Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (29): 4736-4742.doi: 10.3969/j.issn.2095-4344.0634
Previous Articles Next Articles
Bone marrow mesenchymal stem cells in the treatment of spinal cord injury: research hotspots and directions
Lu Yu-bao1, Yang Yang1, Wang Yun-chang1, Chen Guo-hu2, Yuan Lin-hui1, Li Chun-yan1, Guan Xin1, Ma Zhan-jun1
- 1The Second Hospital of Lanzhou University, Lanzhou 730030, Gansu Province, China; 2The First Hospital of Lanzhou University, Lanzhou 730000, Gansu Province, China
-
Revised:2018-05-31Online:2018-10-18Published:2018-10-18 -
Contact:Ma Zhan-jun, the Second Hospital of Lanzhou University, Lanzhou 730030, Gansu Province, China -
About author:Lu Yu-bao, the Second Hospital of Lanzhou University, Lanzhou 730030, Gansu Province, China -
Supported by:the Scientific Culture Plan of the Second Hospital of Lanzhou University, No. CYXZ-09
CLC Number:
Cite this article
Lu Yu-bao, Yang Yang, Wang Yun-chang, Chen Guo-hu, Yuan Lin-hui, Li Chun-yan, Guan Xin, Ma Zhan-jun. Bone marrow mesenchymal stem cells in the treatment of spinal cord injury: research hotspots and directions[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(29): 4736-4742.
share this article

2.1 BMSCs的生物学效应 BMSCs在不同诱导条件下可分化为骨髓基质细胞、骨细胞、肌腱细胞和脂肪细胞等多种间充质细胞,并且大量的研究发现BMSCs还具有在不同诱导条件下分化为神经细胞和心肌细胞等实质性细胞的能力[2],自20世纪70年代首次报道以来,BMSCs一直是国内外各课题组的研究热点。与此同时,BMSCs还可以分泌白细胞介素、集落刺激因子、肿瘤坏死因子α、干细胞因子和神经生长因子等多种细胞因子并表达其受体[3]。根据相关研究结果,BMSCs可在体外环境中被诱导分化为神经元样细胞,在移植入体内后仍保持分化稳定性[4]。此外,Lee课题组[5]已成功在体外环境下将BMSCs诱导分化为不同亚型的神经胶质细胞和神经元。Satake等[6]在脊髓损伤模型大鼠脊髓中注射了荧光标记的BMSCs,发现大鼠在神经和运动功能方面均得到一定程度的改善。Ye课题组[7]在实验研究中发现,通过向脊髓损伤模型大鼠侧脑室注射经脑脊液诱导BMSCs产生的神经元样细胞(BMSC-Ns)干预8周后,注射BMSCs和BMSC-Ns均能使大鼠Ntf-3水平显着上调,并且2组大鼠后肢功能恢复情况均明显优于对照组。以上实验结果均表明,BMSCs在脊髓损伤的治疗过程中具有积极的作用和意义,有可能成为一种新的脊髓损伤治疗方法和策略。 2.2 BMSCs在脊髓损伤治疗中的作用机制 目前,BMSCs治疗脊髓损伤尚处于实验阶段,并未得到广泛的普及,但是随着动物实验和临床试验的不断研究,可以发现BMSCs在脊髓损伤治疗方面有着极为乐观的应用前景。但是,现阶段BMSCs治疗脊髓损伤的相关机制尚不明确,且多数研究者认为该机制是多方面的,目前大家较为认可的机制有以下几种。 2.2.1 轴突再生重建神经通路 大量实验证实,在脊髓损伤患者的损伤部位移植BMSCs后,可在一定条件下被诱导分化为星形胶质细胞和少突胶质细胞等神经胶质细胞及神经元,从而使脊髓损伤患者的神经通路得以重建[8-9]。与此同时,BMSCs还可以表达多种细胞黏附相关的细胞因子,如Ninjurin 1和2、Netrin 4、Robo 1和Robo 4等,这些细胞因子可以通过刺激神经元轴突再生及促进细胞迁移,从而在神经修复方面起着至关重要的作用。此外,有研究显示BMSCs可以分泌具有降解Ⅰ型胶原作用的基质金属蛋白酶1和具有促血管生长作用的基质金属蛋白酶2从而降低硫酸软骨素的浓度,来促进轴突生长和抑制瘢痕形成[10]。 2.2.2 神经营养作用 BMSCs还可以分泌干细胞因子、白细胞介素、粒细胞集落刺激因子、肿瘤坏死因子α、神经生长因子和血小板衍生生长因子等众多细胞因子并表达其相关受体,从而调节体内多种生理作用。研究显示,BMSCs可以分泌脑源性神经营养因子、神经生长因子和血管内皮生长因子等多种具有神经保护作用的营养因子,以促进损伤部位的神经重塑和血管再生,达到细胞修复的目的[11-12]。 