Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (35): 5702-5708.doi: 10.12307/2021.303
Previous Articles Next Articles
Role of calcium ions in bone repair and osteogenesis
Lu Haiping1, Lang Xuemei2, Cao Jin1, Ma Yaping1, Xiao Yin3, Wang Xin1
- 1Department of Orthopedics, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China; 2Pre-hospital Emergency Department, Chongqing Emergency Center, Chongqing 400010, China; 3Department of Bone Tissue Engineering, The Queensland University of Technology, Brisbane 4059, Australia
-
Received:2021-01-13Revised:2021-01-15Accepted:2021-01-30Online:2021-12-18Published:2021-08-05 -
Contact:Xiao Yin, MD, Professor, Doctoral supervisor, Department of Bone Tissue Engineering, The Queensland University of Technology, Brisbane 4059, Australia -
About author:Lu Haiping, Master candidate, Department of Orthopedics, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China -
Supported by:the National Natural Science Foundation, No. 31960209 and 31760266 (both to WX); Basic Research Plan of Guizhou Provincial Department of Science and Technology, No. [2020]1Y093 (to WX)
CLC Number:
Cite this article
Lu Haiping, Lang Xuemei, Cao Jin, Ma Yaping, Xiao Yin, Wang Xin. Role of calcium ions in bone repair and osteogenesis[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(35): 5702-5708.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
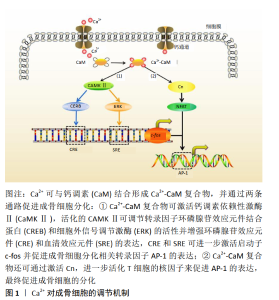
2.1 钙离子对间充质干细胞的影响 间充质干细胞具有向多种细胞(包括成骨细胞)分化的潜能,Ca2+在间充质干细胞的分化过程中起着至关重要的作用[12-13]。KAWANO等[9]首先发现间充质干细胞的分化与胞内钙震荡(Ca2+以浓度振荡的方式转导信息的过程)有关。钙振荡所利用的Ca2+主要有两种来源,其中一种是通过质膜进入,另一种是细胞内储存Ca2+的释放。细胞内Ca2+储存在内质网中,当Ca2+降低时,其主要通过两个通道释放,即肌醇1,4,5-三磷酸受体(InsP3R)和兰尼碱受体(RyR)[14]。值得注意的是,通过质膜来源的Ca2+并不是直接对钙震荡产生影响,而是通过补充间充质干细胞胞内的Ca2+储备,进而影响钙震荡的强度来传递信号[15],故对Ca2+内流产生影响的多种钙通道,如电压门控钙离子通道、非选择性钙离子通道、Na+-Ca2+交换体、钙泵等均能对钙振荡产生影响进而影响间充质干细胞的分化[16]。 间充质干细胞分化形成成骨细胞的过程大致可分为3个阶段:起始、增殖和迁移[17]。有研究表明在间充质干细胞分化的起始阶段,大导电钙钾通道的敲除会显著降低骨钙素、骨桥蛋白和Runx2基因的表达,故大导电钙钾通道被认为是决定间充质干细胞分化起始阶段的因素之一[18]。在间充质干细胞增殖阶段,有报道中导电钙钾通道的抑制或敲除会导致细胞周期蛋白D1和E表达减少,从而抑制间充质干细胞增殖[19]。目前,没有足够的证据表明钙钾通道与间充质干细胞迁移阶段有关[20]。但Ca2+浓度对间充质干细胞迁移的影响却有证据可寻,AQUINO-MARTíNEZ 等[21]制备了含不同浓度Ca2+的硫酸钙-琼脂糖-明胶复合材料,将其作为Ca2+释放体并接种小鼠来源的间充质干细胞培养7周,发现 3-5 mmol/L的Ca2+浓度最有助于小鼠间充质干细胞的迁移;在小鼠5 mm颅骨缺损模型中,他们进一步证实了Ca2+是通过减弱骨形态发生蛋白2对蛋白激酶B的激活来调节骨髓间充质干细胞的迁移过程。 2.2 钙离子对成骨细胞影响 成骨细胞是形成骨组织的主要功能细胞,在骨再生修复时,成骨细胞被招募迁移至骨损伤部位,合成分泌骨基质蛋白、胶原、骨桥蛋白等,并促进其矿化[22]。Ca2+是磷酸钙替代材料的主要组成成分,也是重要的第2信使,调控表型基因和转录因子的转录,控制着成骨细胞的分化过程。有研究表明,2-4 mmol/L的Ca2+有利于小鼠原代成骨细胞的增殖和存活,而稍高浓度(6-8 mmol/L)的Ca2+可促进小鼠原代成骨细胞分化和基质矿化[8]。另有研究表明,较高浓度的Ca2+(<14 mmol/L)可促进成骨细胞形成圆形形态,而低浓度的Ca2+(≤8 mmol/L)可促进人成骨细胞的迁移[23-24]。 Ca2+促进成骨细胞分化的机制被研究者们进一步探索。研究发现,Ca2+促进成骨细胞的分化与钙调素(Calmodulin,CaM)和钙调素依赖性激酶Ⅱ(Calmodulin dependent kinase Ⅱ,CaMKⅡ)途径的激活密切相关[25-27]。在成骨细胞分化过程中,Ca2+首先结合到钙调素上,形成钙/钙调素复合物(Ca2+/CaM),当结合多达4个Ca2+时,钙调素发生构象变化,使其能够结合特定的蛋白质如钙调素依赖性激酶。钙调素依赖性激酶是一种拥有30多种亚型的丝氨酸/苏氨酸特性的蛋白激酶,其中α-CaMKⅡ亚型在成骨细胞分化过程中发挥作用,它可以被Ca2+/CaM复合物激活,激活的α-CaMKⅡ可促进苏氨酸-286的自磷酸化,而苏氨酸-286的自磷酸化可以反过来使CaMKⅡ与Ca2+/CaM复合物结合的亲和力提高约1 000倍并使α-CaMKⅡ处于部分活化状态[28]。随后,活化的α-CaMKⅡ可调节多种成骨细胞相关转录因子的活性,如环磷腺苷效应元件结合蛋白(cAMP-response element-binding protein,CREB)和细胞外信号调节激酶(extracellular signal-regulated kinase,ERK)[29]。CREB和ERK可增加环磷腺苷效应元件(CRE)和血清效应元件(serum-response element,SRE)的转录,CRE和SER进一步激活启动子c-fos,并促进成骨细胞分化相关转录因子AP-1的表达,最终促进成骨细胞的分化[30-31]。 此外,研究发现Ca2+/CaM除了可激活CaMKⅡ之外,其存在的另一个激活靶点是钙调磷酸酶(Calcineurin,Cn)。钙调磷酸酶是一种丝氨酸/苏氨酸磷酸酶,是由催化亚单位(CnA)和调节亚单位(CnB)组成的异二聚体蛋白复合物。钙调磷酸酶被激活后,直接结合并去磷酸化细胞质中激活T细胞的核因子(nuclear factor of activated T cells,NFAT),使其转移到细胞核并参与基因表达的调节[32],见图1。"
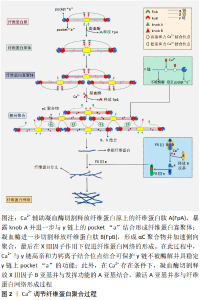
