Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (23): 3739-3745.doi: 10.3969/j.issn.2095-4344.1322
Previous Articles Next Articles
Regulation of osteoclasts derived coupling factors sphingosine-1-phosphate and platelet-derived growth factor-BB on osteoblasts
Liu Hui1, 2, Chen Huihong2, Liao Hongbing1
- (1Department of Prosthodontics, College and Hospital of Stomatology, Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China; 2Guangxi Key Laboratory of Oral and Maxillofacial Rehabilitation and Reconstruction, Guangxi Clinical Research Center for Craniofacial Deformity, Guangxi Key Laboratory of Oral and Maxillofacial Surgery Disease Treatment, Nanning 530021, Guangxi Zhuang Autonomous Region, China)
-
Received:2019-03-15Online:2019-08-18Published:2019-08-18 -
Contact:Liao Hongbing, MD, Professor, Department of Prosthodontics, College and Hospital of Stomatology, Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
About author:Liu Hui, Master candidate, Department of Prosthodontics, College and Hospital of Stomatology, Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China; Guangxi Key Laboratory of Oral and Maxillofacial Rehabilitation and Reconstruction, Guangxi Clinical Research Center for Craniofacial Deformity, Guangxi Key Laboratory of Oral and Maxillofacial Surgery Disease Treatment, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
Supported by:the National Natural Science Foundation of China, No. 81560190 and 81860201 (both to LHB); the Natural Science Foundation of Guangxi Zhuang Autonomous Region, No. 2016GXNSFAA380289 (to LHB)
CLC Number:
Cite this article
Liu Hui1, 2, Chen Huihong2, Liao Hongbing1. Regulation of osteoclasts derived coupling factors sphingosine-1-phosphate and platelet-derived growth factor-BB on osteoblasts[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(23): 3739-3745.
share this article
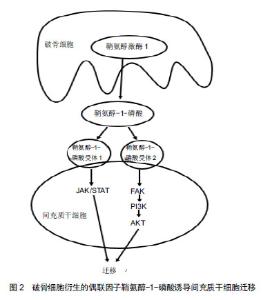
