Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (35): 5709-5715.doi: 10.12307/2021.304
Previous Articles Next Articles
Regulatory role of microRNAs in the occurrence and development of osteoarthritis
Wang Weikang, Liu Xiaodong, Zhou Changlin, Tian Jun
- Department of Orthopedics, The Second Affiliated Hospital of Harbin Medical University, Harbin 150000, Heilongjiang Province, China
-
Received:2020-11-07Revised:2020-11-26Accepted:2020-12-31Online:2021-12-18Published:2021-08-05 -
Contact:Tian Jun, Master, Professor, Chief physician, Department of Orthopedics, The Second Affiliated Hospital of Harbin Medical University, Harbin 150000, Heilongjiang Province, China -
About author:Wang Weikang, Master candidate, Department of Orthopedics, The Second Affiliated Hospital of Harbin Medical University, Harbin 150000, Heilongjiang Province, China
CLC Number:
Cite this article
Wang Weikang, Liu Xiaodong, Zhou Changlin, Tian Jun. Regulatory role of microRNAs in the occurrence and development of osteoarthritis[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(35): 5709-5715.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
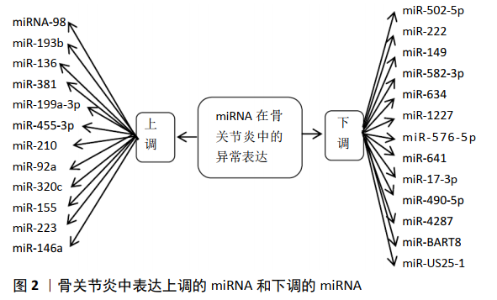
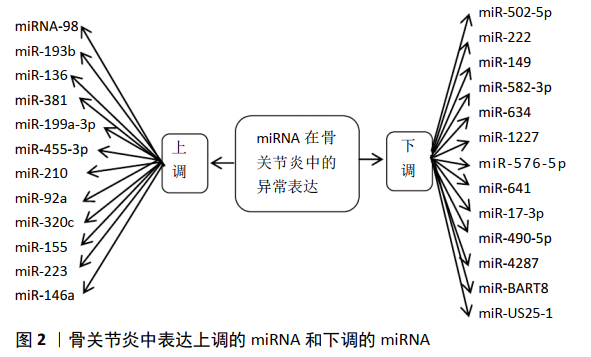
2.1 骨关节炎的发病机制 骨关节炎发病率和致残性较高,对患者机体功能和生活质量产生严重影响,但到目前为止其发病机制尚不明确。骨关节类的致病因素复杂多样,包括机械、遗传和环境因素,如年龄、性别、肥胖、吸烟、饮酒、激素、感染、创伤、炎症等,这些因素都会使骨关节炎的患病风险增加[6],可能导致了软骨细胞的破坏,细胞外基质降解和软骨下骨合成失衡,致软骨损伤和骨关节炎的形成[7]。在遗传方面,骨关节炎遗传性较强,遗传率可高达40%-65%。在分子水平致病机制方面,与微小RNA、炎症因子、基质金属蛋白酶、脂肪因子、信号通路、生长因子等息息相关。骨关节炎的病理表现主要为软骨和软骨下骨退变、滑膜炎症及骨赘形成。软骨的变性是最根本且较早表现在骨关节炎中的,软骨细胞是唯一存在关节软骨中的细胞类型,其作用表现在维持骨、软骨、关节的结构及其功能等方面[8]。软骨细胞自噬也调控着软骨细胞凋亡启动过程,其病理发生发展过程还包括细胞外基质降解、骨赘、软骨下硬化症和骨髓损害。在骨关节炎发病机制尚未明确的情况下,调节和控制相关因素、了解生物分子的贡献和关节组织之间的相互作用就尤为重要。 2.2 miRNA与骨关节炎发生的关系 2.2.1 miRNA的概念 微小RNA(microRNA,miRNA)是一种小的非编码的单链RNA,其长度为21-23个核苷酸。在秀丽隐杆线虫中鉴定出首个miRNA后[9],已有超过1 000个miRNA在人类和小鼠中被发现,这些miRNA与疾病和正常细胞生理有关。人类60%以上的蛋白质编码基因受miRNAs调控,这意味着miRNAs在基因表达调控中起着重要而核心的作用[10]。在人类中,miRNAs既由编码基因的内含子编码,也由非编码基因的内含子和外显子编码。在其成熟过程中,miRNA首先由RNA聚合酶Ⅱ转录形成初级miRNAs(pri-miRNAs), pri-miRNAs由5’端的甲基鸟嘌呤核苷帽和3’聚(A)尾组成。随后,pri-miRNAs在DROSHA和DGCR8 (DiGeorge syndrome critical region 8 homologue) 微处理器酶的作用下识别并切割,产生一个约70个核苷酸长度的含有茎环发夹结构的前体miRNAs(pre-miRNAs)。pre-miRNAs在转运蛋白Exportin 5协助下进入细胞质,在细胞质中通过RNAase酶Ⅲ加工成约22个核苷酸长度的双链miRNA结构。以双链中的一条链作为导链整合形成蛋白质复合体,即miRNA诱导沉默复合体(MiRISC),MiRISC产物是成熟的、具有功能的miRNA[11]。研究表明,miRNAs通过碱基配对到非翻译区(3’-UTR)的互补序列来调节许多基因的表达,从而对翻译起到抑制作用[12-13]。miRNAs在细胞的生长、增殖、分化、凋亡、代谢和稳态等病理生理过程中起到重要调控作用[14]。近年来,研究发现miRNA能影响软骨发生和软骨修复,miRNA可调节干细胞的自我更新或是对转录后的基因进行调控进而达到对细胞周期的控制,被认为是骨关节炎新的有效治疗工具[15]。 2.2.2 miRNA与骨关节炎的关系概述 大量临床研究表明, miRNAs的表达水平与骨关节炎发生发展进程中关节软骨破坏、软骨细胞凋亡、滑膜炎症以及疼痛等病理变化关系密切。miRNA调控胶原蛋白和基质金属蛋白酶的表达,其调控结果为软骨细胞的衰老与凋亡及细胞外基质降解,软骨细胞凋亡主要是指线粒体凋亡、内质网应激相关性死亡和死亡受体凋亡,miRNA最终对骨关节炎发生发展起到了作用[16]。近年来,990 种已知的miRNA和1 621种潜在 miRNA在骨关节炎软骨细胞中发现[17]。miRNA的异常表达参与影响着软骨细胞增殖与凋亡、自噬及代谢。miRNA 参与关节软骨的发育,对关节软骨内平衡、骨赘形成及滑膜炎都发挥重要作用,在预防治疗关节疼痛及关节衰老中也发挥了重要作用,所以miRNA与骨关节炎的关系密不可分。 2.2.3 miRNA在骨关节炎中的异常表达 随着人们对miRNA研究的逐步深入,越来越多的miRNA被证实在骨关节炎中异常表达,miRNA的表达上调或者下调与骨关节炎的发生发展有着紧密的联系,miRNA调节失控会导致骨关节炎的发生,同时也能影响到炎症和肥胖的发生。有相关研究表明,在研究分析了365例正常人和骨关节炎患者软骨组织中miRNA的表达水平,结果发现存在16个不同的miRNA表达异常[18]。这些表达差异的miRNA可以用来区别骨关节炎患者的软骨细胞和正常人的软骨细胞。JONES 等[19]研究了157人的组织中的miRNA,发现与正常组织相比,一些 miRNA在骨和软骨组织中表达存在差异。ZHANG等[20]报道发现8个miRNA在骨关节炎软骨组织呈现高表达,4个miRNA在骨关节炎软骨组织中呈现低表达状态。骨关节炎中表达上调的miRNA和下调的miRNA见图2。 (1)骨关节炎中表达上调的miRNA:WANG等[21]发现miRNA-98在骨关节炎患者的软骨组织中表达明显高于正常软骨组织中。