Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (8): 1284-1289.doi: 10.3969/j.issn.2095-4344.3035
Previous Articles Next Articles
Relationship between hypoxia inducible factor 1 alpha and hypoxia signaling pathway
Gu Xia, Zhao Min, Wang Pingyi, Li Yimei, Li Wenhua
- School of Medicine, Xizang Minzu University, Xianyang 712082, Shanxi Province, China
-
Received:2020-03-16Revised:2020-03-20Accepted:2020-04-21Online:2021-03-18Published:2020-12-14 -
Contact:Li Wenhua, Professor, School of Medicine, Xizang Minzu University, Xianyang 712082, Shaanxi Province, China -
About author:Gu Xia, Master candidate, School of Medicine, Xizang Minzu University, Xianyang 712082, Shaanxi Province, China -
Supported by:the National Natural Science Foundation of China, No. 81760332 (to LWH); Scientific Research Project of Shaanxi Provincial Department of Education
CLC Number:
Cite this article
Gu Xia, Zhao Min, Wang Pingyi, Li Yimei, Li Wenhua. Relationship between hypoxia inducible factor 1 alpha and hypoxia signaling pathway[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1284-1289.
share this article
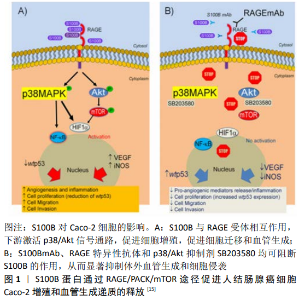
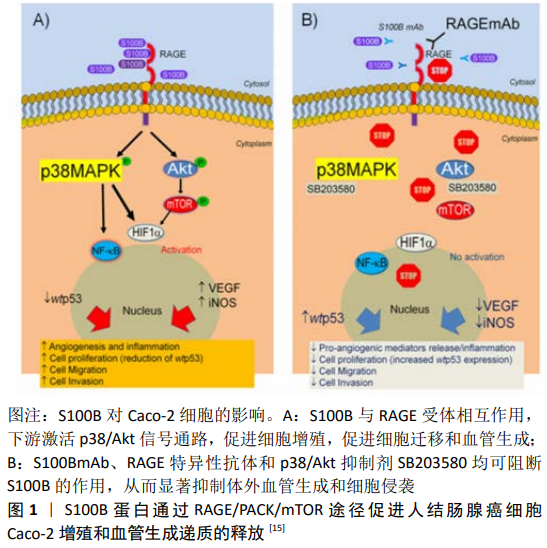
2.1 HIF-1α与经典PHDs/HIF-1α/pVHL通路 HIF-1α几乎在所有哺乳动物细胞内都表达,但却基本不能在常氧细胞内观察到,这是因为在氧气正常存在的情况下,脯氨酸羟化酶(PHDs)将HIF-1α的第564位和第402位脯氨酸羟基化,同时ODDD域与肿瘤抑制蛋白(pVHL)结合,并与募集的多种泛素蛋白结合共同组成泛素连接蛋白酶复合体,作用于HIF-1α亚基并使之降解。常氧时在细胞内还存在另外一种阻碍HIF-1α作用的因子,即HIF-1抑制因子(FIH-1)。FIH-1使HIF-1α C端反式激活结构域第803位天冬氨酸残基羟基化,从而阻止了HIF-1α与CBP/P300的结合[3],使下游靶基因不能表达。当细胞低氧时,HIF-1α进入核内与稳定存在于核内的HIF-1β结合,构成异源二聚体形式的HIF-1,并结合到基因的低氧反应原件HRE上诱导靶基因表达,影响下游因子,这一过程依赖于转录激活辅助因子(CBP/P300)。HIF的激活影响下游一系列因子活动,包括促红细胞生成素、血管内皮生长因子、活性氧等因子,调节无氧糖酵解和氧化磷酸化过程[11],在细胞的各种生命活动中发挥重要作用。 2.2 HIF-1α与其他低氧相关信号通路 2.2.1 HIF-1α与PI3K/Akt/mTOR通路 膜磷脂肌醇被磷脂酰肌醇-3激酶(phosphoinositide 3-kinase,PI3K)磷酸化而激活,生成3,4二磷酸磷脂酰肌醇(phosphatidylinositol-3, 4-biphosphate,PI-3,4P2)及3,4,5三磷酸磷脂酰肌醇(phosphatidylinositol 3,4,5-trisphosphate,PI-3,4,5P3),与Akt结合后定位到质膜上,使后者充分磷酸化后完全激活,启动下游底物哺乳动物雷帕霉素靶向蛋白( mammalian target of rapamycin,mTOR)的翻译过程。作为细胞中经典的再灌注损伤救援激酶信号之一,PI3K/Akt/mTOR通路在细胞内广泛存在,参与细胞增殖分化、代谢、炎症和保护过程[12],并与促进细胞凋亡、改变组织重塑和转移等相关的信号有关[13]。 已有研究表明,PI3K/Akt途径主要通过增强HIF-1α mRNA的蛋白质翻译过程增加HIF-1α蛋白质水平,这种方式被证明是依赖或独立于下游mTOR[14]。SEGUELLA等[15]在S100B蛋白以及S100B蛋白阻断剂存在的情况下研究Akt、mTOR、HIF-1α等分子蛋白在人结肠癌Caco-2细胞中的表达,证明S100B可以激活Akt/mTOR/HIF-1α信号通路引起血管生成炎症级联,上调血管内皮生长因子及其受体,刺激细胞增殖和血管生成递质的释放,见图1。TONG等[16]在多发性骨髓瘤细胞中研究发现长非编码RNA(LncRNAs) DARS-AS1的表达水平与HIF-1α的表达水平呈正相关,且HIF-1α蛋白表达水平反过来受DARS-AS1的调控。随后他们在DARS-AS1的启动子区域发现了2种可能的HRE区域,并用芯片验证了HIF-1与预测的这2个区域结合,同时他们发现 DARS-AS1在低氧环境下通过RNA结合蛋白(RBM39)调节mTOR信号传导,以此推测DARS-AS1通过抑制mTOR途径来抑制HIF-1α的翻译。