A Preliminary Study of the Therapeutic Role of Human Early Fetal Aorta-derived Endothelial Progenitor Cells in Inhibiting Carotid Artery Neointimal Hyperplasia. Chin Med J (Engl). 2015;128(24):3357-3362. [13] LIU NM, SIU KL, YOUN JY, et al. Attenuation of neointimal formation with netrin-1 and netrin-1 preconditioned endothelial progenitor cells. J Mol Med (Berl). 2017;95(3):335-348. [14] 曾贵荣,罗桂芳,周仕达,等.内皮祖细胞对实验兔动脉粥样硬化再狭窄模型调控功能的研究[J].中国药理学与毒理学杂志,2016, 30(10):1005. [15] 丁蓉蓉,赵国峰,许荣睿,等.携人血栓调节蛋白基因内皮祖细胞预防动脉成形术后再狭窄[J].中华实验外科杂志,2017,34(5):740-743. [16] BARSOTTI MC, DI STEFANO R, SPONTONI P, et al. Role of endothelial progenitor cell mobilization after percutaneous angioplasty procedure. Curr Pharm Des. 2009;15(10):1107-1122. [17] HU A, HUANG J, LI S, et al. Involvement of stromal cell-derived factor-1α (SDF-1α), stem cell factor (SCF), fractalkine (FKN) and VEGF in TSG protection against intimal hyperplasia in rat balloon injury. Biomed Pharmacother. 2019;110:887-894. [18] KLOMP M, BEIJK MA, DE WINTER RJ. Genous endothelial progenitor cell-capturing stent system: a novel stent technology. Expert Rev Med Devices. 2009;6(4):365-375. [19] DUAN Y, YU S, XU P, et al. Co-immobilization of CD133 antibodies, vascular endothelial growth factors, and REDV peptide promotes capture, proliferation, and differentiation of endothelial progenitor cells. Acta Biomater. 2019;96:137-148. [20] XU WY, TANG WL, YUAN M, et al. The Adverse Events Rate of Endothelial Progenitor Cell Capturing Stent in the Treatment of CAD Patients. Comb Chem High Throughput Screen. 2018;21(10):725-733. [21] BEREBICHEZ-FRIDMAN R, MONTERO-OLVERA PR. Sources and Clinical Applications of Mesenchymal Stem Cells: State-of-the-art review. Sultan Qaboos Univ Med J. 2018;18(3):e264-e277. [22] 王俊力,许慧芳,冯莉,等.不同组织来源间充质干细胞向血管内皮细胞诱导分化的研究与进展[J].中国组织工程研究,2016,20(36): 5466-5472. [23] ISO Y, USUI S, TOYODA M, et al. Bone marrow-derived mesenchymal stem cells inhibit vascular smooth muscle cell proliferation and neointimal hyperplasia after arterial injury in rats. Biochem Biophys Rep. 2018;16:79-87. [24] SONG BW, KIM IK, LEE S, et al. 1H-pyrrole-2,5-dione-based small molecule-induced generation of mesenchymal stem cell-derived functional endothelial cells that facilitate rapid endothelialization after vascular injury. Stem Cell Res Ther. 2015;6(1):174. [25] 景涛,何国祥,苗莉,等.间充质干细胞移植实现AT2R基因在体可调控表达在再狭窄形成的作用及机制[J].中国介入心脏病学杂志, 2010,18(5):284-289. [26] 王庆元,吴向军,张晓燕,等.骨髓间充质干细胞局部移植对兔腹主动脉成形术后再狭窄和平滑肌细胞增殖的影响[J].中国老年学杂志,2017,37(4):814-816. [27] 刘志江,石蓓,许官学,等.骨髓间充质干细胞移植对兔颈动脉球囊损伤后再狭窄的影响[J].中国动脉硬化杂志,2013,21(1):22-27. [28] 吴向军, 宋艳丽, 耿文真, 等.骨髓间充质干细胞移植对兔腹主动脉成形术后血管内膜的影响[J]. 中华老年心脑血管病杂志,2015, 17(6):634-637. [29] 束波,范芳.基质细胞衍生因子1(SDF-1)预处理的骨髓间充质干细胞移植减轻大鼠颈动脉内皮和平滑肌细胞增生[J].细胞与分子免疫学杂志,2019,5(4):320-326. [30] 赵然尊,龙仙萍,许官学,等.受体活性修饰蛋白-1转染间充质干细胞对兔血管成形术后血管平滑肌细胞增殖和凋亡的影响[J].中华老年医学杂志,2014,33(10):1127-1131. [31] 龙仙萍,赵然尊,石蓓,等. hRAMP1修饰的骨髓间充质干细胞移植对兔球囊损伤血管再狭窄及心功能的影响[J].中华医学杂志, 2012,92(30):2134-2139. [32] CHEN P, HE F, LIU T, et al. Construction of calcitonin gene-related peptide-modified mesenchymal stem cells and analysis of their effects on the migration and proliferation of vascular smooth muscle cells. In Vitro Cell Dev Biol Anim. 2020;56(2):181-191. [33] CHANG HK, KIM PH, KIM DW, et al. Coronary stents with inducible VEGF/HGF-secreting UCB-MSCs reduced restenosis and increased re-endothelialization in a swine model. Exp Mol Med. 2018;50(9):114. [34] KIM AK, KIM MH, KIM DH, et al. Inhibitory effects of mesenchymal stem cells in intimal hyperplasia after balloon angioplasty. J Vasc Surg. 2016;63(2):510-517. [35] 郭艳,石蓓,王正龙,等.