Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (28): 7465-7474.doi: 10.12307/2026.815
Previous Articles Next Articles
Pathogenesis and potential therapeutic targets of idiopathic pulmonary fibrosis: analysis of data from a large-scale genome-wide association study
Fan Zhiliang1, Chai Yihui1, Chen Guanglei1, Li Qian1, Gu Chunsong2, Chen Yunzhi1, Li Wen1, Wu Damei1, Pu Xiang1
- 1School of Basic Chinese Medicine, Guizhou University of Traditional Chinese Medicine, Guiyang 550025, Guizhou Province, China; 2Second Affiliated Hospital, Guizhou University of Traditional Chinese Medicine, Guiyang 550003, Guizhou Province, China
-
Received:2025-08-01Revised:2025-12-11Online:2026-10-08Published:2026-02-26 -
Contact:Pu Xiang, MD, Professor, School of Basic Chinese Medicine, Guizhou University of Traditional Chinese Medicine, Guiyang 550025, Guizhou Province, China -
About author:Fan Zhiliang, MD candidate, School of Basic Chinese Medicine, Guizhou University of Traditional Chinese Medicine, Guiyang 550025, Guizhou Province, China Chai Yihui, MD, Senior experimentalist, School of Basic Chinese Medicine, Guizhou University of Traditional Chinese Medicine, Guiyang 550025, Guizhou Province, China Fan Zhiliang and Chai Yihui contributed equally to this article. -
Supported by:Guizhou Provincial Science and Technology Program, No. ZK[2022]456 (to CYH); National Natural Science Foundation of China, No. 82060913 (to PX); Guizhou Provincial Key Laboratory of Miao Medicine, No. [2025]018 (to PX); Scientific Research Project of Guizhou University of Traditional Chinese Medicine, No. [2025]48 (to PX)
CLC Number:
Cite this article
Fan Zhiliang, Chai Yihui, Chen Guanglei, Li Qian, Gu Chunsong, Chen Yunzhi, Li Wen, Wu Damei, Pu Xiang. Pathogenesis and potential therapeutic targets of idiopathic pulmonary fibrosis: analysis of data from a large-scale genome-wide association study[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7465-7474.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

