Chinese Journal of Tissue Engineering Research ›› 2016, Vol. 20 ›› Issue (34): 5089-5097.doi: 10.3969/j.issn.2095-4344.2016.34.012
Previous Articles Next Articles
Nano-hydroxyapatite coated biphasic calcium phosphate for posterolateral lumbar fusion: combined magnetic field plays an auxiliary role
Zhuo Xiang-long1, Lv Hong-bin2, Hu Jian-zhong3, Li Bing1, Liu Yong4, Wu Zu-tong3
- 1Department of Spine Surgery, Liuzhou Worker’s Hospital, Liuzhou 545001, Guangxi Zhuang Autonomous Region, China; 2Department of Sport Medicine, 3Department of Spine Surgery, Xiangya Hospital of Central South University, Changsha 410008, Hunan Province, China; 4Powder Metallurgy Research Institute, Central South University, Changsha 410008, Hunan Province, China
-
Received:2016-06-04Online:2016-08-19Published:2016-08-19 -
Contact:Hu Jian-zhong, Department of Spine Surgery, Xiangya Hospital of Central South University, Changsha 410008, Hunan Province, China -
About author:Zhuo Xiang-long, M.D., Associate chief physician, Master’s supervisor, Department of Spine Surgery, Liuzhou Worker’s Hospital, Liuzhou 545001, Guangxi Zhuang Autonomous Region, China -
Supported by:the National Natural Science Foundation of China, No. 81171699
CLC Number:
Cite this article
Zhuo Xiang-long, Lv Hong-bin, Hu Jian-zhong, Li Bing, Liu Yong, Wu Zu-tong. Nano-hydroxyapatite coated biphasic calcium phosphate for posterolateral lumbar fusion: combined magnetic field plays an auxiliary role[J]. Chinese Journal of Tissue Engineering Research, 2016, 20(34): 5089-5097.
share this article
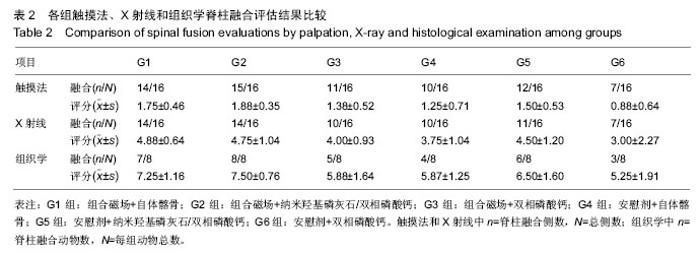
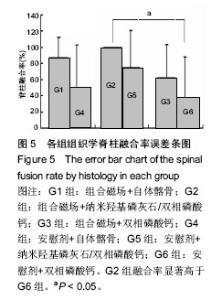
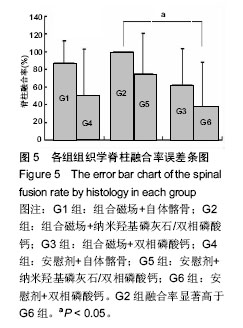
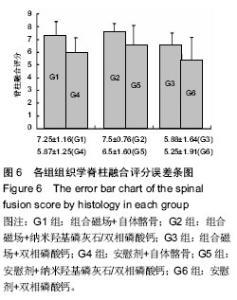
