Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (11): 1761-1766.doi: 10.3969/j.issn.2095-4344.1096
Previous Articles Next Articles
Myokine, adipo-myokine and adipokine: recognition of the function of skeletal muscle and adipose tissue
Liao Shuaixiong1,Zhang Guodong2,Song Gang1,3
- (1College of Physical Education, Southwest University, Chongqing 400715, China; 2State Key Laboratory of Physical Fitness Evaluation and Sports Function Monitoring, Chongqing 400715, China; 3Institute for Sports Rehabilitation of Southwest University, Chongqing 400715, China)
-
Received:2018-10-07Online:2019-04-18Published:2021-04-28 -
Contact:Song Gang, PhD, Professor, College of Physical Education, Southwest University, Chongqing 400715, China; Institute for Sports Rehabilitation of Southwest University, Chongqing 400715, China -
About author:Liao Shuaixiong, Master candidate, College of Physical Education, Southwest University, Chongqing 400715, China -
Supported by:the Special Physical Education Research, No. SWU1709116 (to SG)
CLC Number:
Cite this article
Liao Shuaixiong, Zhang Guodong, Song Gang, . Myokine, adipo-myokine and adipokine: recognition of the function of skeletal muscle and adipose tissue[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(11): 1761-1766.
share this article

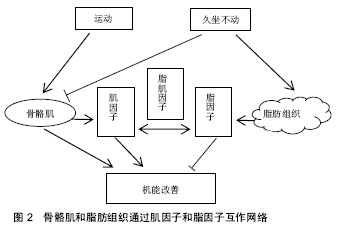
2.2 骨骼肌和脂肪组织生物学功能 2.2.1 骨骼肌和脂肪组织内分泌功能的认识 人们对骨骼肌和脂肪组织功能的认识是一个逐步深入的过程。最初大家认为骨骼肌只是运动器官,或者只被当作身体内最大的能量产生和消耗的器官,在姿势控制、运动和能量控制中起到关键作用;脂肪组织最初被认为是一种惰性的能量储存组织。近20年来,骨骼肌也被认为是最大的内分泌器官,对其内分泌功能的研究引起了广泛的关注,肌肉收缩释放的肌因子起到改善机体健康的作用[7]。骨骼肌和脂肪组织释放出数以百计的分泌蛋白,很多的肌因子和脂因子在不同的组织中都有表达和分泌。这些物质对人体大部分器官具有调节作用。缺乏体力活动会导致内脏脂肪积累,从而激活炎症信号途径,促进胰岛素抵抗、动脉粥样硬化、神经退行性疾病和肿瘤生长,从而引起一系列慢性病。肌因子是联系器官间复杂网络的一部分,它能够通过脏器间通信产生自分泌、旁分泌和内分泌的作用[7]。其主要功能是对抗慢性和炎症性疾病,如2型糖尿病,胰岛素抵抗,代谢综合征等。肌因子的生物学效应主要表现为在运动中起到改善功能和促进健康的作用。目前,对这一领域的研究方兴未艾,为体育科学的基础学科提供了一个新的研究领域。尤其是关于肌肉如何与其他器官沟通,如与脂肪组织、肝脏和胰腺等,以及身体活动如何介导众多因子对代谢综合征的影响等。 长期运动锻炼具有多种健康益处,骨骼肌通过分泌肌因子,对机体产生良性作用,其效果可以抵消脂肪组织分泌的成炎性细胞因子对机体的损伤[8]。肌因子能对抗因身体活动不足所增加的多种疾病的风险,如:2型糖尿病,心血管疾病,结肠癌,乳腺癌和骨质疏松症等[9-11]。 总之,< 3 h的单次运动,肌因子和脂-肌因子主要由骨骼肌分泌,并产生内分泌、自分泌或旁分泌作用。分泌的肌因子像白细胞介素6、肌肉生长抑制素、白细胞介素7、核心蛋白多糖和白血病抑制因子等细胞因子参与肌肉损伤的修复,促进肌肉的生成,提高胰岛素敏感性,增加内皮的功能,促进血管生成,加速脂肪组织分解并减少内脏脂肪,有助于肝糖释放。大于8周有规律的运动训练有助于降低脂肪组织促炎性细胞因子水平,促进全身胰岛素敏感性,减少内脏脂肪,降低炎症[12]。 2.2.2 骨骼肌和脂肪组织内分泌功能的再认识——“阴阳”学说 阴阳学说认为:对于人体具有推进、温煦、兴奋等作用的物质和功能统归于“阳”,对于人体具有凝聚、滋润、抑制等作用的物质和功能归于“阴”。“阴阳”是相互关联的一种事物或是一个事物的两个方面。阴阳学说认为:阴阳之间的对立制约、互根互用,并不是处于静止不变的状态,而是始终处在不断的运动变化之中[13]。 肌因子(来自骨骼肌)和脂因子(来自脂肪组织)的相互作用似乎可以用中国传统的“阴阳”平衡来解释。比如在肥胖患者中,脂肪组织分泌的某些脂因子,能促进慢性炎症内环境的形成,加快病理形成过程,此为“阴”。而运动时骨骼肌能够产生一些肌因子,这样的肌因子可能抵消炎症因子的有害影响[14],此为“阳”。将运动训练小鼠的皮下白色脂肪组织(subcutaneous white adipose tissue,scWAT)移植入久坐的小鼠体内,能改善小鼠骨骼肌摄取葡萄糖的能力,提示运动训练scWAT以内分泌的方式改善骨骼肌功能,说明脂肪组织能在运动训练的干预下产生“阳”性因子,或减少“阴”性因子。动物实验表明,一次剧烈运动可以改变血中的脂因子。这提示,脂肪组织分泌脂因子作为正/负反馈信号,是肌细胞和脂肪细胞相互作用的开关,一旦肌因子和脂因子分泌失调,会产生全身性的胰岛素抵抗[15]。 多种器官会出现因运动而产生适应,其中的骨骼肌和脂肪组织相互作用,形成网络,起到改善机体新陈代谢的作用。如骨骼肌适应包括改善葡萄糖摄取,增加葡萄糖转运蛋白4型(GLUT4)转位和表达,增强线粒体活性,增加脂肪作为燃料,增加肌因子进入血液循环 等[16-18]。肌因子和脂因子通过自分泌、旁分泌、内分泌的作用,调节机体的糖、脂代谢和能量平衡。 如图2所示,骨骼肌通过肌因子,脂肪组织通过脂因子对机体器官产生影响,形成互相作用的网络,包括骨骼肌、脂肪组织相互的作用;在网络中,“阳”是骨骼肌分泌肌因子,“阴”为脂肪组织分泌的脂因子。有些肌因子也由脂肪组织分泌,称为脂-肌因子。肌因子和脂因子体现为矛盾统一的两方面,通过脂-肌因子相互转化。运动激发骨骼肌释放肌因子起到促进健康的作用,而与久坐不动引发脂肪组织所分泌的脂因子相互拮抗。"