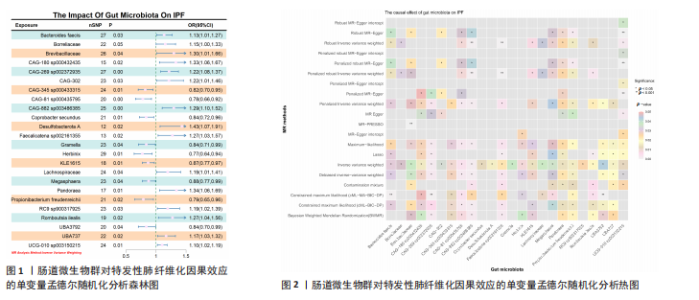
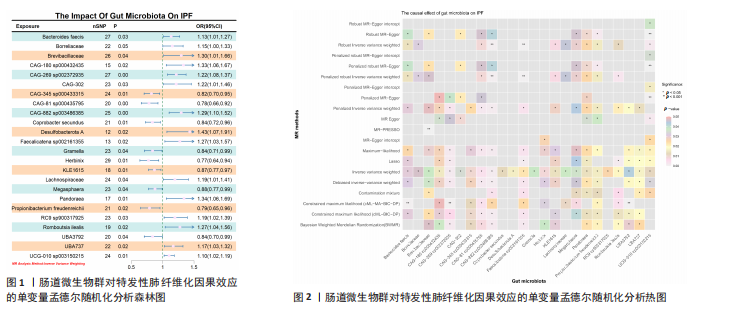
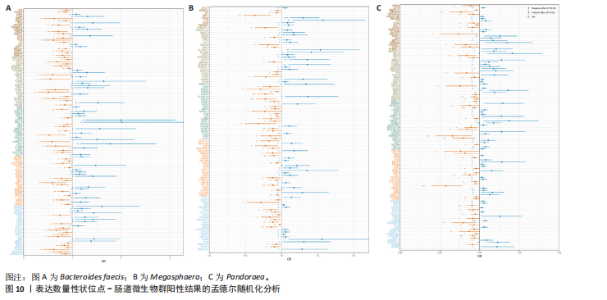
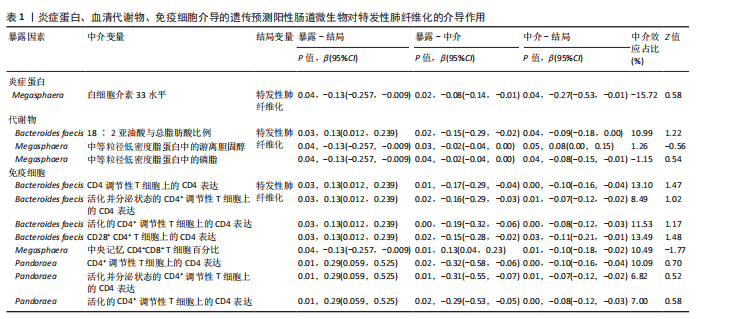
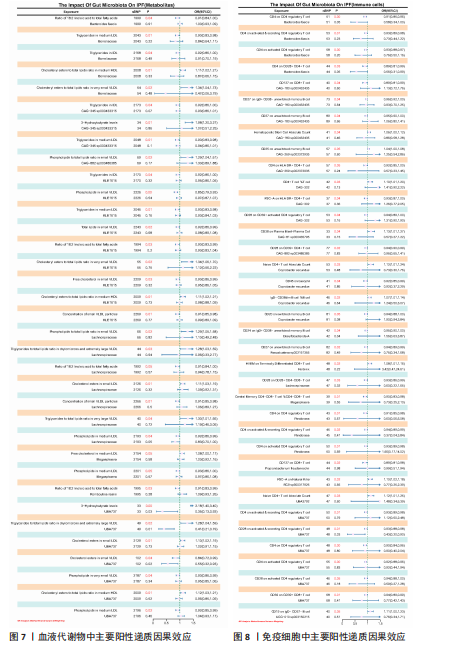
2.2 特发性肺纤维化对肠道微生物群的因果效应 通过逆方差加权法初步筛选出11种与特发性肺纤维化具有反向因果关系的肠道微生物群(图3,4),随后采用了20种不同的方法来排除异常值。经严格筛选有2种显著肠道微生物群被认为对特发性肺纤维化具有显著的反向因果效应,但与2.1结果无重叠,提示先前鉴定的3种肠道微生物群不存在与特发性肺纤维化的反向因果关系。 2.3 肠道微生物群对免疫细胞、炎症蛋白及血清代谢物的因果效应 为进一步探究肠道微生物群与特发性肺纤维化因果关系中可能存在的机制,该研究引入了233种代谢物、731种免疫细胞和91种炎症蛋白进行单变量孟德尔随机化分析。结果显示,Bacteroides faecis、Megasphaera、Pandoraea与以下生物标志物存在因果关系:代谢物方面包括18∶2亚油酸/总脂肪酸比值、中等粒径低密度脂蛋白中的游离胆固醇与磷脂;免疫细胞方面涉及包括CD4在多种调节性T细胞亚群中的表达水平、中央记忆CD4?CD8? T细胞的比例;炎症蛋白方面则以白细胞介素33为主。相关性分析显示,Bacteroides faecis与亚油酸比值及调节性T细胞中CD4表达均呈负相关;而Pandoraea在免疫细胞表达方面亦显示出负相关趋势;Megasphaera与白细胞介素33、低密度脂蛋白相关代谢物呈负相关,与中央记忆T细胞呈正相关。这些结果提示肠道微生物群可能通过调控代谢途径和免疫炎症通路影响特发性肺纤维化的发展(图5-8)。 2.4 免疫细胞、炎症蛋白及血清代谢物的中介效应 在该研究中,中央记忆CD4?CD8? T细胞占CD4?CD8? T细胞的比率(10.49%)以及中等粒径低密度脂蛋白中的游离胆固醇(1.26%)被鉴定为Megasphaera影响特发性肺纤维化风险的介导因子。 而Bacteroides faecis的中介结果显示CD4在CD28+ CD4+ T细胞(13.49%)的表达水平、CD4在CD4 Tregs(13.10%)的表达水平、CD4在活化的CD4 Tregs (11.53%)的表达水平、CD4在活化并分泌的CD4 Tregs (8.49%)的表达水平以及18∶2亚油酸/总脂肪酸比率(10.99%)均"
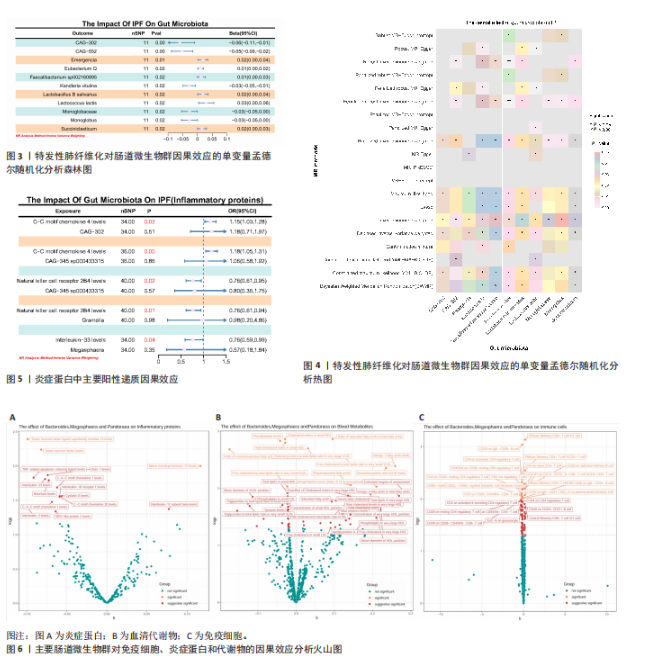
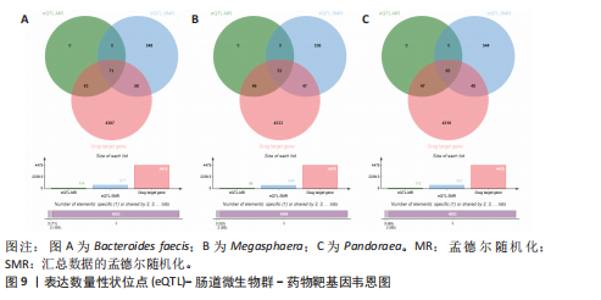
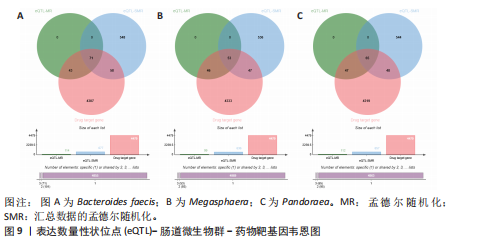
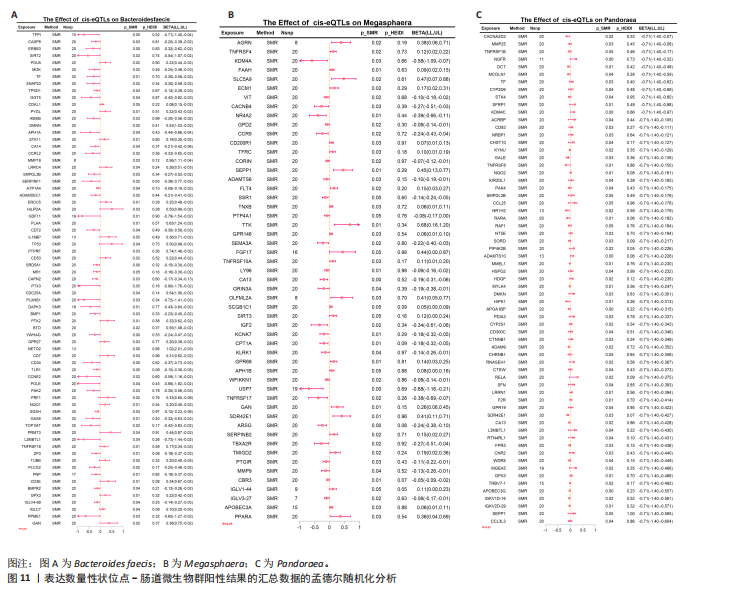
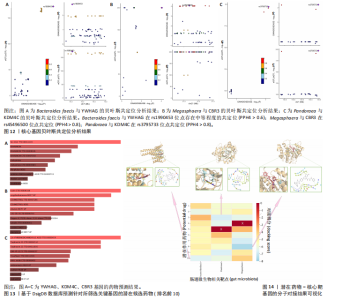
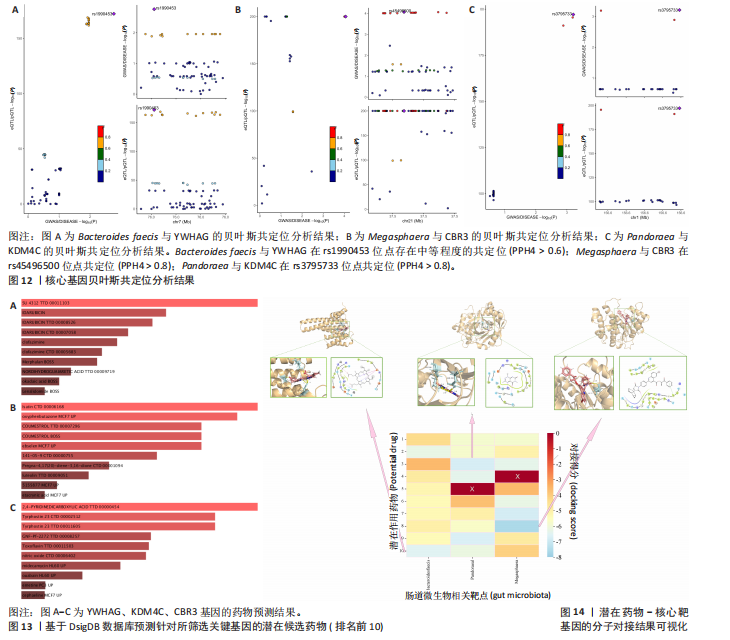
介导了其对特发性肺纤维化风险的预测。介导效应比率均超过8%,提示T细胞和脂肪酸可能是Bacteroides faecis影响特发性肺纤维化的关键介导因素。值得注意的是,这4种免疫细胞均与CD4 T细胞相关。此外,CD4在CD4 Tregs (10.09%)的表达水平、CD4在活化的CD4 Tregs (7.00%)的表达水平、CD4在活化并分泌的CD4 Tregs (6.82%)的表达水平介导了Pandoraea对特发性肺纤维化风险的影响。综合考虑结果认为CD4 T细胞在Bacteroides faecis、Megasphaera、Pandoraea与特发性肺纤维化的因果关系中起到了介导作用(表1)。 2.5 孟德尔随机化-汇总数据的孟德尔随机化-药物靶点交集分析及贝叶斯共定位分析 基于孟德尔随机化-汇总数据的孟德尔随机化的药靶分析研究结果发现,与Bacteroides faecis相关的基因表达产生了114个显著的孟德尔随机化结果,汇总数据的孟德尔随机化分析则识别出677个显著结果。将这些结果与4 479个药物靶点基因交集后,筛选出71个关键基因。对于Megasphaera,孟德尔随机化分析识别出99个显著结果,汇总数据的孟德尔随机化分析获得636个显著结果,交集后筛选出53个关键基因。对于Pandoraea,孟德尔随机化分析识别出112个显著结果,汇总数据的孟德尔随机化分析识别出657个显著结果,交集后筛选出65个关键基因(图9-11)。 