Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (15): 2443-2449.doi: 10.3969/j.issn.2095-4344.1183
Previous Articles Next Articles
Osteochondral lesions of the talus: etiology, diagnosis, treatment and prospects
Han Yu, Chang Fei, Jiang Zhende, Feng Naibo
- (Second Hospital of Jilin University, Changchun 130000, Jilin Province, China)
-
Received:2019-01-29Online:2019-05-28Published:2019-05-28 -
Contact:Chang Fei, MD, Professor, Second Hospital of Jilin University, Changchun 130000, Jilin Province, China -
About author:Han Yu, Master candidate, Second Hospital of Jilin University, Changchun 130000, Jilin Province, China -
Supported by:the Natural Science Foundation of China, No. 81671804 and 81701811 (both to CF), the Development and Reform Commission of Jilin Province, No. 2018C052-4 (to CF); the Health and Family Planning Commission of Jilin Province, No. 20165061 (to CF); the Science and Technology Development Project of Jilin Province, No. 20160101109JC (to CF)
CLC Number:
Cite this article
Han Yu, Chang Fei, Jiang Zhende, Feng Naibo. Osteochondral lesions of the talus: etiology, diagnosis, treatment and prospects[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(15): 2443-2449.
share this article
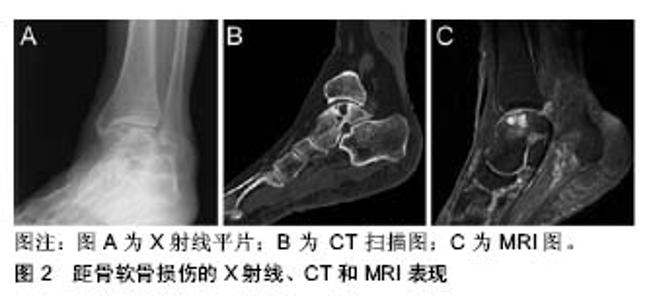
2.1 病因和发病机制 距骨软骨损伤的病因一直以来都存在争议,在1924年,Phemister率先提出创伤性因素是造成距骨软骨损伤的主要原因;随后在1933年,这一观点得到了Fairbank支持,后来又在1959年得到了Berndt和Harty的肯定[4-5]。然而在1966年,Campbell等[6]提出由缺血导致的骨坏死造成了距骨的病理性骨折,这样创伤就不是距骨软骨损伤的唯一致病因素,除创伤外的其他致病因素包括血管功能不全、滑膜损伤、软组织撞击、慢性踝关节不稳、遗传易感性和内分泌或代谢异常等[7],尤其是软骨下血管闭塞导致距骨囊变,最终造成了距骨病理性骨折。目前,一般认为创伤造成了64%的距骨内侧病变和98%的外侧病变[8]。外侧的距骨软骨损伤通常与剪切力机制的创伤有关,所以外侧损伤直径小、深度浅、更靠前;与此相对,内测损伤呈杯状,更深、更靠后;然而,一般认为损伤位置并不是判断损伤机制的可靠指标。 2.2 临床表现 距骨软骨损伤常发生于日常运动量较大的人群(如运动员等),且并无明显性别差异,基本上所有距骨软骨损伤的患者都会有明显的踝关节创伤或踝关节不稳相关病史,患者多数由于无明显特异性的踝关节深部间歇性疼痛就诊,其多与负重相关。 体格检查对于距骨软骨损伤是十分重要的。当踝关节极度跖屈时,按压距骨顶的内外侧,会出现明显的压痛,这是距骨软骨损伤最明显的体征,然而局部压痛可能并不总会呈阳性;胫距关节和距下关节的运动范围不会出现明显的限制,跖屈和背伸可能会引起弹响。 2.3 诊断和鉴别诊断 尽管距骨软骨损伤的症状和体征可以为最终的临床诊断提供指向性的线索,但是这些模糊的临床线索并不足以确诊距骨软骨损伤,因此相应的影像学检查和分析是诊断的主要依据。 对于怀疑距骨软骨损伤的患者,首需要通过X射线踝关节负重位平片进行初步的诊断和评估(图2A),并排除其他可能的骨性病变[9]。然而,虽然距骨软骨损伤传统上用X射线检查,但是大概有50%的病变是无法通过X射线发现的[10],且骨坏死、囊变以及骨碎片等具体情况很难在X射线平片上进行评估确认。 与X射线平片相比,CT扫描可以提供更为详尽和形象的信息(图2B),尤其是在估算损伤面积、计算骨量和鉴定病变相关囊肿等方面,这对距骨软骨损伤的手术治疗方案有很大的作用;近些年来,CT关节造影的发展也有有利于病损的诊断[11]。 相较于放射线检查,MRI在分辨软骨损伤、骨挫伤等方面具有其独特的优势(图2C)[12]。此外,MRI对于早期的距骨软骨相关病变和确认关节软骨完整性中具有相当大的鉴别作用;同时,也有人利用MRI对关节进行术后评估[13]。近些年来,T2 mapping的应用使得可以对软骨的情况进行量化分析[14-16]。然而,在急性的距骨软骨损伤诊断中,MRI可能对病变的严重程度难以做出准确的评估[17]。"
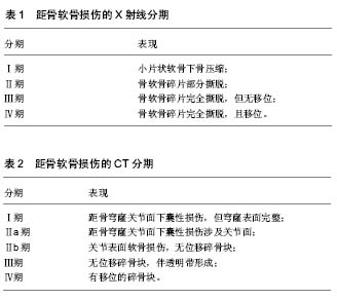
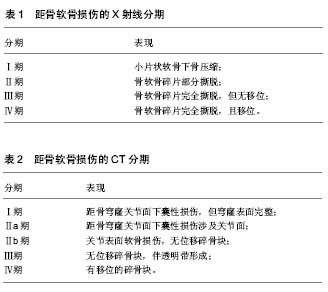