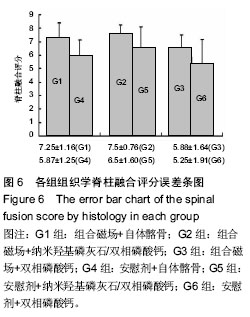
2.1 实验动物数量分析 共有48只成年家兔纳入实验,随机分成6组,每组8只。在实验早期(术后2周内)有2只兔手术中死亡,按序补入。 2.2 大体观察及触摸法融合评估结果 触摸法融合率和评分见表2。各组融合率以G2组最高(94%),G6组最低(44%),2组相比差异有显著性意义(P=0.006),G1组也显著高于G6组(P=0.023),G5和G3组略高于G4组,但差异无显著性意义。析因分析显示A和B两因素没有交互作用(F=0.56,P=0.949),A因素主效应提高融合率23%,差异有显著性意义(F=6.747,P=0.011),B因素主效应是提高融合率19%,差异也有显著性意义(F=3.513, P=0.034)。 G1,2,5组融合评分都显著高于G6组(P=0.003,0.001,0.028),G2组也高于G4组(P=0.028),余各组间比较差异无显著性意义。析因分析结果显示A因素和B因素没有交互作用(F=0.073,P=0.930),A因素主效应提高融合评分0.33,差异有显著性意义(F=4.667,P=0.037);B因素的主效应提高0.38,差异有显著性意义(F=4.594,P=0.016)。 2.3 X射线片结果 术后5周各组均可见到横突间有多少不等的骨痂形成,但不连续;术后7周,在G1和G2组中可见到横突间连续骨痂出现,各有3只实验动物达到了影像学融合标准,但各组间骨痂形成量没有明显差别。术后9周,组合磁场治疗组形成的骨痂量相对要多一些,G1,G2和G5组骨痂形成稍多,G3,G4和G6组形成较少。 2.3.1 X射线融合评价 X射线融合率和评分见表2。"
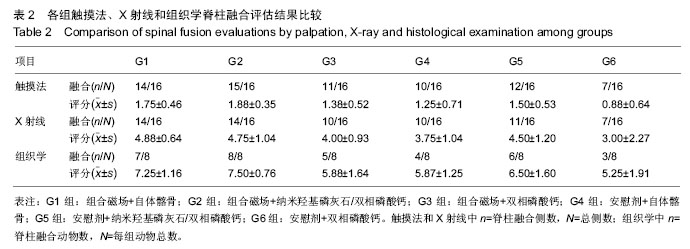
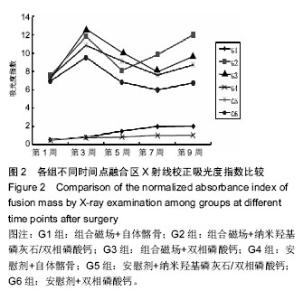
G1和G2组融合率最高,显著高于G6组(P=0.023),其他各组之间差异无显著性意义。析因分析结果显示A因素和B因素没有交互作用(F=0.51,P=0.95),A因素(组合磁场治疗)主效应提高融合率为21%,差异有显著性意义(F=5.102,P=0.026);B因素提高融合率为17%,差异无显著性意义(F=2.908,P=0.06)。 比较各组间融合评分结果显示G1,G2,G5组融合评分显著高于G6组(P=0.006,0.01,0.025),余各组间比较差异无显著性意义(P > 0.05)。析因分析显示A因素和B因素没有交互作用(F=0.538,P=0.588),A和B因素主效应分别为0.79和0.75,差异有显著性意义(F=4.521,P=0.039;F=3.243,P=0.49)。 2.3.2 融合区校正吸光度指数 由图2所示融合区校正吸光度指数度指数,自体髂骨组(G1组和G4组)校正吸光度指数始终显著低于其他各组(P < 0.05),但保持增高的趋势。而G2,G3,G5和G6四组校正吸光度指数经历升高、下降后再缓慢升高的过程。第5周G3组校正吸光度指数高于其他各组,此后G2组校正吸光度指数升高加快,第7,9周始终明显高于其他各组(P < 0.05)。"
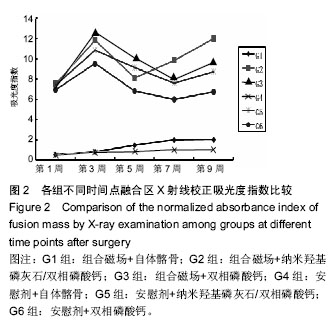
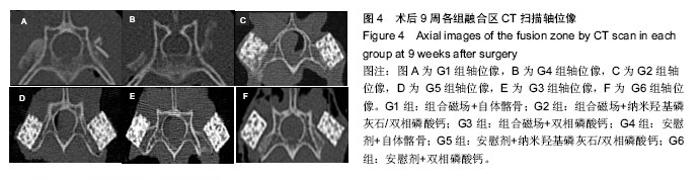
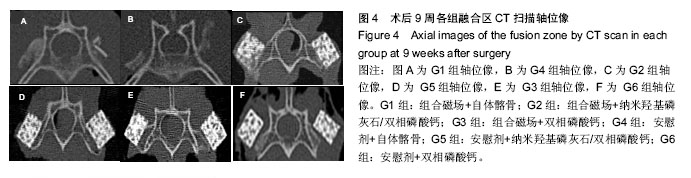
2.4 CT分析结果 CT扫描显示自体髂骨组植骨大部分被吸收,在相邻横突之间新生骨痂形成骨桥连接。在G1组新生骨痂量要多于G4组,密度也高于G4组,这与X射线的校正吸光度指数结果一致(图4A,B)。人工骨组可见到新生骨自横突和小关节突与人工骨紧密连接并长入人工骨内部,形成连续骨桥连接相邻横突。G2组与其他组相比,人工骨变得稀疏、松散,人工骨残留减少明显,有更多的骨痂形成包绕人工骨,人工骨与自体骨形成紧密骨整合(图4C)。G3和G5组人工骨与自体骨形成骨整合,但新生骨痂相对G2组少,人工骨稍松散,孔隙变大,结构尚规整(图4D,E),G6组新生骨骨痂更少,有时在界面区可见到线状缝隙,这说明人工骨与自体骨之间新生骨矿化不完全,密度稍低于周围组织,人工骨较规整(图4F)。"