LEE等[17]在研究O-环磷酰肌酐-1-磷酸(cP1P)对骨髓间充质干细胞治疗潜力的影响及其调控机制的过程中,发现由cP1P触发的mTOR信号通路通过S6K1途径调节HIF-1α的翻译,同时cP1P触发的mTOR途径诱导双耳D同源蛋白1表达,导致HIF-1α核转位。他们认为cP1P通过mTOR依赖性HIF-1α的翻译和核转位增强了骨髓间充质干细胞的治疗潜力。SHI等[18]以阻塞性黄疸大鼠为研究对象,观察右美托咪啶对其肺损伤的影响机制,发现右美托咪啶通过PI3K/Akt/HIF-1α信号通路减轻阻塞性黄疸大鼠肺损伤。PEI-YUAN等[19]将受体酪氨酸激酶AXL过表达慢病毒转染EAhy926细胞用R428处理,通过分析各种信号分子的激活情况,确定了AXL在低氧高糖条件下通过PI3K/Akt/mTOR/p70S6K依赖性方式调节HIF-1α表达。以上各种研究结果表明,HIF-1α在低氧环境下确实受PI3K/Akt信号通路调控,从而导致下游分子表达水平升高或降低,而这种调控是否依赖于mTOR还有待证实。 2.2.2 HIF-1α与MAPK通路 目前已发现的MAPK通路家族除了细胞外信号调控的蛋白激酶(ERK),还有P38 MAPK、c-Jun N端激酶(JNK)和ERK5/BMK1。Ras/Raf/MEK/ERK通路是MAPK信号通路的主要途径,也是目前研究相对较清楚的通路。Ras将Raf募集到质膜上,并将其激活,Raf随后激活MEK,MEK又激活ERK。ERK的激活使下游其他蛋白激酶和核中部分基因调节蛋白磷酸化,使基因表达和蛋白质活性发生改变。细胞的增殖分化和生长发育等病理生理过程离不开Ras/Raf/MEK/ERK通路,JNK通路作为细胞的重要调节靶点在细胞凋亡过程中发挥不容忽视的作用,P38 MAPK是细胞转录因子合成、骨架识别和转录调节过程中的重要因子,而ERK5/BMK1则对血管生成和肿瘤增殖分化过程有决定性影响[20]。 万军等[21]研究人员证明Akt1、ERK1/2和HIF-1α均在肺癌组织中高表达,且3者具有明显相关性,都与肺癌分化程度、淋巴结转移及TNM分期有关,并推断HIF-1α可能是ERK1/2的主要下游调节成员之一。已有研究表明ERK1/2通过介导HIF-1α磷酸化来调节HIF-1α转录活性,其过程涉及2个丝氨酸残基的修饰和核输出信号(NES)的掩盖,都在被称为ERK靶向域(ETD)内完成。ETD变体的过度表达,包括野生型、磷酸化(SE)、磷酸缺乏(SA)形式或无NES(IA)突变体形式。KARAGIOTA等[22]将ETD与HIV-TAT序列融合成TAT-ETD肽添加到Huh7细胞的培养基中,发现除ETD-SA以外,其他型ETD都聚集在细胞核内,导致内源性HIF-1α在向细胞质定位的过程中出现错误,从而显著抑制了HIF-1的活动和靶点表达。WU等[23]通过HIF-1α调节TET1和TET3的表达,协同作用TNFα-p38-MAPK通路,导致乳腺肿瘤起始细胞特性,在乳腺癌的发展进程中发挥重要作用。KOLIBABKA等[24]建立了氧诱导的视网膜病变动物模型,并给予皮下注射雷纳格力丁,发现血管内皮生长因子和HIF-1α的表达增加,并且雷纳格力丁通过抑制血管内皮生长因子诱导的MAPK/ERK途径发挥抗血管生成的作用,保留对糖尿病视网膜病变患者潜在的益处。TALWAR等[25]通过在RAW264.7细胞中过表达MEK2蛋白进行功能获得实验,并研究了MEK1和MEK2双次和三次突变对ERK磷酸化、HIF-1α表达和IL-1β产生的作用,发现了MEK2独立于ERK激活反比例调节HIF-1α和IL-1β的表达,且MEK2通过调节HIF-1α的表达产生IL-1β来响应TLR4的激活。GONG等[26]证明香草酸在人结肠癌HCT116细胞中通过抑制mTOR/p70S6K/4E-bp1和raf/mek/erk途径降低HIF-1α蛋白合成,并阻断细胞周期进程于G1期,抑制人结肠癌HCT116细胞的增殖,从而起到抑制肿瘤血管生成的作用。在某些研究中,HIF-1α与MAPK表现为协同作用,同时受ERK激活;而在另一些研究中,HIF-1α的表达却受到ERK信号通路的抑制,且MEK2确实被证实独立于ERK反比例调节HIF-1α的表达。MAPK对HIF-1α的调节作用到底是促进还是抑制,还需要大量实验来证实,但毋庸置疑的是,HIF-1α确实存在受MAPK信号通路调节的情况。 2.2.3 HIF-1α与JAK/STAT通路 JAK家族由JAK1、JAK2、JAK3和Tyk2亚型组成,而STAT家族则由STAT1、STAT2、STAT3、STAT4、STAT5a、STAT5b及STAT6亚型组成。STAT蛋白被JAK激酶募集到受体磷酸位点上并激活后,与受体分离形成STAT-STAT二聚体进入核内,调控靶基因表达。JAK/STAT通路通过介导细胞增殖分化、免疫调节、凋亡等过程的精细调控维持着内环境的稳态。当JAK/STAT通路在病理过程中失控,反而会促进一系列疾病的发生和发展,包括各种炎性疾病、实体肿瘤和白血病[27]。 蒲霞等[28]通过研究推测低氧可能通过JAK/STAT1信号通路诱导肾小管上皮细胞发生表型化,从而促进肾间质纤维化的发展。JACOBSSON等[29]研究发现JAK/STAT相关因子无论在乳腺癌雌激素受体阳性低氧细胞还是雌激素受体阴性低氧细胞中都显著过量表达,而IL-6/JAK2/STAT3轴则在雌激素受体阴性低氧细胞行为中特别重要。已有研究表明低氧条件下STAT1可被HIF-1激活[30]。ZHANG等[31]模拟低氧条件(体积分数为1%O2),并使用慢病毒在U251细胞和U373细胞中过表达STAT1,进行一系列功能实验,阐明了STAT1过表达通过抑制HIF-1α活性部分影响脑胶质瘤细胞中血管内皮生长因子A的表达,从而抑制血管生成。COULIBABY等[32]将数学建模与实验室分析相结合,并模拟缺氧和炎症的条件,结果显示在白细胞介素15刺激的NK细胞中,STAT3是HIF-1α富集的重要调节因子,以此推测在癌症治疗中STAT3的抑制效应可能在于减少NK细胞中依赖白细胞介素15富集的HIF-1α。