关节镜技术对于距骨软骨损伤来说,既是诊断方法,也是治疗手段。通过镜下观察,医生可以直观的评估软骨的损伤范围大小、深度和损伤程度,并可以对其进行清理等镜下治疗。但是作为一项检查来说,关节镜造成的组织创伤和昂贵的费用依旧是个巨大的问题。 综上,影像学检查是距骨软骨损伤的主要诊断方法,X射线主要用于距骨软骨损伤的筛查和早期诊断,CT对于诊断和手术方案的设计具有极其重要的意义,而MRI可以在早期发现软骨病变并用于对预后的评估。 2.4 病变分区和分期评估 距骨软骨损伤常发生于距骨顶,为了确定软骨病变位置,Elias等[17]将距骨顶的关节面分为9区域网格,然后回顾了428例距骨软骨损伤患者的MRI,发现超过半数的距骨软骨损伤发生在中间内侧区域,另外又有接近1/4发生在中间内侧区域。 距骨软骨损伤的临床分期基本都是通过影像学检查来确定的, Berndt和Harty最早提出利用X射线平片对距骨软骨损伤进行分期(见表1)[4]。Ferkel等[3]于1990年提出了一个基于CT的分期标准(见表2),这为距骨软骨损伤的临床诊断和治疗提供了十分有力的支持;与此同时,MRI也被用于距骨软骨损伤的分期(见表3)[18]。除此以外,Pritsch和Ferkle又分别提出了关节镜3期(见表4)和扩展的关节镜分期(见表5)[19]。"
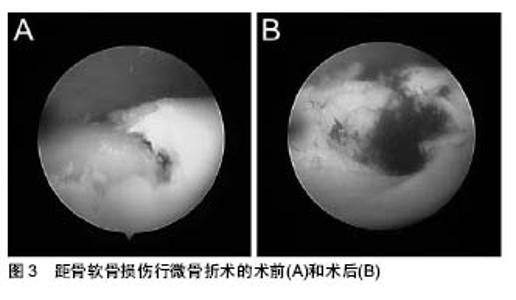
2.5 治疗 由于透明软骨固有的不可再生特性,即其无血管性、无神经,这就限制了关节软骨的愈合潜力,所以距骨软骨损伤的治疗一直都是一个巨大的难题[20]。目前临床上的疗法包括非手术治疗和手术治疗两大类,其中又以手术治疗为主。此次综述除主要系统介绍目前临床常用的非手术治疗和手术治疗方法外,还会列举出一些新的治疗理念、物理疗法、细胞疗法以及组织工程技术在距骨软骨损伤治疗上的应用,对其进行评估讨论,并对距骨软骨损伤治疗未来的发展方向进行展望。 2.5.1 非手术治疗 非手术疗法主要针对的是临床症状较轻且无明显移位的病变损伤,具体方法主要包括减少患者活动、减轻患者负重、使用石膏、行走靴等支具和使用非类固醇类抗炎药等,但是具体效果却难以得到有效的评估。通过Verhagen等[21]在2004年对距骨软骨损伤的系统评价中可以发现:在14项研究中,共计201位患者采用了非手术治疗,其中91位患者治愈,非手术治疗的成功率仅为45%。然而,在最短2年的随访中,约有86%的非手术治疗患者表示出疼痛的缓解甚至消失。Klammer等[22]的研究显示对于临床症状较轻且无明显病变移位的距骨软骨损伤患者,非手术治疗不会导致病变深度或者病变面积的进一步发展。在评估距骨软骨损伤自愈的指标中,损伤的位置无疑是很重要,例如,中外侧的损伤就很难通过非手术疗法得以治愈。总体来说,目前还没有出现针对距骨软骨损伤的理想非手术疗法,且保守治疗的期限最好不要超过半年。 2.5.2 手术治疗 (1)踝关节灌洗和清理:踝关节灌洗和清理的机制是通过去除踝关节腔内的能造成距骨软骨损伤的炎性因子(如基质金属蛋白酶等)和游离的碎块,进而延缓病变进展,最终达到痊愈的目的。尤其是随着关节镜技术的发展,踝关节灌洗和清理得到了更为广泛有效的利用,并且提高了手术成功率。对于慢性微小损伤和病变来说,踝关节灌洗和清理显示出了较好的效果,但单纯灌洗的疗效并不稳定,且踝关节灌洗和清理后加用刮除术会明显提升手术效果[21]。 (2)微骨折术:微骨折术,又称骨髓刺激,即从软骨损伤区域向软骨下骨钻孔以沟通髓腔(见图3)。由于软骨无血供,这使得软骨的修复变得十分困难;通过微骨折术沟通髓腔后,来源于骨髓的富含营养物质、生长因子和骨髓间充质干细胞的血凝块充盈于软骨损伤部位,随后骨髓间充质干细胞开始增殖分化为软骨细胞,最终形成纤维软骨型修复组织,但这样产生的多数是纤维软骨,且这样的组织以Ⅰ型胶原为主,Ⅱ型胶原水平相对较低[23]。"
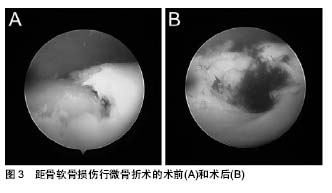
对于距骨软骨损伤来说,微骨折术的预后较为明确,具有操作难度低、效果良好等特点,尤其是关节镜技术的应用,更是提高了距骨软骨损伤的成功率[3,24]。最近,一项针对关节镜下微骨折术的术后5年随访显示:近半数患者没有任何症状,23.1%的患者在行走超过2 h后或在竞技体育活动后出现疼痛,且所有患者均无严重关节炎表现[25]。Ferkel等[3]于2008年报道了利用关节镜下微骨折术治疗距骨软骨损伤,其治愈率超过70%。然而,微骨折术只对小面积的损伤有效,Choi等[26]对120个踝关节行微骨折术,当病变面积小于1.5 cm2时,手术成功率达到了90%;当病变面积大于1.5 cm2时,微骨折术的成功率则小于20%。此外,微骨折术最终形成的是一种纤维软骨瘢痕组织,其生物力学特性远低于正常透明软骨,且不耐磨,这同样是微骨折术对于较大面积软骨缺损效果较差的原因之一[27]。 除大面积的软骨缺损外,存在有较大的软骨下骨囊肿也是不被建议应用微骨折术;然而,同样有研究显示软骨下囊变并不影响微骨折术的治疗结果。Lee等[28]对伴或不伴有软骨下囊变的距骨软骨损伤患者进行了一项前瞻性队列研究分析并进行评分,发现囊变和非囊变两组在评分上较术前评分均有显著提高,但两组之间并无明显差异。对于关节面完整的病损,有人提出逆行微骨折术要优于顺行钻孔,但是近些年来也有研究者提出用生物制剂辅助微骨折术,但其效果并不明确。 (3)切开复位内固定术:切开复位内固定术是另一种治疗距骨软骨损伤的方法,它主要是通过利用埋头钉或者可吸收钉将游离的骨块固定于距骨。对于具有较大游离碎片的急性距骨软骨损伤来说,切开复位内固定术具有令人吃惊的良好效果,Kumai等[29]通过7年的随访发现:切开复位内固定术对距骨软骨损伤的治愈率达到了89%;此外,Shank等[30]提出及时地内固定可以有效延缓关节炎的进程,更好的避免了后期地关节融合;然而,对于伴有硬化边缘的慢性病损来说该疗法的效果并不理想。 与其他疗法最大的不同就是,切开复位内固定术可以保留患者的固有软骨。Kumai等[29]的研究显示在对切开复位内固定术治疗距骨软骨损伤的术后随访中,取得超过90%的良好的影像学证据和临床结果,只有少数出现了软骨损伤处的塌陷。 (4)骨软骨移植:对于伴有巨大软骨缺损的距骨软骨损伤,目前主要应用的是骨软骨移植技术,即在充分暴露距骨损伤区域后,清理并修整病损区域的软骨和软骨下骨,随后将移植物植入缺损区域,最终达到修复距骨软骨损伤的目的(见图4)[31-32]。"
