Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (24): 3914-3920.doi: 10.12307/2021.102
Previous Articles Next Articles
Role and mechanism of Nel-like molecule-1 in promoting bone fusion after spinal fusion
Tang Xiaokai1, Li Weiming2
- 1Harbin Medical University, Harbin 150001, Heilongjiang Province, China; 2Department of Orthopedics, First Affiliated Hospital of Harbin Medical University, Harbin 150001, Heilongjiang Province, China
-
Received:2020-10-26Revised:2020-10-29Accepted:2020-11-28Online:2021-08-28Published:2021-03-18 -
Contact:Li Weiming, MD, Chief physician, Department of Orthopedics, First Affiliated Hospital of Harbin Medical University, Harbin 150001, Heilongjiang Province, China -
About author:Tang Xiaokai, Master candidate, Harbin Medical University, Harbin 150001, Heilongjiang Province, China -
Supported by:the China Postdoctoral Science Foundation Project, No. 2016M601449 (to LWM)
CLC Number:
Cite this article
Tang Xiaokai, Li Weiming. Role and mechanism of Nel-like molecule-1 in promoting bone fusion after spinal fusion[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3914-3920.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
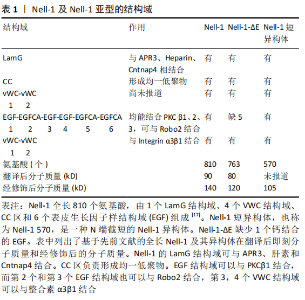
2.1 对Nell-1蛋白结构和功能相关性的进一步认识 自从首次报道Nell-1过度表达与人类颅缝融合症有关以来[15],在前10年主要集中在3个方面:①Nell-1的成骨特性;②Nell-1在骨再生中的潜在应用;③Nell-1介导的丝裂原激活蛋白激酶(mitogen-activated protein kinase,MAPK)信号通路[16-17]。在之后的20年里,有关Nell-1调节骨骼发育和生长以及促进骨软骨组织再生方面的作用和机制研究取得了较大进步。 2.1.1 Nell-1的结合伴侣 由于Nell-1主要以蛋白的形式发生作用,因此人们在寻找Nell-1的特定功能性受体方面付出了巨大的努力,见表1。Nell-1最初被认为是一种与蛋白激酶Cβ1(protein kinase C β1,PKCβ1)结合的蛋白,它们的结合导致Nell-1磷酸化[18],后来ZOU等[19]发现凋亡相关蛋白3(apoptosis-related protein 3,APR3)与Nell-1共同定位于人骨肉瘤细胞系的核膜上,这在功能上可能与促进成骨细胞分化和抑制细胞增殖有关,再往后NAKAMURA 等[20]发现Nell-1可以利用其C末端的最后2个冯?威勒布兰德因子C(von Willebrand factor C,vWC)结构域与Integrin β1结合,促进成骨细胞的黏附和分化,最后YAMAMOTO等[21]发现Nell-1通过其第2个和第3个表皮细胞生长因子(epidermal growth factor,EGF)结构域与环形交叉受体2(roundabout receptor 2,Robo 2)结合。但是以上结合伴侣都不是Nell-1的特异性功能受体,这是因为细胞膜蛋白样整合素β1能与广泛的分子结合,而Nell-1与Robo2和APR3的结合不是天然存在于细胞表面,见表1。 "
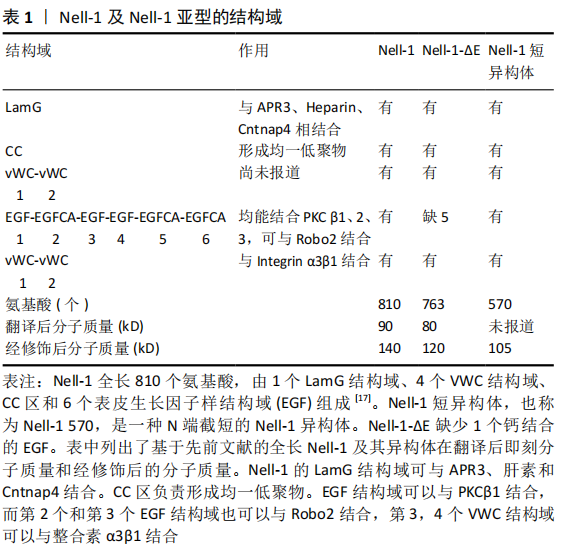
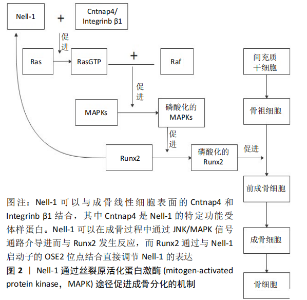
2.1.2 Nell-1特异性受体的确定 最近,在识别Nell-1的细胞表面特异性受体方面取得了突破性进展。在不施加人为因素的情况下,确定了接触素相关蛋白样蛋白4 (contactin-associated protein-like 4,Cntnap4)与Nell-1之间的物理高亲和性配体样受体结合。在功能上,Cntnap4表达缺失可使Nell-1诱导的成骨减少,却不影响骨形态发生蛋白2的成骨作用。此外,表达Wnt1的小鼠细胞中特异性Cntnap4失活在新生期具有锁骨、颅骨发育不良的表型,这与N-乙基-N-亚硝基诱导的Nell-1缺陷小鼠颅骨缺陷非常相似[22]。因此,Cntnap4可以被定义为迄今为止发现的唯一与成骨有关的Nell-1细胞表面特异性受体。 2.1.3 具有不同功能的Nell-1亚型 到目前为止,已经报道了2种Nell-1亚型,第一个是N端截短的Nell-1短异构体,也被称为Nell-1 570,见表1。Nell-1 570可以显著刺激间充质基质细胞的增殖和成骨分化[23]。Nell-1的另一个亚型称为Nell-1-ΔE,它缺少1个钙结合的EGF样结构域,表现出与Nell-1相似的亚细胞定位和表达模式。Nell-1-ΔE能够与成骨线性细胞系的胞外间隙中的烯醇化酶1相互作用 [24]。 2.2 脊柱融合术后的骨性融合 脊柱融合术后的骨性融合过程主要依赖于骨重建,加强骨融合需要将骨稳态转变为增加骨形成和减少骨吸收。这一过程经历了复杂的骨代谢过程,类似于骨折愈合过程[25]。推动这一过程的是成骨细胞和生长因子,它们激活一系列细胞因子和炎症因子来诱导骨形成[8]。骨形成是一个复杂的过程,在细胞水平上迁移、增殖、血管生成、成骨分化和随后的矿化是使骨形成和修复发生的基本过程,触发成骨的主要事件是骨髓间充质干细胞向成骨细胞分化[26]。 2.3 Nell-1具有促进脊柱融合的能力 为了探究脊柱融合术后Nell-1与脊柱骨性融合的关系。