林建国等[33]对JAK2/STAT3/HIF-1α信号通路与脑卒中的关系进行了详细的阐述。他们通过查阅总结资料,发现JAK2/STAT3/HIF-1α信号通路的激活或抑制对脑缺血再灌注的影响是复杂的,并不仅仅是单一的保护或损害作用,并提出将脑卒中的急性期和康复期分阶段研究,分别给予JAK2/STAT3/HIF-1α通路抑制和激活干预,从而更好地应对脑缺血再灌注疾病的预后和康复。另外一方面,PARK等[34]证明了人胶质瘤细胞中STAT6的下调会反过来激活mTOR信号通路,从而促进缺氧条件下HIF-1α蛋白的合成;而恢复STAT6将抑制HIF-1α蛋白的合成,并诱导STAT6调节的免疫反应和凋亡。以上结果表明,STAT家族不同成员对HIF-1α的作用并不一致,且同一成员对HIF-1α的作用在时间上也表现出一定的差异性。目前关于STAT与HIF-1α的关系研究并不十分全面,因此若想深入了解两者关系,必须加大这方面的实验力度。 2.2.4 HIF-1α与TGF-β/Smad通路 转化生长因子β(transforming growth factor β,TGF-β)至少包含TGF-β1、TGF-β2、TGF-β3、TGF-β4、TGF-β5这5种亚型。TGF-β作为细胞活性多功能的调节因子,广泛参与胚胎发育,影响细胞增殖和分化,且通过诱导肿瘤上皮间质化调节肿瘤的发生发展和迁移过程。TGF可使一些上皮细胞停止在G1期,表现为增殖抑制,从而对各种肿瘤细胞和上皮细胞等细胞的增殖活动进行负性调控。Smad是TGF-β受体途径的下游转录因子,有受体调节Smad(receptor-regulated Smad,R-Smad)、辅Smad(co-Smad)和抑制性Smad(inhibitory Smad,I-smad)3种类型。激活的TGF-β受体使下游的R-Smad(Smad3或Smad4)磷酸化。磷酸化的R-Smad与未磷酸化的co-Smad和核输入蛋白β(Imp-β)形成一个大的复合物,进入核内发挥作用。 有研究表明肿瘤源性TGF-β信号转导与癌症恶病质患者的纤维化有关[35]。徐荣伟等[36]通过检测TGF-β1、HIF-1α和VEGF在100例大肠癌及癌旁组织中的表达,发现3者在大肠癌组织中的表达明显高于癌旁组织,且3者呈正相关。LEI等[37]研究发现HIF-1α激活TGF-β/Smad信号通路,诱导和刺激血管内皮生长因子的表达,从而导致异常的血管增生,促进瘢痕疙瘩的形成。LIU等[38]表示TGF-β可以在非低氧条件下诱导牙周膜干细胞中的HIF-1α稳定,HIF-1α反过来又可以负调节牙周膜干细胞中的TGF-β/Smad3信号通路。此外,他们还发现HIF-1α是通过降低TGF-β mRNA和蛋白的表达以及减少Smad3磷酸化来干扰TGF-β/Smad的信号转导。在另一项研究中,YOSHIMOTO等[39]发现缺氧诱导的HIF-1α在牙成釉细胞瘤中的上皮间质转化过程中起关键作用,而上皮间质转化这一过程依赖于TGF-β信号分子。ROSELL-GARCíA等[40]研究人员通过缺失作图和诱变分析确定了人PLOD2基因启动子上HIF和Smad蛋白的特异性结合位点,还揭示了HIF信号在Smad信号途径中起主要作用,完整的HIF位点对于TGF-β1发挥作用是必需的。这些研究证明TGF-β信号通路在介导细胞增殖和分化的过程中受HIF-1α调控,而低氧环境下TGF-β与HIF-1α是否相互影响以及如何影响,这都需要更多实验和研究去证实。 2.2.5 HIF-1α与NF-κB通路 NF-κB家族的成员包括 NF-κB1(p50)、NF-κB2(p52)、RelA(p65)、RelB和cRel这5种。NF-κB常以p65/p50这样的二聚体形式稳定存在于细胞内。静止状态下NF-κB与抑制蛋白I-κB绑定在一起,抑制NF-κB活性。当I-κB激酶存在时并作用于I-κB使之磷酸化后降解,NF-κB的抑制作用被解除,即可进入核内进行转位。NF-κB家族的各个成员分别在淋巴器官、淋巴结结构中发挥作用,调节细胞免疫功能[41]。当NF-κB信号通路失控,机体的平衡遭到破坏,就会引起各种炎症和肿瘤疾病[42]。"

完整的NF-κB途径是低氧时炎症基因表达所必需的,而HIF-1α和NF-κB信号之间高度依赖[43]。已有报道指出,活化的NF-κB在时间上调节HIF-1α的表达,p65、p50可以直接与HIF-1α启动子结合,促进HIF-1α的转录[44]。符智慧课题组[45]研究表明脂多糖在低氧条件下可通过NF-κB活化诱导RAW264.7细胞的HIF-1α表达,且应用NF-κB抑制剂会导致HIF-1α表达下降。GUO等[46]通过对8种非小细胞癌细胞的研究发现NF-κB和HIF-1α在EGFR突变体调节程序性细胞死亡配体(PD-L1)表达中有协同作用,且HIF-1α的蛋白表达与I-κB的磷酸化水平高度相关。JIN等[47]首次表明缺氧可以诱导内皮细胞自分泌肿瘤坏死因子α,进而通过NF-κB依赖过程激活HIF通路,促进内皮细胞产生血管内皮生长因子导致血管生成;而LDL下调NF-κB通路的多个关键成分关闭TNF-α/NF-κB/HIF信号级联反应,导致一系列依赖HIF的下游事件失活,减弱缺氧诱导的血管生成。ZHANG等[48]表示过表达的长非编码RNA CPS1-IT通过降低白细胞介素1β从而减轻阻塞性睡眠呼吸暂停大鼠的肺动脉高压,其机制与抑制依赖于NF-κB通路的HIF-1α转录活性有关。杨娟等[49]在研究植物雌激素影响缺氧肺部炎症浸润的机制时,发现植物雌激素削弱缺氧对膜联蛋白A1(AnxA1)基因表达的上调效应与HIF-1α和NF-κB的调节机制有关。根据以上结果完全有理由猜测,在低氧环境下,NF-κB通路对HIF-1α的表达起促进作用,并且通过介导HIF-1α的蛋白表达过程,影响下游血管内皮生长因子的表达水平,从而诱导血管生成,改善炎症反应,见表1。但反过来,HIF-1α如何调节NF-κB却知之甚少。BANDARRA等[50]提出HIF-1α在调控促炎反应过程中对NF-κB通路起抑制作用,从而保护机体免受过度炎症反应的损害。 