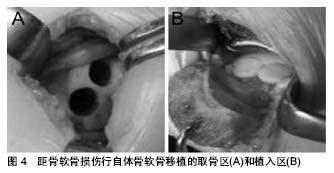
| [1] Leontaritis N, Hinojosa L, Panchbhavi VK. Arthroscopically detected intra-articular lesions associated with acute ankle fractures. J Bone Joint Surg Am. 2009; 91(2): 333-339.[2] Saxena A, Eakin C. Articular talar injuries in athletes: results of microfracture and autogenous bone graft. Am J Sports Med.2007; 35(10):1680-1687.[3] Ferkel RD, Zanotti RM, Komenda GA, et al.Arthroscopic treatment of chronic osteochondral lesions of the talus: long-term results. Am J Sports Med.2008;36(9): 1750-1762.[4] Berndt AL, Harty M.Transchondral fractures (osteochondritis dissecans) of the talus. J Bone Joint Surg Am. 1959;41-a: 988-1020.[5] Cheng JC, Ferkel RD. The role of arthroscopy in ankle and subtalar degenerative joint disease. Clin Orthop Relat Res. 1998; (349): 65-72.[6] Campbell CJ, Ranawat CS.Osteochondritis dissecans: the question of etiology. J Trauma.1966; 6(2): 201-221.[7] Prado MP, Kennedy JG, Raduan F,et al.Diagnosis and treatment of osteochondral lesions of the ankle: current concepts. Rev Bras Ortop. 2016;51(5): 489-500.[8] Flick AB, Gould N.Osteochondritis dissecans of the talus (transchondral fractures of the talus): review of the literature and new surgical approach for medial dome lesions. Foot Ankle.1985; 5(4): 165-185.[9] van Bergen CJ, Gerards RM, Opdam KT, et al.Diagnosing, planning and evaluating osteochondral ankle defects with imaging modalities. World J Orthop.2015; 6(11): 944-953.[10] Donnenwerth MP, Roukis TS.Outcome of arthroscopic debridement and microfracture as the primary treatment for osteochondral lesions of the talar dome. Arthroscopy.2012;28(12): 1902-1907.[11] Kirschke JS, Braun S, Baum T, et al., Diagnostic Value of CT Arthrography for Evaluation of Osteochondral Lesions at the Ankle. Biomed Res Int.2016;2016: 3594253.[12] Weber MA, Wünnemann F, Jungmann PM,et al.Modern Cartilage Imaging of the Ankle. Rofo.2017;189(10): 945-956.[13] Albano D, Martinelli N, Bianchi A,et al.Evaluation of reproducibility of the MOCART score in patients with osteochondral lesions of the talus repaired using the autologous matrix-induced chondrogenesis technique. Radiol Med.2017;122(12):909-917.[14] Van Ginckel A, Witvrouw EE.In vivo deformation of thin cartilage layers: Feasibility and applicability of T2* mapping. J Orthop Res. 2016;34(5): 771-778.