Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (21): 3292-3299.doi: 10.3969/j.issn.2095-4344.3873
Previous Articles Next Articles
Biological response of Co2+ to preosteoblasts during aseptic loosening of the prosthesis
Jiang Shengyuan1, Li Dan2, Jiang Jianhao1, Shang-you Yang3, 4, Yang Shuye1
- 1Department of Traumatic Orthopedics, Binzhou Medical University Hospital, Binzhou 256600, Shandong Province, China; 2Chongqing Wushan County People’s Hospital, Chongqing 404700, China; 3Department of Orthopedic Surgery, the University of Kansas School of Medicine-Wichita, Wichita, KS, USA; 4Department of Biological Sciences, Wichita State University, Wichita, KS, USA
-
Received:2019-12-06Revised:2019-12-10Accepted:2020-07-20Online:2021-07-28Published:2021-01-23 -
Contact:Yang Shuye, MD, Department of Traumatic Orthopedics, Binzhou Medical University Hospital, Binzhou 256600, Shandong Province, China Shang-you Yang, Professor, Department of Orthopedic Surgery, the University of Kansas School of Medicine-Wichita, Wichita, KS, USA; Department of Biological Sciences, Wichita State University, Wichita, KS, USA -
About author:Jiang Shengyuan, Master candidate, Department of Traumatic Orthopedics, Binzhou Medical University Hospital, Binzhou 256600, Shandong Province, China Li Dan, Nurse-in-charge, Chongqing Wushan County People’s Hospital, Chongqing 404700, China Jiang Shengyuan and Li Dan contributed equally to this work. -
Supported by:the Medical and Health Technology Development Plan Project of Shandong Province, No. 2016WS0023 (to YSY); the Scientific Research Plan from the Binzhou Medical University, No. BY2016KYQD19 (to YSY)
CLC Number:
Cite this article
Jiang Shengyuan, Li Dan, Jiang Jianhao, Shang-you Yang, Yang Shuye. Biological response of Co2+ to preosteoblasts during aseptic loosening of the prosthesis[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(21): 3292-3299.
share this article
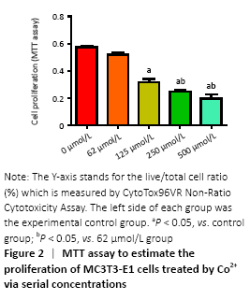
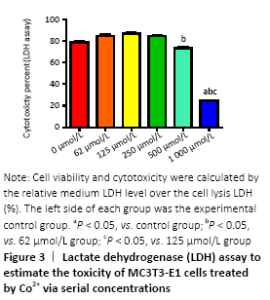
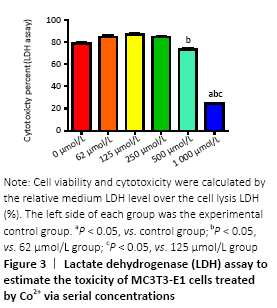
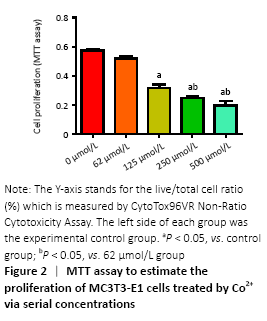
Cytotoxicity of divalent cobalt ions and their effects on cell proliferation (in vitro) Cells were cultured with Co2+ of different concentrations for 72 hours. MTT cell proliferation assay showed that the experimental groups which included 125, 250, 500 μmol/L Co2+ inhibited the proliferation of MC3T3-E1 cells in varying degrees (P < 0.05; Figure 2). LDH assay showed significant toxicity when Co2+ reached 1 000 μmol/L and Co2+ showed slight toxicity at 500 μmol/L (P < 0.05; Figure 3). "