2.3 钙离子对破骨细胞影响 骨吸收和骨形成之间的微妙平衡对于维持骨完整性至关重要,其中破骨细胞在骨吸收中起着核心作用[33]。破骨细胞是单核前体细胞融合形成的特殊骨吸收细胞,其形成与成骨细胞和骨细胞分泌的单核细胞集落刺激因子和核因子κB受体活化因子配体有关[34]。研究证明,单核细胞集落刺激因子可促进破骨细胞的增殖分化,而核因子κB受体活化因子配体通路的激活可引发细胞内发生钙振荡,导致激活T细胞核因子1(NFATC-1)的表达上调,其产物激活T细胞核因子1蛋白作为一种钙依赖性神经钙蛋白,具有活化T细胞并促进单核细胞融合成多核破骨细胞的功能[35-36]。 骨吸收可导致大量Ca2+释放到细胞外液中,而细胞外Ca2+浓度的变化亦可影响破骨细胞的形成和吸收。CHENG 等[37]将羊来源的CD14+单核细胞与不同浓度Ca2+在体外共培养14 d,发现破骨细胞的形成量随着Ca2+浓度的不断升高(1.8-20 mmol/L)而逐渐减少,当培养液中Ca2+浓度达到 20 mmol/L时,成骨细胞的生成完全被抑制[38]。在造血干细胞培养中,细胞外高钙被证明能抑制破骨细胞的形成和活性。这表明胞外Ca2+浓度可以直接抑制破骨细胞的形成,影响骨重建动态平衡。 此外,Ca2+浓度对破骨细胞的迁移能力有着重要影响。单核巨噬细胞表面的钙通道敏感受体(CaR)被证明对破骨细胞的迁移起着决定性作用,当Ca2+与CaR结合后,使肌醇磷酸盐去磷酸化,进一步诱发胞内钙振荡并激活(磷脂酰肌醇-3激酶/蛋白激酶B,PI3K/Akt)通路使细胞骨架变形,导致破骨细胞的迁移[39]。有研究将不同浓度的Ca2+(1.8-20 mmol/L)与小鼠来源的破骨前体细胞(RAW 264.7)共培养,发现当细胞外Ca2+浓度为10 mmol/L时,RAW 264.7细胞迁移速度最快,其迁移距离是对照组的10倍[40]。 2.4 钙离子对血管化的影响 研究表明,成骨细胞必须位于直径为150-200 μm的毛细血管内才能获得充足的氧气和营养供应,而血管生成不足会导致骨组织中心区域坏死,并延迟骨再生过程[41]。因此,骨缺损处新生血管生长的速度和范围被认为是有效骨愈合的决定因素之一[42]。另有研究表明,细胞内Ca2+浓度与内皮细胞的增殖和运动有关,Ca2+波驱动内皮祖细胞的增殖、归巢和小管发生[43-44]。Ca2+不仅可充当各种细胞内酶的变构激活剂或抑制剂,在细胞质、细胞器和细胞核中表现出独特的调节作用(增殖、分化、凋亡等),它还可以与其他蛋白质相互作用,调节钙依赖性酶和离子通道[45-47]。最重要的例子是钙调素,它是一种细胞增殖的钙解码器,参与调节CaMKⅡ家族和几个膜通道[48]。研究表明,CaMKⅡ直接参与细胞周期进程中的几个转变点(G1至S期)[49]。此外,钙依赖性酶可激活与DNA分裂相关的核因子,如与内皮细胞增殖密切相关的细胞周期蛋白,从而促进内皮细胞的增殖[50-51]。 作为血管形成过程中最具调节性的因子之一,血管内皮生长因子不仅可通过Ras/丝裂原活化蛋白激酶(Ras/MAPK)途径刺激内皮细胞中毛细血管样结构的增殖、迁移、分化和形成,同时,在Ca2+存在下,血管内皮生长因子还可通过激活磷脂酰肌醇3-激酶(PI3K/AKT)途径和磷脂酶Cγ/三磷酸肌醇(PLCγ/IP3)信号途径结合内皮生长因子受体2促进内皮细胞增殖[52]。有报道,在兔胫骨缺损模型中,含钙量高的材料组(羟基磷灰石和β-磷酸三钙组)平均新血管生成数分别为 6.58±0.59和4.47±0.2,显著高于生物活性玻璃组(1.58±0.246)(P < 0.05) [53]。此外,在5 mm×8 mm的兔胫骨骨缺损模型中,β-磷酸三钙同样展现出良好的促进血管生成能力[54]。这些结果表明,β-磷酸三钙能有效地促进血管生成,其可能的原因是其释放的Ca2+促进了新血管的生成[55-56]。 2.5 钙离子促进生长因子的释放 骨折后各类生长因子释放并协调作用,对骨再生过程起着至关重要的作用[57-58]。骨折发生后,巨噬细胞清除坏死组织并释放肿瘤坏死因子,从而提高破骨细胞的活性,破骨细胞吸收断裂的骨基质并释放胰岛素样生长因子和骨形态发生蛋白,导致祖细胞的成骨分 化[59-60]。与此同时,血管内皮生长因子通过驱动内皮细胞增殖引起新血管再生[61]。血小板衍生生长因子、转化生长因子β (transforming growth factor-β,TGF-β)及成纤维细胞生长因子(fibroblast growth factor,FGF)等从浆细胞、巨噬细胞和成骨细胞中释放出来促进骨再生进程[60-62]。 骨损伤后,Ca2+促进血小板聚集黏附在伤口处,加速止血过程并为血小板释放生长因子提供前提条件。在随后过程中,Ca2+可进一步激活血小板并促进血小板释放生长因子如血小板衍生生长因子、表皮生长因子(epidermal growth factor,EGF)和血管内皮生长因子等[63-64]。有报道,将浓度为22.8 mmol/L的CaCl2加入富血小板血浆中15-30 min,测得转化生长因子β的最大释放量(约是对照组的2倍)、血小板衍生生长因子的最大释放量(约是对照组1.2倍)及内皮生长因子的最大释放量(约是对照组3倍)均显著升高,P < 0.05[65]。另有研究表明,血小板释放生长因子血小板衍生生长因子和表皮生长因子不仅随着Ca2+浓度增高而增高,还受到电生理的调节,并可通过Ca2+浓度和电生理双重控制实现生长因子的定量释放[66]。此外,Ca2+在生长因子发挥作用的过程中也起着不可忽视的作用,如血管内皮生长因子促进内皮细胞增殖的PLCγ-IP3通路是在Ca2+存在的条件下进行 的[67]。以上数据表明,Ca2+可促进骨折处血小板释放生长因子并协同生长因子共同促进骨再生过程。 2.6 钙离子通过影响血凝块结构来影响成骨 2.6.1 骨缺损处血凝块的结构对骨愈合的影响 在骨损伤部位血管破裂后,骨缺损处形成富含血小板-纤维蛋白的血肿,在局部止血中发挥重要作用[68]。骨缺损发生后的早期阶段,血凝块充当着“天然支架”的角色。有报道,移除最初的骨折血肿会损害骨修复过程,而植入血肿可以在大鼠模型中产生新的骨形成[10]。早期血肿不仅被认为是间充质干细胞发挥功能的初始纤维蛋白支架,而且是持续释放生长因子的临时“储库”,而生长因子的释放(特别是血小板衍生生长因子AB和转化生长因子β1 是骨再生的主要影响因素[69-71]。 纤维蛋白凝块中的结构参数包括纤维直径、密度、分支点的数量和性质、分支点之间的距离和孔隙尺寸等[72]。有研究表明,纤维蛋白的直径与纤维密度成反比,与孔径成正比[73]。据此,骨缺损处血凝块结构大致可分为两种:一种是由粗纤维(直径大)组成的具有孔径大、密度低、容易被纤溶酶降解的纤维蛋白;另一种是由细纤维(直径小)组成的具有孔径小、高密度、不容易被纤溶酶降解的纤维蛋 白[10]。粗纤维形成的纤维蛋白网络多孔的结构有助于间充质干细胞的迁移和黏附,其较厚的结构还可以促进间充质干细胞的增殖和分化,及时地降解有助于生长因子的释放和新血管的长入[74-75]。与之相反的是,致密的纤维蛋白凝块的渗透性较低,显著延缓了间充质干细胞从邻近组织的迁移[76]。此外,纤维密度被认为是纤维蛋白溶解速率的主要决定因素,细纤维形成的致密纤维蛋白网络密度高,具有较强的抵抗纤维蛋白溶解能力,阻碍了生长因子的释放和间充质干细胞的迁移,会导致延迟的骨折愈合[77-78]。因而,较小的骨缺损 (1 mm)(被观察具有粗纤维的纤维蛋白网络结构)能够在无任何干预的情况下自然愈合而较大的骨缺损(5 mm)(具有较细的纤维蛋白网络结构)不能自愈的这一实验现象得到合理的解释[79-80]。 2.6.2 钙离子对血凝块结构的影响 纤维蛋白原是一个分子质量约为340 kD的糖蛋白,长约45 μm,由2组3条多肽链(AαBβγ)2组成。在纤维蛋白聚合过程中,Ca2+辅助凝血酶首先裂解Aα链释放纤维蛋白肽A(fibrinopeptide A,FpA),暴露knob A,形成纤维蛋白单体并纵向聚合形成纤维蛋白寡聚体;其次,凝血酶继续裂解原纤维上的 Bβ链,暴露knob B并释放纤维蛋白肽B(fibrinopeptide B,FpB),促进侧向聚合过程形成纤维蛋白纤维并分支,最后在凝血XⅢ因子的参与下交联形成纤维蛋白网络结构[81]。 纤维蛋白分子结构中含有3个高亲和力钙结合位点[82],其中2个高亲和力钙离子结合位点存在于γ链中,并被证明与4个相应的氨基酸残基相关,包括γAsp318,γAsp320,γGly324和γPhe322[83]。研究者们在β链中发现了另外一个高亲和力Ca2+结合位点,位置在β381–385[81]。