2.2.3 抑制氧化应激,减轻炎症反应 BMSCs可以分泌具有抗氧化作用的细胞因子和促血管内皮生长的细胞因子[13],进一步通过自身分化为血管平滑肌细胞和内皮细胞来恢复受损细胞的线粒体而重塑损伤部位的血供,从而减轻脊髓损伤患者受损部位的炎症反应和氧化应激。 2.2.4 填充、替代作用 Zhang等[14]通过磁共振成像追踪技术证实,在脊髓损伤模型动物体内移植BMSCs后,BMSCs可迅速并准确地迁移至损伤坏死部位并诱导分化为神经元和星形胶质细胞替代受损组织而填充囊性区,有效减小神经空洞面积以阻止神经瘢痕的进一步扩大,为神经元轴突再生提供有利条件,促进脊髓损伤后的神经修复。 2.3 BMSCs诱导分化为神经元样细胞相关研究 BMSCs具有较强的可塑性和分化能力,可在不同微环境的诱导下增殖分化为多种细胞;由于取材方便以及易于培养和无伦理学问题,自体移植后无明显的免疫排斥反应,故而成为了脊髓损伤移植治疗中的优选材料。 2.3.1 机制方面研究 BMSCs来源于胚胎中胚层而神经元来源于胚胎外胚层,因此这种跨胚层分化的方式被定义为“转分化”。目前,BMSCs向神经元诱导分化的相关研究较多,但仍然处于初步探索阶段,未出现突破性研究进展。Woodbury等[15]于2000年首次在体外环境下将人与大鼠的BMSCs通过二甲基亚砜、4-羟基茴香醚以及β-巯基乙醇成功诱导分化为神经胶质细胞和神经元。研究结果显示,BMSCs诱导分化产生的神经元样细胞可表达神经元特异性蛋白烯醇化酶、神经生长因子受体Trk A、神经丝和巢蛋白,其中神经元特异性蛋白烯醇化酶和神经丝表达率高达80%,部分细胞在形态学上表现出由双极向多极分化的趋势,并且逐渐出现了次级分支结构和锥样生长末端。与此同时,少部分表达神经元特异性蛋白烯醇化酶的细胞经有丝分裂形成成熟神经元并明显表达神经元特异性核内抗原(NeuN)。该方法自发现以来已经被广泛应用于体外实验中将BMSCs向神经元的诱导分化。研究发现,神经营养因子可以结合细胞膜上的高亲和力酪氨酸激酶受体和低亲和力的P75受体来激活酪氨酸蛋白激酶,进而提高磷酯酶Cγ、PI3激酶和连接蛋白质Shc的活性,并通过Ras-MAP激酶途径调节基因的表达而诱导细胞分化。 此外,Rivera等[16]分别从海马、小脑皮质及大脑皮质提取了可溶性神经营养因子用于BMSCs的体外诱导分化实验,结果显示与海马中提取的神经营养因子共培养的BMSCs分化程度最高,不仅发生了形态学改变而且表达GAP-43、NF和nestin等蛋白。随后Jiang等[17]将BMSCs与脑源性神经营养因子、表皮生长因子和成纤维细胞生长因子共培养6 d后,通过免疫荧光技术和RT-PCR方法发现66%的BMSCs有树突形成,并表现出神经元的特征,同时还检测出大部分神经元标记物,且这些标记物分别属于神经元的不同分化阶段。 以上研究结果均表明BMSCs可在体外环境下被诱导分化为具有神经元形态学特征和标记物的细胞,但是并未发现这些细胞具有神经元生理功能的相关证据,因此仅可将这些细胞定义为“神经元样细胞”,而非真正意义上的神经元。 2.3.2 基因方面研究 Blondheim等[18]发现人类间充质干细胞可表达神经元的部分基因,但是表达水平较低,其中EVI-1、GAG和FKHR-1等8个基因在多巴胺能神经系统中发挥着重要作用,因此间充质干细胞通过诱导可成功分化为神经元和神经胶质细胞。杨晓苏等[19]利用碱性纤维生长因子将BMSCs进行预诱导后再用二甲基亚砜和丁基羟基茴香醚进行诱导,通过免疫组化发现BMSCs高表达神经元特异性烯醇化酶、神经丝和运动神经元生存基因,而RT-PCR显示运动神经元生存基因在BMSCs中也有所表达,并且运动神经元生存基因在诱导后出现过表达现象。 Liu等[20]利用慢病毒转染技术使BMSCs过表达脑源性神经营养因子,进行超顺磁性氧化铁纳米粒子标记,发现BMSCs成功分化为神经元样细胞,并高表达脑源性神经营养因子和神经元标志蛋白,这说明基因修饰技术可以使BMSCs分化为具有神经元功能的神经元样细胞。 2.4 BMSCs诱导分化在脊髓损伤治疗中的应用 Moviglia等[21]研究发现在脊髓损伤患者内,自身反应性T细胞可将BMSCs诱导分化为神经干细胞从而促进损伤部位的功能修复。Coronel课题组[22]将标记后的BMSCs注射入脊髓损伤大鼠模型中,免疫组化结果显示BMSCs首先迁移到脊髓损伤部位。Hellmann等[23]在脑缺血、脑损伤和脊髓损伤动物模型移植BMSCs后,发现BMSCs可增强损伤部位的神经营养作用,从而提高神经元的可塑性;随后的星形孢菌素诱导BMSCs凋亡实验中发现诱导剂可增强Erk1/2和Akt磷酸化水平,而使用MEK1抑制剂PD98059和PI3-K抑制剂Ly294002后,神经保护出现消除现象,进而导致了BMSCs的凋亡。 2.5 BMSCs联合治疗在脊髓损伤中的应用 由于通过BMSCs单纯诱导分化在脊髓损伤患者的治疗预后中效果并不理想,因此BMSCs与其他细胞、细胞因子等多种方法的联合治疗方案逐渐成为了新的研究方向,研究表明不同的BMSCs联合治疗方案在脊髓损伤的修复中发挥着不同程度的积极作用,见表1。"

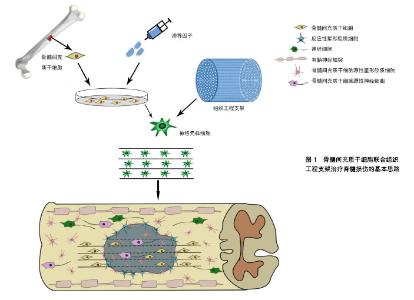
