Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (13): 2114-2120.doi: 10.3969/j.issn.2095-4344.2184
Previous Articles Next Articles
Mesenchymal stem cells in the treatment of spinal cord injury: cell therapy and combination of new drugs and biomaterials
Qian Nannan1, Zhang Qian2, Yang Rui1, Ao Jun3, Zhang Tao1
- 1Key Laboratory of Cell Engineering in Guizhou Province, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China; 2Department of Human Anatomy, School of Basic Medicine, Zunyi Medical University, Zunyi 563003, Guizhou Province, China; 3Department of Spinal Surgery, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China
-
Received:2020-03-30Revised:2020-04-03Accepted:2020-05-09Online:2021-05-08Published:2020-12-29 -
Contact:Zhang Tao, PhD, Professor, Master’s supervisor, Key Laboratory of Cell Engineering in Guizhou Province, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China -
About author:Qian Nannan, Master candidate, Key Laboratory of Cell Engineering in Guizhou Province, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China -
Supported by:the National Natural Science Foundation of China, No. 81960299 (to ZT); the Special Project of Academic New Seedling Cultivation and Innovation Exploration of Zunyi Medical University, No. Qian Ke He Platform Talents [2017]5733-029 (to ZQ)
CLC Number:
Cite this article
Qian Nannan, Zhang Qian, Yang Rui, Ao Jun, Zhang Tao. Mesenchymal stem cells in the treatment of spinal cord injury: cell therapy and combination of new drugs and biomaterials[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2114-2120.
share this article
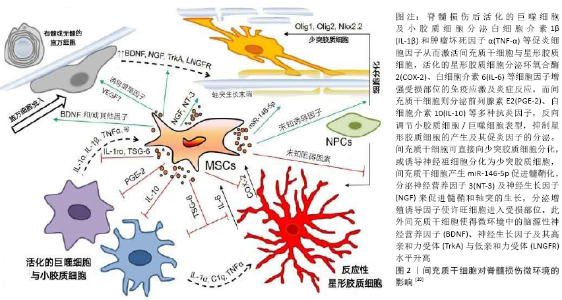
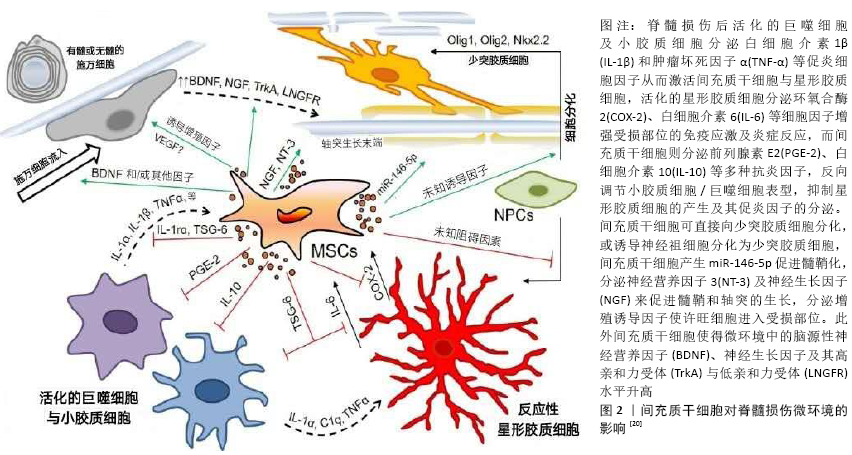
2.1 脊髓损伤概述 脊髓损伤多为高空坠落及交通意外导致的脊髓挫伤或脊髓压迫,临床表现为损伤平面以下感觉和运动神经功能受损甚至完全丧失。脊髓损伤疾病分为原发性损伤和继发性损伤两个阶段:原发性损伤主要是指脊柱受到外力后脊髓完整性遭到破坏,导致血管损伤引起血供受阻,血脊髓屏障通透性升高,组织压迫和缺血水肿等,使得脊髓内环境稳态遭到破坏;继发性损伤是在原发性损伤基础上,血脊髓屏障破坏及局部缺血再灌注损伤引起巨噬细胞、中性粒细胞、星形胶质细胞、淋巴细胞等炎症细胞浸润[5-6],炎症细胞释放促炎性细胞因子、趋化因子、氧自由基、蛋白水解酶及一氧化氮等物质触发炎症级联反应,造成神经元功能障碍或丧失,最终导致脊髓神经元死亡、组织溶解等,并破坏血管内皮细胞造成微血管损伤[7]。脊髓受损部位持续的炎症微环境不利于神经功能细胞的再生和修复[8]。脊髓损伤后中性粒细胞弹性蛋白酶、基质金属蛋白酶9等物质,参与继发性组织损伤、出血性损伤及之后的细胞外基质分子重塑(形成瘢痕组织等),严重影响神经元、轴突等的再生[9-11]。原发性损伤通过手术治疗对受损部位进行异物清除、解除脊髓压迫并恢复脊柱结构完整性,但炎症途径所释放的细胞毒性物质仍使受损脊髓在原发性损伤的基础上进一步损伤,因此及时干预继发性损伤的发生发展对于脊髓损伤的治疗及预后尤为重要。抑制炎症因子的释放,调节免疫反应,诱导血管内皮细胞、神经功能细胞再生及抑制胶质瘢痕的形成等可作为干预治疗脊髓继发性损伤的切入点[12]。 2.2 间充质干细胞的来源与生物学特性 间充质干细胞几乎存在所有组织器官中,但不同组织来源的间充质干细胞其分化潜能、诱导血管新生和免疫调节能力等方面存在差异 性[13-15]。间充质干细胞的生物学特性主要体现在以下几方面:①间充质干细胞体外扩增能力强[3];②间充质干细胞具有多向分化潜能,除可分化为成骨、成软骨、成脂等中胚层组织细胞外,适宜诱导培养条件下还可跨胚层分化为神经元、神经胶质细胞等[16];③间充质干细胞通过自分泌和旁分泌效应与内环境中的其他细胞相互作用,参与炎症反应、免疫功能的调节,促进血管的生成和组织损伤的再生修复[17]。 