2.2.6 HIF-1α与Notch通路 Notch通路本质为2个细胞间的接触反应。相邻细胞的Notch受体配体相互作用,经过分步切割后将有活性的胞内段NICD(Notch intracellular domain)释放入胞内,并进入细胞核与转录因子CSL(CBF-1,Suppressor of hairless,Lag)结合,形成NICD/CSL复合物,启动下游基因,限制血管生长和有丝分裂过程[51]。Notch通路广泛参与调节信号中枢和外周淋巴器官中T细胞和B细胞的多个阶段的发育过程,并在先天免疫和炎症反应中发挥关键作用[52],一旦失调将会导致许多疾病的发生。 KIM等[53]对HIF和Notch信号通路在中枢神经系统的神经元、血管中的密切联系和相互作用得到了详细的阐述。李德龙等[54]表明Notch信号通路在缺血再灌注损伤中被激活,其机制可能与HIF-1α有关。MIRANDA等[55]发现在牙源性角化囊肿中靠近囊腔的副基底层的低氧情况更严重,且这些区域HIF-1α呈现高表达状态,同时高表达的还有Notch1,这一结果提示HIF-1α激活了Notch1。ZOU等[56]也表示Notch1信号是低氧时HIF-1α诱导T细胞急性淋巴白血病增殖、侵袭和耐药过程中所必需的。PARK等[57]认为HIF-1α对Notch信号通路不是直接激活,而是由血管内皮生长因子介导的间接激活。LI等[58]研究认为HIF-1α通过结合与稳定NICD,激活Notch下游基因的转录和翻译,从而参与癫痫发生过程。另外一项研究表明Notch信号通过增强HIF-1α的转录活性刺激糖酵解,而在常氧和低氧条件下敲除HIF-1α基因可以减弱NICD1上调的糖酵解基因[59]。MENG等[60]使用暴露于电离辐射的细胞评估FIH抑制剂NOFD的作用,发现NOFD显著增加了下游Notch2基因的表达。与之前报道不同的是,ITOH等[61]研究结果表明低氧增加了特定细胞系中HIF-1α和HIF-2α的表达,而抑制了Notch1的激活。大多数证据表明,HIF-1的上调促进了Notch的表达,甚至可以推测HIF-1通过调控下游血管内皮生长因子的表达间接调节Notch的表达。但仍有一部分实验结果与这一结论不同,虽然没有直接的证据表明HIF-1上调直接抑制了Notch的活性,但HIF-1的上调不总是导致Notch激活的这一事实确确实实存在。"

| [1] SEMENZA GL, WANG GL. A nuclear factor induced by hypoxia via de novo protein synthesis binds to the human erythropoietin gene enhancer at a site required for transcriptional activation. Mol Cell Biol. 1992;12(12):5447-5454. [2] 杨梦思,周娜,王志钢,等.转录因子HIF-1α及其信号通路在疾病发生中的作用研究进展[J].生物技术通报,2016,32(8):8-13. [3] 刘川川,马兰,格日力.HIF-1调控低氧性肺动脉高压[J].生理科学进展, 2018,49(6):423-427. [4] GRAHAM AM, MCCRACKEN KG. Convergent evolution on the hypoxia-inducible factor (HIF) pathway genes EGLN1 and EPAS1 in high-altitude ducks. Heredity (Edinb). 2019;122(6):819-832. [5] XU Y, TAN M, MA X, et al. Human mesenchymal stem cells-derived conditioned medium inhibits hypoxia-induced death of neonatal porcine islets by inducing autophagy. Xenotransplantation. 2020;27(1):e12556. [6] CAO X, FANG X, MALIK WS, et al. TRB3 interacts with ERK and JNK and contributes to the proliferation, apoptosis, and migration of lung adenocarcinoma cells. J Cell Physiol. 2020;235(1):538-547. [7] 赵吉清,林海,吴强,等.缺氧PC12细胞中JAK1/STAT1、STAT3介导的信号通路变化[J].第三军医大学学报,2009,31(5):391-393. [8] CHO S, YU SL, KANG J, et al. NADPH oxidase 4 mediates TGF-β1/Smad signaling pathway induced acute kidney injury in hypoxia. PLoS One. 2019; 14(7):e0219483. [9] KOLB R, KLUZ P, TAN ZW, et al. Obesity-associated inflammation promotes angiogenesis and breast cancer via angiopoietin-like 4. Oncogene. 2019; 38(13):2351-2363. [10] XU ZX, XU L, WANG JQ, et al. Expression changes of the notch signaling pathway of PC12 cells after oxygen glucose deprivation. Int J Biol Macromol. 