外周血间充质干细胞移植对球囊损伤兔血管平滑肌细胞凋亡的影响[J].中国生物医学工程学报,2010,29(2): 288-294. [36] 郭艳,石蓓,赵然尊,等.外周血间充质干细胞移植防治兔血管成形术后再狭窄的探讨[J].中华老年心脑血管病杂志,2009,11(12):968-971. [37] 石蓓,郭艳,王正龙,等.缺氧预处理外周间充质干细胞对兔血管成形术后再狭窄的影响[J].中国病理生理杂志,2009,25(9):1686-1691. [38] HOMMA J, SEKINE H, MATSUURA K, et al. Mesenchymal Stem Cell Sheets Exert Antistenotic Effects in a Rat Arterial Injury Model. Tissue Eng Part A. 2018;24(19-20):1545-1553. [39] CAI C, KILARI S, ZHAO C, et al. Therapeutic Effect of Adipose Derived Mesenchymal Stem Cell Transplantation in Reducing Restenosis in a Murine Angioplasty Model. J Am Soc Nephrol. 2020;31(8):1781-1795. [40] 郑仕杰,周敬群,杨维华,等.血管损伤后TGF-β1/Smad信号通路在诱导BMSCs分化参与血管再狭窄的相关性研究[J].中国医药科学,2019,9(17):50-54. [41] 魏翠蕾,邹泓,段军仓,等. TGF-β1/Smad信号通路的激活对招募间充质干细胞在血管损伤中的表达变化及意义[J].石河子大学学报(自然科学版),2014,32(6):720-726. [42] PANG L, WEI C, DUAN J, et al. TGF-β1/Smad signaling, MMP-14, and MSC markers in arterial injury: discovery of the molecular basis of restenosis. Int J Clin Exp Pathol. 2014;7(6):2915-2924. [43] ZHAO W, WANG C, LIU R, et al. Effect of TGF-β1 on the Migration and Recruitment of Mesenchymal Stem Cells after Vascular Balloon Injury: Involvement of Matrix Metalloproteinase-14. Sci Rep. 2016;6:21176. [44] 刘志江,石蓓,许官学,等.CXCR4基因修饰骨髓间充质干细胞移植对损伤血管内皮化的影响[J].临床心血管病杂志,2014,30(12): 1094-1098. [45] 刘志江,石蓓,许官学,等.骨髓间充质干细胞移植后颈动脉损伤球囊核转录因子κBp65和增殖细胞核抗原的表达[J].中国组织工程研究与临床康复,2011,15(10):1814-1818. [46] SKERRETT IM, WILLIAMS JB. A structural and functional comparison of gap junction channels composed of connexins and innexins. Dev Neurobiol. 2017;77(5):522-547. [47] LAO KH, ZENG L, XU Q. Endothelial and smooth muscle cell transformation in atherosclerosis. Curr Opin Lipidol. 2015;26(5):449-456. [48] ZHANG Y, HU YW, ZHENG L, et al. Characteristics and Roles of Exosomes in Cardiovascular Disease. DNA Cell Biol. 2017;36(3):202-211. [49] Pankajakshan D, Agrawal DK. Mesenchymal Stem Cell Paracrine Factors in Vascular Repair and Regeneration. J Biomed Technol Res. 2014;1(1):10. [50] 王刚,时光旭,韩骅,等.内皮祖细胞来源外泌体在血管生成中作用的研究进展[J].心脏杂志,2019,31(5):588-591. [51] 潘满昌,林晓莹,汪虹,等.血管内皮祖细胞源性外泌体在创面修复中的作用研究进展[J].中华烧伤杂志,2020,36(9):883-886. [52] 谢川平,陈佳树,陈天,等.内皮祖细胞治疗脑创伤的研究进展[J].创伤外科杂志,2019,21(3):77-80. [53] 郭桢楠,马雪,丁文刚.内皮祖细胞在急性肺损伤中应用的研究进展[J].中国现代医学杂志,2021,31(3):65-69. [54] MALANEY P, UVERSKY VN, DAVÉ V. PTEN proteoforms in biology and disease. Cell Mol Life Sci. 2017;74(15):2783-2794. [55] ZHANG X, LU A, LI Z, et al. Exosomes secreted by endothelial progenitor cells improve the bioactivity of pulmonary microvascular endothelial cells exposed to hyperoxia in vitro. Ann Transl Med. 2019;7(12):254. [56] WU X, LIU Z, HU L, et al. Exosomes derived from endothelial progenitor cells ameliorate acute lung injury by transferring miR-126. Exp Cell Res. 2018;370(1):13-23. [57] HERGENREIDER E, HEYDT S, TRÉGUER K, et al. Atheroprotective communication between endothelial cells and smooth muscle cells through miRNAs. Nat Cell Biol. 2012;14(3):249-256. [58] TOGLIATTO G, DENTELLI P, ROSSO A, et al. PDGF-BB Carried by Endothelial Cell-Derived Extracellular Vesicles Reduces Vascular Smooth Muscle Cell Apoptosis in Diabetes. Diabetes. 2018;67(4):704-716. [59] LI B, ZANG G, ZHONG W, et al. Activation of CD137 signaling promotes neointimal formation by attenuating TET2 and transferrring from endothelial cell-derived exosomes to vascular smooth muscle cells. Biomed Pharmacother. 2020;121:109593. [60] LIN X, LI S, WANG YJ, et al. Exosomal Notch3 from high glucose-stimulated endothelial cells regulates vascular smooth muscle cells calcification/aging. Life Sci. 2019;232:116582. [61] 桑宏飞.