[15] Tao H, Hu Y, Qiao Y, et al.T2 -Mapping evaluation of early cartilage alteration of talus for chronic lateral ankle instability with isolated anterior talofibular ligament tear or combined with calcaneofibular ligament tear. J Magn Reson Imaging.2018; 47(1):69-77.[16] Kim HS, Yoon YC.Comparison of T2 Relaxation Values in Subtalar Cartilage between Patients with Lateral Instability of the Ankle Joint and Healthy Volunteers. Eur Radiol.2018;28(10): 4151-4162.[17] Elias I, Jung JW, Raikin SM, et al.Osteochondral lesions of the talus: change in MRI findings over time in talar lesions without operative intervention and implications for staging systems. Foot Ankle Int. 2006;27(3):157-166.[18] Anderson IF, Crichton KJ, Grattan-Smith T, et al.Osteochondral fractures of the dome of the talus. J Bone Joint Surg Am.1989; 71(8): 1143-1152.[19] Cheng MR. Ferkel, and G. Applegate. Osteochondral lesions of the talus: a radiologic and surgical comparison. in Oral paper presented at the Annual Meeting of the Academy of Orthopaedic Surgeons (AAOS) New Orleans. 1995.[20] Huey DJ, Hu JC, Athanasiou KA.Unlike bone, cartilage regeneration remains elusive. Science.2012; 338(6109):917-921.[21] Verhagen RA, Struijs PA, Bossuyt PM,et al.Systematic review of treatment strategies for osteochondral defects of the talar dome. Foot Ankle Clin.2003;8(2): 233-242, viii-ix.[22] Klammer G, Maquieira GJ, Spahn S, et al.Natural history of nonoperatively treated osteochondral lesions of the talus. Foot Ankle Int.2015; 36(1): 24-31.[23] Murawski CD, Kennedy JG.Operative treatment of osteochondral lesions of the talus. J Bone Joint Surg Am. 2013;95(11): 1045-1054.[24] Grambart ST.Arthroscopic Management of Osteochondral Lesions of the Talus. Clin Podiatr Med Surg.2016; 33(4): 521-530.[25] Polat G, Er?en A, Erdil ME, et al.Long-term results of microfracture in the treatment of talus osteochondral lesions. Knee Surg Sports Traumatol Arthrosc.2016; 24(4): 1299-1303.[26] Choi WJ, Park KK, Kim BS, et al.Osteochondral lesion of the talus: is there a critical defect size for poor outcome? Am J Sports Med. 2009; 37(10): 1974-1980.[27] Shimozono Y, Coale M, Yasui Y, et al.Subchondral Bone Degradation After Microfracture for Osteochondral Lesions of the Talus: An MRI Analysis. Am J Sports Med.2018;46(3):642-648.[28] Lee KB, Park HW, Cho HJ, et al.Comparison of Arthroscopic Microfracture for Osteochondral Lesions of the Talus With and Without Subchondral Cyst. Am J Sports Med.2015;43(8): 1951-1956.