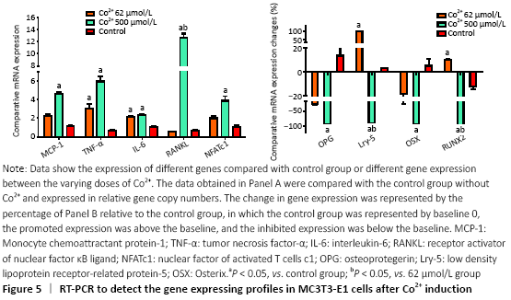
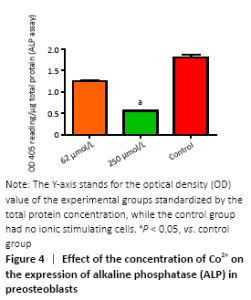
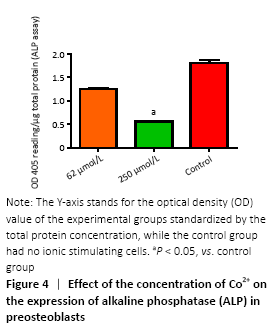
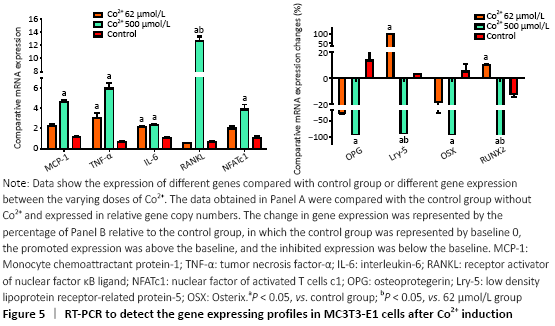
Gene expression in preosteoblasts (in vitro) RT-PCR was used to detect the expression of a variety of genes to further investigate the molecular biological mechanisms underlying the effects of different concentrations of Co2+ (62 μmol/L and 500 μmol/L) on MC3T3-E1 cells. The data showed that Co2+ significantly promoted MCP-1 gene expression at 500 μmol/L compared with the control group (P < 0.05). Both groups significantly promoted the expression of TNF-α and IL-6, and they also promoted the expression of RANKL. When Co2+ was at 500 μmol/L, the expression of RANKL was significantly increased compared with that of 62 μmol/L group. The expression of NFATc1 stimulated by Co2+ was significantly enhanced at 500 μmol/L compared with the control group (P < 0.05). The expression levels of OPG and OSX were inhibited in both experimental groups. When Co2+ was at 500 μmol/L, there were significantly differences with the control group; thus, the RANKL/OPG ratio was much higher than the control value (P < 0.05). Same trend with Lrp-5 and Runx2, both of them promoted the expression of Co2+ at 62 μmol/L and inhibited the expression at 500 μmol/L compared with the control group (P < 0.05; Figure 5). "

General situation and surgical results of experimental mice (in vivo) The mice in each group grew and moved well, and generally responded well. No mice died in each group. The uneven distribution of cobalt and chromium particles around the prosthesis and the knee joint was observed after the knee joint was severed, and no significant anatomical changes were observed in the gross morphology of the knee joint (Figure 6)."

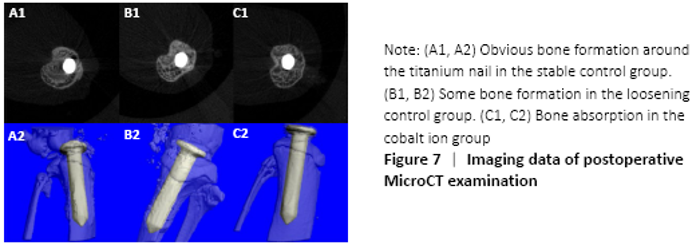
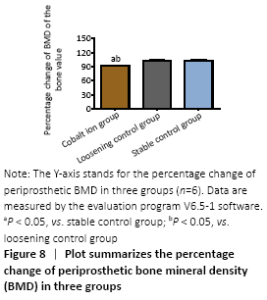
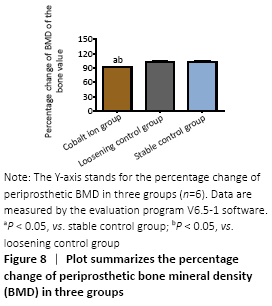
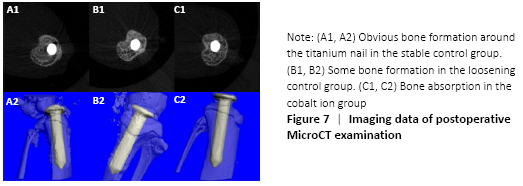
MicroCT evaluation results (in vivo) MicroCT scans showed that mice in the stable control group had good fixation and no displacement, bone ingrowth around the titanium nail could be seen obviously. There was visible bone resorption around the titanium nail in the cobalt ion group (Figure 7), and the percentage of changes of bone mineral density before and after treatment was significantly lower than that in the loose control group and the stable control group (P < 0.05; Figure 8)."