SIU等[27]在一个骨骼成熟的哥伦布母羊腰椎后外侧模型中研究了Nell-1对脊柱融合的影响,术后第3个月及第4个月的结果显示:添加Nell-1的实验组较不添加Nell-1的对照组,脊柱融合率明显增加;YUAN等[10]在雄性无菌大鼠腰椎后外侧(L4-5)模型上研究了Nell-1的作用,他们发现脱钙骨基质与Nell-1(10或50 μg)混合后,术后4周的融合率为100%,相比之下,使用PBS的融合率为20%,使用自体髂骨移植的融合率为0;JAMES等[28]在5-7岁恒河猴腰椎后外侧模型中研究了Nell-1对脊柱融合的影响,术后4个月时对生理盐水对照组、低剂量Nell-1组、高剂量Nell-1组进行CT检查,3位研究员在不知道分组的情况下对CT显示的脊柱融合率进行评分,结果显示高剂量Nell-1组得分最高,低剂组Nell-1组和生理盐水对照组得分相当,但是免疫荧光染色显示重组Nell-1处理组较生理盐水对照组SCA-1+CD31-CD45-基质细胞增多,通过对实验结果的分析,说明Nell-1可以提高脊柱融合率。 2.3.1 Nell-1具有诱导骨形成的能力 首先Nell-1是在人类颅缝融合活跃的骨形成部位被首次发现[29],然后在包括骨质疏松大鼠模型在内的动物模型中均可诱导骨愈合,且无不良反应[30];在骨质疏松小鼠和绵羊身上进行的研究表明,Nell-1甚至可能被用作治疗骨质疏松骨丢失的潜在药物[31];其次Nell-1在小鼠模型中表现出具有与骨形态发生蛋白类似的成骨能力,并且还可能与骨形态发生蛋白协同作用,促进质量更好的局部骨形成[17];最后,一项临床前研究将pDNA-Nell-1与骨髓间充质干细胞和三维打印生物活性玻璃/壳聚糖纳米复合材料联合应用于恒河猴牙槽骨再生,这不仅显著地促进了牙槽骨的再生,而且再生的新骨组织在质量上与正常骨非常接近[32]。 2.3.2 Nell-1促进前成骨细胞分化的能力 为了确定Nell-1是否能以诱导间充质干细胞迁移的形式将祖细胞招募到损伤部位,FAHMY-GARCIA等[12]评估了Nell-1对间充质干细胞迁移的影响,当把Nell-1蛋白加入到含有间充质干细胞的培养基时,结果显示:Nell-1能够诱导间充质干细胞迁移,且在质量浓度为0-100 μg/L的范围内,间充质干细胞的迁移率随Nell-1的质量浓度增加而增加,而骨形态发生蛋白2却无此功效;与对照组相比,Nell-1组的迁移均增加了2倍及以上。虽然通过钙离子沉积评估显示出骨形态发生蛋白2可以促进间充质干细胞成骨分化,而Nell-1对其无影响,但是当Nell-1以低剂量添加到前成骨细胞时,尽管不同培养物之间有很高的变异性,但仍有成骨趋势,并且Nell-1即使在低剂量下也可以有成骨诱导作用,而骨形态发生蛋白2则表现出相反的作用。 在上述基础上,为了确定Nell-1是否可以进一步促进前成骨细胞分化。BOKUI等[16]在人骨肉瘤细胞系、ZOU等[19]在小鼠成骨样细胞中进行了关于Nell-1成骨分化的研究。研究表明:当转染Nell-1时,这些细胞表现出了向晚期成骨分化的趋势;之后PANG等[33]鉴定出Nell-1的功能转录本对于小鼠间充质干细胞系的前成骨细胞分化具有良好的诱导作用。FAHMY-GARCIA等[12]首次通过使用人类来源的2种类型的祖细胞:人骨髓间充质干细胞和人脐静脉内皮细胞进行研究,其主要目的是将Nell-1、骨形态发生蛋白2并列比较,评估它们是否为促进骨形成所必需,即在诱导细胞迁移、前成骨分化和新生血管中起作用,进而研究他们对于脊柱融合的意义,结果显示Nell-1可以将祖细胞招募到损伤部位,促进骨髓间充质干细胞迁移;Nell-1刺激人脐静脉内皮细胞募集和分化进而刺激新生血管的形成;以及Nell-1可以刺激骨祖细胞迁移并促进其前成骨细胞分化,见图2。 2.3.3 Nell-1促进新生血管形成的能力 JAMES等[28]研究显示:通过Nell-1的治疗,非人灵长类腰椎脊柱融合模型动物的血管有明显增加。那么Nell-1对人脊柱融合成骨过程中具有重要作用的间充质干细胞及血管内皮细胞是否有此作用呢?对此,FAHMY-GARCIA等[12]研究结果给予了肯定的回答。在严格的剂量范围内,Nell-1以剂量依赖的方式促进内皮细胞及间充质干细胞的迁移,当达到最高质量浓度100 μg/L时,迁移的细胞增加2倍以上;同时在培养基中加入100 μg/L Nell-1时,显示出Nell-1强大的体外促进内皮细胞形成管状结构的能力。此外,ASKARINAM等 [34]研究显示:Nell-1不仅可以促进人外周细胞分泌血管内皮生长因子,而且促进了高表达血管内皮生长因子的血管形成。所有这些都表明了Nell-1对于脊柱融合过程中早期新生血管形成的重要意义。 2.3.4 Nell-1可以与骨形态发生蛋白2联合促进脊柱融合的能力 FAHMY-GARCIA等[12]在实验中没有观察到Nell-1对成骨分化的显著影响,这与 LIU等[35]的体外实验结果一致,即单独使用Nell-1确实不能增强骨髓间充质干细胞的成骨分化能力,因此在体内也不能完全达到治疗效果。FAHMY-GARCIA等[12]的体内实验结果表明,在缺乏脱钙骨基质载体的情况下,Nell-1不能诱导脊柱融合,因为脱钙骨基质含有有益的骨诱导生长因子,主要是骨形态发生蛋白2,估计每克脱钙骨基质中含有6 μg骨形态发生蛋白2[36],这与已发表的研究非常一致[37],与骨形态发生蛋白2不同的是,Nell-1对细胞增殖和细胞迁移的影响可以忽略不计[17]。因此,它不能诱导细胞的募集和增殖来启动原位骨再生。 在体外成骨方面,Nell-1和骨形态发生蛋白2表现出相互促进的效果[38]。不仅如此,在体内实验中,Nell-1还表现出可以抑制骨形态发生蛋白2诱导的炎症,促进骨形态发生蛋白2诱导的骨形成[39],这为先前的研究提供了有力的证据[40-42]。 LIU等[35]在研究中使用的超低剂量的骨形态发生蛋白2,通过μ-CT骨组织形态计量学分析和组织学评估显示:缺乏显著的骨形成,这表明单独使用这种超低剂量的骨形态发生蛋白2导致了不理想的骨融合。相反,在加入低剂量或者高剂量的Nell-1时,结果显示都实现了坚实的脊柱融合,并显著增强了生物力学刚度且避免了大剂量使用骨形态发生蛋白2导致的异位成骨和成脂的现象[43]。与之前的研究相比,这使得Nell-1和骨形态发生蛋白2能够以低得多的剂量正常使用,同时发挥了更有效的脊柱融合作用。 2.4 Nell-1蛋白诱导成骨的机制 2.4.1 Nell-1蛋白诱导成骨过程中的主要信号通路 目前发现Nell-1蛋白诱导成骨过程中的主要信号通路主要有3条,第1条如ZHANG等[17]所述:在成骨过程中,Nell-1和Runx2相互调节,Nell-1通过RAS磷酸化JNK和ERK1/2发挥作用,而不是通过Smad信号或p38发挥作用,见图2;第2条如JAMES等[44]所述:Nell-1增加了成骨细胞和破骨细胞中β-catenin的表达,从而抑制了破骨细胞定向的骨吸收,并促进了成骨细胞向骨细胞的分化;此外,Nell-1还依赖Wnt/β-catenin信号诱导Sca-1+间充质祖细胞在骨稳定和修复中的表达[28];第3条如JAMES等[45]所述Nell-1诱导成骨的Hedgehog信号通路:除了促成骨和抗破骨作用外,Nell-1还通过抑制成脂转录因子来抑制3T3-L1前脂肪细胞和间充质基质细胞的成脂分化。 