2018;118(Pt B):1984-1988. [11] SIM J, COWBURN AS, PALAZON A, et al. The Factor Inhibiting HIF Asparaginyl Hydroxylase Regulates Oxidative Metabolism and Accelerates Metabolic Adaptation to Hypoxia. Cell Metab. 2018;27(4):898-913. [12] LI Y, YANG W, QUINONES-HINOJOSA A, et al. Interference with Protease-activated Receptor 1 Alleviates Neuronal Cell Death Induced by Lipopolysaccharide-Stimulated Microglial Cells through the PI3K/Akt Pathway. Sci Rep. 2016;6:38247. [13] YOU S, LI W, GUAN Y. Tunicamycin inhibits colon carcinoma growth and aggressiveness via modulation of the ERK-JNK-mediated AKT/mTOR signaling pathway. Mol Med Rep. 2018;17(3):4203-4212. [14] STEGEMAN H, SPAN PN, KAANDERS JH, et al. Improving chemoradiation efficacy by PI3-K/AKT inhibition. Cancer Treat Rev. 2014;40(10): 1182-1191. [15] SEGUELLA L, CAPUANO R, PESCE M, et al. S100B Protein Stimulates Proliferation and Angiogenic Mediators Release through RAGE/pAkt/mTOR Pathway in Human Colon Adenocarcinoma Caco-2 Cells. Int J Mol Sci. 2019; 20(13):3240. [16] TONG J, XU X, ZHANG Z, et al. Hypoxia-induced long non-coding RNA DARS-AS1 regulates RBM39 stability to promote myeloma malignancy. Haematologica. 2019 Jul 9. doi: 10.3324/haematol.2019.218289. Online ahead of print. [17] LEE HJ, JUNG YH, CHOI GE, et al. O-cyclic phytosphingosine-1-phosphate stimulates HIF1α-dependent glycolytic reprogramming to enhance the therapeutic potential of mesenchymal stem cells. Cell Death Dis. 2019; 10(8):590. [18] SHI L, GUO C, XIE Y, et al. Dexmedetomidine Attenuates Lung Injury in Obstructive Jaundice Rats Through PI3K/Akt/HIF-1α Signaling Pathway. Arch Med Res. 2019;50(5):233-240. [19] PEI-YUAN Z, YU-WEI L, XIANG-NAN Z, et al. Overexpression of Axl reverses endothelial cells dysfunction in high glucose and hypoxia. J Cell Biochem. 2019;120(7):11831-11841. [20] 陈建勇,王聪,王娟,等.MAPK信号通路研究进展[J].中国医药科学, 2011,1(8):32-34. [21] 万军,车云,康宁宁.信号通路蛋白Akt1、ERK1/2与HIF-1α在肺癌组织中的表达及其临床意义[J].实用癌症杂志,2014,29(12):1515-1517. [22] KARAGIOTA A, KOURTI M, SIMOS G, et al. HIF-1α-derived cell-penetrating peptides inhibit ERK-dependent activation of HIF-1 and trigger apoptosis of cancer cells under hypoxia. Cell Mol Life Sci. 2019;76(4):809-825. [23] WU MZ, CHEN SF, NIEH S, et al. Hypoxia Drives Breast Tumor Malignancy through a TET-TNFα-p38-MAPK Signaling Axis. Cancer Res. 2015;75(18): 3912-3924. [24] KOLIBABKA M, DIETRICH N, KLEIN T, et al. Anti-angiogenic effects of the DPP-4 inhibitor linagliptin via inhibition of VEGFR signalling in the mouse model of oxygen-induced retinopathy. Diabetologia. 2018;61(11):2412-2421. [25] TALWAR H, BOUHAMDAN M, BAUERFELD C, et al. MEK2 Negatively Regulates Lipopolysaccharide-Mediated IL-1β Production through HIF-1α Expression. J Immunol. 