内皮祖细胞对血管平滑肌细胞功能及静脉血栓后管壁重塑的影响[D].苏州:苏州大学,2014. [62] 方立.内皮祖细胞对血管平滑肌细胞增殖与表型转化的抑制作用及其机制研究[D].长沙:湘雅医院,2010. [63] LI X, CHEN C, WEI L, et al. Exosomes derived from endothelial progenitor cells attenuate vascular repair and accelerate reendothelialization by enhancing endothelial function. Cytotherapy. 2016;18(2):253-262. [64] HU H, JIANG C, LI R, et al. Comparison of endothelial cell- and endothelial progenitor cell-derived exosomes in promoting vascular endothelial cell repair. Int J Clin Exp Pathol. 2019;12(7):2793-2800. [65] HOU YC, LI JA, ZHU SJ, et al. Tailoring of cardiovascular stent material surface by immobilizing exosomes for better pro-endothelialization function. Colloids Surf B Biointerfaces. 2020;189:110831. [66] KONG J, WANG F, ZHANG J, et al. Exosomes of Endothelial Progenitor Cells Inhibit Neointima Formation After Carotid Artery Injury. J Surg Res. 2018;232:398-407. [67] HARRELL CR, JOVICIC N, DJONOV V, et al. Mesenchymal Stem Cell-Derived Exosomes and Other Extracellular Vesicles as New Remedies in the Therapy of Inflammatory Diseases. Cells. 2019;8(12):1605. [68] OTERO-ORTEGA L, GÓMEZ DE FRUTOS MC, LASO-GARCÍA F, et al. Exosomes promote restoration after an experimental animal model of intracerebral hemorrhage. J Cereb Blood Flow Metab. 2018;38(5):767-779. [69] LIU H, LIANG Z, WANG F, et al. Exosomes from mesenchymal stromal cells reduce murine colonic inflammation via a macrophage-dependent mechanism. JCI Insight. 2019;4(24):e131273. [70] YANG J, LIU XX, FAN H, et al. Extracellular Vesicles Derived from Bone Marrow Mesenchymal Stem Cells Protect against Experimental Colitis via Attenuating Colon Inflammation, Oxidative Stress and Apoptosis. PLoS One. 2015;10(10):e0140551. [71] COSENZA S, RUIZ M, TOUPET K, et al. Mesenchymal stem cells derived exosomes and microparticles protect cartilage and bone from degradation in osteoarthritis. Sci Rep. 2017;7(1):16214. [72] SONG Y, DOU H, LI X, et al. Exosomal miR-146a Contributes to the Enhanced Therapeutic Efficacy of Interleukin-1β-Primed Mesenchymal Stem Cells Against Sepsis. Stem Cells. 2017;35(5):1208-1221. [73] SUZUKI E, FUJITA D, TAKAHASHI M, et al. Therapeutic Effects of Mesenchymal Stem Cell-Derived Exosomes in Cardiovascular Disease. Adv Exp Med Biol. 2017;998:179-185. [74] DENG S, ZHOU X, GE Z, et al. Exosomes from adipose-derived mesenchymal stem cells ameliorate cardiac damage after myocardial infarction by activating S1P/SK1/S1PR1 signaling and promoting macrophage M2 polarization. Int J Biochem Cell Biol. 2019;114:105564. [75] TENG X, CHEN L, CHEN W, et al. Mesenchymal Stem Cell-Derived Exosomes Improve the Microenvironment of Infarcted Myocardium Contributing to Angiogenesis and Anti-Inflammation. Cell Physiol Biochem. 2015;37(6):2415-2424. [76] CHEN Q, LIU Y, DING X, et al. Bone marrow mesenchymal stem cell-secreted exosomes carrying microRNA-125b protect against myocardial ischemia reperfusion injury via targeting SIRT7. Mol Cell Biochem. 2020;465(1-2):103-114. [77] LIANG X, ZHANG L, WANG S, et al. Exosomes secreted by mesenchymal stem cells promote endothelial cell angiogenesis by transferring miR-125a. J Cell Sci. 2016;129(11):2182-2189. [78] KONG LY, LIANG MY, LIU JP, et al. Mesenchymal Stem Cell-derived Exosomes Rescue Oxygen-Glucose Deprivation-induced Injury in Endothelial Cells. Curr Neurovasc Res. 2020;17(2):155-163. [79] WANG D, GAO B, YUE J, et al. Exosomes from mesenchymal stem cells expressing miR-125b inhibit neointimal hyperplasia via myosin IE. J Cell Mol Med. 2019;23(2):1528-1540. [80] 龙禹哲,郭鸿浩,杨双亚,等.