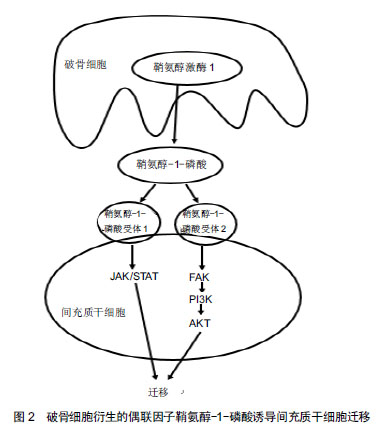
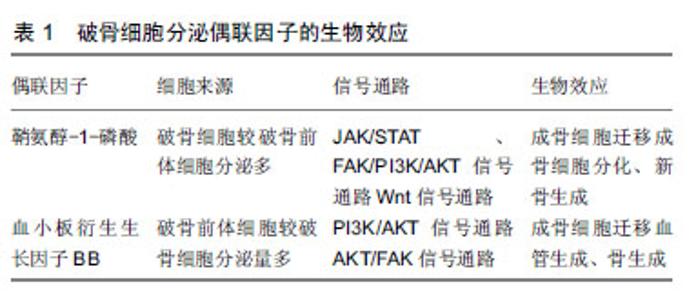
2.1 破骨细胞对成骨细胞的调节作用及促进骨生成 破骨细胞主要来源于骨髓中特定的破骨前体细胞,并在一系列的信号分子,如趋化因子、细胞因子等的指导下迁移至骨吸收单位,发挥骨降解的作用。另外,破骨细胞对成骨细胞的形成、增殖、活化、迁移[9],骨形成也具有十分重要的调节作用[10]。 在缺乏破骨细胞的动物模型中,发现其成骨细胞的数量减少和活性降低,从而导致了成骨细胞的调节活动发生异常,最终形成了不正常的骨结构,如骨皮质结构中断,缺乏有规律的板层骨结构[11-12],而且骨的矿化作用也减少、骨量明显减少[13]。反之,在破骨细胞存在的小鼠或者骨硬化病的患者可以观察到骨形成量正常或者增加[14-15]。Thudium等[16]研究发现,富含破骨细胞的骨硬化症小鼠比缺乏破骨细胞的骨硬化症小鼠的骨小梁更多、骨长度更长及骨生成量明显更多。这些研究发现与传统的认知形成鲜明的对比,即破骨细胞维持在一定数量范围内,有利于促进骨的形成,甚至提高了骨的强度。表明了破骨细胞的存在对于骨质量和骨形成是必不可少的,骨形成的启动与破骨细胞息息相关。那么破骨细胞是怎么与成骨细胞交流的呢? 在不断的探索过程中,体外研究发现破骨细胞独立于它的骨吸收作用,可以合成并分泌或者释放某些代谢信号分子,通过这些信号分子与成骨系细胞交流,从而促进成骨系细胞在骨吸收陷窝处募集、分泌骨基质,促进骨形成,从而获得最佳的骨质量和骨量[17-19]。Henriksen等[20]报道接种在骨片上的成熟破骨细胞条件培养基中,第5天就发现存在代谢因子,16 d达到高峰,直到第19天因子才丢失。研究者用破骨细胞条件培养基培养成骨前体细胞,观察破骨细胞对成骨前体细胞的作用。研究发现破骨细胞条件培养基可以诱导成骨前体细胞成骨分化,形成钙结节,而非破骨细胞条件培养基则无此现象[20-21]。此外,破骨细胞条件培养基诱导骨形成的量与破骨细胞的数量呈正相关,在一定范围内,破骨细胞越多,骨形成量越多[20]。这些研究表明了破骨细胞可以分泌某些信号分子,从而促进成骨前体细胞成骨分化,诱导钙结节的形成。 据报道,骨形态发生蛋白6、Wnt10b已经被证实是破骨细胞分泌的代谢因子[6-9],破骨细胞分泌的骨形态发生蛋白6、Wnt10b在破骨细胞介导的骨吸收后,促进成骨系细胞活化、增殖,并且引导成骨系细胞迁移至骨吸收陷窝,促进新骨生成填充骨缺损。近些年,越来越多的研究发现破骨细胞还可以衍生其他生长因子,如鞘氨醇-1-磷酸[22-25]、血小板衍生生长因子BB[8,17,25-26],这2个偶联因子在调节成骨系细胞的迁移、增殖、分化和矿化也扮演着非常重要的作用。 2.2 破骨细胞衍生的鞘氨醇-1-磷酸介导成骨细胞迁移与成骨作用 鞘氨醇-1-磷酸是细胞膜上鞘脂代谢过程中产生的一种具有生物活性的信号分子[27]。鞘脂在一系列水解酶和神经酰胺酶的作用下形成鞘氨醇,鞘氨醇在鞘氨醇激酶的作用下磷酸化形成鞘氨醇-1-磷酸。鞘氨醇-1-磷酸主要由红细胞和血小板分泌[28],破骨细胞也可以衍生鞘氨醇-1-磷酸[22-25],鞘氨醇-1-磷酸既可以作为细胞内第二信使,又可以与细胞表面特定的G蛋白偶联受体结合后,调节细胞生存、增殖、分化、迁移和凋亡[24,29-32],从而调节心血管、免疫、骨骼系统的功能[29-31,33]。目前发现鞘氨醇-1-磷酸存在5种不同的受体(S1PR1-S1PR5),成骨系细胞可以表达鞘氨醇-1-磷酸受体[34],破骨细胞衍生的鞘氨醇-1-磷酸与成骨系细胞表达鞘氨醇-1-磷酸受体结合后对成骨系细胞产生多种作用,如促进成骨细胞存活、增殖、迁移和分化等[27]。 2.2.1 鞘氨醇-1-磷酸/鞘氨醇-1-磷酸受体1与鞘氨醇- 1-磷酸/鞘氨醇-1-磷酸受体2轴调节成骨细胞的迁移 趋化性和细胞迁移是骨组织形成和骨重建所必需的关键环节。间充质干细胞的迁移与成骨细胞向分化、及成骨细胞在骨吸收陷窝募集是骨重建和骨修复的必要条件。那么,破骨细胞衍生的偶联因子鞘氨醇-1-磷酸对成骨系细胞的迁移作用是否有影响呢?Pederson等[22]利用基因芯片技术、RT-PCR、WB等检测手段证实了破骨系细胞可以分泌鞘氨醇在鞘氨醇激酶1,鞘氨醇在鞘氨醇激酶1磷酸化形成鞘氨醇-1-磷酸,并且破骨细胞较其前体细胞表达更多鞘氨醇在鞘氨醇激酶1,进而破骨细胞比破骨前体细胞衍生更多的鞘氨醇-1-磷酸。