ZHANG等[20]通过研究发现,有8个miRNA在骨关节炎患者的软骨组织中呈上调趋势,分别是miR-193b 、miR-136、miR-381、miR-199a-3p 、miR-455-3p、miR-210、miR-92a及miR-320c。研究发现,miR-155、miR-223、miR-146a等在骨关节炎患者中也呈现高表达状态。其中miR-381促进了软骨形成过程中基质金属蛋白酶13的表达,从而影响软骨退变。抑制miR-381的合成,继而影响血管细胞间黏附分子1(VCAM1)的表达,血管细胞间黏附分子1表达的增加促进了单核细胞与OASF的黏附,加重骨关节炎的发展。MiR-136-5p可促进软骨细胞迁移,增加Ⅱ型胶原、胶原蛋白多糖和SOX9的表达,降低基质金属蛋白酶13的表达。经研究证实,miR-136-5p能靶向作用下调ELF3的表达。在骨关节炎软骨组织中miR-136-5p表达降低,ELF3表达增加,抑制骨关节炎软骨细胞退化。另外,miR210可能通过直接靶向HIF3α,促进软骨细胞增殖,促进细胞外基质沉积,对骨关节炎的发生发展起负调控作用。miR-193B被确定为ST3GAL4的直接调控靶点,可以逆转ST3GAL4的调控。ST3GAL4的过表达修饰CD44。最后,唾液酸化的CD44a降低了与润滑素的结合量,并介导了核因子кB途径的活性,miR-193B/ST3GAL4通过核因子кB途径调节CD44a,从而影响骨关节炎的进展[22]。 (2)骨关节炎中表达下调的miRNA:ZHANG等[23]经过研究发现,在骨关节炎患者的软骨组织中,miR-502-5p呈低表达。SONG等[24]研究发现,miR-222在骨关节炎患者的软骨组织中呈低表达状态。研究发现,在正常的软骨细胞和骨关节炎软骨细胞中,miRNA具有差异性表达,其中miR-149、miR-582-3p、miR-634、miR-1227、miR-576-5p 和 miR-641 在 骨关节炎软骨细胞中呈低表达,表明miRNA参与了关节软骨细胞的发育与破坏[25]。其中miR-582-3p可抑制软骨细胞的凋亡,减少促炎细胞因子的产生,抑制细胞外基质的降解。在分子机制研究表明,YAP1是miR-582-3p的下游靶点。YAP1扩增可以逆转miR-582-3p调控软骨细胞凋亡、炎症反应和细胞外基质降解。miR-17-3p表达下调,减轻成骨细胞分化作用。sox6基因作为miR-17-3p的靶基因,起到负向抑制作用,刺激骨的形成。ZHANG等[20]发现,4种miRNA在骨关节炎患者的软骨组织中呈下调趋势,分别是miR-490-5p、miR-4287、miR-BART8 和miR-US25-1。 2.2.4 miRNA在软骨细胞增殖与凋亡中的作用 骨关节炎疾病的发生与软骨细胞的增殖和凋亡有着密切的联系,当软骨细胞增殖能力遭到破坏,细胞数目就会减少,进而导致骨关节炎的发生,软骨细胞的凋亡亦是促进骨关节炎进展的标志性分子事件。CAO等[26]通过研究发现,软骨细胞中过表达miR-15b可抑制胰岛素样生长因子、胰岛素样生长因子1受体和bcl-2的表达,当miR-15b被替除后,胰岛素样生长因子1、胰岛素样生长因子1受体和bcl-2的表达增强。此外miR-15b能靶向胰岛素样生长因子1和胰岛素样生长因子1受体抑制软骨细胞的增殖,通过靶向bcl-2促进软骨细胞的凋亡。有研究表明,在骨关节炎患者的软骨组织中miR-34a的表达增加,过表达的miR-34a能直接抑制δ样蛋白1(DLL1),进而导致骨关节炎软骨细胞中DLL1、总PI3K和p-AKT浓度下降,诱导软骨细胞凋亡。在手术诱导的骨关节炎大鼠的膝关节中,通过向关节中注射miR-34a能减轻关节软骨细胞的死亡及软骨的丢失[27]。有研究表明miR-520d-5p在人骨髓间充质干细胞软骨形成过程中表达增强。MiR-520d-5p的过表达和下调促进和抑制了软骨的形成,调节了软骨细胞的代谢。组蛋白脱乙酰酶1(histone deacetylase 1,HDAC1)在人骨髓间充质干细胞软骨形成过程中表达降低,HDAC1是miR-520d-5p的靶基因。HDAC1抑制剂CI994可上调软骨特异性基因的表达,促进软骨形成。在白细胞介素1β处理的人软骨细胞中,CI994抑制了基质金属蛋白酶13的表达,降低了白细胞介素1β对PHC的作用。miR-520d-5p通过靶向HDAC1促进人骨髓间充质干细胞软骨形成,调节软骨细胞的代谢[28]。TU等[29]发现,miR-127-5p能参与软骨细胞增殖调控过程,当上调miR-127-5p可明显抑制软骨细胞的增殖过程,加速骨关节炎的发展。此外有研究报道,下调miR-139能促进软骨细胞增殖,miR-210的过表达亦能促进软骨细胞的增殖。SHEN等[30]研究得出结论,miR-30a-5p在骨关节炎患者的软骨组织中呈高表达,可通过靶向Akt基因阻滞G0/G1期的软骨细胞,进而诱导软骨细胞的凋亡。另外还有研究表明,miR-29b-3p通过靶向颗粒体蛋白前体(PGRN)促进软骨细胞的凋亡和骨关节炎的发生,关节注射miR-29b-3p拮抗剂可延缓骨关节的进展过程。miR-29b-3p或颗粒体蛋白前体可能成为治疗骨关节炎的潜在靶点[31]。YAN等[32]研究发现,miR-34a主要通过抑制SIRT1/p53信号通路来促进软骨细胞的凋亡。在软骨细胞增殖、凋亡的过程中,上调或下调的miRNA起到了调控的作用。 2.2.5 miRNA对骨关节炎关节软骨细胞外基质的作用 大量文献已经证实,人血小板反应蛋白解整合素金属肽酶5(ADAMTS-5)与基质金属蛋白酶13是导致关节软骨细胞外基质降解的2种主要水解酶,在基质水解酶的作用下,软骨细胞的细胞外基质降解大于合成,进而导致细胞稳态失衡,诱发骨关节炎的发生。LI等[33]研究发现,miR-27a-3p在骨关节炎患者中呈低表达,白细胞介素1β通过MAPK和核因子κB途径抑制miR-27a-3p的活性,进而增加ADAMTS-5的表达,导致软骨细胞外基质降解,促进骨关节炎的发生。MIYAKI等[34]研究发现,miR-140对ADAMTS-5的表达有重要的调控作用,下调miR-140能使ADAMTS-5的表达增加,引发软骨基质的降解导致骨关节炎的发生。JI等[35]研究发现,miR-30a与ADAMTS-5的表达呈负相关性,miR-30a的过表达可显著抑制ADAMTS-5的表达。WANG等[36]研究发现,在骨关节炎的软骨组织中,miR-411和基质金属蛋白酶13呈负相关,基质金属蛋白酶13是miR-411的靶基因,miR-411的过表达能明显抑制基质金属蛋白酶13的表达,进而延缓骨关节炎的发生进程。ZHANG等[37]研究发现,在骨关节炎患者的软骨组织中miR-9的表达水平显著降低,向骨关节炎的大鼠关节腔内注射miR-9拮抗剂,能够抑制骨关节炎大鼠软骨组织中基质金属蛋白酶13的表达,增加胶原蛋白合成减少其降解,表明miR-9通过对基质金属蛋白酶13表达水平的抑制作用,降低基质金属蛋白酶13对胶原蛋白的抑制,进而延缓骨关节炎进展。 2.2.6 miRNA对软骨细胞自噬的作用 细胞自噬是一种分解代谢的方式,主要指双层膜液泡吞噬需降解的细胞成分成为自噬小体,然后与溶酶体融合形成自噬溶酶体并降解细胞内物质的过程。降解后的产物重新用于机体合成和代谢,自噬是机体在应激状态下防止细胞死亡的保护机制,且有利于维持细胞活性与稳态[38-39]。软骨细胞的异常自噬会导致骨关节软骨组织的病变,从而引发骨关节炎。有大量研究表明,自噬的活性随年龄增长而下降,抑制细胞自噬性蛋白降解会导致衰老加速,而提升自噬性可以延长寿命[40]。自噬被认为是保护关节软骨的重要机制[41-42]。越来越多证据表明miRNA参与调节软骨细胞自噬过程,在其过程中发挥着作用。WAN 等[43]研究发现,miR-155在骨关节炎软骨中高度上调,其通过mTOR信号通路诱导自噬。