2.3 间充质干细胞治疗脊髓损伤的临床前研究 近年来,间充质干细胞移植通过进行炎症反应调节及发挥间充质干细胞干性潜能诱导分化为神经功能细胞等治疗脊髓损伤取得显著进展,除此之外,间充质干细胞来源外泌体的治疗研究也备受瞩目,随着间充质干细胞联合生物材料研究的不断深入,移植细胞生存率低、移植细胞定植困难等问题有望解决。 2.3.1 间充质干细胞移植 脊髓损伤后激活的星形胶质细胞及其产生的细胞外基质硫酸软骨素蛋白多糖是构成胶质瘢痕的主要成分,受损部位胶质瘢痕的形成会严重阻碍轴突细胞的迁移及神经纤维再生[18-19]。骨髓间充质干细胞移植能够抑制胶质细胞活化增殖,为神经细胞得以存活营造良好的微环境[20]。间充质干细胞在体内外还可诱导分化为神经元或星形细胞填充脊髓受损部位,并可通过分泌纤维连接蛋白在脊髓横断损伤中诱导轴突延伸,促进神经纤维再生[21]。研究采用外周血间充质干细胞经局部注射移植于脊髓损伤大鼠,发现移植组受损脊髓空腔面积变小,判断运动功能的BBB评分明显提高,白细胞介素6和白细胞介素17a表达下降,转化生长因子β表达增强[22]。诸多研究表明,移植至脊髓受损部位的间充质干细胞会分泌多种抗炎因子,并调节脊髓受损组织中巨噬细胞表型由M1型(促炎性)重编程为M2型(抗炎 型)[20,23],也促使M0型转化为M2型,发挥间接的损伤组织炎症调节作用[24-26]。脊髓损伤大鼠尾静脉输注间充质干细胞,可降低血脊髓屏障的通透性,修复血管完整性,有效抑制了受损部位的出血量及病变体积,脾脏中白细胞介素10水平显著提升,从而抑制全身性炎症反应[27]。研究应用骨髓间充质干细胞联合电针刺激脊髓损伤大鼠脊膜支传入神经纤维,使受损部位的神经营养素3合成及分泌增加,改善受损脊髓微环境,促进神经元轴突再生及其髓鞘形成,修复感觉运动神经功能[28]。 2.3.2 间充质干细胞来源外泌体的应用 间充质干细胞来源外泌体的无细胞疗法作为近年来的研究热点,其中以外泌体的应用研究最受关注。间充质干细胞来源外泌体中含有脂质、蛋白质、mRNA和miRNA等多种物质,这些细胞因子通过旁分泌作用在组织修复中发挥免疫调节、促血管新生以及诱导再生等功能[29-31]。SUN等[32]在小鼠脊髓损伤后30 min将200 μg直径为70 nm的人脐带间充质干细胞来源外泌体经尾静脉注射至脊髓损伤小鼠体内,相较于对照组,移植组脊髓损伤小鼠炎症因子肿瘤坏死因子α、白细胞介素6及γ-干扰素的表达明显下降,移植组受损部位的巨噬细胞从M1型向M2型极化且BMS评分高于对照组,说明人脐带间充质干细胞来源外泌体在脊髓损伤中发挥抗炎的免疫调节作用,并促进运动功能恢复。LIU等[33]在大鼠脊髓损伤后立即尾静脉注200 μL(外泌体含量为200 μg)骨髓间充质干细胞来源外泌体,28 d后与对照组比较,MRI和组织病理切片显示移植组的病灶面积缩小近60%,神经细胞的凋亡和胶质瘢痕的形成显著降低,BBB评分有明显升高,小管形成实验显示新生血管的密度增加。LI等[34]在大鼠脊髓损伤后24 h将100 μg骨髓间充质干细胞来源外泌体通过尾静脉注射至大鼠体内,4 d后移植组脊髓受损部位神经元数量显著增加,BBB评分明显高于对照组,证明骨髓间充质干细胞来源外泌体移植能有效恢复脊髓损伤大鼠的神经功能。 2.3.3 间充质干细胞联合支架移植 组织工程生物支架的应用可改善移植细胞生存率低、移植细胞定植困难的问题,并起到维持组织结构稳定性的作用[35]。应用于脊髓损伤的生物材料种类较多,研究中最常用的3类支架材料为:①胶原蛋白支架:胶原蛋白来源于细胞外基质,是人体结构蛋白的重要组成部分,具有低抗原型、低免疫原性和可降解特性,良好的生物相容性使胶原蛋白支架广泛应用于干细胞移植的载体研究。将人脐带间充质干细胞种植于胶原蛋白支架并移植至脊髓损伤动物模型中,发现可改善皮质运动诱发电位和体感诱发电位,修复大鼠运动神经功能,促进神经细胞和髓鞘再生,并阻止病变区域外的星形胶质细胞生长[36]。②水凝胶:水凝胶有利于移植细胞的植入,半流体的特性可更好地填充受损脊髓,减少创伤后脊髓空洞的形成,并为受损组织提供持续的神经营养素。将载有人脐带间充质干细胞的水凝胶移植至脊髓受损部位,水凝胶为细胞黏附提供了更便利的条件,使细胞密度显著增高,并且植入的水凝胶可向损伤部位流体扩散,使营养因子在损伤部位释放,从而提高治疗效果[37]。③聚乳酸支架:聚乳酸支架具有良好的降解性,其结构有利于移植细胞的贴壁分化并可诱导神经纤维再生,对脊髓受损部位的修复起到支持和桥接作用。多孔聚乳酸-羟基乙酸共聚物/小肠黏膜下层支架支持骨髓间充质干细胞并允许其在体内生长,促进了营养物质的转运,骨髓间充质干细胞在聚乳酸-羟基乙酸共聚物/小肠黏膜下层支架中的存活时间相较于对照组有所提升,提示采用生物支架可延长移植细胞存活时间,此外根据组织学图像追踪到植入支架中的骨髓间充质干细胞可诱导神经形成[38]。间充质干细胞联合生物材料不仅可以促进脊髓损伤患者的运动功能恢复,而且比单独使用支架或间充质干细胞更有效。尽管有研究显示脊髓损伤急性期进行间充质干细胞联合支架治疗比亚急性期和慢性期更为有效,但考虑到脊髓损伤急性损伤期的免疫应激反应,移植至损伤部位的间充质干细胞存活率可能难以维持[39-40]。 2.4 间充质干细胞改善脊髓损伤的治疗机制 间充质干细胞治疗脊髓损伤的作用机制错综复杂,研究表明NF-κB信号通路在调节炎症、免疫应激等方面发挥着重要作用。脊髓受损后白细胞介素1β和肿瘤坏死因子α等促炎细胞因子激活NF-κB转导通路,使得间充质干细胞中白细胞介素1Ra表达水平增加,白细胞介素1Ra与白细胞介素1受体结合特异性抑制白细胞介素1的信号传导从而发挥抗炎作用[41];在肿瘤坏死因子α刺激后应用NF-κB抑制剂PDTC,间充质干细胞中白细胞介素1Ra的表达和分泌明显降低;间充质干细胞还分泌TSG-6调节NF-κB转导通路信号,从而减少促炎因子释放[42]。