在RAW264.7细胞分化成熟的破骨细胞条件培养基中添加鞘氨醇在鞘氨醇激酶1拮抗剂,发现鞘氨醇在鞘氨醇激酶1拮抗剂明显抑制了人骨髓间充质干细胞的趋化性,也抑制了人骨髓间充质干细胞的成骨分化作用。Ryu等[24]报道,在破骨前体细胞分化的过程中产生的鞘氨醇-1-磷酸以旁分泌的方式作用于成骨细胞,刺激成骨细胞的迁移和存活。浓度为0.01-1 μmol/L的鞘氨醇-1-磷酸可以加强成骨细胞的迁移;FTY-720是一种鞘氨醇模拟物,磷酸化形成FTY-720-P,FTY-720-P与除鞘氨醇-1-磷酸受体2外的4个鞘氨醇-1-磷酸受体特异性结合而竞争性抑制鞘氨醇- 1-磷酸与鞘氨醇-1-磷酸受体结合,添加FTY-720,抑制成骨细胞的迁移。并且通过病毒转染使骨髓来源的巨噬细胞过表达或者沉默鞘氨醇在鞘氨醇激酶1,从而上调或者下调鞘氨醇-1-磷酸产生的量,进而促进或者抑制了成骨细胞的迁移作用。表明了鞘氨醇-1-磷酸在一定的浓度下可以促进成骨细胞的迁移。 鞘氨醇-1-磷酸通过鞘氨醇-1-磷酸/鞘氨醇-1-磷酸受体1与鞘氨醇-1-磷酸/鞘氨醇-1-磷酸受体2轴调控成骨系细胞的迁移。据研究报道,激酶驱动信号可以调节许多类型细胞的迁移,PI3K/AKT信号通路、JAK/STAT信号通路均可调控细胞的迁移[35-38]。鞘氨醇- 1-磷酸受体1和鞘氨醇-1-磷酸受体2是鞘氨醇-1-磷酸的受体,成骨系细胞可以表达鞘氨醇-1-磷酸受体1和鞘氨醇-1-磷酸受体2[39]。Quint等[40]报道,破骨细胞衍生的趋化因子鞘氨醇-1-磷酸活化鞘氨醇-1-磷酸受体1和鞘氨醇-1-磷酸受体2,阻断鞘氨醇-1-磷酸受体1时,仅仅活化了鞘氨醇-1-磷酸受体2,反之,阻断鞘氨醇-1-磷酸受体2则仅活化鞘氨醇-1-磷酸受体1。鞘氨醇-1-磷酸结合鞘氨醇-1-磷酸受体1和鞘氨醇-1-磷酸受体2分别激活JAK/STAT和FAK/PI3K/AKT信号通路,诱导了间充质干细胞的迁移,见图2。阻断JAK1、STAT3抑制了鞘氨醇-1-磷酸与鞘氨醇-1-磷酸受体1结合,但是不影响鞘氨醇-1-磷酸与鞘氨醇-1-磷酸受体2结合后激活FAK/PI3K/AKT信号通路。反之,阻断FAK、PI3K或者AKT抑制了鞘氨醇-1-磷酸受体2与鞘氨醇-1-磷酸结合,但并没有影响鞘氨醇-1-磷酸结合鞘氨醇-1-磷酸受体1后激活JAK/STAT通路,从而调节间充质干细胞的迁移作用,表明了鞘氨醇-1-磷酸是通过鞘氨醇-1-磷酸/鞘氨醇-1-磷酸受体1与鞘氨醇-1-磷酸/鞘氨醇-1-磷酸受体2轴的2个独立的信号通路促进间充质干细胞迁移。这些研究结果证明了破骨细胞衍生的偶联因子鞘氨醇- 1-磷酸与成骨系细胞表达的鞘氨醇-1-磷酸受体结合后,引导成骨系细胞的迁移作用。"
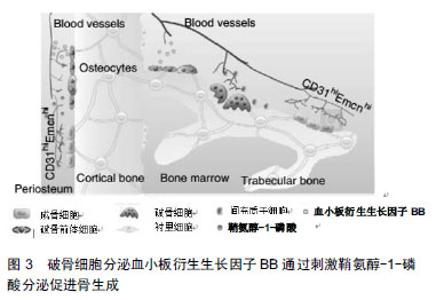
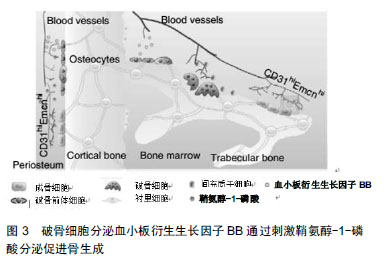
2.2.2 鞘氨醇-1-磷酸通过Wnt信号通路调节成骨分化及骨形成 破骨前体细胞和破骨细胞衍生的鞘氨醇-1-磷酸,促进骨生成[8]。研究发现鞘氨醇-1-磷酸可以诱导间充质干细胞分化为脂肪细胞、成骨细胞,并且认为鞘氨醇-1-磷酸促进间充质干细胞成脂分化时将导致成骨分化受到抑制[41-42]。Hashimoto等[43]学者发现鞘氨醇-1-磷酸介导间充质干细胞成脂和成骨分化存在负反馈调节回路,鞘氨醇-1-磷酸通过抑制间充质干细胞的成脂分化,从而促进间充质干细胞的成骨分化,提高了碱性磷酸酶和骨钙蛋白的表达,促进矿化作用,但是鞘氨醇-1-磷酸促进间充质干细胞成骨分化的机制仍然不清楚。为了进一步了解鞘氨醇-1-磷酸促进间充质干细胞成骨分化的机制,Hashimoto等[44]对成骨前体细胞成骨诱导前24 h在培养基中加入鞘氨醇-1-磷酸(浓度为1 μmol/L),发现鞘氨醇- 1-磷酸进一步促进成骨前体细胞在成骨分化期间Wnt5a的表达量,从而提高了Wnt受体LRP5和LRP6的表达,促进了成骨前体细胞成骨分化作用,促进骨形成。此外,在Wnt拮抗剂Dkk1、抗体以及鞘氨醇-1-磷酸拮抗剂的作用下,破骨细胞条件培养基诱导人成骨前体细胞成骨分化、钙结节的形成受到了抑制[22],从侧面反应了破骨细胞通过鞘氨醇-1-磷酸募集骨祖细胞至骨吸收陷窝,并通过提高Wnt通路刺激其分化及骨形成。 