miR-30b在调控软骨细胞自噬过程有突出的表现作用,CHEN 等[44]证实了与miR-30b抑制作用最终导致自噬上调,并抑制了细胞凋亡和软骨降解。近年来,miR-17-5p在自噬过程中受到广泛的关注。选择性自噬适应蛋白-固位体1对蛋白降解有影响,也参与了软骨细胞的自噬并起到了保护的作用。有相关研究表明,沉默信息调节因子1(silent information regulator 1,SIRT1)是miR-9和miR-449a直接作用靶点,通过影响SIRT1来调控软骨细胞的自噬[45]。SUN等[46]研究发现,miR-4262过表达同样可调控SIRT1。通过激活AKT/mTOR通路,抑制SIRT1,进而抑制软骨细胞自噬,最终导致骨关节炎的发生。ZHANG等[47] 研究发现,miR-27a 的过表达对软骨细胞的增殖具有促进作用,靶向抑制核因子κB信号通路,激活软骨细胞自噬,调节软骨细胞的活性。软骨细胞自噬是早期预防骨关节炎及治疗软骨细胞损伤的潜在靶点,通过分析研究微小RNA在自噬中发挥的重要作用,同样对探究骨关节炎的发病机制也是至关重要的。 2.3 miRNA与关节疼痛 关节疼痛是骨关节炎患者常出现的主要临床症状,其也是关节活动受限的原因之一。目前,骨关节炎的治疗主要集中在如何缓解或减轻患者疼痛。疼痛主要是由周围疼痛感受器与中枢感觉系统之间的信号传导导致的。研究显示,miRNA与骨关节炎疼痛的产生有相关性,存在多种miRNAs的失调与疼痛相关,已被报道参与介导炎症和疼痛途径,其中包括miR-9、miR-27和miR-34a。有最新研究表明,抑制miRNA-27b-3p可增强赖氨酸脱甲基酶4B(Lysine demethylase 4B,KDM4B)和DLX5(Distal-Less Homeobox 5)基因表达,促进骨髓间充质干细胞的成骨分化,抑制成脂分化,最终通过KDM4B脱甲基化减轻骨关节炎患者疼痛[48]。KDM4B是KDM4家族中的一员,是一组组蛋白去甲基化酶,与疾病发展、精子和肿瘤发生有密切的联系[49]。其高表达与KDM4B的抑制和炎症有关,可成为导致骨质破坏高炎类疾病的新治疗靶点[50]。miR-558在软骨组织中呈低表达,抑制炎症因子,减轻患者疼痛。miR-199a-3P却与之作用相反,其可以使炎症因子表达上调,进而产生疼痛效果。WANG等[51]发现,miR-146a的异常表达加剧关节疼痛并影响了骨关节炎病程的进展。 2.4 miRNA与软骨衰老 年龄因素影响着骨关节炎的发生,随着年龄增长,可以引起关节慢性损伤、合成分解代谢紊乱、调节异常等情况。虽然没有明确证实miRNA与衰老之间的相互关系,但大量研究表明,miRNA与软骨衰老息息相关。软骨细胞的衰老是导致骨关节炎发展过程中基质代谢失衡的重要原因[52]。在关节软骨中含有大量丰富的硫酸化蛋白多糖(proteoglycans,PGS),蛋白多糖是由核心蛋白结合的糖胺聚糖链组成[53]。有研究表明,上调miR-204的同时针对蛋白多糖生物合成途径的多个组分,可以有效地关闭PG的合成代谢。异位表达的miR-204触发了自发性软骨丢失和骨关节炎的发展,而抑制miR-204,同时伴随着软骨PG合成的增加和抑制炎性衰老相关分泌表型因子的表达[54]。另外,有学者研究,在骨关节炎患者中miR-204表达显著增高,在软骨细胞的损伤中起重要作用,miR-204可能为临床骨关节炎治疗提供一个新方法[55]。UKAI等[56]研究发现miR-193b、miR-199a-3p和 miR-320c 均参与软骨细胞衰老的调控。miR-21调控表达可以增加软骨的衰老[57]。MIYAKI等[34]研究表明,miR-140 是软骨发育和维持内稳态以及软骨正常代谢的调控者,由于骨关节炎发病过程中的某因素存在使miR-140水平下降,引起年龄相关性关节软骨的病变,最终导致骨关节炎的发生。有研究发现,HDAC2作为靶点,miR-92a-3P可以作用其来调节软骨的发育和衰老,且在软骨细胞外基质增加中起到了一定的作用。MiR-138-5P可促进软骨细胞中的白细胞介素1β诱导软骨降解,这表明miR-138能够促进软骨降解[58]。 2.5 miRNA与骨关节炎早期诊断 骨关节炎的疼痛、肿胀、功能障碍困扰并折磨着患者,所以预防和早期诊断对于临床诊治来说是至关重要的。由于miRNA可以稳定存在人类的体液如血液、关节液、尿液以及唾液中,所以它可以作为诊断多种疾病的生物学标志物[59]。目前,对于骨关节炎的早期诊断在临床上还尚未有有效的方法,在确诊的骨关节炎患者中,大多数患者关节软骨已破坏,达到了不可逆转的阶段,为了减轻患者痛苦和能达到良好的预后效果,早期诊断并加以干预治疗就更加必要了。研究发现,在骨关节炎患者中,miR-98、miR-25、 miR-9的表达均为上升,miR-98、miR-9与许多免疫性疾病(包括骨关节炎)具有相关的基因,高表达 miR-98、miR-9、miR-146,可以使白细胞介素1β调控的表达产物减少。高表达miR-9可以通过调控白细胞介素1β诱导基质金属蛋白酶13 的释放,这些表明 miRNA在骨关节炎的发展过程中起作用。因此,通过检测患者 miR-98、 miR-25、 miR-9 等基因表达水平,可作为临床诊断骨关节炎提供新的途径,并可以作为辅助检测可以提高诊断的正确率[60]。 SOYOCAK等[61]研究表明,骨关节炎患者血清中的miR-146a和miR-155表达远高于健康人,且随病程的加重而增加,miR-146a和miR-155可作为早期诊断骨关节炎的分子水平的标志物,其表达水平越高代表骨关节炎病程越严重。同样有研究显示,miR-1282和miR-4284也可作为诊断骨关节炎的辅助诊断标志物[62]。LIOPOULOS 等[63]发现在正常人关节液中的miR-132表达要明显高于骨关节炎的患者。CHAO 等[64]研究分析比较正常人与早、中及晚期的骨关节炎患者膝关节滑膜中miR-140 的表达水平,结果发现骨关节炎患者关节滑液中 miR-140表达量较正常人少。早期诊断不仅可以为不可逆的关节损害提供干预机会,而且很大程度上避免了患者晚期的痛苦。随着分子水平技术的进步,体液中的部分生物标志物在骨关节炎早期诊断及预防方面的作用受到广大学者的广泛关注。某些miRNAs在体液中持续稳定的表达,可以作为临床用于监测骨关节炎病情进展的可控指标。 根据上述研究成果可以得出结论:miRNA在人的关节液中存在并稳定表达,可以根据不同 miRNA在骨关节炎患者的关节液中表达水平不同,来区分骨关节炎患者与正常人,并以辅助诊断骨关节炎。miRNA对骨关节炎的早期诊断具有非常重要的临床意义。 2.6 miRNA与骨关节炎治疗 miRNA可能为骨关节炎治疗提供新靶点。软骨细胞来源于软骨组织中的细胞外基质,其在维持软骨的结构和功能方面起重要作用[65]。Zust同源增强子2(enhancer of zeste homolog 2,Ezh2 )是一种组蛋白甲基转移酶,参与多种类型癌症的发生,包括结肠癌、乳腺癌和前列腺癌[66]。有证据表明,EZH2在许多疾病的转录和转录后水平上发生失调,包括前列腺癌、白血病[67],但miR-17-5p与EZH2的关系未知。最近有文献报道,在骨关节炎软骨组织和白细胞介素1β诱导的软骨细胞中,miR-17-5p的表达下调,而ezh2的表达上调。miR-17-5P模拟物可抑制白细胞介素1β诱导的软骨细胞凋亡和细胞外基质降解。在软骨细胞中,EZH2是miR-17-5p和miR-19b-3p的靶点,可促进白细胞介素1β诱导的软骨细胞凋亡和细胞外基质降解。挽救实验表明,EZH2过表达可逆转抑制miR-17-5p或miR-19b-3p模拟诱导的骨关节炎进展,即miR-17-5p和miR-19b-3p通过EZH2靶向抑制骨关节炎的进展,从而达到临床治疗效果[68]。