间充质干细胞通过调节Treg/Th17动态平衡发挥作用,间充质干细胞通过Toll样受体诱导部分淋巴细胞的Foxp3表达增加,促进Treg细胞增殖和转化,此外转化生长因子β信号通路激活亦可诱导T淋巴细胞向Treg细胞方向分化[43]。而间充质干细胞对Th17细胞的抑制作用则通过前列腺素E2信号途径介导,但应用前列腺素E2抑制剂对Th17细胞的抑制作用并未完全消除,提示间充质干细胞对Th17细胞的调控也存在其他途径,如STAT信号转导途径[44]。MMP2/STAT3信号转导通路参与胶质瘢痕形成的调节,改善受损部位的微环境。间充质干细胞移植后基质金属蛋白酶2增加,并通过STAT3途径调节星形胶质细胞,从而减少胶质瘢痕的形成[45], 脊髓受损使星形胶质细胞被激活分泌环氧合酶2、白细胞介素6,间充质干细胞移植后环氧合酶2和白细胞介素6水平降低,进而减少氧化应激、促进神经保护。白细胞介素10也可通过STAT3介导免疫调节反应,间充质干细胞移植后分泌的白细胞介素10与其受体结合形成复合物,激活细胞内的Janus激酶(JAKs),进而STAT3被活化的JAKs磷酸化形成二聚体进入细胞核,调节巨噬细胞表型,发挥抗炎作用[46-47]。通过Rho/ROCK/PTEN信号通路可发挥调控神经退行性变、诱导轴突再生的作用,使用ROCK选择性抑制剂联合间充质干细胞移植后,皮质脊髓束再生轴突的数量显著增加,脊髓异常空洞明显减少,激活神经生长因子、神经营养因子和脑源性神经营养因子的产生,促炎因子白细胞介素1β和肿瘤坏死因子α分泌减少[20]。除上述机制外,间充质干细胞还通过其他信号通路发挥作用治疗脊髓损伤,如MAPK、TGF-β1/p-SMAD2等通路[48],且有许多通路尚未被阐明,仍需进一步研究,见图2。 "

2.5 间充质干细胞治疗脊髓损伤的临床研究 2.5.1 骨髓间充质干细胞 骨髓间充质干细胞为最早发现的间充质干细胞,是现在研究最广泛和深入的成体干细胞类型。骨髓间充质干细胞具有较强的增殖能力、分化潜能,但其数量和增殖分化能力随着供者年龄的增加而明显减少,其应用也存在一定的局限性。在评估自体骨髓间充质干细胞安全性的Ⅰ期临床试验中,对脊髓损伤患者进行鞘内注射自体骨髓间充质干细胞后证明其安全无副作用(表1;NCT 02482194)。自体骨髓间充质干细胞的Ⅰ/Ⅱ期临床试验中,通过鞘内注射途径移植治疗脊髓损伤患者同时联合物理治疗,随访18个月ASIS评分显示有持续的神经功能改善,显著提高脊髓损伤患者的生活质量(表1;NCT 00816803)。在KAPANADZE等[49]的研究中,18例脊髓损伤患者接受至少7.5×109个自体骨髓间充质干细胞鞘内注射移植,其中9例患者运动和感觉功能改善,按照美国脊髓损伤协会(ASIA)的评分标准,其中7例为一级改善,2例为二级改善,无严重并发症。MENDONCA等[50]按照5×106个/cm3将自体骨髓间充质干细胞注入14例慢性完全性脊髓损伤患者病变部位(通过MRI分析来估计病变体积),其中7例患者从完全性损伤转变为不完全性损伤,且运动和感觉、临床疼痛和尿动力学方面都有不同程度的改善,AISA评分从A级提高到B级或C级。DAI等[51]选择40例慢性完全性颈髓损伤患者进行研究(治疗组与对照组各20例),将25 μL细胞悬液(8×1011 L-1)分次缓慢注射到治疗组患者损伤脊髓与正常脊髓交界处,随访6个月治疗组中10例患者在运动、轻触、针刺感觉、残余尿量等方面均有明显改善,9例患者AISA评分由A级改善为B级,神经生理学检查与临床观察一致,而对照组上述神经功能均无改善,证实骨髓间充质干细胞移植能有效恢复慢性完全性颈椎脊髓损伤患者的神经功能障碍。 2.5.2 脐带间充质干细胞 脐带间充质干细胞由于发育更为原始,表现出更强的细胞增殖能力和细胞因子分泌能力,且免疫原性更低,近年来备受研究者关注。一项脐带间充质干细胞的Ⅱ期临床试验,以ASIA评分、肌电图、国际神经修复协会脊髓损伤功能评定量表(IANR-SCIRFS) 评估脐带间充质干细胞鞘内移植的安全性和有效性,预计研究将于2022年底完成(表1;NCT03521336)。另一项关于脐带间充质干细胞鞘内注射的Ⅲ期临床试验采取AISA评分、日常生活能力评分、体感诱发电位和运动诱发电位变化等量化脐带间充质干细胞移植的有效性,目前试验结束但成果尚未发布(表1;NCT01873547)。刘静等[52]对35例脊髓损伤患者(25例不完全性损伤,10例完全损伤)进行脐带间充质干细胞移植,将10 mL的脐带间充质干细胞 (1×106个/kg)注入蛛网膜下腔,25例不完全性脊髓损伤患者中22例感觉和运动能力评分明显提高,10例完全性脊髓损伤患者治疗无改善,证明鞘内注射人脐带间充质干细胞的安全性,并可改善部分不完全性脊髓损伤患者的神经功能。YAO等[53]对25例脊髓损伤时间超过6个月的患者进行了人脐带间充质干细胞静脉注射和鞘内注射移植,剂量为4 mL人脐带间充质干细胞[共含(1-3)×107个],术后随访12个月,4例患者ASIA评分提高,7例患者痉挛减少,8例患者自主神经功能改善,9例患者体感诱发电位潜伏期缩短,证实人脐带间充质干细胞治疗慢性创伤性脊髓损伤患者的安全性和有效性。张陆等[54]对50例急性脊髓损伤患者进行人脐带间充质干细胞鞘内注射,伤后72 h内进行首次注射,每次细胞注射量为(3.0-4.0)×107个,4-6次为1个疗程,术后随访显示移植组的ASIA运动评分与感觉评分均明显提升且高于对照组,提示人脐带间充质干细胞可促进急性脊髓损伤患者神经功能恢复。 2.5.3 脂肪间充质干细胞 脂肪间充质干细胞具有来源广泛、含量丰富和易于采集等优势,成为极具应用前景的种子细胞新来源。尽管脂肪间充质干细胞移植已在早期的动物实验中表现出了较好的治疗效果,但脂肪间充质干细胞的临床试验仍然很少。FAB 117-HC是由同种异体脂肪间充质干细胞扩增而来,通过脊髓内注射移植治疗脊髓损伤的Ⅰ/Ⅱ期临床试验正在进行中,以体感诱发电位、运动诱发电位等评估FAB 117-HC的安全性和有效性,预计2022年3月完成。