此外,纤维蛋白单体的β链和γ链中还存在8-10个低亲和力Ca2+结合位点,其中4个(2对)得到了准确定位,一对在β链的BβAsp381、BβAsp383和BβTrp385区域;另一对在γGlu132,BβAsp261及BβAsp398区域[81]。有证据表明Ca2+与γ链高亲和力钙结合位点结合能保证pocket“a”在纤维蛋白聚合过程发挥正常功能,能够形成稳定的A∶a结构,而γ链高亲和力钙结合位点基因突变会严重损坏pocket “a”的功能并导致纤维蛋白原无法完成聚合反应[84-85]。此外,Ca2+与γ链高亲和力钙结合位点结合还能保护γ链免受酶降解[85-86]。目前,Ca2+和低亲和力钙结合位点的结合被认为对纤维蛋白侧向聚合有重要作用,但具体机制仍需进一步研究[87]。 在侧向聚合过程中,Ca2+辅助凝血酶切割释放FpB,FpB的释放会导致knob B的暴露并与β链pocket‘b’结合,促进纤维蛋白侧向聚合,从而形成更加致密的纤维蛋白网络结构,最终导致骨愈合延迟[70]。WANG等[88]将不同浓度的β-磷酸三钙溶液加入大鼠3 mm下颌骨骨缺损模型中,10 d后,测得骨缺损处纤维蛋白的平均直径在对照组(139.5±40.85) nm和50 g/L β-磷酸三钙溶液组 (112.1±43.27) nm明显大于100 g /L β-磷酸三钙溶液组(76.94±27.5) nm和 200 g/L β-磷酸三钙溶液组(71.27±25.79),P < 0.05;他们还观察到纤维蛋白网络的密度随着β-磷酸三钙溶液浓度的增加而增加(P < 0.05)。相似地,在绵羊髂骨临界大小缺损模型中,β-磷酸三钙延迟了早期骨愈合的过程[89]。这可能的解释是由于β-磷酸三钙通过其释放的Ca2+成分对纤维蛋白的侧向聚合过程产生了影响,从而形成了较细的、密度高的纤维蛋白网络,延迟了早期骨愈合过程。 此外, 凝血因子XIII(FXIII)是凝血途径最后一个因子,它可以通过增强纤维蛋白聚合物的机械稳定性来保护纤维蛋白免受剪切应力剪切和血凝块过早降解[90-91]。凝血因子XⅢ含有两个亚基(A亚基和B亚基),在Ca2+存在条件下,凝血酶剪切并释放B亚基,暴露A亚基上的高亲和力钙结合位点并结合Ca2+激活凝血因子XⅢ[92-93]。有趣的是,发挥主要作用的A亚基可以被凝血酶直接水解而失活,但是由于Ca2+的存在,A亚基即使在很高浓度的凝血酶条件下也不会被水 解[94]。这表明,Ca2+可以通过影响凝血因子XⅢ来调节纤维蛋白稳定性,进而形成更加稳定的血凝块结构,具有影响成骨的潜力,见图2。"

| [1] ROBERTS TT, ROSENBAUM AJ. Bone grafts, bone substitutes and orthobiologics: the bridge between basic science and clinical advancements in fracture healing. Organogenesis. 2012;8(4):114-124. [2] ZHENG ZW, CHEN YH, WU DY, et al. Development of an Accurate and Proactive Immunomodulatory Strategy to Improve Bone Substitute Material-Mediated Osteogenesis and Angiogenesis. Theranostics. 2018; 8 (19):5482-5500. [3] KANG H J, MAKKAR P, PADALHIN AR, et al. Comparative study on biodegradation and biocompatibility of multichannel calcium phosphate based bone substitutes. Mater Sci Eng C Mater Biol Appl. 2020;110:110694. [4] GILEV MV, BAZARNY VV, VOLOKITINA EA, et al. Laboratory Monitoring of Bone Tissue Remodeling after Augmentation of Impression Intraarticular Fracture with Different Types of Bone Graft. Bull Exp Biol Med. 2019;167(5):681-684. [5] UDA Y, AZAB E, SUN N, et al. Osteocyte Mechanobiology. Curr Osteoporos Rep. 2017;15(4):318-325. [6] CLAPHAM D E. Calcium signaling. Cell. 2007;131(6):1047-1058. [7] BOOTMAN MD. Calcium signaling. Cold Spring Harb Perspect Biol. 2012;4(7):a011171. [8] MAENO S, NIKI Y, MATSUMOTO H, et al. The effect of calcium ion concentration on osteoblast viability, proliferation and differentiation in monolayer and 3D culture. Biomaterials. 2005;26(23):4847-4855. [9] KAWANO S, SHOJI S, ICHINOSE S, et al. Characterization of Ca(2+) signaling pathways in human mesenchymal stem cells. Cell Calcium. 2002;32(4):165-174. [10] WANG X, ZHANG Y, JI W, et al. Categorising bone defect hematomas - Enhance early bone healing. Med Hypotheses. 2018;113:77-80. [11] YANG Y, XIAO Y. Biomaterials Regulating Bone Hematoma for Osteogenesis. Adv Healthc Mater.2020;e2000726. [12] VITI F, LANDINI M, MEZZELANI A, et al. Osteogenic Differentiation of MSC through Calcium Signaling Activation: Transcriptomics and Functional Analysis. PLoS One. 2016;11(2):e0148173. [13] YE G, GUAN H, KARUSH J, et al. Effects of Ca2+-activated potassium and inward rectifier potassium channel on the differentiation of endothelial progenitor cells from human peripheral blood. Mol Biol Rep. 2014; 41(5):3413-3423. [14] BERRIDGE MJ, LIPP P, BOOTMAN MD. The versatility and universality of calcium signalling. Nat Rev Mol Cell Biol. 2000;1(1):11-21. [15] KAWANO S, OTSU K, KURUMA A, et al. ATP autocrine/paracrine signaling induces calcium oscillations and NFAT activation in human mesenchymal stem cells. Cell Calcium. 