有研究显示,miR-410-3p在骨关节炎小鼠的关节组织和脂多糖诱导的软骨细胞中表达明显下调。miR-410-3p的上调可明显抑制被脂多糖诱导的软骨细胞的凋亡、高迁移率族蛋白B1(high mobility group protein box-1,HMGB1)的表达、核因子kB活性和促炎细胞因子的产生。脂多糖诱导的HMGB1表达上调明显抑制miR-410-3p的表达。说明miR-410-3p/HMGB1可能是通过核因子κB信号通路在骨关节炎中发挥抗炎作用。这就更加说明miR-410-3p在骨关节炎中的作用机制,为骨关节炎的治疗提供有力支撑[69]。LIU等[70]研究发现在骨关节炎软骨中,miR-1207-5p表达降低,人趋化因子Fractalkine受体(C-X3-C motif chemokine receptor 1,CX3CR1)表达增加。从细胞的增殖、凋亡和细胞外基质降解的角度看,miR-1207-5p表达增加,可以减轻脂多糖诱导细胞的损伤。CX3CR1是miR-1207-5p的靶点,其下调增加了miR-1207-5p模拟物的作用,促进细胞增殖,减轻凋亡。在CX3CR1基因敲除后,miR-1207-5p在骨关节炎中的调节作用将增强。miR-1207-5p/CX3CR1轴可能为骨关节炎的治疗带来新进展。SI等[71]研究发现,miR-14能够调节其细胞外基质稳态性,从而抑制骨关节炎发生进展,miR-14可能成为治疗骨关节炎的一个新的靶点。同样有文章报道,在动物模型关节腔内注射锁核酸-抗-miR-449a能够增加软骨再生,在骨关节炎中,miR-449a表达上调,Ⅱ型胶原蛋白和聚集蛋白聚糖的表达升高,骨关节炎进展延缓[72]。研究显示,miR-142-3p可以抑制HMGB1的表达,从而抑制软骨细胞凋亡,HMGB1是骨关节炎重要的炎症因子,在大鼠骨关节炎模型的关节腔注射慢病毒过表达miR-142-3p 可以阻断或延缓大鼠骨关节炎的进展[73]。WANG 等[74]研究结果显示,miR-483-5p在软骨中表达上调,向关节腔内注射慢病毒介导miR-483-5p,使其上调表达,可以发现骨关节炎的病理变化加重,又将antago-miR-483-5p沉默内源性miR-483-5p注射骨关节炎关节腔内,则病变程度减轻,这又为临床治疗提供了新的思路。KANG等[75]研究发现,Ⅱ型胶原和聚集蛋白聚糖表达受miR-23a-3p高表达的影响,SMAD基因的敲除可逆转miR-23a-3p抑制剂对其表达的影响,这表明miR-23a-3p可直接作用于SMAD基因以影响骨关节炎发生发展,可以为骨关节炎治疗提供潜在的治疗靶点。"
| [1] ROSS AK, COUTINHO DE ALMEIDA R, RAMOS YFM, et al. The miRNA-mRNA interactome of murine induced pluripotent stem cell-derived chondrocytes in response to inflammatory cytokines. FASEB J. 2020;34(9):11546-11561. [2] WANG K, XU J, CAI J, et al.SerumleveIsofinterIeu-kin-l7 and adiponectina reassociated with in frapa-tellarfatpad volumeand signal intensity alteration inpatients with knee osteoarthritis. Arthritis Res Ther. 2016;(18):193. [3] LOESER RF. The Role of Aging in the Development of Osteoarthritis. Trans Am Clin Climatol Assoc. 2017;128:44-54. [4] WANG T, HAO Z, LIU C, et al. LEF1 mediates osteoarthritis progression through circRNF121/miR-665/MYD88 axis via NF-кB signaling pathway. Cell Death Dis. 2020;11(7):598. [5] MENDEII JT. MicroRNAs:critical regulatorsofdeveI-opment,cellularphysiology and malignancy. Cell Cycle. 2005;4(9):1179-1184. [6] KOPAŃSKA M, SZALA D, CZECH J, et al. MiRNA expression in the cartilage of patients with osteoarthritis. J Orthop Surg Res. 2017;12(1):51. [7] HUSSAIN SM, DAWSON C, WANG Y, et al. Vascular Pathology and Osteoarthritis: A Systematic Review. J Rheumatol. 2020;47(5):748-760. [8] NIELSEN FK, EGUND N, JØRGENSEN A, et al. Risk factors for joint replacement in knee osteoarthritis; a 15-year follow-up study. BMC Musculoskelet Disord. 2017;18(1):510. [9] LEE RC, FEINBAUM RL, AMBROS V. The C.elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell. 1993;75(5):843-854. [10] ENGELS BM, HUTVAGNER G. Principles and effects of microRNA-mediated post-transcriptional gene regulation. Oncogene. 2006;25(46):6163-6169. [11] Soltanzadeh-Yamchi M, Shahbazi M, Aslani S, et al. MicroRNA signature of regulatory T cells in health and autoimmunity. Biomed Pharmacother. 2018; 100:316-323. [12] Giraldez AJ, Mishima Y, Rihel J, et al. Zebrafish MiR-430 promotes deadenylation and clearance of maternal mRNAs. Science. 2006;312(5770):75-79. [13] Filipowicz W, Jaskiewicz L, Kolb FA, et al. Post-transcriptional gene silencing by siRNAs and miRNAs. Curr Opin Struct Biol. 2005;15(3):331-341. [14] Wang J, Chen J, Sen S. MicroRNA as Biomarkers and Diagnostics. J Cell Physiol. 2016;231(1):25-30. [15] Valinezhad Orang A, Safaralizadeh R, Kazemzadeh-Bavili M. Mechanisms of miRNA-Mediated Gene Regulation from Common Downregulation to mRNA-Specific Upregulation. Int J Genomics. 