2.4.2 Nell-1蛋白诱导成骨分化过程中的环状RNA 越来越多的证据证明,环状RNA(circular RNAs,circRNAs)在正常组织和各种疾病的发育过程中发挥着重要作用,它们作为潜在的生物标志物在癌症和神经退行性疾病中逐渐被发 现[46-51]。HUANG等[52]通过RNA测序,研究了重组Nell-1诱导成骨分化过程中CircRNAs的表达谱,并鉴定了2个关键的CircRNAs,即CircRFWD2和CircINO80,这2个CircRNA在重组Nell-1诱导成骨过程中表达上调,它们的表达下调影响了Nell-1对成骨的积极作用。CircRFWD2和CircINO80可与 hsa-miR-6817-5p相互作用,抑制成骨。沉默hsa-miR-6817-5p可部分逆转si-CircRFWD2和si-CircINO80对成骨的负面影响。因此,CircRFWD2和CircINO80可以调节hsa-miR-6817-5p的表达,影响重组Nell-1诱导的人脂肪干细胞成骨分化,这为更好地了解Nell-1在人脂肪干细胞成骨分化中的作用开辟了新的窗口,并为高效、安全地进行骨再生提供了潜在的分子靶点和新的方法。 2.4.3 Nell-1蛋白诱导成骨分化过程中的长非编码RNA 已有研究表明长非编码RNA(long noncooding RNA,LncRNAs)在间充质干细胞的成骨分化过程中发挥着不可或缺的作用[53]。WU等[54]和XIAO等[55]进一步研究后发现:lncRNA KCNQ1OT1通过介导Wnt/β-catenin途径促进成骨分化;而lncRNA H19则通过MAPK和核因子κB(NF-κB)信号通路发挥作用。最近的几项研究表明,HIF1A-AS2、HOTAIRM1-1和ZBED3-AS1也通过这些关键的成骨途径参与间充质干细胞的分化[56-58]。为此XIA等[59]在先前研究的基础上,鉴定了323个在成骨和Nell-1诱导成骨过程中差异表达的LncRNA,其中3个LncRNA(ENST00000602964、ENST00000326734和TCONS_00006792)被鉴定为核心调控基因。在Nell-1诱导成骨过程中,Hedgehog通路标志物(IHH和GLI1)受到调控,而该通路的拮抗剂(GLI3和HHIP)表达上调;在此过程中,Wnt的拮抗剂SFRP1表达下调。由此可见,lncRNAs通过Hedgehog和Wnt通路之间的串扰在Nell-1诱导成骨过程中发挥重要作用。这些发现为研究Nell-1诱导人脂肪干细胞成骨分化的分子机制提供了新的视角。 2.5 Nell-1用于脊柱融合研究的不足之处及前景 目前Nell-1用于促进脊柱融合的研究还存在一些不足之处。首先给药途径方面:Nell-1的局部给药在大多数实验动物模型中都是有效的,包括促进长骨节段性缺损的愈合[60],防止股骨头塌 陷[61],以及恢复骨质疏松动物局部的低骨密度[44]。但是在骨形态发生蛋白2不良反应的研究中发现:不良反应的产生与否通常与剂量大小有着密切的关系,骨形态发生蛋白2的超生理浓度被认为会引发不必要的不良反应[62]。虽然目前的小样本量临床前实验中关于Nell-1的研究尚未表现出与剂量相关的不良反应,但是仍然存在发生的可能。通过全身给药的静脉注射方式,虽然在Nell-1缺乏症的骨质疏松小鼠中显著诱导骨形成[28],然而静脉注射的频率很高,以及Nell-1 在体内的半衰期短[63],使得这种方法在潜在的临床应用中不切实际。为了加速和优化骨重建,使用多种参与骨形成的生长因子可能会更好地减少单因素治疗时由于大剂量使用而可能产生的不良反应。用于骨形成所需的蛋白质不仅剂量大,而且持续的时间也对结果有很大影响,而蛋白质控释载体能够控制蛋白质在单位时间内释放的量以及总的释放时间,因此未来需要探索多种生长因子的联合运用以及寻求合适的控释载体,用于牢固的脊柱融合。 目前的研究中存在争议的部分:一些研究表明,Nell-1由非载体脱钙骨基质-羟基磷灰石包裹的聚乳酸-羟基乙酸诱导的骨再生相当有限[64]。但是,含有Nell-1的聚乳酸-羟基乙酸支架能够促进颅骨缺损的愈合,与骨形态发生蛋白2相比,经Nell-1治疗的缺损处骨密度接近90%,矿化程度与骨形态发生蛋白2治疗的结果相当[62]。因此,Nell-1的疗效是否独立于脱钙骨基质或其对应物仍存在争议,这是在未来的研究中需要明确的部分。 目前关于Nell-1促进成骨的机制尚不完全清楚:例如Nell-1如何调控miR-6817-5p;重组Nell-1诱导人脂肪干细胞成骨分化过程中lncRNAs的作用机制;特定的核心LncRNA仍需鉴定和分析等。 最后,对于Nell-1是否会造成肿瘤的形成,这对Nell-1的应用很重要。WANG等[65]在Barrett食管中发现了Nell-1的高甲基化;SHEN等[66]在所有类型的骨肿瘤和软骨形成肿瘤中也检测到Nell-1的表达。在良性骨肿瘤中,Nell-1呈强弥漫性染色;而在肿瘤间差异较大的骨肉瘤中却观察到Nell-1染色相对减少。这些Nell-1表达与肿瘤组织的相关性使得Nell-1在肿瘤发生中的作用尚不确定。然而,Nell-1增加了95-D肺癌干细胞样细胞的化疗敏感性 [67],并抑制肾细胞癌细胞的迁移和黏附[68]。这些有限的功能研究表明,Nell-1可能是一个候选的肿瘤抑制因子。因此,未来需要进一步确定Nell-1与肿瘤相关的功能特性,特别是它在促进人类骨肉瘤切除后骨再生方面的利弊,以及Nell-1的量是否与肿瘤形成存在相关性。"
| [1] BUCHBINDER R, VAN TULDER M, ÖBERG B, et al. Low back pain: a call for action. Lancet. 2018;391(10137):2384-2388. [2] CHUN DS, BAKER KC, HSU WK. Lumbar pseudarthrosis: a review of current diagnosis and treatment. Neurosurg Focus. 2015;39(4):E10. [3] MAKINO T, KAITO T, FUJIWARA H, et al. Risk Factors for Poor Patient-Reported Quality of Life Outcomes After Posterior Lumbar Interbody Fusion: An Analysis of 2-Year Follow-up. Spine (Phila Pa 1976). 2017; 42(19):1502-1510. [4] OLABISI R. Cell-based therapies for spinal fusion. Adv Exp Med Biol. 2012;760:148-173. [5] NAKAO S, MINAMIDE A, KAWAKAMI M, et al. The influence of alendronate on spine fusion in an osteoporotic animal model. Spine (Phila Pa 1976). 