2006;39(4):313-324. [16] YE B. Ca2+ oscillations and its transporters in mesenchymal stem cells. Physiol Res. 2010;59(3):323-329. [17] CHEN H, TANG QL, WU XY, et al. Differentiation of human umbilical cord mesenchymal stem cells into germ-like cells in mouse seminiferous tubules. Mol Med Rep. 2015;12(1):819-828. [18] HEI H, GAO J, DONG J, et al. BK Knockout by TALEN-Mediated Gene Targeting in Osteoblasts: KCNMA1 Determines the Proliferation and Differentiation of Osteoblasts. Mol Cells. 2016;39(7):530-535. [19] TAO R, LAU CP, TSE HF, et al. Regulation of cell proliferation by intermediate-conductance Ca2+-activated potassium and volume-sensitive chloride channels in mouse mesenchymal stem cells. Am J Physiol Cell Physiol. 2008;295(5):C1409-1416. [20] PCHELINTSEVA E, DJAMGOZ MBA. Mesenchymal stem cell differentiation: Control by calcium-activated potassium channels. J Cell Physiol. 2018;233(5):3755-3768. [21] AQUINO-MARTíNEZ R, ANGELO AP, PUJOL FV. Calcium-containing scaffolds induce bone regeneration by regulating mesenchymal stem cell differentiation and migration. Stem Cell Res Ther. 2017;8(1):265. [22] DIRCKX N, VAN HUL M, MAES C. Osteoblast recruitment to sites of bone formation in skeletal development, homeostasis, and regeneration. Birth Defects Res C Embryo Today. 2013;99(3):170-191. [23] NAKAMURA S, MATSUMOTO T, SASAKI J, et al. Effect of calcium ion concentrations on osteogenic differentiation and hematopoietic stem cell niche-related protein expression in osteoblasts. Tissue Eng Part A. 2010;16(8):2467-2473. [24] LEI Q, LIN D, HUANG WX, et al. [Effects of calcium ion on the migration and osteogenic differentiation of human osteoblasts]. Hua Xi Kou Qiang Yi Xue Za Zhi. 2018;36(6):602-608. [25] JUNG GY, PARK YJ, HAN JS. Effects of HA released calcium ion on osteoblast differentiation. J Mater Sci Mater Med. 2010;21(5):1649-1654. [26] CARY R L, WADDELL S, RACIOPPI L, et al. Inhibition of Ca²⁺/calmodulin-dependent protein kinase kinase 2 stimulates osteoblast formation and inhibits osteoclast differentiation. J Bone Miner Res. 2013;28(7): 1599-1610. [27] LIU W, LE CC, WANG D, et al. Ca(2+)/CaM/CaMK signaling is involved in cadmium-induced osteoclast differentiation. Toxicology. 2020;441: 152520. [28] ZAYZAFOON M, FULZELE K, MCDONALD JM. Calmodulin and calmodulin-dependent kinase IIalpha regulate osteoblast differentiation by controlling c-fos expression. J Biol Chem. 2005;280(8):7049-7059. [29] KUKUSHKIN AN, ABRAMOVA MV, SVETLIKOVA SB, et al. Downregulation of c-fos gene transcription in cells transformed by E1A and cHa-ras oncogenes: a role of sustained activation of MAP/ERK kinase cascade and of inactive chromatin structure at c-fos promoter. Oncogene. 2002; 21(5):719-730. [30] WAGNER EF. Functions of AP1 (Fos/Jun) in bone development. Ann Rheum Dis. 2002;61 Suppl 2(Suppl 2):ii40-42. [31] BABU RL, NAVEEN KUMAR M, PATIL RH, et al. Effect of estrogen and tamoxifen on the expression pattern of AP-1 factors in MCF-7 cells: role of c-Jun, c-Fos, and Fra-1 in cell cycle regulation. Mol Cell Biochem. 2013;380(1-2):143-151. [32] ZAYZAFOON M. Calcium/calmodulin signaling controls osteoblast growth and differentiation. J Cell Biochem. 2006;97(1):56-70. [33] MANOLAGAS SC. Birth and death of bone cells: basic regulatory mechanisms and implications for the pathogenesis and treatment of osteoporosis. Endocr Rev. 2000;21(2):115-137. [34] BOYLE WJ, SIMONET WS, LACEY DL. Osteoclast differentiation and activation. Nature. 2003;423(6937):337-342. [35] TAKAYANAGI H, KIM S, TANIGUCHI T. Signaling crosstalk between RANKL and interferons in osteoclast differentiation. Arthritis Res. 2002;4 Suppl 3 (Suppl 3):S227-232. [36] 李平,马超, 李小娇,等. 钙离子信号在破骨细胞中作用的研究进展[J].