贝叶斯共定位分析结果表明,Bacteroides faecis与YWHAG在rs1990453位点存在中等程度的共定位(PPH4 > 0.6);Megasphaera与CBR3在rs45496500位点共定位(PPH4 > 0.8);Pandoraea与KDM4C在rs3795733位点共定位(PPH4 > 0.8)(图12)。 2.6 治疗靶点及药物预测 评估药物-蛋白互作是判断基因能否作为潜在治疗靶点的重要途径,该研究借助DSigDB数据库预测了针对所筛选关键基因的潜在候选药物(预测结果见图13),对排名前10的组合进行了分子对接和可视化分析,结果提示大多数对接组合显示出良好的对接得分。基于分子对接结果推测YWHAG的潜在靶向药物为protoporphyrin IX,KDM4C的潜在靶向药物为GNF-Pf-2272,CBR3的潜在靶向药物为6-溴-2-[4-(3,5-二氧亚基-4-氮杂四环[5.3.2.08,10.02,6]十二-11-烯-4-基)苯基]喹啉-4-甲酸-1-氧亚基-1-苯基丙-2-基酯(5155877)(图14)。"
| [1] GLASPOLE I, BONELLA F, BARGAGLI E, et al. Efficacy and safety of nintedanib in patients with idiopathic pulmonary fibrosis who are elderly or have comorbidities. Respir Res. 2021;22(1):125. [2] 程雪,荆焕熙,张运克,等.肺痹方干预肺纤维化小鼠肺泡上皮细胞线粒体途径凋亡的机制[J].中国组织工程研究,2025,29(11):2334-2339. [3] CANI PD. Human gut microbiome: hopes, threats and promises. Gut. 2018;67(9):1716-1725. [4] ROOKS MG, GARRETT WS. Gut microbiota, metabolites and host immunity. Nat Rev Immunol. 2016;16(6):341-352. [5] CLARKE TB. Early innate immunity to bacterial infection in the lung is regulated systemically by the commensal microbiota via nod-like receptor ligands. Infect Immun. 2014;82(11):4596-4606. [6] YANG D, CHEN X, WANG J, et al. Dysregulated Lung Commensal Bacteria Drive Interleukin-17B Production to Promote Pulmonary Fibrosis through Their Outer Membrane Vesicles. Immunity. 2019;50(3):692-706.e7. [7] KLOEPFER KM, LEE WM, PAPPAS TE, et al. Detection of pathogenic bacteria during rhinovirus infection is associated with increased respiratory symptoms and asthma exacerbations. J Allergy Clin Immunol. 2014;133(5):1301-1307. [8] VASAKOVA M, STRIZ I, SLAVCEV A, et al. Th1/Th2 cytokine gene polymorphisms in patients with idiopathic pulmonary fibrosis. Tissue Antigens. 2006;67(3):229-232. [9] MUTSAERS SE, MILES T, PRÊLE CM, et al. Emerging role of immune cells as drivers of pulmonary fibrosis. Pharmacol Ther. 2023;252:108562. [10] ROQUE W, ROMERO F. Cellular metabolomics of pulmonary fibrosis, from amino acids to lipids. Am J Physiol Cell Physiol. 