骨髓间充质干细胞源外泌体对大鼠颈动脉球囊损伤后lncRNAs表达的影响[J].第三军医大学学报, 2020,42(23):2320-2329. [81] LIU Z, WU C, ZOU X, et al. Exosomes derived from mesenchymal stem cells inhibit neointimal hyperplasia by activating the Erk1/2 signalling pathway in rats. Stem Cell Res Ther. 2020;11(1):220. [82] PHINNEY DG, PITTENGER MF. Concise Review: MSC-Derived Exosomes for Cell-Free Therapy. Stem Cells. 2017;35(4):851-858. [83] WERNLY B, MIRNA M, REZAR R, et al. Regenerative Cardiovascular Therapies: Stem Cells and Beyond. Int J Mol Sci. 2019;20(6):1420. |
| [1] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1316-1322. |
| [2] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1323-1329. |
| [3] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhang Qiang, Liu Jing, Shao Ming. Acupuncture for Parkinson’s disease: an insight into the action mechanism in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1330-1335. |
| [4] | Tian Chuan, Zhu Xiangqing, Yang Zailing, Yan Donghai, Li Ye, Wang Yanying, Yang Yukun, He Jie, Lü Guanke, Cai Xuemin, Shu Liping, He Zhixu, Pan Xinghua. Bone marrow mesenchymal stem cells regulate ovarian aging in macaques [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1033-1039. |
| [5] | Hou Jingying, Guo Tianzhu, Yu Menglei, Long Huibao, Wu Hao. Hypoxia preconditioning targets and downregulates miR-195 and promotes bone marrow mesenchymal stem cell survival and pro-angiogenic potential by activating MALAT1 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1053-1059. |
| [6] | Zhou Ying, Zhang Huan, Liao Song, Hu Fanqi, Yi Jing, Liu Yubin, Jin Jide. Immunomodulatory effects of deferoxamine and interferon gamma on human dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1060-1067. |
| [7] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1183-1190. |
| [8] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1191-1197. |
| [9] | Liang Xuezhen, Yang Xi, Li Jiacheng, Luo Di, Xu Bo, Li Gang. Bushen Huoxue capsule regulates osteogenic and adipogenic differentiation of rat bone marrow mesenchymal stem cells via Hedgehog signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1068-1074. |
| [10] | Wang Jifang, Bao Zhen, Qiao Yahong. miR-206 regulates EVI1 gene expression and cell biological behavior in stem cells of small cell lung cancer [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1075-1079. |
| [11] | Liu Feng, Peng Yuhuan, Luo Liangping, Wu Benqing. Plant-derived basic fibroblast growth factor maintains the growth and differentiation of human embryonic stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1080-1085. |
| [12] | Wang Xinmin, Liu Fei, Xu Jie, Bai Yuxi, Lü Jian. Core decompression combined with dental pulp stem cells in the treatment of steroid-associated femoral head necrosis in rabbits [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1122-1127. |
| [13] | Fang Xiaolei, Leng Jun, Zhang Chen, Liu Huimin, Guo Wen. Systematic evaluation of different therapeutic effects of mesenchymal stem cell transplantation in the treatment of ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1133-1140. |
| [14] | Guo Jia, Ding Qionghua, Liu Ze, Lü Siyi, Zhou Quancheng, Gao Yuhua, Bai Chunyu. Biological characteristics and immunoregulation of exosomes derived from mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1141-1150. |
| [15] | Wu Weiyue, Guo Xiaodong, Bao Chongyun. Application of engineered exosomes in bone repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1151-1155. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||