2.5.1 BMSCs联合其他细胞方案 嗅鞘细胞是一类来源于嗅基板的神经胶质细胞,其功能介于许旺细胞和少突胶质细胞之间,终生具有神经再生功能,同时还能够释放多种神经营养因子和神经黏附分子,从而在神经营养、成鞘和抑制胶质增生及瘢痕形成等生理活动中起到重要作用,因此嗅鞘细胞在脊髓损伤治疗中发挥着广阔的应用前景。Wu等[24]研究结果表明,在脊髓损伤大鼠模型体内联合移植BMSCs和嗅鞘细胞后,联合移植组大鼠不仅在后肢功能恢复等方面显著高于单纯BMSCs移植组,并且联合移植组细胞凋亡率明显低于单纯BMSCs移植组。 许旺细胞具有形成有髓神经纤维髓鞘的功能,并可吞噬细胞残渣进而为神经元再生提供空间。与此同时,许旺细胞还可以释放多种细胞因子,从而在神经元再生和抑制神经瘢痕形成中发挥着重要作用。研究结果显示,BMSCs联合许旺细胞的治疗方案在脊髓损伤的修复过程中具有协同作用,且修复效果优于单一BMSCs移植。 2.5.2 BMSCs联合神经营养因子 神经营养因子是一族由机体自身通过内分泌的方式作用于神经系统,并在神经元的发育、生长和分化中起到重要作用的蛋白质,其主要包括神经生长因子、脑源性神经营养因子、胶质细胞源性神经营养因子、神经营养因子3和神经营养因子4以及神经营养因子5,均具有营养神经元和促进轴突生成的功能。 Karimi-Abdolezaee等[25]将BMSCs与神经生长因子联合植入脊髓损伤部位后,发现神经生长因子可促进BMSCs向神经元样细胞分化并促进轴突生成,同时发现大鼠脊髓空洞面积也有明显缩小的现象。步星耀等[26]将BMSCs移植入91例脊髓损伤患者体内后通过肌肉注射的方式给予胶质细胞源性神经营养因子辅助治疗,除1例严重复合伤患者经积极抢救无效死亡外,其余90例患者均在运动功能、感觉功能、神经电生理和影像学方面有明显改善,并治愈出院。Shahrezaie等[27]通过腺病毒转染法将胶质细胞源性神经营养因子基因转染到BMSCs后移植入脊髓损伤大鼠模型体内,结果显示大鼠后肢运动功能评分和轴突再生数量高于对照组,说明胶质细胞源性神经营养因子基因转染的BMSCs在脊髓损伤后神经修复中具有更显著的疗效。 2.5.3 BMSCs联合药物 炎症因子和轴突再生抑制分子的释放、神经瘢痕的形成以及免疫细胞的活化是阻碍脊髓损伤治疗的原因。硫酸软骨素酶ABC是一种由星形胶质细胞产生的酶类物质,研究发现其主要通过调节创伤后炎症和氧化应答来发挥其神经保护作用,并对中枢神经系统损伤后轴突再生与神经功能恢复起到促进作用[28]。因此,利用BMSCs与硫酸软骨素酶ABC的联合治疗方案,脊髓损伤模型大鼠运动功能恢复水平明显优于对照组,且胶质瘢痕与脊髓空洞面积显著减小。 米诺环素(MH)是一种半合成的四环素衍生物,是一种在临床上广泛使用的抗生素和抗炎药物,同时米诺环素被证实具有明显的神经保护作用,其主要机制是通过抗炎、抗氧化和抗凋亡来有效缓解脊髓损伤后的级联炎症反应[29]。Chen等[30]将米诺环素与BMSCs共同植入SD大鼠脊髓损伤部位后,结果显示BMSCs联合米诺环素在脊髓损伤大鼠模型抗炎、激活VEGF、BDNF和proNGF表达、抗凋亡、抑制p38MAPK依赖性以及抑制p75NTR和RhoA的功能改善中具有积极作用。 2.5.4 BMSCs联合组织工程支架 脊髓损伤后组织细胞坏死形成的脊髓空洞和胶质细胞分泌神经胶质形成的胶质瘢痕所共同构成的物理、化学屏障是阻碍脊髓损伤后神经修复的主要原因,而单纯的BMSCs移植存在定向分化率低和异位迁移等问题,显著影响了细胞移植的疗效。而组织工程支架能够在损伤脊髓部位起到桥接作用,并为移植细胞提供黏附、增殖和分化的三维环境,与此同时组织工程支架还可以作为药物和神经因子的载体为细胞提供营养或诱导微环境,从多方面促进脊髓损伤后的神经修复。组织工程支架负载BMSCs逐渐成为脊髓损伤研究的热点和最具前景的方案,为脊髓损伤患者的康复带来了新的曙光(图1)。 Onuma-Ukegawa等[31]将GFP标记的BMSCs负载于蜂窝状胶原蛋白海绵支架中移植入脊髓损伤大鼠体内,结果显示联合治疗组的感觉和运功功能均优于单纯BMSCs移植组,说明蜂窝状胶原蛋白海绵支架能够增强BMSCs在脊髓损伤中的神经修复作用。Wang等[32]将BMSCs种植在脱细胞脊髓支架(ASC)中,结果表明BMSCs可通过抑制免疫炎症反应和细胞凋亡来促进脊髓损伤部位的神经修复。Raynald等[33]将负载BMSCs的透明质酸水凝胶支架(HA-PLL)移植入SD大鼠脊髓损伤模型体内,结果显示实验组大鼠BBB运动功能评分及电生理检测均优于对照组,且损伤部位再生轴突数量更多,因此透明质酸水凝胶支架负载BMSCs可有效促进脊髓损伤后的神经修复。Günther等[34]将BMSCs种植于藻酸盐水凝胶支架后植入半横断大鼠脊髓损伤模型体内,结果显示再生轴突的数量进一步增加,表明藻酸盐水凝胶支架可以提供体内机械引导和促进细胞生长的基质从而增强脊髓损伤部位的轴突再生。Chen等[35]将接种BMSCs的脱细胞脊髓支架移植入半横断脊髓损伤部位,以研究移植物在脊髓损伤修复中的作用,该研究表明,脱细胞脊髓支架与宿主脊髓组织相容性良好,并且植入的BMSCs至少可以在体内存活8周左右,其原因是BMSCs接种的脱细胞脊髓支架可以通过保护天然神经组织并增强组织再髓鞘化作用来促进脊髓损伤大鼠的神经功能恢复。 2.5.5 BMSCs联合电针疗法 电针疗法是利用神经电刺激将传统中医针灸疗法进行改良而形成的一种新的治疗方法,主要是将针灸针刺入腧穴得气后,在针具上施加与人体生物电强度相近的微弱电流,通过结合针刺和电流的双重刺激,提高治疗效果。Paul等[36]和Yamasaki等[37]研究结果均证实电针疗法能够提高神经营养因子的表达水平,并促进神经生长、存活及修复,改善脊髓损伤。 Ding等[38-41]在脊髓损伤大鼠体内移植过表达Trk C基因的BMSCs的基础上联合使用了电针技术。实验结果显示,实验组大鼠移植部位及周围组织中的神经营养因子3水平均显著增高,并且电针刺激能够提高Trk C修饰的BMSCs的存活率以及向神经元样细胞和少突胶质样细胞定向分化的能力,同时电针刺激还可以在抑制硫酸软骨素蛋白多糖和胶质纤维酸性蛋分泌的同时,促进层粘连蛋白的产生及降钙素基因相关肽阳性神经纤维、5-羟色胺阳性神经纤维和皮质脊髓束神经纤维的再生,指导再生神经纤维进入损伤区域,进而全面改善患肢运动和神经修复情况。Liu等[42]对比单纯BMSCs组与植入式电针(iEA)联合BMSCs移植组的实验结果,联合治疗组细胞活性和后肢功能恢复情况均高于单纯BMSCs移植组,因此植入式电针可以有效提高BMSCs在脊髓损伤中的治疗效果。"