2011;36(18):1446-1452. [6] PINHEIRO AL, SANTOS NR, OLIVEIRA PC, et al. The efficacy of the use of IR laser phototherapy associated to biphasic ceramic graft and guided bone regeneration on surgical fractures treated with wire osteosynthesis: a comparative laser fluorescence and Raman spectral study on rabbits.Lasers Med Sci. 2013;28(3):815-822. [7] MULCONREY DS, BRIDWELL KH, FLYNN J, et al. Bone morphogenetic protein (RhBMP-2) as a substitute for iliac crest bone graft in multilevel adult spinal deformity surgery: minimum two-year evaluation of fusion. Spine (Phila Pa 1976). 2008;33(20):2153-2159. [8] BODEN SD. Overview of the biology of lumbar spine fusion and principles for selecting a bone graft substitute. Spine (Phila Pa 1976). 2002;27(16 Suppl 1):S26-31. [9] ZHANG X, TING K, BESSETTE CM, et al. Nell-1, a key functional mediator of Runx2, partially rescues calvarial defects in Runx2(+/-) mice. J Bone Miner Res. 2011;26(4):777-791. [10] YUAN W, JAMES AW, ASATRIAN G, et al. NELL-1 based demineralized bone graft promotes rat spine fusion as compared to commercially available BMP-2 product. J Orthop Sci. 2013;18(4):646-657. [11] LEE S, ZHANG X, SHEN J, et al. Brief Report: Human Perivascular Stem Cells and Nel-Like Protein-1 Synergistically Enhance Spinal Fusion in Osteoporotic Rats. Stem Cells. 2015;33(10):3158-3163. [12] FAHMY-GARCIA S, VAN DRIEL M, WITTE-BUOMA J, et al. NELL-1, HMGB1, and CCN2 Enhance Migration and Vasculogenesis, But Not Osteogenic Differentiation Compared to BMP2. Tissue Eng Part A. 2018;24(3-4):207-218. [13] PAKVASA M, ALVERDY A, MOSTAFA S, et al. Neural EGF-like protein 1 (NELL-1): Signaling crosstalk in mesenchymal stem cells and applications in regenerative medicine. Genes Dis. 2017;4(3):127-137. [14] QI H, KIM JK, HA P, et al. Inactivation of Nell-1 in Chondrocytes Significantly Impedes Appendicular Skeletogenesis. J Bone Miner Res. 2019;34(3):533-546. [15] TING K, VASTARDIS H, MULLIKEN JB, et al. Human NELL-1 expressed in unilateral coronal synostosis. J Bone Miner Res. 1999;14(1):80-89. [16] BOKUI N, OTANI T, IGARASHI K, et al. Involvement of MAPK signaling molecules and Runx2 in the NELL1-induced osteoblastic differentiation. FEBS Lett. 2008;582(2):365-371. [17] ZHANG X, ZARA J, SIU RK, et al. The role of NELL-1, a growth factor associated with craniosynostosis, in promoting bone regeneration. J Dent Res. 2010;89(9):865-878. [18] KURODA S, TANIZAWA K. Involvement of epidermal growth factor-like domain of NELL proteins in the novel protein-protein interaction with protein kinase C. Biochem Biophys Res Commun. 