2.3 破骨细胞衍生的血小板衍生生长因子BB调节成骨细胞迁移与成骨作用 血小板衍生生长因子主要由血小板产生,亦可以由血管平滑肌细胞、血管内皮细胞、成骨细胞、破骨细胞等细胞分泌[45]。血小板衍生生长因子是一种二聚体阳离子糖蛋白,包括血小板衍生生长因子AA、血小板衍生生长因子BB、血小板衍生生长因子AB、血小板衍生生长因子CC及血小板衍生生长因子DD 5种分型[46]。血小板衍生生长因子的受体是一种细胞膜酪氨酸激酶,主要有2种类型血小板衍生生长因子受体α和血小板衍生生长因子受体β,血小板衍生生长因子通过激活血小板衍生生长因子受体对细胞进行调节,研究发现血小板衍生生长因子对间充质干细胞的迁移、分化、矿化等有重要的调节作用[8,47-48]。 2.3.1 血小板衍生生长因子BB/血小板衍生生长因子受体β轴调节成骨细胞的迁移 破骨前体细胞和破骨细胞衍生的血小板衍生生长因子BB是介导间充质干细胞迁移的另一个偶联因子。在破骨前体细胞分化成破骨细胞期间,血小板衍生生长因子BB的表达上调[17,25-26],血小板衍生生长因子BB刺激骨髓来源的人骨髓间充质干细胞[17]、鼠成骨前体细胞和成骨细胞的迁移并促进其存活[26]。研究发现,破骨前体细胞和破骨细胞可以分泌血小板衍生生长因子BB,加入骨片后可以提高血小板衍生生长因子BB的分泌量[8,25]。并且破骨前体细胞条件培养基较破骨细胞条件培养基能诱导更多的间充质干细胞的迁移,将骨片加入到培养基后,进一步刺激间充质干细胞的迁移[8]。 血小板衍生生长因子BB通过血小板衍生生长因子BB/血小板衍生生长因子受体β轴介导成骨系细胞的迁移。成骨前体细胞和成骨细胞表面低表达血小板衍生生长因子受体β,血小板衍生生长因子受体β与血小板衍生生长因子BB结合后,促发了成骨前体细胞和成骨细胞的趋化性,引导细胞迁移[26,49],并且成骨前体细胞的迁移作用较成骨细胞更明显[26],这种现象可能是因为成骨前体细胞分化成成骨细胞期间,血小板衍生生长因子受体β的表达下调,因此血小板衍生生长因子BB/血小板衍生生长因子受体β信号通路刺激成骨前体细胞迁移,同时抑制其本身的分化,更有效地促进成骨前体细胞在特定位点活化,从而促进成骨分化和骨重建、骨修复。血小板衍生生长因子BB与血小板衍生生长因子受体β结合后激活PI3K/AKT信号通路,从而促进成骨前体细胞和成骨细胞的迁移,PI3K抑制剂可以阻断这个信号通路,从而抑制成骨系细胞迁移[32]。这些研究说明了破骨前体细胞较破骨细胞分泌更多的血小板衍生生长因子BB,并且成骨系细胞能表达血小板衍生生长因子受体β,血小板衍生生长因子BB通过血小板衍生生长因子BB/血小板衍生生长因子受体β轴激活PI3K/AKT信号通路调节成骨系细胞的迁移作用。 2.3.2 血小板衍生生长因子BB通过鞘氨醇-1-磷酸介导AKT/FAK信号通路诱导骨生成 血小板衍生生长因子BB对成骨系细胞具有促有丝分裂作用使其增殖,促进细胞趋化作用及成骨作用,以促进骨折的愈合[50-51]。在动物模型实验中,血小板衍生生长因子BB可以促进牙槽骨生成[45,52-53]。成骨前体细胞迁移至正在发育中的骨的时机与血管的侵入密切相关,在骨发育和骨折修复期间,成骨前体细胞与血管一起侵入骨缺损区,促进新骨生成[54]。许多研究证明,血管生长不仅在新形成的骨骼中形成局部循环,从而获得营养和氧气,而且直接促进骨形成,血管内的血管窦是骨生成的起始位置[55-56],充足的血液供应对维护骨骼的健康至关重要,它运输的营养物质、氧气、矿物质直接促进成骨基质的形成和矿化作用[55-57]。破骨系细胞衍生的血小板衍生生长因子BB在破骨细胞生成期间上调血管内皮生长因子刺激血管生成,改善骨重塑过程中的血管的发生[8,25],从而更有利于骨的生成[8]。 CTSK是一种半胱氨酸蛋白酶,在破骨细胞中高度表达[58],CTSK基因敲除与CTSK抑制剂处理的小鼠的骨生成量增加,是因为在骨重建单元中存在某些偶联因 子[59]。研究发现,CTSK缺失或者缺乏的破骨细胞更有利于促进自身的鞘氨醇在鞘氨醇激酶1的表达,从而提高鞘氨醇-1-磷酸的分泌量[58],并且CTSK缺失或者缺乏的小鼠,其骨小梁和骨膜TRAP+PDGFBB+细胞数量增多,骨髓和骨膜中CD31hiEmcnhi细胞数量明显增多[8],促进血管生成和骨生成[8,60],见图3。表明了CTSK缺失的破骨细胞主要是通过提高鞘氨醇-1-磷酸和血小板衍生生长因子BB的分泌量,从而诱导血管生成,进而更有利于促进骨生成。那么,鞘氨醇-1-磷酸与血小板衍生生长因子BB之间是通过什么信号途径进行信息对话呢?"