中国骨与关节外科,2011,4(5):417-421. [37] CHENG X, HOOKWAY ES, KASHIMA T, et al. The role of calcium and nicotinic acid adenine dinucleotide phosphate (NAADP) in human osteoclast formation and resorption. Calcif Tissue Int. 2015;96(1):73-79. [38] SHIN MM, KIM YH, KIM SN, et al. High extracellular Ca2+ alone stimulates osteoclast formation but inhibits in the presence of other osteoclastogenic factors. Exp Mol Med. 2003;35(3):167-174. [39] MENTAVERRI R, YANO S, CHATTOPADHYAY N, et al. The calcium sensing receptor is directly involved in both osteoclast differentiation and apoptosis. FASEB J. 2006;20(14):2562-2564. [40] BOUDOT C, SAIDAK Z, BOULANOUAR A K, et al. Implication of the calcium sensing receptor and the Phosphoinositide 3-kinase/Akt pathway in the extracellular calcium-mediated migration of RAW 264.7 osteoclast precursor cells. Bone. 2010;46(5):1416-1423. [41] NGUYEN LH, ANNABI N, NIKKHAH M, et al. Vascularized bone tissue engineering: approaches for potential improvement. Tissue Eng Part B Rev. 2012; 18 (5): 363-382. [42] LIU Y, CHAN JK, TEOH SH. Review of vascularised bone tissue-engineering strategies with a focus on co-culture systems. J Tissue Eng Regen Med. 2015;9(2):85-105. [43] MOCCIA F, TANZI F, MUNARON L. Endothelial remodelling and intracellular calcium machinery. Curr Mol Med. 2014;14(4):457-480. [44] MOCCIA F, BERRA-ROMANI R, TANZI F. Update on vascular endothelial Ca(2+) signalling: A tale of ion channels, pumps and transporters. World J Biol Chem. 2012;3(7):127-158. [45] PINTO MC, KIHARA AH, GOULART VA, et al. Calcium signaling and cell proliferation. Cell Signal. 2015;27(11):2139-2149. [46] PINTON P, GIORGI C, SIVIERO R, et al. Calcium and apoptosis: ER-mitochondria Ca2+ transfer in the control of apoptosis. Oncogene. 2008;27(50):6407-6418. [47] WESTERLUND AM, DELEMOTTE L. Effect of Ca2+ on the promiscuous target-protein binding of calmodulin. PLoS Comput Biol. 2018;14(4): e1006072. [48] TOUSSAINT F, CHARBEL C, BLANCHETTE A, et al. CaMKII regulates intracellular Ca²⁺ dynamics in native endothelial cells. Cell Calcium. 2015;58(3):275-285. [49] KAHL CR, MEANS AR. Regulation of cell cycle progression by calcium/calmodulin-dependent pathways. Endocr Rev. 2003;24(6):719-736. [50] LI XX, LIU YM, LI YJ, et al. High glucose concentration induces endothelial cell proliferation by regulating cyclin-D2-related miR-98. J Cell Mol Med. 2016;20(6):1159-1169. [51] HUI S, CHOI J, ZAIDI S, et al. Peptide-mediated disruption of calmodulin-cyclin E interactions inhibits proliferation of vascular smooth muscle cells and neointima formation. Circ Res. 2011;108(9):1053-1062. [52] JIN Y, MUHL L, BURMAKIN M, et al. Endoglin prevents vascular malformation by regulating flow-induced cell migration and specification through VEGFR2 signalling. Nat Cell Biol. 2017;19(6):639-652. [53] ANGHELESCU VM, NECULAE I, DINCA O, et al. Inflammatory-Driven Angiogenesis in Bone Augmentation with Bovine Hydroxyapatite, B-Tricalcium Phosphate, and Bioglasses: A Comparative Study. J Immunol Res. 2018;2018:9349207. [54] KUMAGAI H, MAKIHARA T, FUNAYAMA T, et al. Angiogenesis and new bone formation in novel unidirectional porous beta-tricalcium phosphate: a histological study. J Artif Organs. 2019;22(4):294-299. [55] KUSUMBE AP, RAMASAMY SK, ADAMS RH. Coupling of angiogenesis and osteogenesis by a specific vessel subtype in bone. Nature. 2014; 507(7492):323-328. [56] RATHER HA, JHALA D, VASITA R. Dual functional approaches for osteogenesis coupled angiogenesis in bone tissue engineering. Mater Sci Eng C Mater Biol Appl. 2019;103:109761. [57] PIKUŁA M, LANGA P, KOSIKOWSKA P, et al. Stem cells and growth factors in wound healing. Postepy Hig Med Dosw (Online). 