1999;265(3):752-757. [19] ZOU X, SHEN J, CHEN F, et al. NELL-1 binds to APR3 affecting human osteoblast proliferation and differentiation. FEBS Lett. 2011;585(15): 2410-2418. [20] NAKAMURA Y, HASEBE A, TAKAHASHI K, et al. Oligomerization-induced conformational change in the C-terminal region of Nel-like molecule 1 (NELL1) protein is necessary for the efficient mediation of murine MC3T3-E1 cell adhesion and spreading. J Biol Chem. 2014; 289(14):9781-9794. [21] YAMAMOTO N, KASHIWAGI M, ISHIHARA M, et al. Robo2 contains a cryptic binding site for neural EGFL-like (NELL) protein 1/2. J Biol Chem. 2019;294(12):4693-4703. [22] LI C, ZHENG Z, HA P, et al. Neurexin Superfamily Cell Membrane Receptor Contactin-Associated Protein Like-4 (Cntnap4) Is Involved in Neural EGFL-Like 1 (Nell-1)-Responsive Osteogenesis. J Bone Miner Res. 2018;33(10):1813-1825. [23] MEYERS CA, SUN Z, CHANG L, et al. Age dependent effects of NELL-1 isoforms on bone marrow stromal cells. J Orthop. 2019;16(2):175-178. [24] ZHAO H, QIN X, ZHANG Q, et al. Nell-1-ΔE, a novel transcript of Nell-1, inhibits cell migration by interacting with enolase-1. J Cell Biochem. 2018;119(7):5725-5733. [25] HIRSCH BP, UNNANUNTANA A, CUNNINGHAM ME, et al. The effect of therapies for osteoporosis on spine fusion: a systematic review. Spine J. 2013;13(2):190-199. [26] BLONDER J, XIAO Z, VEENSTRA TD. Proteomic profiling of differentiating osteoblasts. Expert Rev Proteomics. 2006;3(5):483-496. [27] SIU RK, LU SS, LI W, et al. Nell-1 protein promotes bone formation in a sheep spinal fusion model. Tissue Eng Part A. 2011;17(7-8):1123-1135. [28] JAMES AW, SHEN J, TSUEI R, et al. NELL-1 induces Sca-1+ mesenchymal progenitor cell expansion in models of bone maintenance and repair. JCI Insight. 2017;2(12):e92573. [29] TRUONG T, ZHANG X, PATHMANATHAN D, et al. Craniosynostosis-associated gene nell-1 is regulated by runx2. J Bone Miner Res. 2007;22(1):7-18. [30] KWAK J, ZARA JN, CHIANG M, et al. NELL-1 injection maintains long-bone quantity and quality in an ovariectomy-induced osteoporotic senile rat model. Tissue Eng Part A. 2013;19(3-4):426-436. [31] PARK SB, PARK SH, KIM NH, et al. BMP-2 induced early bone formation in spine fusion using rat ovariectomy osteoporosis model. Spine J. 2013;13(10):1273-1280. [32] ZHANG J, CHEN Y, XU J, et al. Tissue engineering using 3D printed nano-bioactive glass loaded with NELL1 gene for repairing alveolar bone defects. Regen Biomater. 2018;5(4):213-220. [33] PANG S, SHEN J, LIU Y, et al. Proliferation and osteogenic differentiation of mesenchymal stem cells induced by a short isoform of NELL-1. Stem Cells. 2015;33(3):904-915. [34] ASKARINAM A, JAMES AW, ZARA JN, et al. Human perivascular stem cells show enhanced osteogenesis and vasculogenesis with Nel-like molecule I protein. Tissue Eng Part A. 2013;19(11-12):1386-1397. [35] LIU L, LAM WMR, NAIDU M, et al. Synergistic Effect of NELL-1 and an Ultra-Low Dose of BMP-2 on Spinal Fusion. Tissue Eng Part A. 2019; 25(23-24):1677-1689. [36] CHIBA H, SAWADA N, ONO T, et al. Establishment and characterization of a simian virus 40-immortalized osteoblastic cell line from normal human bone. Jpn J Cancer Res. 1993;84(3):290-297. [37] TAKIGAWA M. CCN2: a master regulator of the genesis of bone and cartilage. J Cell Commun Signal. 