2021;320(5):C689-C695. [11] YAMADA T, NAKASHIMA T, MASUDA T, et al. Intestinal overgrowth of Candida albicans exacerbates bleomycin-induced pulmonary fibrosis in mice with dysbiosis. J Pathol. 2023;261(2):227-237. [12] 殷月,冷思逸,靳攀,等.肠道菌群、运动干预与呼吸系统疾病[J].中国组织工程研究,2025, 29(14):3034-3042. [13] VÕSA U, CLARINGBOULD A, WESTRA HJ, et al. Large-scale cis- and trans-eQTL analyses identify thousands of genetic loci and polygenic scores that regulate blood gene expression. Nat Genet. 2021;53(9):1300-1310. [14] FINAN C, GAULTON A, KRUGER FA, et al. The druggable genome and support for target identification and validation in drug development. Sci Transl Med. 2017;9(383):eaag1166. [15] QIN Y, HAVULINNA AS, LIU Y, et al. Combined effects of host genetics and diet on human gut microbiota and incident disease in a single population cohort. Nat Genet. 2022;54(2):134-142. [16] ORRÙ V, STERI M, SIDORE C, et al. Complex genetic signatures in immune cells underlie autoimmunity and inform therapy. Nat Genet. 2020;52(10): 1036-1045. [17] ZHAO JH, STACEY D, ERIKSSON N, et al. Genetics of circulating inflammatory proteins identifies drivers of immune-mediated disease risk and therapeutic targets. Nat Immunol. 2023;24(9):1540-1551. [18] KARJALAINEN MK, KARTHIKEYAN S, OLIVER-WILLIAMS C, et al. Genome-wide characterization of circulating metabolic biomarkers. Nature. 2024;628(8006):130-138. [19] ZHAO G, CAI Y, WANG Y, et al. Genetically predicted blood metabolites mediate the association between circulating immune cells and pancreatic cancer: A Mendelian randomization study. J Gene Med. 2024;26(5):e3691. [20] GONG Y, WANG J, PAN M, et al. Harmine inhibits pulmonary fibrosis through regulating DNA damage repair-related genes and activation of TP53-Gadd45α pathway. Int Immunopharmacol. 2024;138:112542. [21] QI T, WU Y, ZENG J, et al. Identifying gene targets for brain-related traits using transcriptomic and methylomic data from blood. Nat Commun. 2018; 9(1):2282. [22] ZHU Z, ZHANG F, HU H, et al. Integration of summary data from GWAS and eQTL studies predicts complex trait gene targets. Nat Genet. 