2015;69: 874-885. [58] WALSH DP, RAFTERY RM, CHEN G, et al. Rapid healing of a critical-sized bone defect using a collagen-hydroxyapatite scaffold to facilitate low dose, combinatorial growth factor delivery. J Tissue Eng Regen Med. 2019;13(10):1843-1853. [59] LUGINBUEHL V, ZOIDIS E, MEINEL L, et al. Impact of IGF-I release kinetics on bone healing: a preliminary study in sheep. Eur J Pharm Biopharm. 2013;85(1):99-106. [60] WU M, CHEN G, LI Y P. TGF-β and BMP signaling in osteoblast, skeletal development, and bone formation, homeostasis and disease. Bone Res. 2016;4:16009. [61] LADDHA AP, KULKARNI YA. VEGF and FGF-2: Promising targets for the treatment of respiratory disorders. Respir Med. 2019;156:33-46. [62] CAPLAN AI, CORREA D. PDGF in bone formation and regeneration: new insights into a novel mechanism involving MSCs. J Orthop Res. 2011; 29(12):1795-1803. [63] NESBITT WS, GIULIANO S, KULKARNI S, et al. Intercellular calcium communication regulates platelet aggregation and thrombus growth. J Cell Biol. 2003;160(7):1151-1161. [64] MARTíNEZ CE, SMITH PC, PALMA ALVARADO VA. The influence of platelet-derived products on angiogenesis and tissue repair: a concise update. Front Physiol. 2015;6:290. [65] CAVALLO C, ROFFI A, GRIGOLO B, et al. Platelet-Rich Plasma: The Choice of Activation Method Affects the Release of Bioactive Molecules. Biomed Res Int. 2016;2016:6591717. [66] GARNER AL, FRELINGER AL, 3RD, GERRITS AJ, et al. Using extracellular calcium concentration and electric pulse conditions to tune platelet-rich plasma growth factor release and clotting. Med Hypotheses. 2019; 125:100-105. [67] WANG M, TAN J, MIAO Y, et al. Role of Ca²⁺ and ion channels in the regulation of apoptosis under hypoxia. Histol Histopathol. 2018;33(3): 237-246. [68] ECHEVERRI LF, HERRERO MA, LOPEZ JM, et al. Early stages of bone fracture healing: formation of a fibrin-collagen scaffold in the fracture hematoma. Bull Math Biol. 2015;77(1):156-183. [69] KOLAR P, SCHMIDT-BLEEK K, SCHELL H, et al. The early fracture hematoma and its potential role in fracture healing. Tissue Eng Part B Rev. 2010;16(4):427-434. [70] WANG X, FRIIS T, GLATT V, et al. Structural properties of fracture haematoma: current status and future clinical implications. J Tissue Eng Regen Med. 2017;11(10):2864-2875. [71] SHIU HT, GOSS B, LUTTON C, et al. Controlling whole blood activation and resultant clot properties by carboxyl and alkyl functional groups on material surfaces: a possible therapeutic approach for enhancing bone healing. J Mater Chem B. 2014;2(20):3009-3021. [72] WEISEL JW, LITVINOV RI. Mechanisms of fibrin polymerization and clinical implications. Blood. 2013;121(10):1712-1719. [73] SISSON K, ZHANG C, FARACH-CARSON MC, et al. Fiber diameters control osteoblastic cell migration and differentiation in electrospun gelatin. J Biomed Mater Res A. 2010;94(4):1312-1320. [74] O’KEEFE RJ. Fibrinolysis as a Target to Enhance Fracture Healing. N Engl J Med. 2015;373(18):1776-1778. [75] BADAMI AS, KREKE MR, THOMPSON MS, et al. Effect of fiber diameter on spreading, proliferation, and differentiation of osteoblastic cells on electrospun poly(lactic acid) substrates. Biomaterials. 2006;27(4): 596-606. [76] DAS S, JHINGRAN R, BAINS VK, et al. Socket preservation by beta-tri-calcium phosphate with collagen compared to platelet-rich fibrin: A clinico-radiographic study. Eur J Dent. 2016;10(2):264-276. [77] COLLET J P, PARK D, LESTY C, et al. Influence of fibrin network conformation and fibrin fiber diameter on fibrinolysis speed: dynamic and structural approaches by confocal microscopy. Arterioscler Thromb Vasc Biol. 2000;20(5):1354-1361. [78] PETERS A, SCHELL H, BAIL HJ, et al. Standard bone healing stages occur during delayed bone healing, albeit with a different temporal onset and spatial distribution of callus tissues. Histol Histopathol. 