2014;2014:970607. [16] Soyocak A, Kurt H, Ozgen M, et al. miRNA-146a, miRNA-155 and JNK expression levels in peripheral blood mononuclear cells according to grade of knee osteoarthritis. Gene. 2017;627:207-211. [17] Rasheed Z, Al-Shobaili HA, Rasheed N, et al. Integrated study of globally expressed microRNAs in IL-1β-stimulated human osteoar- thritis chondrocytes and osteoarthritis relevant genes: a microarray and bioinformatics analysis.Nucleosides Nucleotides Nucleic Acids. 2016;35(7):335-355. [18] IUOPOULOS D, MALIZOS K N, OIKONOMOU P, et al. lntegrative microRNA and proteomic approaches identify novel osthritis gen and their collaborative metabolic and inflammatory networks. PLoS One. 2008;3(11):e3740. [19] JONES SW, WATKINS G, LE GOOD N, et al. The identification of differentially expressed microRNA in osteoarthritic tissue that modulate the production of TNF - alpha and MMP13. Osteoarthritis Cartilage. 2009;17(4):464-472. [20] ZHANG Z, KANG Y, ZHANG Z, et al. Expression of microRNAs during chondrogenesis of human adipose-derived stem cells. Osteoarthritis Cartilage. 2012;20(12):1638-1646. [21] WANG GL, WU YB, LIU JT, et al. Upregulation of miR-98 Inhibits Apoptosis in Cartilage Cells in Osteoarthritis.Genet Test Mol Biomarkers. 2016;20(11):645-653. [22] WANG T, HAO Z, LIU C, et al. MiR-193b modulates osteoarthritis progression through targeting ST3GAL4 via sialylation of CD44 and NF-кB pathway. Cell Signal. 2020;76:109814. [23] ZHANG G, SUN Y, WANG Y, et al. MiR-502-5p inhibits IL-1β-induced chondrocyte injury by targeting TRAF2. Cell Immunol. 2016;302:50-57. [24] SONG J, JIN EH, KIM D, et al. MicroRNA-222 regulates MMP-13 via targeting HDAC-4 during osteoarthritis pathogenesis. Bba Clinical. 2015;3(1):79-89. [25] DÍAZ-PRADO S, CICIONE C, MUIÑOS-LÓPEZ E, et al. Characterization of microRNA expression profiles in normal and osteoarthritic human chondrocytes. BMC Musculoskelet Disord. 2012;13:144. [26] CAO P, FENG Y, DENG M, et al. MiR-15b is a key regulator of proliferation and apoptosis of chondrocytes from patients with condylar hyperplasia by targeting IGF1, IGF1R and BCL2. Osteoarthritis Cartilage. 2019;27(2):336-346. [27] ZHANG W, HSU P, ZHONG B, et al. MiR-34a Enhances Chondrocyte Apoptosis, Senescence and Facilitates Development of Osteoarthritis by Targeting DLL1 and Regulating PI3K/AKT Pathway. Cell Physiol Biochem. 2018;48(3):1304-1316. [28] LU J, ZHOU Z, SUN B, et al. MiR-520d-5p modulates chondrogenesis and chondrocyte metabolism through targeting HDAC1.Aging (Albany NY). 2020; 12(18):18545-18560. [29] TU M, LI Y, ZENG C, et al. MicroRNA-127-5p regulates osteopontin expression and osteopontin-mediated proliferation of human chondrocytes. Rep. 2016;6:25032. [30] SHEN PF, QU YX, WANG B, et al. miR-30a-5p promotes the apoptosis of chondrocytes in patients with osteoarthritis by targeting protein kinase B. Zhonghua Yi Xue Za Zhi. 2017;97(39):3079-3084. [31] CHEN L, LI Q, WANG J, et al. MiR-29b-3p promotes chondrocyte apoptosis and facilitates the occurrence and development of osteoarthritis by targeting PGRN. J Cell Mol Med. 2017;21(12):3347-3359. [32] YAN S, WANG M, ZHAO J, et al. MicroRNA-34a affects chondrocyte apoptosis and proliferation by targeting the SIRT1/p53 signaling