2019;202(6):1815-1825. [26] GONG J, ZHOU S, YANG S. Vanillic Acid Suppresses HIF-1α Expression via Inhibition of mTOR/p70S6K/4E-BP1 and Raf/MEK/ERK Pathways in Human Colon Cancer HCT116 Cells. Int J Mol Sci. 2019;20(3):465. [27] 薛翔,刘红梅,邵旦兵,等.JAK/STAT信号通路调节机制的研究进展[J].现代生物医学进展,2015,15(11):2161-2165. [28] 蒲霞,郭庆喜,周鹏飞,等.JAK/STAT1信号途径在低氧诱导肾小管上皮-间质转化中的作用[J].泸州医学院学报,2016,39(1):34-37. [29] JACOBSSON H, HARRISON H, HUGHES É, et al. Hypoxia-induced secretion stimulates breast cancer stem cell regulatory signalling pathways. Mol Oncol. 2019;13(8):1693-1705. [30] HUANG S, BUCANA CD, VAN ARSDALL M, et al. Stat1 negatively regulates angiogenesis, tumorigenicity and metastasis of tumor cells. Oncogene. 2002;21(16):2504-2512. [31] ZHANG Y, JIN G, ZHANG J, et al. Overexpression of STAT1 suppresses angiogenesis under hypoxia by regulating VEGF‑A in human glioma cells. Biomed Pharmacother. 2018;104:566-575. [32] COULIBALY A, BETTENDORF A, KOSTINA E, et al. Interleukin-15 Signaling in HIF-1α Regulation in Natural Killer Cells, Insights Through Mathematical Models. Front Immunol. 2019;10:2401. [33] 林建国,刘冰冰,梁飞,等.JAK2/STAT3/HIF-1α信号通路对脑卒中的影响[J].新医学,2020,51(1):6-11. [34] PARK SJ, KIM H, KIM SH, et al. Epigenetic downregulation of STAT6 increases HIF-1α expression via mTOR/S6K/S6, leading to enhanced hypoxic viability of glioma cells. Acta Neuropathol Commun. 2019;7(1):149. [35] LIMA JDCC, SIMOES E, DE CASTRO G, et al. Tumour-derived transforming growth factor-β signalling contributes to fibrosis in patients with cancer cachexia. J Cachexia Sarcopenia Muscle. 2019;10(5):1045-1059. [36] 徐荣伟,王金申,李乐平,等.大肠癌组织中TGF-β1和HIF-1α和VEGF的表达及其临床意义[J].中国现代普通外科进展,2014,17(7):505-510. [37] LEI R, LI J, LIU F, et al. HIF-1α promotes the keloid development through the activation of TGF-β/Smad and TLR4/MyD88/NF-κB pathways. Cell Cycle. 2019;18(23):3239-3250. [38] LIU Z, GUO L, LI R, et al. Transforming growth factor-β1 and hypoxia inducible factor-1α synergistically inhibit the osteogenesis of periodontal ligament stem cells. Int Immunopharmacol. 2019;75:105834. [39] YOSHIMOTO S, TANAKA F, MORITA H, et al. Hypoxia-induced HIF-1α and ZEB1 are critical for the malignant transformation of ameloblastoma via TGF-β-dependent EMT. Cancer Med. 2019;8(18):7822-7832. [40] ROSELL-GARCÍA T, PALOMO-ÁLVAREZ O, RODRÍGUEZ-PASCUAL F. A hierarchical network of hypoxia-inducible factor and SMAD proteins governs procollagen lysyl hydroxylase 2 induction by hypoxia and transforming growth factor β1. J Biol Chem. 2019;294(39):14308-14318. [41] 赵运旺.NF-κB信号通路研究进展[J].