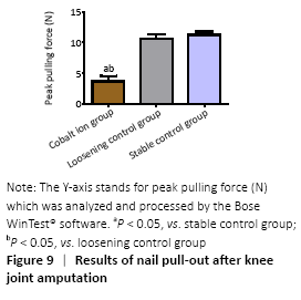
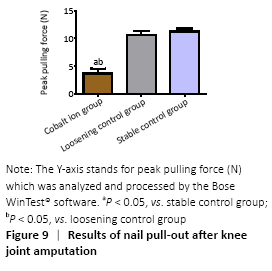
Stability evaluation of titanium nail prosthesis-nail pulling test (in vivo) The blade holding the distal tibia of the mice which were fixed well by bone cement was able to withstand at least 100 Newtons (N) tension. In the stable control group, the maximum shear force needed at the moment of pullout was (11.58±2.47) N and (10.92±2.44) N in the loose control group, while in the cobalt ion group, the pullout force of the titanium nail was further reduced to (5.93±1.91) N. There were significant differences between the experimental group and the control group (P < 0.05; Figure 9)."
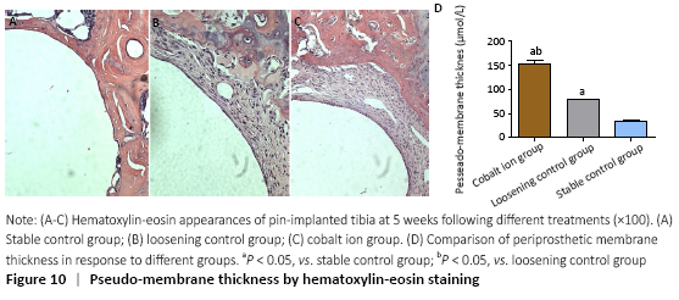
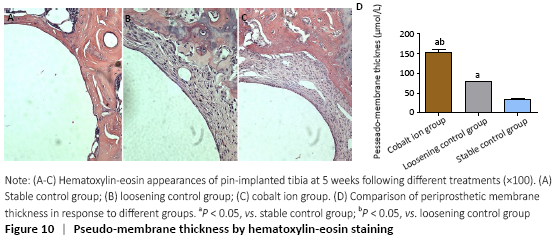
Observation of histomorphology (in vivo) Hematoxylin-eosin staining showed that there was obvious bone ingrowth and few inflammatory cells infiltration around the titanium nail in the stable control group. There was obvious inflammatory reaction membrane formation and a large number of inflammatory cell infiltration around the prosthesis in the cobalt ion group (Figure 10A-C). The comparison of periprosthetic membrane thickness between the above groups was calculated by computer image analysis software. The thickness of inflammatory reaction membrane around prosthesis in cobalt ion group was significantly thicker than that other two control groups (P < 0.05; Figure 10D). Differentiation and quantity of osteoclasts at the titanium nail prosthesis-bone interface were evaluated by TRAP staining, which showed that the degree of them in the surrounding tissues of the cobalt ion group was significantly higher than that of the control group (P < 0.05; Figure 11)."
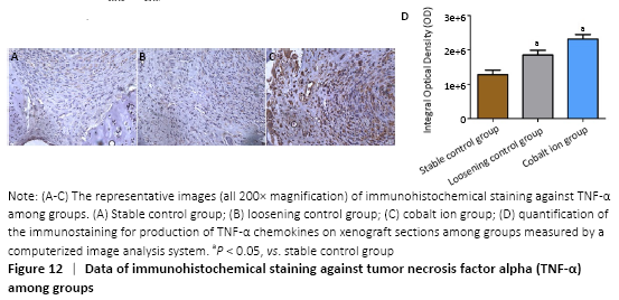
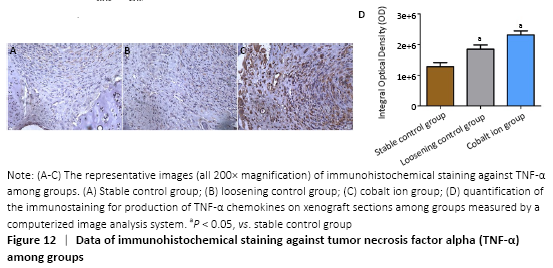
Immunohistochemical assessments (in vivo) The expression of the positive-staining of TNF-α among groups is shown in the Figure 12A-C. There was almost no expression of TNF-α in the stable control group, but the cobalt ion group was the most obvious. All staining conditions were summarized, and it was concluded that the cobalt ion group and loosening control group were significantly higher than the stable control group in staining (P < 0.05; Figure 12D)."