2016;48(5):481-487. [23] GIAMBARTOLOMEI C, VUKCEVIC D, SCHADT EE, et al. Bayesian test for colocalisation between pairs of genetic association studies using summary statistics. PLoS Genet. 2014;10(5):e1004383. [24] SI S, LIU H, XU L, et al. Identification of novel therapeutic targets for chronic kidney disease and kidney function by integrating multi-omics proteome with transcriptome. Genome Med. 2024;16(1):84. [25] YOO M, SHIN J, KIM J, et al. DSigDB: drug signatures database for gene set analysis. Bioinformatics. 2015;31(18):3069-3071. [26] KULESHOV MV, JONES MR, ROUILLARD AD, et al. Enrichr: a comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res. 2016;44(W1):W90-97. [27] WONGKARNJANA A, YANAGIHARA T, KOLB MR. Treatment of idiopathic pulmonary fibrosis with Nintedanib: an update. Expert Rev Respir Med. 2019;13(12):1139-1146. [28] 陈雯,陈丽萍.肺部微生物群在肺纤维化疾病中的研究进展[J].中国当代医药,2023,30(12):44-49. [29] TANG J, XU L, ZENG Y, et al. Effect of gut microbiota on LPS-induced acute lung injury by regulating the TLR4/NF-kB signaling pathway. Int Immunopharmacol. 2021;91:107272. [30] HILTY M, BURKE C, PEDRO H, et al. Disordered microbial communities in asthmatic airways. PLoS One. 2010;5(1):e8578. [31] SEELIGER B, CARLEO A, WENDEL-GARCIA PD, et al. Changes in serum metabolomics in idiopathic pulmonary fibrosis and effect of approved antifibrotic medication. Front Pharmacol. 2022; 13:837680. [32] MOHEBALI N, WEIGEL M, HAIN T, et al. Faecalibacterium prausnitzii, Bacteroides faecis and Roseburia intestinalis attenuate clinical symptoms of experimental colitis by regulating Treg/Th17 cell balance and intestinal barrier integrity. Biomed Pharmacother. 2023;167:115568. [33] NARAYANA JK, ALIBERTI S, MAC AOGÁIN M, et al. Microbial Dysregulation of the Gut-Lung Axis in Bronchiectasis. Am J Respir Crit Care Med. 2023;207(7):908-920. [34] DICKSON RP, SINGER BH, NEWSTEAD MW, et al. Enrichment of the lung microbiome with gut bacteria in sepsis and the acute respiratory distress syndrome. Nat Microbiol. 