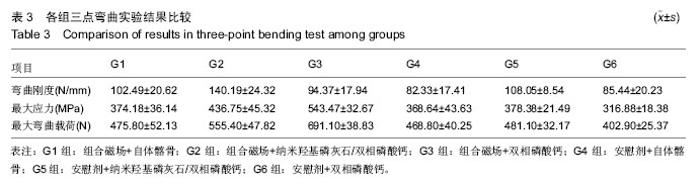
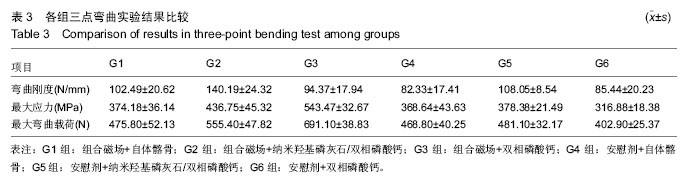
2.5 生物力学结果 如表3所示,G2组脊柱弯曲刚度最高,显著高于其他各组(P < 0.05),G4组脊柱弯曲刚度最低,亦显著低于G1和G5组(P < 0.05),G5组脊柱弯曲刚度还显著高于G6组(P < 0.05)。余各组间比较差异无显著性意义(P > 0.05)。析因分析结果显示A因素和B因素没有交互作用(F=1.521,P=0.068),A因素(组合磁场治疗)主效应为提高弯曲刚度20.41 N/mm,差异有显著性意义(F=14.12,P=0.01);B因素的主效应是提高弯曲刚度23.85 N/mm,差异有显著性意义(F=16.43, P=0.000)。 由表3可见,G3组的最大应力和最大弯曲载荷最大,显著高于其他各组(P < 0.05);G6组最大应力和最大弯曲载荷最小,显著小于其他各组(P < 0.05);G2组显著高于G1,G4和G5组(P < 0.05),余各组间比较差异无显著性意义。G1,G4和G5组之间差异无显著性意义(P > 0.05)。组合磁场治疗组可显著提高人工骨组最大应力和最大弯曲载荷(P < 0.05),但对自体髂骨组没有明显作用。 "
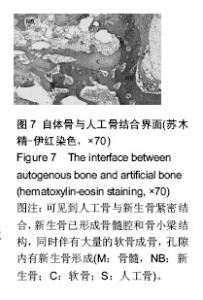
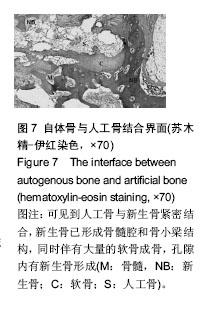
横突间可见连续骨桥形成,人工骨周围生成新生骨痂,并长入人工骨内,形成骨性相连,有成熟骨髓腔和骨小梁结构。界面区域内有大量的成骨细胞、软骨细胞存在、骨髓(图7)。人工骨孔隙内由新生骨、纤维组织组成,可见骨细胞陷窝和骨细胞,有成熟骨小梁并相互连通,可见到骨小管,人工骨变得稀疏,松散,边缘模糊(图8),成骨细胞围绕在新生骨周围,破骨细胞和软骨细胞少见,并可见到较多血管长入,骨样基质沉积在成骨样细胞周围并逐渐钙化,在人工骨周围和内部可见到多核细胞围绕。G2组人工骨自体骨整合效果最佳,有连续新生骨小梁长入人工骨孔隙内,可见到成熟新生骨小梁结构形成的网络,而且孔内新生骨形成也高于其他人工骨组,周围骨痂以成熟骨组织为主。G6组新生骨小梁最少,孔隙内以纤维组织为主,伴有新生骨形成,在新生骨边缘可见成骨细胞和破骨细胞围绕,人工骨周围骨痂纤维组织较多。G3和G5组相对G6组来说,新生骨增加,胶原纤维减少,新生骨与纤维组织的比例在50%左右。人工骨周围包绕的骨痂以软骨为主。"
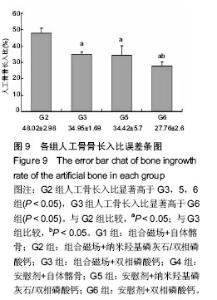
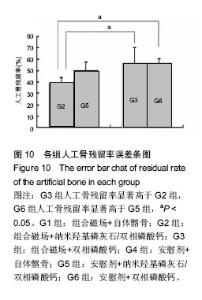
2.6.2 人工骨孔隙内骨长入比 G2组骨长入比最高并显著高于其他各组(P < 0.05),G6组最低并显著低于G3组(P < 0.05)和G5组(P=0.054)。析因分析结果显示A和B因素的主效应分别为10.305和9.865(P=0.001)(图9)。 2.6.3 人工骨残留率 在新生骨形成的同时伴随着人工骨的降解,IPP软件计算的人工骨残留面积百分率结果显示(图10),G2组人工骨残留率最少为38%,显著低于G3和G6组(P < 0.05),余各组残留率比较差异无显著性意义(P > 0.05)。析因分析结果显示,A因素对人工骨降解没有显著促进作用(P=0.13),B因素能够显著促进人工骨降解(P=0.003)。"

| [1] Morone MA, Boden SD. Experimental posterolateral lumbar spinal fusion with a demineralized bone matrix gel. Spine (Phila Pa 1976). 1998;23(2): 159-167.