| [1] Albersen M, Fandel TM, Lin G, et al. Injections of adipose tissue-derived stem cells and stem cell lysate improve recovery of erectile function in a rat model of cavernous nerve injury. J Sex Med. 2010;7(10):3331-3340.[2] Chhabra HS, Sarda K.Clinical translation of stem cell based interventions for spinal cord injury - Are we there yet. Adv Drug Deliv Rev. 2017;120:41-49.[3] Dominici M, Le Blanc K, Mueller I, et al. Minimal criteria for defining multipotentmesenchymal stromal cells. The International Society for Cellular Therapy position statement.Cytotherapy. 2006;8(4): 315-317.[4] Li SH, Guo PD, Wang WJ.Research progress of bone marrow mesenchymal stem cells differentiation into nerve-like cells induced by traditional Chinese medicine.ZhongguoGu Shang. 2010;23(3): 233-235.[5] Lee J, Elkahloun AG, Messina SA, et al. Cellular and genetic characterization of human adult bone marrow-derived neural stem-like cells: a potential antiglioma cellular vector. Cancer Res. 2003;63(24):8877-8889.[6] Satake K, Lou J, Lenke LG.Migration of mesenchymal stem cells through cerebrospinal fluid into injured spinal cord tissue.Spine (Phila Pa 1976). 2004;29(18):1971-1979.[7] Ye Y, Feng TT, Peng YR, et al. The treatment of spinal cord injury in rats using bone marrow-derived neural-like cells induced by cerebrospinal fluid.NeurosciLett. 2018;666:85-91.[8] Crigler L, Robey RC, Asawachaicharn A, et al. Human mesenchymal stem cell subpopulations express a variety of neuro-regulatory molecules and promote neuronal cell survival and neuritogenesis.Exp Neurol. 2006;198(1):54-64.[9] Phinney DG, Baddoo M, Dutreil M, et al. Murine mesenchymal stem cells transplanted to the central nervous system of neonatal versus adult mice exhibit distinct engraftment kinetics and express receptors that guide neuronal cell migration.Stem Cells Dev. 2006; 15(3):437-447.[10] Son BR, Marquez-Curtis LA, Kucia M, et al. Migration of bone marrow and cord blood mesenchymal stem cells in vitro is regulated by stromal-derived factor-1-CXCR4 and hepatocyte growth factor-c-met axes and involves matrix metalloproteinases.Stem Cells. 2006;24(5):1254-1264.