研究报道,破骨细胞生成期间上调鞘氨醇-1-磷酸和血小板衍生生长因子BB的表达[25],破骨细胞通过血小板衍生生长因子在鞘氨醇-1-磷酸介导AKT/FAK信号通路中诱导血管生成和骨形成[8]。黏着斑激酶(FAK)的活化作用对细胞的迁移和血管生成至关重要,其激活是由几种激酶和磷酸酶相互作用产生的复杂机制协调的[61-62]。在Xie等[8]的报道中,破骨前体细胞条件培养基诱导间充质干细胞和内皮祖细胞的血小板衍生生长因子受体-β磷酸化,其下游的PI3K/AKT/FAK轴因子也被快速磷酸化作用。添加血小板衍生生长因子受体抑制剂阻断了其下游因子的磷酸化作用,同时也抑制了间充质干细胞、内皮祖细胞的迁移和血管生成,表明了血小板衍生生长因子通过AKT/FAK信号通路诱导血管生成和细胞迁移。而且血小板衍生生长因子b-/-小鼠比血小板衍生生长因子bflox/flox小鼠的破骨前体细胞鞘氨醇激酶1表达和鞘氨醇- 1-磷酸的分泌量低,表明了鞘氨醇-1-磷酸是血小板衍生生长因子的下游因子,鞘氨醇-1-磷酸1,3受体拮抗剂不能抑制破骨前体细胞条件培养基诱导间充质干细胞和内皮祖细胞的迁移[8],说明了血小板衍生生长因子是通过鞘氨醇-1-磷酸介导AKT/FAK信号通路诱导血管生成和骨生成。 "
| [1]Hattner R, Epker BN, Frost HM.Suggested sequential mode of control of changes in cell behaviour in adult bone remodelling. Nature.1965; 206(983):489-490.[2]Martin TJ, Sims NA. Osteoclast-derived activity in the coupling of bone formation to resorption. Trends Mol Med. 2005;11(2):76-81.[3]Sims NA, Martin TJ. Coupling signals between the osteoclast and osteoblast: how are messages transmitted between these temporary visitors to the bone surface? Front Endocrinol. 2015;6: 41.[4]Zhu S, Yao F, Qiu H, et al. Coupling factors and exosomal packaging microRNAs involved in the regulation of bone remodelling. Biol Rev Camb Philos Soc. 2018;93(1):469-480.[5]Sims NA, Martin TJ.Coupling the activities of bone formation and resorption: a multitude of signals within the basic multicellular unit. Bonekey Rep. 2014;3:481.[6]Furuya M, Kikuta J, Fujimori S, et al. Direct cell-cell contact between mature osteoblasts and osteoclasts dynamically controls their functions in vivo. Nature Commun. 2018;9(1):300.[7]Keller J, Catala-Lehnen P, Huebner AK, et al.Calcitonin controls bone formation by inhibiting the release of sphingosine 1-phosphate from osteoclasts. Nature Commun. 2014;5:5215.[8]Xie H, Cui Z, Wang L, et al. PDGF-BB secreted by preosteoclasts induces angiogenesis during coupling with osteogenesis. Nat Med. 2014;20(11):1270-1278.[9]Troen BR. Molecular mechanisms underlying osteoclast formation and activation. Exp Gerontol. 2003;38(6):605-614.[10]Han D, Zhang Q. An essential requirement for osteoclasts in refined bone-like tissue reconstruction in vitro. Med Hypotheses. 2006; 67(1): 75-78.[11]Dai XM, Zong XH, Akhter MP, et al.Osteoclast deficiency results in disorganized matrix, reduced mineralization, and abnormal osteoblast behavior in developing bone. J Bone Miner Res. 2004;19(9): 1441-1451.[12]Sakagami N, Amizuka N, Li M, et al. Reduced osteoblastic population and defective mineralization in osteopetrotic (op/op) mice. Micron. 2005;36(7-8):688-695.[13]Demiralp B, Chen HL, Koh AJ, et al. Anabolic actions of parathyroid hormone during bone growth are dependent on c-fos. Endocrinology. 2002;143(10):4038-4047.[14]Alatalo SL.Osteoclast-derived serum tartrate-resistant acid phosphatase 5b in albers-schonberg disease (type ii autosomal dominant osteopetrosis). Clin Chem. 2004;50(5):883-890.[15]Del Fattore A. Clinical, genetic, and cellular analysis of 49 osteopetrotic patients: implications for diagnosis and treatment. J Med Genet. 2005; 43(4):315-325.[16]Thudium CS, Moscatelli I, Flores C, et al.A comparison of osteoclast-rich and osteoclast-poor osteopetrosis in adult mice sheds light on the role of the osteoclast in coupling bone resorption and bone formation. Cal Tissue Int. 2014;95(1):83-93.[17]Kreja L, Brenner RE, Tautzenberger A, et al.Non-resorbing osteoclasts induce migration and osteogenic differentiation of mesenchymal stem cells. J Cell Biochem. 2009.[18]Kim BJ, Lee YS, Lee SY, et al.Afamin secreted from nonresorbing osteoclasts acts as a chemokine for preosteoblasts via the Akt-signaling pathway. Bone. 2012;51(3):431-440.[19]Karsdal MA, Martin TJ, Bollerslev J, et al. Are nonresorbing osteoclasts sources of bone anabolic activity? J Bone Miner Res. 2007;22(4): 487-494.