pathway during the pathogenesis of osteoarthritis. Int J Mol Med. 2016;38(1):201-209. [33] LI X, HE P, LI Z, et al. Interleukin 1β mediated suppression of microRNA 27a 3p activity in human cartilage via MAPK and NF κB pathways: A potential mechanism of osteoarthritis pathogenesis. Mol Med Rep. 2018;18(1):541-549. [34] MIYAKI S, SATO T, INOUE A, et al. MicroRNA-140 plays dual roles in both cartilage development and homeostasis. Genes Dev. 2010;24(11):1173-1185. [35] JI Q, XU X, ZHANG Q, et al. The IL-1β/AP-1/miR-30a/ADAMTS-5 axis regulates cartilage matrix degradation in human osteoarthritis. J Mol Med. 2016;94(7): 771-785. [36] WANG G, ZHANG Y, ZHAO X, et al. MicroRNA-411 inhibited matrix metalloproteinase 13 expression in human chondrocytes. Am J Trans Res. 2015; 7(10):2000-2006. [37] ZHANG H, SONG B, PAN Z. Downregulation of microRNA-9 increases matrix metalloproteinase-13 expression levels and facilitates osteoarthritis onset. Mol Med Rep. 2018;17(3):3708-3714. [38] CHOI AM, RYTER SW, LEVINE B. Autophagy in Human Health and Disease. N Engl J Med. 2013;368(7):651-662. [39] LEVINE B, KROEMER G. Autophagy in the pathogenesis of disease. Cell. 2008;132: 27-42. [40] BERGAMINI E, CAVALLINI G, DONATI A, et al. The role of autophagy in aging. Ann N Y Acad Sci. 2010;1114(1):69-78. [41] WANG ZJ, ZHANG HB, CHEN C, et al. Effect of PPARG on AGEs-induced AKT/MTOR signaling-associated human chondrocytes autophagy. Cell Biol Int. 2018;42(7): 841-848. [42] DE FIGUEROA PL, LOTZ MK, BLANCO FJ, et al.Autophagy activation and protection from mitochondrial dysfunction in human chondrocytes. Arthritis Rheumatol. 2015;67(4):966-976. [43] WAN G, XIE W, LIU Z, et al. Hypoxia- induced\r,MIR155\r, is a potent autophagy inducer by targeting multiple players in the MTOR pathway. Autophagy. 2014; 10(1):70-79. [44] CHEN Z, JIN T, LU Y. AntimiR- 30b Inhibits TNF- α Mediated Apoptosis and Attenuated Cartilage Degradation through Enhancing Autophagy. Cell Physiol Biochem. 2016;40(5):883-894. [45] D”ADAMO S, CETRULLO S, GUIDOTTI S, et al. Hydroxytyrosol modulates the levels of microRNA- 9 and its target sirtuin- 1 thereby counteracting oxidative stress- induced chondrocyte death. Osteoarthritis Cartilage. 2017;25(4):600-610. [46] SUN W, LI Y, WEI S. miR-4262 regulates chondrocyte viability, apoptosis, autophagy by targeting SIRT1 and activating PI3K/AKT/mTOR signaling pathway in rats with osteoarthritis. Exp Ther Med. 2018;15(1):1119-1128. [47] ZHANG FQ, WANG Z, ZHANG H, et al. MiR-27a alleviates osteoarthritis in rabbits via inhibiting inflammation. Eur Rev Med Pharmacol Sci. 2019;23(3 Suppl):89-95. [48] ZHANG G, ZHOU Y, SU M, et al. Inhibition of microRNA-27b-3p relieves osteoarthritis pain via regulation of KDM4B-dependent DLX5. Biofactors. 2020;46(5):788-802. [49] WILSON C, KRIEG AJ. KDM4B: A Nail for Every Hammer? Genes (Basel). 2019; 10(2):134. [50] KIRKPATRICK JE, KIRKWOOD KL, WOSTER PM. Inhibition of the histone demethylase KDM4B leads to activation of KDM1A, attenuates bacterial-induced pro-inflammatory cytokine release, and reduces osteoclastogenesis. Epigenetics. 2018;13(5):557-572. [51] WANG JH, SHIH KS, WU YW, et al. Histone deacetylase inhibitors increase microRNA-146a expression and enhance negative regulation of interleukin-1β signaling in osteoarthritis fibroblast-like synoviocytes. Osteoarthritis Cartilage. 