[11] Chen X, Li Y, Wang L, et al. Ischemic rat brain extracts induce human marrow stromal cell growth factor production. Neuropathology. 2002;22(4):275-279.[12] 于德水,吕刚,王岩峰,等.骨髓间充质干细胞移植对大鼠脊髓损伤后血管内皮生长因子表达的影响[J].中国组织工程研究与临床康复, 2008, 12(21):4115-4118.[13] 李洪秋,王哲,阿良.骨髓间充质干细胞移植对大鼠脊髓损伤后氧化应激的影响[J].脊柱外科杂志,2010,8(3):157-161.[14] Zhang H, Wang L, Wen S, et al. Magnetic resonance imaging tracking and assessing repair function of the bone marrow mesenchymal stem cells transplantation in a rat model of spinal cord injury.Oncotarget. 2017;8(35):58985-58999.[15] Woodbury D, Schwarz EJ, Prockop DJ, et al. Adult rat and human bone marrow stromal cells differentiate into neurons.J Neurosci Res. 2000;61(4):364-370.[16] Rivera FJ, Sierralta WD, Minguell JJ, et al. Adult hippocampus derived soluble factors induce a neuronal-like phenotype in mesenchymal stem cells.NeurosciLett. 2006;406(1-2):49-54.[17] Jiang Y, Jahagirdar BN, Reinhardt RL, et al. Pluripotency of mesenchymal stem cells derived from adult marrow.Nature. 2002; 418(6893):41-49.[18] Blondheim NR, Levy YS, Ben-Zur T, et al. Human mesenchymal stem cells express neural genes, suggesting a neural predisposition.Stem Cells Dev. 2006;15(2):141-164.[19] 杨晓苏,吴海香,肖波.人骨髓间充质干细胞分化为神经元样细胞及运动神经元生存蛋白表达的研究[J]. 中华医学杂志,2005,85(16): 1125-1128[20] Liu XL, Zu QQ, Wang B, et al. Differentiation of genetically modified canine bone mesenchymal stem cells labeled with superparamagnetic iron oxide into neural?like cells.Mol Med Rep. 2018;17(6):7902-7910.[21] Moviglia GA, Varela G, Gaeta CA, et al. Autoreactive T cells induce in vitro BM mesenchymal stem cell transdifferentiation to neural stem cells.Cytotherapy. 2006;8(3):196-201.[22] Coronel MF, Musolino PL, Villar MJ.Selective migration and engraftment of bone marrow mesenchymal stem cells in rat lumbar dorsal root ganglia after sciatic nerve constriction.Neurosci Lett. 2006;405(1-2):5-9.[23] Hellmann MA, Panet H, Barhum Y, et al. Increased survival and migration of engrafted mesenchymal bone marrow stem cells in 6-hydroxydopamine-lesioned rodents.Neurosci Lett. 2006;395(2): 124-128.[24] Wu S, Cui G, Shao H, et al. The Cotransplantation of Olfactory Ensheathing Cells with Bone Marrow Mesenchymal Stem Cells Exerts Antiapoptotic Effects in Adult Rats after Spinal Cord Injury. Stem Cells Int. 2015;2015:516215.[25] Karimi-Abdolrezaee S, Eftekharpour E, Wang J, et al. Delayed transplantation of adult neural precursor cells promotes remyelination and functional neurological recovery after spinal cord injury.J Neurosci. 