[20]Henriksen K, Andreassen KV, Thudium CS, et al. A specific subtype of osteoclasts secretes factors inducing nodule formation by osteoblasts. Bone. 2012;51(3):353-361.[21]Karsdal MA, Neutzsky-Wulff AV, Dziegiel MH, et al. Osteoclasts secrete non-bone derived signals that induce bone formation. Biochem Biophys Res Commun. 2008;366(2):483-488.[22]Pederson L, Ruan M, Westendorf JJ, et al. Regulation of bone formation by osteoclasts involves Wnt/BMP signaling and the chemokine sphingosine-1-phosphate. Proc Natl Acad Sci U S A. 2008;105(52): 20764-20769. [23]Henriksen K, Karsdal MA, Martin TJ. Osteoclast-derived coupling factors in bone remodeling. Calcif Tissue Int. 2014;94(1):88-97.[24]Ryu J, Kim HJ, Chang EJ, et al. Sphingosine 1-phosphate as a regulator of osteoclast differentiation and osteoclast-osteoblast coupling. EMBO J. 2006;25(24):5840-5851.[25]Rahman MM, Matsuoka K, Takeshita S, et al. Secretion of PDGF isoforms during osteoclastogenesis and its modulation by anti-osteoclast drugs. Biochem Biophys Res Commun. 2015;462(2): 159-164.[26]Sanchez-Fernandez MA, Gallois A, Riedl T, et al. Osteoclasts control osteoblast chemotaxis via PDGF-BB/PDGF receptor beta signaling. PloS one. 2008;3(10):e3537.[27]Khavandgar Z, Murshed M.Sphingolipid metabolism and its role in the skeletal tissues. Cell Mol Life Sci. 2015;72(5):959-969.[28]Boyce BF.Advances in osteoclast biology reveal potential new drug targets and new roles for osteoclasts. J Bone Miner Rese. 2013;28(4): 711-722.[29]Hla T. Physiological and pathological actions of sphingosine 1-phosphate. Semin Cell Dev Biol. 2004;15(5):513-520.[30]Avery K, Avery S, Shepherd J,et al. Sphingosine-1-phosphate mediates transcriptional regulation of key targets associated with survival, proliferation, and pluripotency in human embryonic stem cells. Stem Cells Dev. 2008;17(6):1195-1205.[31]Patmanathan SN, Wang W, Yap LF, et al. Mechanisms of sphingosine 1-phosphate receptor signalling in cancer. Cell Signal. 2017;34:66-75.[32]Roelofsen T, Akkers R, Beumer W, et al. Sphingosine-1-phosphate acts as a developmental stage specific inhibitor of platelet-derived growth factor-induced chemotaxis of osteoblasts. J Cell Biochem. 2008;105(4): 1128-1138.[33]Dobrosak C, Gooi JH. Increased sphingosine-1-phosphate production in response to osteocyte mechanotransduction. Bone Rep. 2017;7:114-120.[34]Matsuzaki E, Hiratsuka S, Hamachi T, et al. Sphingosine-1- phosphate promotes the nuclear translocation of beta-catenin and thereby induces osteoprotegerin gene expression in osteoblast-like cell lines. Bone. 2013;55(2):315-324.[35]Zhang W, Yin L, Song G, et al. LKB1 loss cooperating with BRAF V600E promotes melanoma cell invasion and migration by up-regulation MMP-2 via PI3K/Akt/mTOR pathway. Oncotarget. 2017;8(69):113847-113857.[36]Huang Y, Mao Y, Li H, et al. Knockdown of Nrf2 inhibits angiogenesis by down-regulating VEGF expression through PI3K/Akt signaling pathway in cerebral microvascular endothelial cells under hypoxic conditions. Biochem Cell Biol. 2018.[37]Badgwell DB, Lu Z, Le K, et al. The tumor-suppressor gene ARHI (DIRAS3) suppresses ovarian cancer cell migration through inhibition of the Stat3 and FAK/Rho signaling pathways. Oncogene. 2011;31(1): 68-79.[38]Niwa Y, Kanda H, Shikauchi Y, et al.Methylation silencing of SOCS-3 promotes cell growth and migration by enhancing JAK/STAT and FAK signalings in human hepatocellular carcinoma. Oncogene. 2005;24(42): 6406-6417.[39]Sartawi Z, Schipani E, Ryan KB, et al. Sphingosine 1-phosphate (S1P) signalling: Role in bone biology and potential therapeutic target for bone repair. Pharmacol Res. 2017;125(Pt B):232-245.