2013;7(3):191-201. [38] COWAN CM, JIANG X, HSU T, et al. Synergistic effects of Nell-1 and BMP-2 on the osteogenic differentiation of myoblasts. J Bone Miner Res. 2007;22(6):918-930. [39] SHEN J, JAMES AW, ZARA JN, et al. BMP2-induced inflammation can be suppressed by the osteoinductive growth factor NELL-1. Tissue Eng Part A. 2013;19(21-22):2390-2401. [40] AGHALOO T, COWAN CM, ZHANG X, et al. The effect of NELL1 and bone morphogenetic protein-2 on calvarial bone regeneration. J Oral Maxillofac Surg. 2010;68(2):300-308. [41] ZHU S, SONG D, JIANG X, et al. Combined effects of recombinant human BMP-2 and Nell-1 on bone regeneration in rapid distraction osteogenesis of rabbit tibia. Injury. 2011;42(12):1467-1473. [42] SHEN J, JAMES AW, ZHANG X, et al. Novel Wnt Regulator NEL-Like Molecule-1 Antagonizes Adipogenesis and Augments Osteogenesis Induced by Bone Morphogenetic Protein 2. Am J Pathol.2016;186(2):419-434. [43] PAN HC, LEE S, TING K, et al. Cyst-Like Osteolytic Formations in Recombinant Human Bone Morphogenetic Protein-2 (rhBMP-2) Augmented Sheep Spinal Fusion. Am J Pathol. 2017;187(7):1485-1495. [44] JAMES AW, SHEN J, ZHANG X, et al. NELL-1 in the treatment of osteoporotic bone loss. Nat Commun. 2015;6:7362. [45] JAMES AW, PAN A, CHIANG M, et al. new function of Nell-1 protein in repressing adipogenic differentiation. Biochem Biophys Res Commun. 2011;411(1):126-131. [46] CARRARA M, FUSCHI P, IVAN C, et al. Circular RNAs: Methodological challenges and perspectives in cardiovascular diseases. J Cell Mol Med. 2018;22(11):5176-5187. [47] CAI H, LI Y, NIRINGIYUMUKIZA JD, et al. Circular RNA involvement in aging: An emerging player with great potential. Mech Ageing Dev. 2019;178:16-24. [48] E S, COSTA MC, KURC S, et al. The circulating non-coding RNA landscape for biomarker research: lessons and prospects from cardiovascular diseases. Acta Pharmacol Sin. 2018;39(7):1085-1099. [49] LI LJ, ZHU ZW, ZHAO W, et al. Circular RNA expression profile and potential function of hsa_circ_0045272 in systemic lupus erythematosus. Immunology. 2018;155(1):137-149. [50] CHEN D, MA W, KE Z, et al. CircRNA hsa_circ_100395 regulates miR-1228/TCF21 pathway to inhibit lung cancer progression. Cell Cycle. 2018;17(16):2080-2090. [51] HE JH, HAN ZP, ZHOU JB, et al. MiR-145 affected the circular RNA expression in prostate cancer LNCaP cells. J Cell Biochem. 2018; 119(11):9168-9177. [52] HUANG X, CEN X, ZHANG B, et al. The roles of circRFWD2 and circINO80 during NELL-1-induced osteogenesis. J Cell Mol Med. 2019;23(12):8432-8441. [53] FATICA A, BOZZONI I. Long non-coding RNAs: new players in cell differentiation and development. Nat Rev Genet. 2014;15(1):7-21. [54] WU R, RUAN J, SUN Y, et al. Long non-coding RNA HIF1A-AS2 facilitates adipose-derived stem cells (ASCs) osteogenic differentiation through miR-665/IL6 axis via PI3K/Akt signaling pathway. Stem Cell Res Ther. 2018;9(1):348. [55] XIAO Y, YAN X, YANG Y, et al. Downregulation of long noncoding RNA HOTAIRM1 variant 1 contributes to osteoarthritis via regulating miR-125b/BMPR2 axis and activating JNK/MAPK/ERK pathway. Biomed Pharmacother. 2019;109:1569-1577. [56] GAO X, GE J, LI W, et al. LncRNA KCNQ1OT1 promotes osteogenic differentiation to relieve osteolysis via Wnt/β-catenin activation. Cell Biosci. 