甘肃科技,2016,32(21):117-123. [42] 曲畅,吴云红,穆靖洲,等.核转录因子-κB在缺氧导致的炎症中的作用[J].生理科学进展, 2018,49(1):39-43. [43] FITZPATRICK SF, TAMBUWALA MM, BRUNING U, et al. An intact canonical NF-κB pathway is required for inflammatory gene expression in response to hypoxia. J Immunol. 2011;186(2):1091-1096. [44] JIANG Y, ZHU Y, WANG X, et al. Temporal regulation of HIF-1 and NF-κB in hypoxic hepatocarcinoma cells. Oncotarget. 2015;6(11):9409-9419. [45] 符智慧,徐凌,黄姣,等.LPS通过p38/MAPK通路和NF-κB的活化诱导RAW264.7细胞HIF-1α的表达[J].重庆医科大学学报,2016,41(10): 1033-1039. [46] GUO R, LI Y, WANG Z, et al. Hypoxia-inducible factor-1α and nuclear factor-κB play important roles in regulating programmed cell death ligand 1 expression by epidermal growth factor receptor mutants in non-small-cell lung cancer cells. Cancer Sci. 2019;110(5):1665-1675. [47] JIN F, ZHENG X, YANG Y, et al. Impairment of hypoxia-induced angiogenesis by LDL involves a HIF-centered signaling network linking inflammatory TNFα and angiogenic VEGF. Aging (Albany NY). 2019;11(2):328-349. [48] ZHANG Z, LI Z, WANG Y, et al. Overexpressed long noncoding RNA CPS1-IT alleviates pulmonary arterial hypertension in obstructive sleep apnea by reducing interleukin-1β expression via HIF1 transcriptional activity. J Cell Physiol. 2019;234(11):19715-19727. [49] 杨娟,李晓栩,陈建,等.植物雌激素通过缺氧诱导因子-1α与核因子- κB下调缺氧肺泡上皮细胞膜联蛋白A1的表达[J].第三军医大学学报, 2013,35(19):2019-2023. [50] BANDARRA D, BIDDLESTONE J, MUDIE S, et al. HIF-1α restricts NF-κB-dependent gene expression to control innate immunity signals. Dis Model Mech. 2015;8(2):169-181. [51] RAMASAMY SK, KUSUMBE AP, WANG L, et al. Endothelial Notch activity promotes angiogenesis and osteogenesis in bone. Nature. 2014;507(7492): 376-380. [52] SHANG Y, SMITH S, HU X. Role of Notch signaling in regulating innate immunity and inflammation in health and disease. Protein Cell. 2016;7(3): 159-174. [53] KIM S, LEE M, CHOI YK. The Role of a Neurovascular Signaling Pathway Involving Hypoxia-Inducible Factor and Notch in the Function of the Central Nervous System. Biomol Ther (Seoul). 2020;28(1):45-57. [54] 李德龙,牛其芳,冯芝恩,等.Notch信号通路中HIF-1α及Notch-1在组织瓣缺血再灌注损伤后的表达变化[J].中国口腔颌面外科杂志,2018, 16(3):209-214. [55] MIRANDA DA COSTA NM, SAAB ABE CT, MITRE GP, et al. HIF-1α is Overexpressed in Odontogenic Keratocyst Suggesting Activation of HIF-1α and NOTCH1 Signaling Pathways. Cells. 2019;8(7):731. [56] ZOU J, LI P, LU F, et al. Notch1 is required for hypoxia-induced proliferation, invasion and chemoresistance of T-cell acute lymphoblastic leukemia cells. J Hematol Oncol. 