2010;25(9): 1149-1162. [79] CAMERON JA, MILNER DJ, LEE JS, et al. Employing the biology of successful fracture repair to heal critical size bone defects. Curr Top Microbiol Immunol. 2013;367:113-132. [80] MEHTA M, SCHELL H, SCHWARZ C, et al. A 5-mm femoral defect in female but not in male rats leads to a reproducible atrophic non-union. Arch Orthop Trauma Surg. 2011;131(1):121-129. [81] LUGOVSKOĬ EV, GRITSENKO PG, KOMISARENKO SV. [Molecular mechanisms of the polymerization of fibrin and the formation of its three-dimensional network]. Bioorg Khim. 2009;35(4):437-456. [82] WEISEL JW, LITVINOV RI. Fibrin Formation, Structure and Properties. Subcell Biochem. 2017;82:405-456. [83] KAMIJO T, MUKAI S, TAIRA C, et al. γD318Y fibrinogen shows no fibrin polymerization due to defective “A-a” and “B-b” interactions, whereas that of γK321E fibrinogen is nearly normal. Thromb Res. 2019;182: 150-158. [84] REMIJN JA, LOUNES KC, HOGAN KA, et al. Mutations on fibrinogen (gamma 316-322) are associated with reduction in platelet adhesion under flow conditions. Ann N Y Acad Sci. 2001;936:444-448. [85] IKEDA M, KOBAYASHI T, ARAI S, et al. Recombinant γT305A fibrinogen indicates severely impaired fibrin polymerization due to the aberrant function of hole ‘A’ and calcium binding sites. Thromb Res. 2014;134(2): 518-525. [86] BRENNAN SO, DAVIS RL, MOSESSON MW, et al. Congenital hypodysfibrinogenaemia (Fibrinogen Des Moines) due to a gamma320Asp deletion at the Ca2+ binding site. Thromb Haemost. 2007;98(2):467-469. [87] ALEMAN MM, BYRNES JR, WANG JG, et al. Factor XIII activity mediates red blood cell retention in venous thrombi. J Clin Invest. 2014;124(8): 3590-3600. [88] WANG X, LUO Y, YANG Y, et al. Alteration of clot architecture using bone substitute biomaterials (beta-tricalcium phosphate) significantly delays the early bone healing process. J Mater Chem B. 2018;6(48):8204-8213. [89] CHOO T, MARINO V, BARTOLD PM. Effect of PDGF-BB and beta-tricalcium phosphate (beta-TCP) on bone formation around dental implants: a pilot study in sheep. Clin Oral Implants Res. 2013;24(2): 158-166. [90] DUVAL C, ALLAN P, CONNELL SD, et al. Roles of fibrin alpha- and gamma-chain specific cross-linking by FXIIIa in fibrin structure and function. Thromb Haemost. 2014;111(5):842-850. [91] VASILYEVA A, YURINA L, INDEYKINA M, et al. Oxidation-induced modifications of the catalytic subunits of plasma fibrin-stabilizing factor at the different stages of its activation identified by mass spectrometry. Biochim Biophys Acta Proteins Proteom. 2018;1866(8):875-884. [92] PROTOPOPOVA A D, RAMIREZ A, KLINOV DV, et al. Factor XIII topology: organization of B subunits and changes with activation studied with single-molecule atomic force microscopy. J Thromb Haemost. 2019; 17(5):737-748. [93] HETHERSHAW EL, CILIA LA CORTE AL, DUVAL C, et al. The effect of blood coagulation factor XIII on fibrin clot structure and fibrinolysis. J Thromb Haemost. 2014;12(2):197-205. [94] MUSZBEK L, BERECZKY Z, BAGOLYZ, et al. Factor XIII: a coagulation factor with multiple plasmatic and cellular functions. Physiol Rev. 2011; 91(3):931-972. |
| [1] | Chen Junyi, Wang Ning, Peng Chengfei, Zhu Lunjing, Duan Jiangtao, Wang Ye, Bei Chaoyong. Decalcified bone matrix and lentivirus-mediated silencing of P75 neurotrophin receptor transfected bone marrow mesenchymal stem cells to construct tissue-engineered bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 510-515. |