| [1] Ruderman NB, Keller C, Richard AM, et al. Interleukin-6 regulation of AMP-activated protein kinase. Potential role in the systemic response to exercise and prevention of the metabolic syndrome. Diabetes.2006; 55 Suppl 2:S48-S54. [2] Wang J, Liu R, Hawkins M, et al. A nutrient-sensing pathway regulates leptin gene expression in muscle and fat. Nature.1998;393(6686): 684-688. [3] Hecksteden A, Wegmann M, Steffen A, et al. Irisin and exercise training in humans - results from a randomized controlled training trial. BMC Med.2013; 11:235. [4] Pedersen BK, Steensberg A, Fischer C, et al. Searching for the exercise factor: is IL-6 a candidate?. J Muscle Res Cell Motil.2003; 24(2-3):113-119. [5] Cook KS, Min HY, Johnson D, et al. Adipsin: a circulating serine protease homolog secreted by adipose tissue and sciatic nerve. Science.1987;237(4813):402-405. [6] Scherer PE. Adipose tissue: from lipid storage compartment to endocrine organ. Diabetes.2006; 55(6):1537-1545. [7] Eckardt K, Gorgens SW, Raschke S, et al. Myokines in insulin resistance and type 2 diabetes. Diabetologia. 2014;57(6): 1087-1099. [8] Pedersen BK. Muscle as a Secretory Organ. Compr Physiol. 2013;3(3):1337-1362. [9] Wolin KY, Yan Y, Colditz GA, et al. Physical activity and colon cancer prevention: a meta-analysis. Br J Cancer. 2009;100(4): 611-616. [10] Monninkhof EM, Elias SG, Vlems FA, et al. Physical activity and breast cancer - A systematic review. Epidemiology. 2007;18(1): 137-157. [11] Borer KT. Physical activity in the prevention and amelioration of osteoporosis in women - Interaction of mechanical, hormonal and dietary factors. Sports Med. 2005;35(9):779-830. [12] Goergens SW, Eckardt K, Jensen J, et al. Exercise and Regulation of Adipokine and Myokine Production[M]//Bouchard C. Progress in Molecular Biology and Translational Science. 2015:313-336. [13] 陈明.读《素问•阴阳应象大论》札记五则[J].中医函授通讯,1995,(2): 2-3.[14] Pedersen BK, Febbraio MA. Muscles, exercise and obesity: skeletal muscle as a secretory organ. Nat Rev Endocrinol. 2012;8(8):457-465. [15] Rodriguez A, Ezquerro S, Mendez-Gimenez L, et al. Revisiting the adipocyte: a model for integration of cytokine signaling in the regulation of energy metabolism. Am J Physiol Endocrinol Metab. 2015;309(8):E691-714. [16] Richter EA, Hargreaves M. Exercise, GLUT4, and skeletal muscle glucose uptake.Physiol Rev. 2013;93(3):993-1017. [17] Little JP, Gillen JB, Percival ME, et al. Low-volume high-intensity interval training reduces hyperglycemia and increases muscle mitochondrial capacity in patients with type 2 diabetes. J Appl Physiol (1985). 