RA等[55] 对8例脊髓损伤患者进行剂量为4×108个自体脂肪间充质干细胞的静脉移植治疗,患者运动能力评分、感觉评分以及自我护理、括约肌管理评分均有不同程度的提高,证实自体脂肪间充质干细胞静脉移植的安全性和初步的有效性。HUR 等[56]分3次(每次间隔1个月)对14例脊髓损伤患者鞘内注射9×107个自体脂肪间充质干细胞,在8个月的随访中5例患者的ASIA运动评分得到改善,2例患者自主性肛门收缩功能改善。1例患者自随访期以来感觉功能一直表现出持续的恢复。此外有1例患者移植后双侧正中神经短潜伏期体感诱发电位趋于正常,提示正中神经功能改善。 间充质干细胞的临床应用潜力逐渐凸显,截至2020-02-22,全球已有31个间充质干细胞治疗脊髓损伤项目在美国NIH临床研究数据库注册(表1,数据来自https://clinicaltrials.gov)。 2.5.4 间充质干细胞联合生物材料 AMR等[57]将骨髓间充质干细胞注入腓肠神经与壳聚糖-层粘连蛋白支架复合,随后移植至14例脊髓损伤所致的慢性截瘫患者的受损脊髓处,发现患者的感觉和运动功能均有不同程度的改善,其中12例患者的AISA评分由A级提升为C级,2例由A级提升为B级,体现了间充质干细胞联合生物材料极佳的应用前景。ZHAO等[58]将载有 4×107个人脐带间充质干细胞的神经再生胶原支架植入8例慢性完全性脊髓损伤患者损伤部位,随访1年观察到感觉水平和运动诱发电位反应区扩大、手指活动增加和自主神经功能改善,表明了人脐带间充质干细胞联合胶原支架治疗具有良好的安全性。 "

| [1] 陈星月,陈栋,陈春慧,等.中国创伤性脊髓损伤流行病学和疾病经济负担的系统评价[J].中国循证医学杂志,2018,18(2):143-150. [2] JENDELOVA P. Therapeutic Strategies for Spinal Cord Injury. Int J Mol Sci. 2018;19(10):3200. [3] HAN Y, LI X, ZHANG Y, et al. Mesenchymal Stem Cells for Regenerative Medicine. Cells. 2019;8(8):886. [4] MUKAI T, TOJO A, NAGAMURA-INOUE T. Mesenchymal stromal cells as a potential therapeutic for neurological disorders. Regen Ther. 2018;9:32-37. [5] FUNES SC, RIOS M, ESCOBAR-VERA J, et al. Implications of macrophage polarization in autoimmunity. Immunology. 2018;154(2):186-195. [6] FAN B, WEI Z, YAO X, et al. Microenvironment Imbalance of Spinal Cord Injury. Cell Transplant. 2018;27(6):853-866. [7] ANWAR MA, AL SHEHABI TS, EID AH. Inflammogenesis of Secondary Spinal Cord Injury. Front Cell Neurosci. 2016;10:98. [8] TRAN AP, WARREN PM, SILVER J. The Biology of Regeneration Failure and Success After Spinal Cord Injury. Physiol Rev. 2018;98(2):881-917. [9] ORR MB, GENSEL JC. Spinal Cord Injury Scarring and Inflammation: Therapies Targeting Glial and Inflammatory Responses. Neurotherapeutics. 2018;15(3):541-553. [10] FLEMING JC, NORENBERG MD, RAMSAY DA, et al. The cellular inflammatory response in human spinal cords after injury. Brain. 2006;129(Pt 12): 3249-3269. [11] KUMAR H, CHOI H, JO MJ, et al. Neutrophil elastase inhibition effectively rescued angiopoietin-1 decrease and inhibits glial scar after spinal cord injury. Acta Neuropathol Commun. 2018;6(1):73. [12] COFANO F, BOIDO M, MONTICELLI M, et al. Mesenchymal Stem Cells for Spinal Cord Injury: Current Options, Limitations, and Future of Cell Therapy. Int J Mol Sci. 2019;20(11):2698. [13] KOZLOWSKA U, KRAWCZENKO A, FUTOMA K, et al. Similarities and differences between mesenchymal stem/progenitor cells derived from various human tissues. World J Stem Cells. 