[40]Quint P, Ruan M, Pederson L, et al.Sphingosine 1-Phosphate (S1P) receptors 1 and 2 coordinately induce mesenchymal cell migration through S1P activation of complementary kinase pathways. J Biol Chem. 2013;288(8):5398-5406.[41]Cawthorn WP, Bree AJ, Yao Y, et al. Wnt6, Wnt10a and Wnt10b inhibit adipogenesis and stimulate osteoblastogenesis through a beta-catenin-dependent mechanism. Bone. 2012;50(2):477-489.[42]Rahman S, Czernik PJ, Lu Y, et al. beta-catenin directly sequesters adipocytic and insulin sensitizing activities but not osteoblastic activity of PPARgamma2 in marrow mesenchymal stem cells. PloS one. 2012; 7(12):e51746.[43]Hashimoto Y, Matsuzaki E, Higashi K, et al. Sphingosine-1- phosphate inhibits differentiation of C3H10T1/2 cells into adipocyte. Mol Cell Biochem. 2015;401(1-2):39-47.[44]Hashimoto Y, Kobayashi M, Matsuzaki E, et al.Sphingosine-1- phosphate-enhanced Wnt5a promotes osteogenic differentiation in C3H10T1/2 cells. Cell Biol Int. 2016;40(10):1129-1136.[45]Takeshita S, Fumoto T, Matsuoka K, et al.Osteoclast-secreted CTHRC1 in the coupling of bone resorption to formation. J Clin Invest. 2013,123(9): 3914-3924.[46]雷文龙,施斌.血小板衍生生长因子-BB在口腔种植领域中的作用[J].国际口腔医学杂志.2014,41(2):199-203. [47]Fiedler J, Etzel N, Brenner RE.To go or not to go: Migration of human mesenchymal progenitor cells stimulated by isoforms of PDGF. J Cell Biochem. 2004;93(5):990-998.[48]Fiedler J, Roderer G, Gunther KP, et al. BMP-2, BMP-4, and PDGF-bb stimulate chemotactic migration of primary human mesenchymal progenitor cells. J Cell Biochem. 2002;87(3): 305-312.[49]Tokunaga A, Oya T, Ishii Y, et al. PDGF receptor beta is a potent regulator of mesenchymal stromal cell function. J Bone Miner Res. 2008;23(9):1519-1528.[50]McCarthy HS, Williams JHH, Davie MWJ, et al. Platelet-derived growth factor stimulates osteoprotegerin production in osteoblastic cells. J Cell Phys. 2009;218(2):350-354.[51]Hollinger JO, Onikepe AO, MacKrell J, et al.Accelerated fracture healing in the geriatric, osteoporotic rat with recombinant human platelet-derived growth factor-bb and an injectable beta-tricalcium phosphate/collagen matrix. J Orthop Res. 2008;26(1):83-90.[52]Camelo M, Nevins ML, Schenk RK, et al. Periodontal regeneration in human Class II furcations using purified recombinant human platelet-derived growth factor-BB (rhPDGF-BB) with bone allograft. Int J Periodontics Restorative Dent. 2003;23(3):213-225.[53]Nevins M, Camelo M, Nevins ML, et al. Periodontal regeneration in humans using recombinant human platelet-derived growth factor-BB (rhPDGF-BB) and allogenic bone. J Periodontol. 2003;74(9): 1282-1292.[54]Maes C, Kobayashi T, Selig MK, et al. Osteoblast precursors, but not mature osteoblasts, move into developing and fractured bones along with invading blood vessels. Dev Cell. 2010;19(2):329-344.[55]Percival CJ, Richtsmeier JT. Angiogenesis and intramembranous osteogenesis. Dev Dyn. 2013;242(8):909-922.[56]Matsuyama J, Ohnishi I, Kageyama T, et al. Osteogenesis and angiogenesis in regenerating bone during transverse distraction. Clin Orthop Relat Res. 2005;(433):243-250.[57]Choi IH, Chung CY, Cho TJ, et al. Angiogenesis and mineralization during distraction osteogenesis. J Korean Med Sci. 2002;17(4): 435-447.[58]Lotinun S, Kiviranta R, Matsubara T, et al. Osteoclast-specific cathepsin K deletion stimulates S1P-dependent bone formation. J Clin Invest. 2013;123(2):666-681.[59]Bonnet N, Brun J, Rousseau JC, et al. Cathepsin K controls cortical bone formation by degrading periostin. J Bone Miner Res. 2017; 32(7):1432-1441.[60]Kusumbe AP, Ramasamy SK, Adams RH.Coupling of angiogenesis and osteogenesis by a specific vessel subtype in bone. Nature. 2014;507(7492):323-328.[61]Zhao X, Guan JL.Focal adhesion kinase and its signaling pathways in cell migration and angiogenesis. Adv Drug Deliv Rev. 2011;63(8): 610-615.[62]Zhao LN, Wang P, Liu YH, et al. MiR-383 inhibits proliferation, migration and angiogenesis of glioma-exposed endothelial cells in vitro via VEGF-mediated FAK and Src signaling pathways. Cell Signal. 2017;30:142-153. |