2011;111(6):1554-1560. [18] Jeukendrup AE. Regulation of fat metabolism in skeletal muscle[M]//Klimes I, Sebokova E, Howard B V, et al. Annals of the New York Academy of Sciences. 2002:217-235. [19] Hoene M, Weigert C. The role of interleukin-6 in insulin resistance, body fat distribution and energy balance. Obes Rev. 2008;9(1):20-29. [20] Pedersen BK, Steensberg A, Fischer C, et al. The metabolic role of IL-6 produced during exercise: is IL-6 an exercise factor? . Proc Nutr Soc.2004;63(2):263-267. [21] Carey AL, Steinberg GR, Macaulay SL, et al. Interleukin-6 increases insulin-stimulated glucose disposal in humans and glucose uptake and fatty acid oxidation in vitro via AMP-activated protein kinase. Diabetes.2006;55(10):2688-2697. [22] Bruce CR, Dyck DJ. Cytokine regulation of skeletal muscle fatty acid metabolism: effect of interleukin-6 and tumor necrosis factor- alpha. Am J Physiol Endocrinol Metab.2004;287(4):E616-E621. [23] Kelly M, Gauthier M, Saha AK, et al. Activation of AMP-Activated Protein Kinase by Interleukin-6 in Rat Skeletal Muscle Association With Changes in cAMP, Energy State, and Endogenous Fuel Mobilization. Diabetes. 2009;58(9):1953-1960. [24] Minokoshi Y, Kim YB, Peroni OD, et al. Leptin stimulates fatty-acid oxidation by activating AMP-activated protein kinase. Nature.2002; 415(6869):339-343. [25] Nehlsen-Cannarella SL, Fagoaga OR, Nieman DC, et al. Carbohydrate and the cytokine response to 2. 5 h of running. J Appl Physiol (1985).1997;82(5):1662-1667. [26] Starkie RL, Angus DJ, Rolland J, et al. Effect of prolonged, submaximal exercise and carbohydrate ingestion on monocyte intracellular cytokine production in humans. J Physiol.2000;528(Pt 3):647-655. [27] Pedersen BK, Febbraio MA. Muscle as an endocrine organ: Focus on muscle-derived interleukin-6. Physiol Rev. 2008;88(4): 1379-1406. [28] Raschke S, Eckel J. Adipo-myokines: two sides of the same coin--mediators of inflammation and mediators of exercise. Mediators Inflamm.2013;2013:320724. [29] Serrano AL, Baeza-Raja B, Perdiguero E, et al. Interleukin-6 is an essential regulator of satellite cell-mediated skeletal muscle hypertrophy. Cell Metab. 2008;7(1):33-44. [30] Willoughby DS, McFarlin B, Bois C. Interleukin-6 expression after repeated bouts of eccentric exercise. Int J Sports Med.2003;24(1):15-21. [31] Steensberg A, Febbraio MA, Osada T, et al. Interleukin-6 production in contracting human skeletal muscle is influenced by pre-exercise muscle glycogen content. J Physiol. 2001;537(Pt 2):633-639. [32] Muenzberg H, Morrison CD. Structure, production and signaling of leptin. Metabolism. 