2019;11(6):347-374. [14] NTEGE EH, SUNAMI H, SHIMIZU Y. Advances in regenerative therapy: A review of the literature and future directions. Regen Ther. 2020;14:136-153. [15] FU Q, ZHANG Q, JIA LY, et al. Isolation and Characterization of Rat Mesenchymal Stem Cells Derived from Granulocyte Colony-Stimulating Factor-Mobilized Peripheral Blood. Cells Tissues Organs. 2015 -2016;201(6): 412-422. [16] RAJABZADEH N, FATHI E, FARAHZADI R. Stem cell-based regenerative medicine. Stem Cell Investig. 2019;6:19. [17] LUZ-CRAWFORD P, DJOUAD F, TOUPET K, et al. Mesenchymal Stem Cell-Derived Interleukin 1 Receptor Antagonist Promotes Macrophage Polarization and Inhibits B Cell Differentiation. Stem Cells. 2016;34(2): 483-492. [18] QU J, ZHANG H. Roles of Mesenchymal Stem Cells in Spinal Cord Injury. Stem Cells Int. 2017;2017:5251313. [19] 王国毓,程志坚,杨保辉,等.脊髓损伤后硫酸软骨素蛋白多糖及GFAP表达的变化[J].生物骨科材料与临床研究,2020,17(1): 43-46. [20] MUKHAMEDSHINA YO, GRACHEVA OA, MUKHUTDINOVA DM, et al. Mesenchymal stem cells and the neuronal microenvironment in the area of spinal cord injury. Neural Regen Res. 2019;14(2):227-237. [21] ZENG X, MA YH, CHEN YF, et al. Autocrine fibronectin from differentiating mesenchymal stem cells induces the neurite elongation in vitro and promotes nerve fiber regeneration in transected spinal cord injury. J Biomed Mater Res A. 2016;104(8):1902-1911. [22] FU Q, LIU Y, LIU X, et al. Engrafted peripheral blood-derived mesenchymal stem cells promote locomotive recovery in adult rats after spinal cord injury. Am J Transl Res. 2017;9(9):3950-3966. [23] MALDONADO-LASUNCIÓN I, VERHAAGEN J, OUDEGA M. Mesenchymal Stem Cell-Macrophage Choreography Supporting Spinal Cord Repair. Neurotherapeutics. 2018;15(3):578-587. [24] 陈龙.外周血间充质干细胞诱导巨噬细胞极化与重编程的体外研究[D]. 遵义:遵义医学院.2016. [25] 高弘斐,张潜,陈龙,等.间充质干细胞与巨噬细胞共培养体系的细胞因子表达模式研究[J].免疫学杂志,2017,33(11): 930-936. [26] ZHENG YH, DENG YY, LAI W, et al. Effect of bone marrow mesenchymal stem cells on the polarization of macrophages. Mol Med Rep. 2018;17(3): 4449-4459. [27] BADNER A, VAWDA R, LALIBERTE A, et al. Early Intravenous Delivery of Human Brain Stromal Cells Modulates Systemic Inflammation and Leads to Vasoprotection in Traumatic Spinal Cord Injury. Stem Cells Transl Med. 2016;5(8):991-1003. [28] 曾园山.电针督脉经穴联合成体干细胞移植策略在修复脊髓损伤中的研究进展[J].中山大学学报(医学科学版),2017,38(6):801-807. [29] ZHANG S, CHUAH SJ, LAI RC, et al. MSC exosomes mediate cartilage repair by enhancing proliferation, attenuating apoptosis and modulating immune reactivity. Biomaterials. 