2013;21(12):1987-1996. [52] LOESER RF, COLLINS JA, DIEKMAN BO. Ageing and the pathogenesis of osteoarthritis. Nat Rev Rheumatol. 2016;12(7):412-420. [53] MOUW JK, OU G, WEAVER VM. Extracellular matrix assembly: a multiscale deconstruction. Nat Rev Mol Cell Biol. 2014;15(12):771-785. [54] KANG D, SHIN J, CHO Y, et al. Stress-activated miR-204 governs senescent phenotypes of chondrocytes to promote osteoarthritis development. Sci Transl Med. 2019;11(486):eaar6659. [55] LIU X, GAO F, WANG W, et al. Expression of miR-204 in patients with osteoarthritis and its damage to chondrocytes. J Musculoskelet Neuronal Interact. 2020;20(2): 265-271. [56] UKAI T, SATO M, AKUTSU H, et al. MicroRNA-199a-3p,microRNA-193b, and microRNA-320c are correlated to aging and regulate human cartilage metabolism.Orthop Res. 2012;30(12):1915-1922. [57] PEFFERS MJ. Transcriptomic signatures in cartilage ageing. Arthritis Res Ther. 2013;21(4):1-17. [58] YUAN Y, ZHANG GQ, CHAI W, et al. Silencing of microRNA-138-5p promotes IL-1β-induced cartilage degradation in human chondrocytes by targeting FOXC1: miR-138 promotes cartilage degradation. Bone Joint Res. 2016;5(10):523-530. [59] MANNE U, SHANMUGAM C, BOVELL L, et al. miRNAs as biomarkers for man-agement of patients with colorectal cancer. Biomark Med. 2010;4(5):761-770. [60] IUOPOULOS D, MALIZOS K N, OIKONOMOU P, et al. lntegrative microRNA and proteomic approaches identify novel osthritis gen and their collaborative metabolic and inflammatory networks. PLoS One. 2008;3(11): e3740. [61] SOYOCAK A, KURT H, OZGEN M, et al. miRNA-146a, miRNA-155 and JNK expression levels in peripheral blood mononuclear cells according to grade of knee osteoarthritis. Gene. 2017;627:207-211. [62] 顾宗欣,王五洲.微小RNA对骨关节炎发生发展影响的研究进展[J].实用骨科杂志,2017,23(12):1102-1105. [63] LIOPOULOS D, MALIZOS KN, OIKONOMOU P, et al. Integrative microRNA and proteomic approaches identify novel osteoarthritis genes and their collaborative metabolic and inflammatory networks. PLoS One. 2008;3(11):e3740. [64] CHAO Y, ZHANG L, ZHANG X, et al. Expression of miR-140 and miR-199 in synovia and its correlation with the progression of knee osteoarthritis. Med Sci Monit. 2020;26:e918174. [65] ŞAHIN Ş, TUNCEL SA, SALIMI K, et al. Advanced Injectable Alternatives for Osteoarthritis. Adv Exp Med Biol. 2018;1077:183-196. [66] YAN KS, LIN CY, LIAO TW, et al. EZH2 in Cancer Progression and Potential Application in Cancer Therapy: A Friend or Foe? Int J Mol Sci. 2017;18(6):1172. [67] YAMAGISHI M, UCHIMARU K. Targeting EZH2 in cancer therapy. Curr Opin Oncol. 2017;29(5):375-381. [68] LI Y, YUAN F, SONG Y, et al. miR-17-5p and miR-19b-3p prevent osteoarthritis progression by targeting EZH2. Exp Ther Med. 2020;20(2):1653-1663. [69] PAN H, DAI H, WANG L, et al. MicroRNA-410-3p modulates chondrocyte apoptosis and inflammation by targeting high mobility group box 1 (HMGB1) in an osteoarthritis mouse model. BMC Musculoskelet Disord. 2020;21(1):486. [70] LIU XC, XU L, CAI YL, et al. MiR-1207-5p/CX3CR1 axis regulates the progression of osteoarthritis via the modulation of the activity of NF-κB pathway. Int J Rheum Dis. 2020;23(8):1057-1065. [71] SI HB, ZENG Y, LIU SY, et al. Intra-articular injection of microRNA-140 (miRNA-140) alleviates osteoarthritis (OA) progression by modulating extracellular matrix (ECM) homeostasis in rats. Osteoarthritis Cartilage. 