2015;64(1):13-23. [33] Wolsk E, Mygind H, Grondahl TS, et al. Human skeletal muscle releases leptin in vivo. Cytokine. 2012;60(3):667-673. [34] Phillips MD, Patrizi RM, Cheek DJ, et al. Resistance training reduces subclinical inflammation in obese, postmenopausal women. Med Sci Sports Exerc.2012; 44(11):2099-2110. [35] Fatouros IG, Chatzinikolaou A, Tournis S, et al. Intensity of resistance exercise determines adipokine and resting energy expenditure responses in overweight elderly individuals. Diabetes Care.2009; 32(12):2161-2167. [36] Varady KA, Bhutani S, Church EC, et al. Adipokine responses to acute resistance exercise in trained and untrained men. Med Sci Sports Exerc.2010;42(3):456-462. [37] Polak J, Klimcakova E, Moro C, et al. Effect of aerobic training on plasma levels and subcutaneous abdominal adipose tissue gene expression of adiponectin, leptin, interleukin 6, and tumor necrosis factor alpha in obese women. Metabolism.2006; 55(10): 1375-1381. [38] Kondo T, Kobayashi I, Murakami M. Effect of exercise on circulating adipokine levels in obese young women. Endocr J.2006;53(2):189-195. [39] Haugen F, Norheim F, Lian H, et al. IL-7 is expressed and secreted by human skeletal muscle cells. Am J Physiol Cell Physiol.2010;298(4):C807-C816. [40] Andersson H, Bohn SK, Raastad T, et al. Differences in the inflammatory plasma cytokine response following two elite female soccer games separated by a 72-h recovery. Scand J Med Sci Sports.2010;20(5):740-747. [41] Kraemer WJ, Hatfield DL, Comstock BA, et al. Influence of HMB supplementation and resistance training on cytokine responses to resistance exercise. J Am Coll Nutr.2014;33(4):247-255. [42] Huang EJ, Reichardt LF. Neurotrophins: Roles in neuronal development and function. Annu Rev Neurosci. 2001;24:677-736. [43] Mattson MP. Energy Intake and Exercise as Determinants of Brain Health and Vulnerability to Injury and Disease. Cell Metab. 2012; 16(6):706-722. [44] Brenmoehl J, Albrecht E, Komolka K, et al. Irisin Is Elevated in Skeletal Muscle and Serum of Mice Immediately after Acute Exercise. Int J Biol Sci. 2014;10(3):338-349. [45] Bostrom P, Wu J, Jedrychowski MP, et al. A PGC1-alpha- dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature.2012;481(7382):463-468. [46] Wu Z, Puigserver P, Andersson U, et al. Mechanisms controlling mitochondrial biogenesis and respiration through the thermogenic coactivator PGC-1. Cell.1999; 98(1):115-124. [47] Jedrychowski MP, Wrann CD, Paulo JA, et al. Detection and Quantitation of Circulating Human Irisin by Tandem Mass Spectrometry. Cell Metab. 