[2] Lobo SE, Glickman R, Da SW, et al. Response of stem cells from different origins to biphasic calcium phosphate bioceramics. Cell and Tissue Res. 2015;36(2):477-495.
[3] Kitayama S, Wong LO, Ma L, et al. Regeneration of rabbit calvarial defects using biphasic calcium phosphate and a strontium hydroxyapatite-containing collagen membrane. Clin Oral Implants Res. 2015.
[4] Fellah BH, Gauthier O, Weiss P, et al. Osteogenicity of biphasic calcium phosphate ceramics and bone autograft in a goat model. Biomaterials. 2008;29(9): 1177-1188.
[5] Pang KM, Lee JK, Seo YK, et al. Biologic properties of nano-hydroxyapatite: An in vivo study of calvarial defects, ectopic bone formation and bone implantation. Biomed Mater Eng. 2015;25(1):25-38.
[6] Juhasz JA, Best SM, Bonfield W. Preparation of novel bioactive nano-calcium phosphate-hydrogel composites. Sci Tech Adv Mat 2010; 11(1):014103.
[7] Ou KL, Wu J, Lai W, et al. Effects of the nanostructure and nanoporosity on bioactive nanohydroxyapatite/ reconstituted collagen by electrodeposition. J Biom Mat Res. 2010;92A(3): 906-912.
[8] Xiao T, Peng C, Luo Y, et al. [Culture of chondrocytes using nano-hydroxyapatite/collagen in vitro]. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2007;32(4): 641-645.
[9] Venkatesan J, Kim SK. Nano-hydroxyapatite composite biomaterials for bone tissue engineering--a review. J Biomed Nanotechnol. 2014;10(10):3124-3140.
[10] Deibert MC, Mcleod BR, Smith SD, et al. Ion resonance electromagnetic field stimulation of fracture healing in rabbits with a fibular ostectomy. J Orthop Res.1994;12(6): 878-885.
[11] Oishi M, Onesti ST. Electrical bone graft stimulation for spinal fusion: a review. Neurosurgery. 2000;47(5): 1041-1055, 1055-1056.