2018;8:19. [57] PAN JX. LncRNA H19 promotes atherosclerosis by regulating MAPK and NF-kB signaling pathway. Eur Rev Med Pharmacol Sci. 2017;21(2):322-328. [58] HU K, JIANG W, SUN H, et al. Long noncoding RNA ZBED3-AS1 induces the differentiation of mesenchymal stem cells and enhances bone regeneration by repressing IL-1β via Wnt/β-catenin signaling pathway. J Cell Physiol. 2019;234(10):17863-17875. [59] XIA K, CEN X, YU L, et al. Long noncoding RNA expression profiles during the NEL-like 1 protein-induced osteogenic differentiation. J Cell Physiol. 2020;235(9):6010-6022. [60] TANJAYA J, LORD EL, WANG C, et al. The Effects of Systemic Therapy of PEGylated NEL-Like Protein 1 (NELL-1) on Fracture Healing in Mice. Am J Pathol. 2018;188(3):715-727. [61] FAN M, JIANG WX, WANG AY, et al. Combined effects of NEL-like type 1 gene and zoledronate in preventing collapse of the femoral head. Zhongguo Yi Xue Ke Xue Yuan Xue Bao. 2013;35(5):553-560. [62] JAMES AW, LACHAUD G, SHEN J, et al. A Review of the Clinical Side Effects of Bone Morphogenetic Protein-2. Tissue Eng Part B Rev. 2016;22(4):284-297. [63] KWAK JH, ZHANG Y, PARK J, et al. Pharmacokinetics and osteogenic potential of PEGylated NELL-1 in vivo after systemic administration. Biomaterials. 2015;57:73-83. [64] LEE S, WANG C, PAN HC, et al. Combining Smoothened Agonist and NEL-Like Protein-1 Enhances Bone Healing. Plast Reconstr Surg. 2017;139(6):1385-1396. [65] WANG Z, KAMBHAMPATI S, CHENG Y, et al. Methylation Biomarker Panel Performance in EsophaCap Cytology Samples for Diagnosing Barrett’s Esophagus: A Prospective Validation Study. Clin Cancer Res. 2019;25(7):2127-2135. [66] SHEN J, LACHAUD G, KHADARIAN K, et al. NELL-1 expression in benign and malignant bone tumors. Biochem Biophys Res Commun. 2015;460(2):368-374. [67] ZHAI Y, WEI R, SHA S, et al. Effect of NELL1 on lung cancer stem‑like cell differentiation. Oncol Rep. 2019;41(3):1817-1826. [68] NAKAMURA R, OYAMA T, TAJIRI R, et al. Expression and regulatory effects on cancer cell behavior of NELL1 and NELL2 in human renal cell carcinoma. Cancer Sci. 2015;106(5):656-664. |
| [1] | Ye Haimin, Ding Linghua, Kong Weihao, Huang Zutai, Xiong Long. Role and mechanism of hierarchical microchanneled bone scaffolds in promoting osteogenesis and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 621-625. |
| [2] | Chen Jiang, Li Jinyu, Zheng Chenying, Bai Chunxiao, Zhang Fan, Liu Chuyin, Zhao Xueqian, Yuan Qiaomei, Di Xueshi, Kang Shengqian, Jia Yusong . Changes in sagittal parameters of cervical spine after double-segment artificial cervical disc replacement and anterior cervical discectomy and fusion [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(15): 2341-2346. |
| [3] | Chen Haoyan, Xiao Zenglin, Wang Dongping, Xie Weixing, Jin Daxiang. Comparative clinical efficacy of cortical bone trajectory screw and pedicle screw fixation in posterior lumbar interbody fusion: a meta‑analysis [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(36): 5871-5879. |