[29] Kumai T, Takakura Y, Kitada C,et al.Fixation of osteochondral lesions of the talus using cortical bone pegs. J Bone Joint Surg Br.2002; 84(3): 369-374.[30] Shank JR, Benirschke SK, Swords MP.Treatment of Peripheral Talus Fractures. Foot Ankle Clin.2017;22(1): 181-192.[31] Ng A, Bernhard K. Osteochondral Autograft and Allograft Transplantation in the Talus. Clin Podiatr Med Surg. 2017; 34(4): 461-469.[32] Zhu Y, Xu X.Osteochondral Autograft Transfer Combined With Cancellous Allografts for Large Cystic Osteochondral Defect of the Talus. Foot Ankle Int.2016; 37(10): 1113-1118.[33] Leumann A, Horisberger M, Buettner O, et al.Medial malleolar osteotomy for the treatment of talar osteochondral lesions: anatomical and morbidity considerations. Knee Surg Sports Traumatol Arthrosc. 2016;24(7): 2133-2139.[34] Hannon CP, Smyth NA, Murawski CD, et al.Osteochondral lesions of the talus: aspects of current management. Bone Joint J.2014; 96-b(2): 164-171.[35] Henak CR, Ross KA, Bonnevie ED, et al.Human talar and femoral cartilage have distinct mechanical properties near the articular surface. J Biomech.2016;49(14):3320-3327.[36] Juras V, Zbýň Š, Mlynarik V, et al.The compositional difference between ankle and knee cartilage demonstrated by T2 mapping at 7 Tesla MR. Eur J Radiol.2016;85(4):771-777.[37] Hangody L.The mosaicplasty technique for osteochondral lesions of the talus. Foot Ankle Clin.2003;8(2): 259-273.[38] Hangody L, Kish G, Kárpáti Z, et al.Treatment of osteochondritis dissecans of the talus: use of the mosaicplasty technique--a preliminary report. Foot Ankle Int.1997;18(10): 628-634.[39] Sammarco GJ, Makwana NK.Treatment of talar osteochondral lesions using local osteochondral graft. Foot Ankle Int.2002;23(8): 693-698.[40] Flynn S, Ross KA, Hannon CP, et al. Autologous Osteochondral Transplantation for Osteochondral Lesions of the Talus. Foot Ankle Int.2016; 37(4):363-372.[41] Kubosch EJ, Erdle B, Izadpanah K, et al.Clinical outcome and T2 assessment following autologous matrix-induced chondrogenesis in osteochondral lesions of the talus. Int Orthop.2016; 40(1): 65-71.[42] Paul J, Sagstetter A, Kriner M, et al. Donor-site morbidity after osteochondral autologous transplantation for lesions of the talus. J Bone Joint Surg Am.2009;91(7): 1683-1688.[43] Gross AE, Agnidis Z, Hutchison CR.Osteochondral defects of the talus treated with fresh osteochondral allograft transplantation. Foot Ankle Int. 2001;22(5): 385-391.[44] Adams SB, Dekker TJ, Schiff AP, et al.Prospective Evaluation of Structural Allograft Transplantation for Osteochondral Lesions of the Talar Shoulder. Foot Ankle Int.2018;39(1): 28-34.