| [2] | Ma Zhijie, Li Jingyu, Cao Fang, Liu Rong, Zhao Dewei. Influencing factors and biological property of novel biomedical materials: porous silicon carbide coated with bioactive tantalum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 558-563. |
| [3] | Shi Xiaoxiu, Mao Shilong, Liu Yang, Ma Xingshuang, Luo Yanfeng. Comparison of tantalum and titanium (alloy) as orthopedic materials: physical and chemical indexes, antibacterial and osteogenic ability [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 593-599. |
| [4] | Su Chengli, Zhu Songsong, Li Yunfeng. Application of transport distraction osteogenesis in reconstruction of mandibular segmental defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(35): 5604-5609. |
| [5] | Huang Minling, Lu Zhaoqi, Shen Zhen, Lin Haixiong, Feng Junjie, Huang Feng, Jiang Ziwei, Cai Qunbin. Effects of total flavone of Rhizoma Drynariae on the coupling of angiogenesis and osteogenesis in bone remodeling through notch signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(32): 5116-5122. |
| [6] | Zhao Qianzeng, Zhao Zhenqun, Liu Wanlin. Endoplasmic reticulum stress regulates autophagy and apoptosis in steroid-induced avascular necrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(29): 4685-4690. |
| [7] | Zhang Chuanhui, Li Jianjun, Yang Jun. Effect of dynamic compression combined with insulin-like growth factor-1 gene transfection on chondrogenic differentiation of rabbit adipose-derived mesenchymal stem cells in chitosan/gelatin scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(28): 4485-4491. |
| [8] | Qu Xiangyang, Song Qinyong. Piezo 1 mediates apoptosis of fibroblast-like synovial cells in rheumatoid arthritis via MAPK/ERK5 signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(26): 4156-4161. |
| [9] | Chen Weijian, Zhang Gangyu, Lin Tianye, Liang Du, Wang Haibin. N6-methyladenosine RNA methylation is involved in orthopedic related diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(26): 4236-4242. |
| [10] | Yang Junhui, Luo Jinli, Yuan Xiaoping. Effects of human growth hormone on proliferation and osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3956-3961. |
| [11] | Li Xinping, Cui Qiuju, Zeng Shuguang, Ran Gaoying, Zhang Zhaoqiang, Liu Xianwen, Fang Wei, Xu Shuaimei. Effect of modification of β-tricalcium phosphate/chitosan hydrogel on growth and mineralization of dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3493-3499. |
| [12] | Zhou Anqi, Tang Yufei, Wu Bingfeng, Xiang Lin. Designing of periosteum tissue engineering: combination of generality and individuality [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3551-3557. |
| [13] | Chen Song, He Yuanli, Xie Wenjia, Zhong Linna, Wang Jian. Advantages of calcium phosphate nanoparticles for drug delivery in bone tissue engineering research and application [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3565-3570. |
| [14] | Ailimaierdan·Ainiwaer, Wang Ling, Gu Li, Dilidaer•Taxifulati, Wang Shan, Yin Hongbin. Effect of transforming growth factor-beta3 on the proliferation and osteogenic capability of osteoblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(17): 2664-2669. |
| [15] | Huang Na, Liu Jiayue, Huang Yingjie, Wen Junmao, Wang Haibin, Zhang Qingwen, Zhou Chi . Bibliometric and visualized analysis of research on osteonecrosis of the femoral head from the Web of Science in the last 5 years [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(17): 2711-2718. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||