2018;156:16-27. [30] JOO HS, SUH JH, LEE HJ, et al. Current Knowledge and Future Perspectives on Mesenchymal Stem Cell-Derived Exosomes as a New Therapeutic Agent. Int J Mol Sci. 2020;21(3):727. [31] ELAHI FM, FARWELL DG, NOLTA JA, et al. Preclinical translation of exosomes derived from mesenchymal stem/stromal cells. Stem Cells. 2020;38(1): 15-21. [32] SUN G, LI G, LI D, et al. hucMSC derived exosomes promote functional recovery in spinal cord injury mice via attenuating inflammation. Mater Sci Eng C Mater Biol Appl. 2018;89:194-204. [33] LIU W, WANG Y, GONG F, et al. Exosomes Derived from Bone Mesenchymal Stem Cells Repair Traumatic Spinal Cord Injury by Suppressing the Activation of A1 Neurotoxic Reactive Astrocytes. J Neurotrauma. 2019;36(3):469-484. [34] LI D, ZHANG P, YAO X, et al. Exosomes Derived From miR-133b-Modified Mesenchymal Stem Cells Promote Recovery After Spinal Cord Injury. Front Neurosci. 2018;12:845. [35] 陈刚,吴骏,于天飞.干细胞结合生物材料治疗脊髓损伤的研究进展[J].生物学教学,2016,41(12): 11-13. [36] WANG N, XIAO Z, ZHAO Y, et al. Collagen scaffold combined with human umbilical cord-derived mesenchymal stem cells promote functional recovery after scar resection in rats with chronic spinal cord injury. J Tissue Eng Regen Med. 2018;12(2):e1154-e1163. [37] CARON I, ROSSI F, PAPA S, et al. A new three dimensional biomimetic hydrogel to deliver factors secreted by human mesenchymal stem cells in spinal cord injury. Biomaterials. 2016;75:135-147. [38] KANG KN, LEE JY, KIM DY, et al. Regeneration of completely transected spinal cord using scaffold of poly(D,L-lactide-co-glycolide)/small intestinal submucosa seeded with rat bone marrow stem cells. Tissue Eng Part A. 2011;17(17-18):2143-2152. [39] YOUSEFIFARD M, NASSERI MALEKI S, ASKARIAN-AMIRI S, et al. A combination of mesenchymal stem cells and scaffolds promotes motor functional recovery in spinal cord injury: a systematic review and meta-analysis. J Neurosurg Spine. 2019;32(2):269-284. [40] 孙佳慧,林欣怡,邹剑龙,等.神经组织工程技术治疗脊髓损伤的研究进展[J].解剖学研究,2016,38(6):480-483. [41] CHEN L, ZHANG Q, CHEN QH, et al. Combination of G-CSF and AMD3100 Improves the Anti-inflammatory Effect of Mesenchymal Stem Cells on Inducing M2 Polarization of Macrophages Through NF-κB-IL1RA Signaling Pathway. Front Pharmacol. 2019;10:579. [42] CHAUBEY S, THUESON S, PONNALAGU D, et al. Early gestational mesenchymal stem cell secretome attenuates experimental bronchopulmonary dysplasia in part via exosome-associated factor TSG-6. Stem Cell Res Ther. 2018;9(1):173. [43] WANG K, SHI YJ, SONG ZL, et al. Regulatory effect of rat bone marrow mesenchymal stem cells on Treg/Th17 immune balance in vitro. Mol Med Rep. 2020;21(5):2123-2130. [44] WANG D, HUANG S, YUAN X, et al. The regulation of the Treg/Th17 balance by mesenchymal stem cells in human systemic lupus erythematosus. Cell Mol Immunol. 