[45] Ahmad J, Maltenfort M. Arthroscopic Treatment of Osteochondral Lesions of the Talus With Allograft Cartilage Matrix. Foot Ankle Int. 2017; 38(8): 855-862.[46] Lanham NS, Carroll JJ, Cooper MT, et al.A Comparison of Outcomes of Particulated Juvenile Articular Cartilage and Bone Marrow Aspirate Concentrate for Articular Cartilage Lesions of the Talus. Foot Ankle Spec.2017; 10(4):315-321.[47] Shimozono Y, Yasui Y, Ross AW, et al.Scaffolds based therapy for osteochondral lesions of the talus: A systematic review. World J Orthop.2017; 8(10): 798-808.[48] Gao F, Chen N, Sun W, et al.Combined Therapy with Shock Wave and Retrograde Bone Marrow-Derived Cell Transplantation for Osteochondral Lesions of the Talus. Sci Rep.2017; 7(1): 2106.[49] Shang XL, Tao HY, Chen SY, et al.Clinical and MRI outcomes of HA injection following arthroscopic microfracture for osteochondral lesions of the talus. Knee Surg Sports Traumatol Arthrosc.2016;24(4): 1243-1249.[50] Chahla J, Cinque ME, Shon JM,et al.Bone marrow aspirate concentrate for the treatment of osteochondral lesions of the talus: a systematic review of outcomes. J Exp Orthop.2016; 3(1): 33.[51] Tahta M, Akkaya M, Gursoy S, et al. Arthroscopic treatment of osteochondral lesions of the talus: Nanofracture versus hyaluronic acid-based cell-free scaffold with concentration of autologous bone marrow aspirate. J Orthop Surg (Hong Kong).2017;25(2): 2309499017717870.[52] Desando G, Bartolotti I, Vannini F, et al.Repair Potential of Matrix-Induced Bone Marrow Aspirate Concentrate and Matrix-Induced Autologous Chondrocyte Implantation for Talar Osteochondral Repair: Patterns of Some Catabolic, Inflammatory, and Pain Mediators. Cartilage.2017;8(1):50-60.[53] Kreulen C, Giza E, Walton J, et al.Seven-Year Follow-up of Matrix-Induced Autologous Implantation in Talus Articular Defects. Foot Ankle Spec.2018;11(2): 133-137.[54] Campagnoli C, Roberts IA, Kumar S,et al.Identification of mesenchymal stem/progenitor cells in human first-trimester fetal blood, liver, and bone marrow. Blood.2001;98(8): 2396-2402.[55] Bougioukli S, Sugiyama O, Pannell W, et al.Gene Therapy for Bone Repair Using Human Cells: Superior Osteogenic Potential of Bone Morphogenetic Protein 2-Transduced Mesenchymal Stem Cells Derived from Adipose Tissue Compared to Bone Marrow. Human Gene Therapy.2018; 29(4): 507-519.