2006;26(13):3377-3389.[26] 步星耀,赵红卫,钱宝延,等.自体骨髓间质干细胞移植联合神经营养因子及综合康复治疗脊髓损伤[J].中华实用诊断与治疗杂志, 2009,23(4): 329-331.[27] Shahrezaie M, Mansour RN, Nazari B, et al. Improved stem cell therapy of spinal cord injury using GDNF-overexpressed bone marrow stem cells in a rat model.Biologicals. 2017;50:73-80.[28] Akbari M, Khaksari M, Rezaeezadeh-Roukerd M, et al. Effect of chondroitinase ABC on inflammatory and oxidative response following spinal cord injury.Iran J Basic Med Sci. 2017;20(7): 806-812.[29] Shultz RB, Zhong Y.Minocycline targets multiple secondary injury mechanisms in traumatic spinal cord injury.Neural Regen Res. 2017;12(5):702-713. [30] Chen D, Zeng W, Fu Y, et al. Bone marrow mesenchymal stem cells combined with minocycline improve spinal cord injury in a rat model.Int J Clin Exp Pathol. 2015;8(10):11957-11969.[31] Onuma-Ukegawa M, Bhatt K, Hirai T, et al. Bone Marrow Stromal Cells Combined With a Honeycomb Collagen Sponge Facilitate Neurite Elongation In Vitro and Neural Restoration in the Hemisected Rat Spinal Cord.Cell Transplant. 2015;24(7):1283-1297.[32] Wang YH, Chen J, Zhou J, et al. Reduced inflammatory cell recruitment and tissue damage in spinal cord injury by acellular spinal cord scaffold seeded with mesenchymal stem cells.ExpTher Med. 2017;13(1):203-207.[33] Raynald, Li Y, Yu H, et al. The hetero-transplantation of human bone marrow stromal cells carried by hydrogel unexpectedly demonstrates a significant role in the functional recovery in the injured spinal cord of rats.Brain Res. 2016;1634:21-33.[34] Günther MI, Weidner N, Müller R, et al. Cell-seeded alginate hydrogel scaffolds promote directed linear axonal regeneration in the injured rat spinal cord.Acta Biomater. 2015;27:140-150.[35] Chen J, Zhang Z, Liu J, et al. Acellular spinal cord scaffold seeded with bone marrow stromal cells protects tissue and promotes functional recovery in spinal cord-injured rats.J Neurosci Res. 2014; 92(3):307-317.[36] Paul C, Samdani AF, Betz RR, et al. Grafting of human bone marrow stromal cells into spinal cord injury: a comparison of delivery methods.Spine (Phila Pa 1976). 2009;34(4):328-334.[37] Yamasaki K, Setoguchi T, Takenouchi T, et al. Stem cell factor prevents neuronal cell apoptosis after acute spinal cord injury.Spine (Phila Pa 1976). 2009;34(4):323-327.