2017;14(5):423-431. [45] KIM C, KIM HJ, LEE H, et al. Mesenchymal Stem Cell Transplantation Promotes Functional Recovery through MMP2/STAT3 Related Astrogliosis after Spinal Cord Injury. Int J Stem Cells. 2019;12(2):331-339. [46] IP WKE, HOSHI N, SHOUVAL DS, et al. Anti-inflammatory effect of IL-10 mediated by metabolic reprogramming of macrophages. Science. 2017; 356(6337):513-519. [47] OUYANG W, RUTZ S, CRELLIN NK, et al. Regulation and functions of the IL-10 family of cytokines in inflammation and disease. Annu Rev Immunol. 2011;29:71-109. [48] 刘克勋,霍洪军,赵岩,等.间充质干细胞移植治疗脊髓损伤的进展及发展趋势[J].中国组织工程研究,2018,22(21):3410-3416. [49] KAKABADZE Z, KIPSHIDZE N, MARDALEISHVILI K, et al. Phase 1 Trial of Autologous Bone Marrow Stem Cell Transplantation in Patients with Spinal Cord Injury. Stem Cells Int. 2016;2016:6768274. [50] MENDONÇA MV, LAROCCA TF, DE FREITAS SOUZA BS, et al. Safety and neurological assessments after autologous transplantation of bone marrow mesenchymal stem cells in subjects with chronic spinal cord injury. Stem Cell Res Ther. 2014;5(6):126. [51] DAI G, LIU X, ZHANG Z, et al. Transplantation of autologous bone marrow mesenchymal stem cells in the treatment of complete and chronic cervical spinal cord injury. Brain Res. 2013;1533:73-79. [52] 刘静,韩冬梅,薛梅,等.脐带间充质干细胞鞘内注射治疗脊髓损伤的长期临床观察[J].组织工程与重建外科,2017,13(6): 313-317. [53] YAO L, HE C, ZHAO Y, et al. Human umbilical cord blood stem cell transplantation for the treatment of chronic spinal cord injury: Electrophysiological changes and long-term efficacy. Neural Regen Res. 2013;8(5):397-403. [54] 张陆,刘志昂,姜岩,等.脐带间充质干细胞移植治疗急性脊髓损伤的临床价值[J].中国脊柱脊髓杂志,2019,29(3):254-260. [55] RA JC, SHIN IS, KIM SH, et al. Safety of intravenous infusion of human adipose tissue-derived mesenchymal stem cells in animals and humans. Stem Cells Dev. 2011;20(8):1297-1308. [56] HUR JW, CHO TH, PARK DH, et al. Intrathecal transplantation of autologous adipose-derived mesenchymal stem cells for treating spinal cord injury: A human trial. J Spinal Cord Med. 2016;39(6):655-664. [57] AMR SM, GOUDA A, KOPTAN WT, et al. Bridging defects in chronic spinal cord injury using peripheral nerve grafts combined with a chitosan-laminin scaffold and enhancing regeneration through them by co-transplantation with bone-marrow-derived mesenchymal stem cells: case series of 14 patients. J Spinal Cord Med. 2014;37(1):54-71. [58] ZHAO Y, TANG F, XIAO Z, et al. Clinical Study of NeuroRegen Scaffold Combined With Human Mesenchymal Stem Cells for the Repair of Chronic Complete Spinal Cord Injury. Cell Transplant. 