[56] Branly T, Bertoni L, Contentin R, et al.Characterization and use of Equine Bone Marrow Mesenchymal Stem Cells in Equine Cartilage Engineering. Study of their Hyaline Cartilage Forming Potential when Cultured under Hypoxia within a Biomaterial in the Presence of BMP-2 and TGF-beta 1. Stem Cell Rev.2017; 13(5): 611-630.[57] Kondo S, Muneta T, Nakagawa Y, et al.Transplantation of Autologous Synovial Mesenchymal Stem Cells Promotes Meniscus Regeneration in Aged Primates. J Orthop Res. 2017;35(6):1274-1282.[58] Ashton BA, Allen TD, Howlett CR,et al.Formation of bone and cartilage by marrow stromal cells in diffusion chambers in vivo. Clin Orthop Relat Res. 1980;(151):294-307.[59] Gao F, Chiu SM, Motan DA, et al.Mesenchymal stem cells and immunomodulation: current status and future prospects. Cell Death Dis. 2016;7:e2062. [60] Aggarwal S, Pittenger MF.Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood. 2005;105(4): 1815-1822.[61] Vannini F, Cavallo M, Ramponi L,et al.Return to Sports After Bone Marrow-Derived Cell Transplantation for Osteochondral Lesions of the Talus. Cartilage.2017;8(1): 80-87.[62] Mehrali M, Thakur A, Pennisi CP, et al.Nanoreinforced Hydrogels for Tissue Engineering: Biomaterials that are Compatible with Load-Bearing and Electroactive Tissues. Advanced Materials. 2017; 29(8): 26.[63] Yang X, Lu Z, Wu H, et al.Collagen-alginate as bioink for three-dimensional (3D) cell printing based cartilage tissue engineering. Mater Sci Eng C Mater Biol Appl. 2018;83:195-201.[64] Dai Y, Shen T, Ma L, et al.Regeneration of osteochondral defects in vivo by a cell-free cylindrical poly(lactide-co-glycolide) scaffold with a radially oriented microstructure. J Tissue Eng Regen Med. 2018;12(3): e1647-e1661. [65] Li X, Ding J, Zhang Z, et al.Kartogenin-incorporated thermogel supports stem cells for significant cartilage regeneration. ACS Appl Mater Interfaces. 2016;8(8):5148-5159.[66] Kanatl? U, Eren A, Eren TK, et al.Single-Step Arthroscopic Repair With Cell-Free Polymer-Based Scaffold in Osteochondral Lesions of the Talus: Clinical and Radiological Results. Arthroscopy.2017; 33(9): 1718-1726.[67] Legendre F, Ollitrault D, Gomez-Leduc T, et al.Enhanced chondrogenesis of bone marrow-derived stem cells by using a combinatory cell therapy strategy with BMP-2/TGF-beta1, hypoxia, and COL1A1/HtrA1 siRNAs. Sci Rep, 2017;7(1): 3406.[68] Crecente-Campo J, Borrajo E, Vidal A, et al. New scaffolds encapsulating TGF-beta3/BMP-7 combinations driving strong chondrogenic differentiation. Eur J Pharm Biopharm.2017; 114: 69-78.