2017;25(10):1698-1707. [72] BAEK D, LEE KM, PARK KW, et al. Inhibition of miR-449a Promotes Cartilage Regeneration and Prevents Progression of Osteoarthritis in In Vivo Rat Models.Mol Ther Nucleic Acids. 2018;13:322-333. [73] WANG X, GUO Y, WANG C, et al. MicroRNA-142-3p Inhibits Chondrocyte Apoptosis and Inflammation in Osteoarthritis by Targeting HMGB1. Inflammation. 2016;39(5):1718-1728. [74] WANG H, ZHANG H, SUN Q, et al. Intra- articular Delivery of Antago- miR-483-5p Inhibits Osteoarthritis by Modulating Matrilin 3 and Tissue Inhibitor of Metalloproteinase 2. MolTher. 2017;25(3):715-727. [75] KANG L, YANG C, SONG Y, et al. MicroRNA-23a-3p promotes the development of osteoarthritis by directly targeting SMAD3 in chondrocytes. Biochem Biophys Res Commun. 2016;478(1):467-473. [76] HONVO G, REGINSTER JY, RABENDA V, et al. Safety of Symptomatic Slow-Acting Drugs for Osteoarthritis: Outcomes of a Systematic Review and Meta-Analysis.Drugs Aging. 2019;36(Suppl 1):65-99. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Peng Zhihao, Feng Zongquan, Zou Yonggen, Niu Guoqing, Wu Feng. Relationship of lower limb force line and the progression of lateral compartment arthritis after unicompartmental knee arthroplasty with mobile bearing [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1368-1374. |
| [3] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [4] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Multiple problems in the selection of implants for patellar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1440-1445. |
| [5] | Wang Debin, Bi Zhenggang. Related problems in anatomy mechanics, injury characteristics, fixed repair and three-dimensional technology application for olecranon fracture-dislocations [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1446-1451. |
| [6] | Huang Dengcheng, Wang Zhike, Cao Xuewei. Comparison of the short-term efficacy of extracorporeal shock wave therapy for middle-aged and elderly knee osteoarthritis: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1471-1476. |
| [7] | Liu Xiangxiang, Huang Yunmei, Chen Wenlie, Lin Ruhui, Lu Xiaodong, Li Zuanfang, Xu Yaye, Huang Meiya, Li Xihai. Ultrastructural changes of the white zone cells of the meniscus in a rat model of early osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1237-1242. |
| [8] | Ji Zhixiang, Lan Changgong. Polymorphism of urate transporter in gout and its correlation with gout treatment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1290-1298. |
| [9] | Yuan Mei, Zhang Xinxin, Guo Yisha, Bi Xia. Diagnostic potential of circulating microRNA in vascular cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1299-1304. |
| [10] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [11] | Wan Ran, Shi Xu, Liu Jingsong, Wang Yansong. Research progress in the treatment of spinal cord injury with mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1088-1095. |
| [12] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [13] | Zhao Min, Feng Liuxiang, Chen Yao, Gu Xia, Wang Pingyi, Li Yimei, Li Wenhua. Exosomes as a disease marker under hypoxic conditions [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1104-1108. |
| [14] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [15] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||