| [1] Holt G, Murnaghan C, Reilly J, et al. The biology of aseptic osteolysis. Clin Orthop Relate Res. 2007;460:240-252. [2] Pajarinen J, Lin TH, Nabeshima A, et al. Mesenchymal stem cells in the aseptic loosening of total joint replacements. J Biomed Mater Res Part A. 2017;105(4):1195-1207. [3] Inacio MC, Ake CF, Paxton EW, et al. Sex and risk of hip implant failure: assessing total hip arthroplasty outcomes in the United States. JAMA Intern Med. 2013;173(6):435-441. [4] Saeed K, Dryden M, Bassetti M, et al. Prosthetic joints : shining lights on challenging blind spots. Int J Antimicrob Agents. 2017;49(2): 153-161. [5] Jiang Y, Jia T, Gong W, et al. Effects of Ti, PMMA, UHMWPE, and Co-Cr wear particles on differentiation and functions of bone marrow stromal cells. J Biomed Mater Res Part A. 2013;101(10):2817-2825. [6] Yang SY, Zhang K, Bai L, et al. Polymethylmethacrylate and titanium alloy particles activate peripheral monocytes during periprosthetic inflammation and osteolysis. J Orthop Res. 2011;29(5):781-786. [7] Lochner K, Fritsche A, Jonitz A, et al. The potential role of human osteoblasts for periprosthetic osteolysis following exposure to wear particles. Int J Mol Med. 2011;28(6):1055-1063. [8] Chen W, Li Z, Guo Y, et al. Wear particles impair antimicrobial activity via suppression of reactive oxygen species generation and ERK1/2 phosphorylation in activated macrophages. Inflammation. 2015;38(3):1289-1296. [9] Jonitz-Heincke A, Lochner K, Schulze C, et al. Contribution of human osteoblasts and macrophages to bone matrix degradation and proinflammatory cytokine release after exposure to abrasive endoprosthetic wear particles. Mol Med Rep. 2016;14(2):1491-500. [10] Tereza D, Jiri G, Eva K. The role of the chemokine system in tissue response to prosthetic by-products leading to periprosthetic osteolysis and aseptic loosening. Front Immunol. 2017;8:1026. [11] Wang W, Ferguson DJP, Quinn JMW, et al. Biomaterial particle phagocytosis by bone-resorbing osteoclasts. Bone Joint J. 1997; 79-B(5):849-856. [12] Ollivere B, Wimhurst J A, Clark IM, et al. Current concepts in osteolysis. J Bone Joint Surg Br Vol. 2012;94(1):10-15. [13] Lass R, Alexander G, Kolb A, et al. Comparison of synovial fluid, urine, and serum ion levels in metal-on-metal total hip arthroplasty at a minimum follow-up of 18 years. J Orthop Res. 2014;32(9):1234-1240. [14] Lombardi JAV, Barrack RL, Berend KR, et al. The hip society: algorithmic approach to diagnosis and management of metal-on-metal arthroplasty. Bone Joint J. 2012;94-B(11 Supple A):14-18. [15] Keegan GM, Learmonth ID, Case CP. Orthopaedic metals and their potential toxicity in the arthroplasty patient: a review of current knowledge and future strategies. J Bone Joint Surg Br Vol. 2007;89(5): 567. [16] Hailer NP, Blaheta RA, Dahlstrand H, et al. Elevation of circulating HLA DR\r, +\r, CD8\r, +\r, T-cells and correlation with chromium and cobalt concentrations 6 years after metal-on-metal hip arthroplasty. Acta Orthop. 2011;82(1):6-12. [17] Caicedo MS, Desai R, Mcallister K, et al. Soluble and particulate Co-Cr-Mo alloy implant metals activate the inflammasome danger signaling pathway in human macrophages: a novel mechanism for implant debris reactivity. J Orthop Res. 2009;27(7):847-854. [18] Jian-Hao J, Peng L, Gang-Qiang D, et al. Cobalt-chromium particles inducing preosteoblasts may aggravate periprosthetic inflammation. Zhongguo Zuzhi Gongcheng Yanjiu. 