2013;6:3. [57] PARK ST, KIM BR, PARK SH, et al. Suppression of VEGF expression through interruption of the HIF‑1α and Akt signaling cascade modulates the anti‑angiogenic activity of DAPK in ovarian carcinoma cells. Oncol Rep. 2014; 31(2):1021-1029. [58] LI Y, WU L, YU M, et al. HIF-1α is Critical for the Activation of Notch Signaling in Neurogenesis During Acute Epilepsy. Neuroscience. 2018;394:206-219. [59] MORIYAMA H, MORIYAMA M, OZAWA T, et al. Notch Signaling Enhances Stemness by Regulating Metabolic Pathways Through Modifying p53, NF-κB, and HIF-1α. Stem Cells Dev. 2018;27(13):935-947. [60] MENG Y, YANG F, LONG W, et al. Radioprotective Activity and Preliminary Mechanisms of N-oxalyl-d-phenylalanine (NOFD) In Vitro. Int J Mol Sci. 2018;20(1):37. [61] ITOH M, OKUHASHI Y, TAKAHASHI Y, et al. Hypoxia Up-regulates HIF Expression While Suppressing Cell Growth and NOTCH Activity in Leukaemia Cells. Anticancer Res. 2019;39(8):4165-4170. [62] 范明,朱玲玲.低氧研究受到关注[J].生理学报,2019,71(5):806-808. |
| [1] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [2] | Jiang Yong, Luo Yi, Ding Yongli, Zhou Yong, Min Li, Tang Fan, Zhang Wenli, Duan Hong, Tu Chongqi. Von Mises stress on the influence of pelvic stability by precise sacral resection and clinical validation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1318-1323. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Yuan Jiawei, Zhang Haitao, Jie Ke, Cao Houran, Zeng Yirong. Underlying targets and mechanism of Taohong Siwu Decoction in prosthetic joint infection on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1428-1433. |
| [5] | Geng Qiudong, Ge Haiya, Wang Heming, Li Nan. Role and mechanism of Guilu Erxianjiao in treatment of osteoarthritis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1229-1236. |
| [6] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [7] | Liu Cong, Liu Su. Molecular mechanism of miR-17-5p regulation of hypoxia inducible factor-1α mediated adipocyte differentiation and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1069-1074. |
| [8] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [9] | Zhao Min, Feng Liuxiang, Chen Yao, Gu Xia, Wang Pingyi, Li Yimei, Li Wenhua. Exosomes as a disease marker under hypoxic conditions [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1104-1108. |
| [10] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [11] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [12] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [13] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [14] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [15] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||