| [1] | Li Cai, Zhao Ting, Tan Ge, Zheng Yulin, Zhang Ruonan, Wu Yan, Tang Junming. Platelet-derived growth factor-BB promotes proliferation, differentiation and migration of skeletal muscle myoblast [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1050-1055. |
| [2] | Zheng Xiaolong, He Xiaoming, Gong Shuidi, Pang Fengxiang, Yang Fan, He Wei, Liu Shaojun, Wei Qiushi. Bone turnover characteristics in patients with alcohol-induced osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 657-661. |
| [3] | Xiao Sha, Gao Chengzhi, Zhou Dongping. Comparison of three kinds of bone replacement materials in the treatment of bone defects around the mandibular posterior teeth with immediate implantation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(34): 5495-5500. |
| [4] | Yang Li, Guan Shuai, Zhang Xingkui, Liu Bing, Li Xiangjun. Role and mechanism of polycystin in bone formation and bone disease treatments [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(29): 4715-4720. |
| [5] | Zhang Mingjin, Zhou Yanheng, Liu Dawei. Reopening the fused suture provides a new idea for maxillary expansion [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(29): 4721-4727. |
| [6] | Wang Qing, Weng Yiping, Liu Hongwei, Zhang Wen, Shi Qin, Zhang Runze, Jiang Junfeng, Wang Caimei. Three-dimensional printed porous titanium alloy scaffolds with different apertures in repair of femoral defects in rabbits: 600 μm aperture is more conducive to osseointegration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(28): 4441-4446. |
| [7] | Li Ruozhen, Tian Yaping, Wen Ning. Biological effect of magnetic fields to promote bone responses to biomaterials [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(28): 4510-4515. |
| [8] | Wei Congcong, Yao Mengxuan, Yang Meng, Li Huijie. Mechanism and treatment of osteolysis around artificial joint prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(27): 4401-4407. |
| [9] | Zheng Feng, Zhang Fucai, Xu Zhe. MicroRNA-98-5p promotes osteoblast proliferation and differentiation: possibilities and mechanisms [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(26): 4112-4117. |
| [10] | You Bin, Huang Xiuzhu, Lin Yu, Huang Xi. Changes in bone morphology, bone mineral density and bone metabolism in different parts of natural aging rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(26): 4118-4122. |
| [11] | Huo Hua, Cheng Yuting, Zhou Qian, Qi Yuhan, Wu Chao, Shi Qianhui, Yang Tongjing, Liao Jian, Hong Wei. Effects of drug coating on implant surface on the osseointegration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3558-3564. |
| [12] | Li Xiaoqun, Xu Kaihang, Ji Fang. Corylin inhibits osteoclastogenesis and attenuates postmenopausal osteoporosis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(2): 186-190. |
| [13] | Wei Qin, Zhang Xue, Ma Lei, Li Zhiqiang, Shou Xi, Duan Mingjun, Wu Shuo, Jia Qiyu, Ma Chuang. Platelet-derived growth factor-BB induces the differentiation of rat bone marrow mesenchymal stem cells into osteoblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 2953-2957. |
| [14] | Guo Zhibin, Wu Chunfang, Liu Zihong, Zhang Yuying, Chi Bojing, Wang Bao, Ma Chao, Zhang Guobin, Tian Faming. Simvastatin stimulates osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 2963-2968. |
| [15] | Wu Yukun, Han Jie, Wen Shuaibo. Mechanism of Runx2 gene in fracture healing [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(14): 2274-2279. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||