2019;23(26):4101-4108. [19] Yang S, Zhang K, Li F, et al. Biological responses of preosteoblasts to particulate and ion forms of Co-Cr alloy. J Biomed Mater Res Part A. 2015;103(11):3564-3571. [20] Yang SY, Yu H, Gong W, et al. Murine model of prosthesis failure for the long-term study of aseptic loosening. J Orthop Res. 2007;25(5): 603-611. [21] Zhang L, Jia TH, Chong ACM, et al. Cell-based osteoprotegerin therapy for debris-induced aseptic prosthetic loosening on a murine model. Gene Ther. 2010;17(10):1262-1269. [22] Goodman SB, Gibon E, Pajarinen J, et al. Novel biological strategies for treatment of wear particle-induced periprosthetic osteolysis of orthopaedic implants for joint replacement. J R Soc Interface. 2014;11(93):20130962. [23] Nakamura-Ota M, Hamanaka R, Yano H, et al. A new murine osteoblastic cell line immortalized with the SV40 large T antigen. Cell Tissue Banking. 2014;15(3):373-380. [24] Cadosch D, Gautschi OP, Chan E, et al. Titanium induced production of chemokines CCL17/TARC and CCL22/MDC in human osteoclasts and osteoblasts. J Biomed Mater Res Part A. 2010;92A(2): 475-483. [25] Chiu R, Smith KE, Ma GK, et al. Polymethylmethacrylate particles impair osteoprogenitor viability and expression of osteogenic transcription factors Runx2, osterix, and Dlx5. J Orthop Res. 2010; 28(5):571-577. [26] Nakamura Y, Hasebe A, Takahashi K, et al. Oligomerization-induced conformational change in the c-terminal region of nel-like molecule 1 (NELL1) protein is necessary for the efficient mediation of murine MC3T3-E1 cell adhesion and spreading. J Biol Chem. 2014; 289(14):9781-9794. [27] Hallab NJ, Anderson S, Caicedo M, et al. Effects of soluble metals on human peri-implant cells. 2005;74A(1):124-140. [28] Gallo J, Goodman SB, Konttinen YT, et al. Osteolysis around total knee arthroplasty: A review of pathogenetic mechanisms. Acta Biomater. 2013;9(9):8046-8058. [29] Haleem-Smith H, Argintar E, Bush C, et al. Biological responses of human mesenchymal stem cells to titanium wear debris particles. J Orthop Res. 2012;30(6):853-863. [30] Dalal A, Pawar V, Mcallister K, et al. Orthopedic implant cobalt-alloy particles produce greater toxicity and inflammatory cytokines than titanium alloy and zirconium alloy-based particles in vitro, in human osteoblasts, fibroblasts, and macrophages. J Biomed Mater Res Part A. 2012;100A(8):2147-2158. [31] Mandelin J, Li TF, Liljeström M, et al. Imbalance of RANKL/RANK/OPG system in interface tissue in loosening of total hip replacement. Bone Joint J. 2003;85(8):1196-1201. [32] Zijlstra WP, Bulstra SK, Raay J, et al. Cobalt and chromium ions reduce human osteoblast-like cell activity in vitro, reduce the OPG to RANKL ratio, and induce oxidative stress. J Orthop Res. 2012;30(5): 740-747. [33] Millett PJ, Allen MJ, Bostrom MPG. Effects of alendronate on particle-induced osteolysis in a rat model. J Bone Joint Surg. 2002; 84-A(2):236-249. [34] Ren WP, Yang SY, Wooley PH. A novel murine model of orthopaedic wear-debris associated osteolysis. Scand J Rheumatol. 2004;33(5): 349-357. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Zhang Lishu, Liu Anqi, He Xiaoning, Jin Yan, Li Bei, Jin Fang. Alpl gene affects the therapeutic effect of bone marrow mesenchymal stem cells on ulcerative colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3970-3975. |
| [12] | Wang Qiufei, Gu Ye, Peng Yuqin, Xue Feng, Ju Rong, Zhu Feng, Wang Yijun, Geng Dechun, Xu Yaozeng. Effect of Wnt/beta-catenin signaling pathway on osteoblasts under the action of wear particles [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3894-3901. |
| [13] | Yang Caihui, Liu Qicheng, Dong Ming, Wang Lina, Zuo Meina, Lu Ying, Niu Weidong. Serine/threonine protein kinases can promote bone destruction in mouse models of chronic periapical periodontitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3654-3659. |
| [14] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [15] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||