2015;22(4):734-740. [48] Huh JY, Panagiotou G, Mougios V, et al. FNDC5 and irisin in humans: I. Predictors of circulating concentrations in serum and plasma and II. mRNA expression and circulating concentrations in response to weight loss and exercise. Metabolism.2012;61(12):1725-1738. [49] Kurdiova T, Balaz M, Vician M, et al. Effects of obesity, diabetes and exercise on Fndc5 gene expression and irisin release in human skeletal muscle and adipose tissue: in vivo and in vitro studies. J Physiol.2014; 592(5):1091-1107. [50] Yamauchi T, Iwabu M, Okada-Iwabu M, et al. Adiponectin receptors: a review of their structure, function and how they work. Best Pract Res Clin Endocrinol Metab. 2014;28(1):15-23. [51] Han SH, Quon MJ, Kim J A, et al. Adiponectin and cardiovascular disease: response to therapeutic interventions. J Am Coll Cardiol. 2007;49(5):531-538. [52] Christiansen T, Bruun JM, Paulsen SK, et al. Acute exercise increases circulating inflammatory markers in overweight and obese compared with lean subjects. Eur J Appl Physiol.2013; 113(6):1635-1642. [53] Bobbert T, Wegewitz U, Brechtel L, et al. Adiponectin oligomers in human serum during acute and chronic exercise: relation to lipid metabolism and insulin sensitivity. Int J Sports Med.2007;28(1):1-8. [54] Bouassida A, Lakhdar N, Benaissa N, et al. Adiponectin responses to acute moderate and heavy exercises in overweight middle aged subjects. J Sports Med Phys Fitness. 2010;50(3): 330-335. [55] Ferguson MA, White L J, McCoy S, et al. Plasma adiponectin response to acute exercise in healthy subjects. Eur J Appl Physiol. 2004;91(2-3):324-329. [56] Jamurtas AZ, Theocharis V, Koukoulis G, et al. The effects of acute exercise on serum adiponectin and resistin levels and their relation to insulin sensitivity in overweight males. Eur J Appl Physiol.2006;97(1):122-126. [57] Saunders TJ, Palombella A, McGuire K A, et al. Acute exercise increases adiponectin levels in abdominally obese men. J Nutr Metab.2012;2012:148729. [58] Polak J, Klimcakova E, Moro C, et al. Effect of aerobic training on plasma levels and subcutaneous abdominal adipose tissue gene expression of adiponectin, leptin, interleukin 6, and tumor necrosis factor alpha in obese women. Metabolism.2006;55(10):1375-1381. [59] Tencerova M, Kracmerova J, Krauzova E, et al. Experimental hyperglycemia induces an increase of monocyte and T-lymphocyte content in adipose tissue of healthy obese women. PLoS One.2015;10(3):e122872. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Li Jun, Zuo Xinhui, Liu Xiaoyuan, Zhang Kai, Han Xiangzhen, He Huiyu, . Effect of over expression of miR-378a on osteogenic and vascular differentiation of bone marrow mesenchymal stem cell sheet [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(31): 4939-4944. |
| [11] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [12] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [13] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [14] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [15] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