[12] Linovitz RJ, Pathria M, Bernhardt M, et al. Combined magnetic fields accelerate and increase spine fusion: a double-blind, randomized, placebo controlled study. Spine (Phila Pa 1976). 2002;27(13): 1383-1389.
[13] Boden SD, Schimandle JH, Hutton WC. An experimental lumbar intertransverse process spinal fusion model. Radiographic, histologic, and biomechanical healing characteristics. Spine (Phila Pa 1976). 1995;20(4): 412-420.
[14] Lopez MJ, Mcintosh KR, Spencer ND, et al. Acceleration of spinal fusion using syngeneic and allogeneic adult adipose derived stem cells in a rat model. J Orthop Res. 2009;27(3): 366-373.
[15] Huang RC, Khan SN, Sandhu HS, et al. Alendronate inhibits spine fusion in a rat model. Spine (Phila Pa 1976). 2005;30(22): 2516-2522.
[16] Motomiya M, Ito M, Takahata M, et al. Effect of Hydroxyapatite porous characteristics on healing outcomes in rabbit posterolateral spinal fusion model. Eur Spine J. 2007;16(12): 2215-2224.
[17] Qiu QQ, Shih MS, Stock K, et al. Evaluation of DBM/AM composite as a graft substitute for posterolateral lumbar fusion. J Biomed Mater Res B Appl Biomater. 2007;82(1): 239-245.
[18] Kim DH, Jahng TA, Fu TS, et al. Evaluation of HealosMP52 osteoinductive bone graft for instrumented lumbar intertransverse process fusion in sheep. Spine (Phila Pa 1976). 2004;29(24): 2800-2808.
[19] Hing KA, Best SM, Tanner KE, et al. Mediation of bone ingrowth in porous hydroxyapatite bone graft substitutes. J Biomed Mater Res A. 2004;68(1): 187-200.
[20] Linovitz RJ, Pathria M, Bernhardt M, et al. Combined magnetic fields accelerate and increase spine fusion: a double-blind, randomized, placebo controlled study. Spine (Phila Pa 1976). 2002;27(13): 1383-1389, 1389.
[21] Marks RA. Spine fusion for discogenic low back pain: outcomes in patients treated with or without pulsed electromagnetic field stimulation. Adv Ther. 2000;17(2): 57-67.
[22] Ito M, Fay L A, Ito Y, et al. The effect of pulsed electromagnetic fields on instrumented posterolateral spinal fusion and device-related stress shielding. Spine (Phila Pa 1976). 1997;22(4): 382-388.
[23] Hu J, Zhang T, Xu D, et al. Combined magnetic fields accelerate bone-tendon junction injury healing through osteogenesis. Scand J Med Sci Sports. 2015;25(3):398-405.
[24] Fini M, Giavaresi G, Giardino R, et al. Histomorphometric and mechanical analysis of the hydroxyapatite-bone interface after electromagnetic stimulation - An experimental study in rabbits. J Bone Joint Surg. 2006;88B(1): 123-128.
[25] Fini M, Cadossi R, Cane V, et al. The effect of pulsed electromagnetic fields on the osteointegration of hydroxyapatite implants in cancellous bone: a morphologic and microstructural in vivo study. J Orthop Res. 2002;20(4): 756-763.
[26] Glazer PA, Heilmann MR, Lotz JC, et al. Use of electromagnetic fields in a spinal fusion. A rabbit model. Spine (Phila Pa 1976). 1997;22(20): 2351-2356. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Song Chengjie, Chang Hengrui, Shi Mingxin, Meng Xianzhong. Research progress in biomechanical stability of lateral lumbar interbody fusion [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 923-928. |
| [4] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [5] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [6] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [7] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [8] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [9] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [10] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [11] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [12] | Tang Xiaokai, Li Weiming. Role and mechanism of Nel-like molecule-1 in promoting bone fusion after spinal fusion [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3914-3920. |
| [13] | Yang Qin, Zhou Honghai, Chen Longhao, Zhong Zhong, Xu Yigao, Huang Zhaozhi. Research status and development trend of pelvic reconstruction techniques: a bibliometric and visual analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3718-3724. |
| [14] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [15] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||