[69] Gugjoo MB, Amarpal, Abdelbaset-Ismail A, et al.Mesenchymal stem cells with IGF-1 and TGF- beta1 in laminin gel for osteochondral defects in rabbits. Biomed Pharmacother.2017; 93:1165-1174.[70] Johnson K, Zhu S, Tremblay MS,et al.A stem cell-based approach to cartilage repair. Science.2012. 336(6082): 717-721.[71] Christensen BB, Foldager CB, Jensen J, et al.Poor osteochondral repair by a biomimetic collagen scaffold: 1- to 3-year clinical and radiological follow-up. Knee Surg Sports Traumatol Arthrosc.2016; 24(7): 2380-2387.[72] Kaipel M, Schreiner M, Kellner R, et al.Beneficial clinical effects but limited tissue quality following osteochondral repair with a cell-free multilayered nano-composite scaffold in the talus. Foot Ankle Surg. 2017;23(4):302-306. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Wu Xun, Meng Juanhong, Zhang Jianyun, Wang Liang. Concentrated growth factors in the repair of a full-thickness condylar cartilage defect in a rabbit [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1166-1171. |
| [3] | Li Jiacheng, Liang Xuezhen, Liu Jinbao, Xu Bo, Li Gang. Differential mRNA expression profile and competitive endogenous RNA regulatory network in osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1212-1217. |
| [4] | Geng Qiudong, Ge Haiya, Wang Heming, Li Nan. Role and mechanism of Guilu Erxianjiao in treatment of osteoarthritis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1229-1236. |
| [5] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [6] | He Xiangzhong, Chen Haiyun, Liu Jun, Lü Yang, Pan Jianke, Yang Wenbin, He Jingwen, Huang Junhan. Platelet-rich plasma combined with microfracture versus microfracture in the treatment of knee cartilage lesions: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 964-969. |
| [7] | Liu Xin, Yan Feihua, Hong Kunhao. Delaying cartilage degeneration by regulating the expression of aquaporins in rats with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 668-673. |
| [8] | Deng Zhenhan, Huang Yong, Xiao Lulu, Chen Yulin, Zhu Weimin, Lu Wei, Wang Daping. Role and application of bone morphogenetic proteins in articular cartilage regeneration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 798-806. |
| [9] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [10] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [11] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [12] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [13] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [14] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [15] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