2017;26(5):891-900. [59] 唐玺和,范益民,蓝胜勇.细胞与生物材料联合治疗脊髓损伤的研究现状[J].中国比较医学杂志,2018, 28(6):127-130. [60] 秦丰伟,焦锋,张兆飞,等.淫羊藿苷联合骨髓间充质干细胞移植对脊髓损伤区域炎症反应的影响[J].广东医学,2020,41(1): 5-8. [61] LIU W, RONG Y, WANG J, et al. Exosome-shuttled miR-216a-5p from hypoxic preconditioned mesenchymal stem cells repair traumatic spinal cord injury by shifting microglial M1/M2 polarization. J Neuroinflammation.2020;17(1): 47. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Min Youjiang, Yao Haihua, Sun Jie, Zhou Xuan, Yu Hang, Sun Qianpu, Hong Ensi. Effect of “three-tong acupuncture” on brain function of patients with spinal cord injury based on magnetic resonance technology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-8. |
| [3] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [4] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [5] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Multiple problems in the selection of implants for patellar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1440-1445. |
| [6] | Wang Debin, Bi Zhenggang. Related problems in anatomy mechanics, injury characteristics, fixed repair and three-dimensional technology application for olecranon fracture-dislocations [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1446-1451. |
| [7] | Ji Zhixiang, Lan Changgong. Polymorphism of urate transporter in gout and its correlation with gout treatment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1290-1298. |
| [8] | Yuan Mei, Zhang Xinxin, Guo Yisha, Bi Xia. Diagnostic potential of circulating microRNA in vascular cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1299-1304. |
| [9] | Jiang Hongying, Zhu Liang, Yu Xi, Huang Jing, Xiang Xiaona, Lan Zhengyan, He Hongchen. Effect of platelet-rich plasma on pressure ulcers after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1149-1153. |
| [10] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [11] | Wan Ran, Shi Xu, Liu Jingsong, Wang Yansong. Research progress in the treatment of spinal cord injury with mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1088-1095. |
| [12] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [13] | Zhao Min, Feng Liuxiang, Chen Yao, Gu Xia, Wang Pingyi, Li Yimei, Li Wenhua. Exosomes as a disease marker under hypoxic conditions [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1104-1108. |
| [14] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [15] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||