| [4] | Li Yongtao, Song Wenhui, Liu Changwen, Liang Kaiheng, Zhang Kaixi, Liu Changfeng. Clinical efficacy of calcium sulphate/demineralized bone matrix versus bone allograft in anterior cervical discectomy and fusion [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5478-5485. |
| [5] | Liu Hao, Liu Jun, Rui Yongjun, Tang Fenglin, Lu Miao, Ding Tao. Co-transfection by Nell-1 and Noggin-shRNA promotes osteoblast differentiation of adipose derived mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 4966-4970. |
| [6] | Jin Liangyu, Li Weiming. Nell-1 in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(20): 3217-3225. |
| [7] | Cao Liangliang, Xu Jianguang, Mei Wei. Three-dimensional finite element analysis of the biomechanical changes of the lumbar spine after the combination of intervertebral fusion with dynamic internal fixation of the interspinous process in the lumbosacral region [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(12): 1905-1910. |
| [8] | Wang Liang, Li Lijun, Zhu Fuliang, Jiang Zhuyan, Wang Shuai, Ni Dongkui . Cortical bone trajectory screw versus pedicle screw fixation after posterior lumbar interbody fusion: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(8): 1291-1298. |
| [9] | Li Qingsong, Liu Shaoyu, Yin Zongsheng. Multiple posterior lumbar interbody fusion and posterolateral fusion instrumentation for thoracolumbar kyphosis and osteoporotic fracture in older adults [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(4): 512-517. |
| [10] | Li Xianzhou, Wang Qian, Zhang Cunxin . Lumbar spondylolisthesis: status and prospects of implant treatment [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(4): 621-627. |
| [11] | Chi Yulei, Bu Xianmin, Zha Yumei, Wang Haibin, Meng Chunyang, Wu Bin. Bone marrow mesenchymal stem cells combined with scaffolds in treatment of bone defects: present and future [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(29): 4749-4756. |
| [12] | Yi Guoliang, Wang Shankun, Song Xizheng. Biomechanical testing of the locking axial lumbosacral interbody fusion [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(28): 4546-4551. |
| [13] | Tang Fubo, Zhou Jinyan, Zhang Yisheng, Zhong Yuanming, Li Zhifei, Mo Yi. Risk factors associated with titanium cage subsidence after anterior single-level subtotal corpectomy and interbody fusion using titanium cage [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(12): 1818-1823. |
| [14] | Li Wen-bo, Zhang Chao, Shi Jie, Huang Qiang, Jia Ding-ding, Gao Qiu-ming. Choke vessel growth in perforator flaps and the conception of angiosome [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(8): 1261-1266. |
| [15] | Tian Yi-xing, Bao Zhao-hua, Zou Jun, Ji Yi-ming, Mei Xin, Pan Jun, He Wen-ye, Yang Hui-lin. Short-term effectiveness of single-level Isobar TTL dynamic internal fixation in the treatment of lumbar degenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(7): 1020-1025. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||