[38] Ding Y, Yan Q, Ruan JW, et al. Electroacupuncture promotes the differentiation of transplanted bone marrow mesenchymal stem cells overexpressing TrkC into neuron-like cells in transected spinal cord of rats.Cell Transplant. 2013;22(1):65-86.[39] Ding Y, Yan Q, Ruan JW, et al. Electro-acupuncture promotes survival, differentiation of the bone marrow mesenchymal stem cells as well as functional recovery in the spinal cord-transected rats. BMC Neurosci. 2009;10:35. [40] Yan Q, Ruan JW, Ding Y, et al. Electro-acupuncture promotes differentiation of mesenchymal stem cells, regeneration of nerve fibers and partial functional recovery after spinal cord injury. Exp Toxicol Pathol. 2011;63(1-2):151-156. [41] Ding Y, Yan Q, Ruan JW, et al. Bone marrow mesenchymal stem cells and electroacupuncturedownregulate the inhibitor molecules and promote the axonal regeneration in the transected spinal cord of rats.Cell Transplant. 2011;20(4):475-491.[42] Liu H, Yang K, Xin T, et al. Implanted electro-acupuncture electric stimulation improves outcome of stem cells' transplantation in spinal cord injury. Artif Cells Blood SubstitImmobil Biotechnol. 2012;40(5): 331-337. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [3] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [4] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [5] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [6] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [7] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [8] | Li Cai, Zhao Ting, Tan Ge, Zheng Yulin, Zhang Ruonan, Wu Yan, Tang Junming. Platelet-derived growth factor-BB promotes proliferation, differentiation and migration of skeletal muscle myoblast [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1050-1055. |
| [9] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [10] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [11] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [12] | Chen Junyi, Wang Ning, Peng Chengfei, Zhu Lunjing, Duan Jiangtao, Wang Ye, Bei Chaoyong. Decalcified bone matrix and lentivirus-mediated silencing of P75 neurotrophin receptor transfected bone marrow mesenchymal stem cells to construct tissue-engineered bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 510-515. |
| [13] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [14] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [15] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||