2016;1(10):16113. [35] LU X, XIONG L, ZHENG X, et al. Structure of gut microbiota and characteristics of fecal metabolites in patients with lung cancer. Front Cell Infect Microbiol. 2023;13:1170326. [36] WANG Z, LIU J, LI F, et al. Mechanisms of Qingyi Decoction in Severe Acute Pancreatitis-Associated Acute Lung Injury via Gut Microbiota: Targeting the Short-Chain Fatty Acids-Mediated AMPK/NF-κB/NLRP3 Pathway. Microbiol Spectr. 2023;11(4):e0366422. [37] ZHANG M, SHEN L, ZHOU X, et al. The Microbiota of Human Lung of Pulmonary Tuberculosis and the Alteration Caused by Anti-tuberculosis Drugs. Curr Microbiol. 2022;79(11):321. [38] WU X, LI RF, LIN ZS, et al. Coinfection with influenza virus and non-typeable Haemophilus influenzae aggregates inflammatory lung injury and alters gut microbiota in COPD mice. Front Microbiol. 2023;14:1137369. [39] FARSI Y, TAHVILDARI A, ARBABI M, et al. Diagnostic, Prognostic, and Therapeutic Roles of Gut Microbiota in COVID-19: A Comprehensive Systematic Review. Front Cell Infect Microbiol. 2022;12:804644. [40] CHAU J, YADAV M, LIU B, et al. Prospective correlation between the patient microbiome with response to and development of immune-mediated adverse effects to immunotherapy in lung cancer. BMC Cancer. 2021;21(1):808. [41] LU HF, LI A, ZHANG T, et al. Disordered oropharyngeal microbial communities in H7N9 patients with or without secondary bacterial lung infection. Emerg Microbes Infect. 2017;6(12):e112. [42] MA S, ZHANG F, ZHOU F, et al. Metagenomic analysis reveals oropharyngeal microbiota alterations in patients with COVID-19. Signal Transduct Target Ther. 2021;6(1):191. [43] LU KY, ALQADERI H, BIN HASAN S, et al. Sputum production and salivary microbiome in COVID-19 patients reveals oral-lung axis. PLoS One. 2024; 19(7):e0300408. [44] DRUMMOND MH. The base sequence of the nifF gene of Klebsiella pneumoniae and homology of the predicted amino acid sequence of its protein product to other flavodoxins. Biochem J. 1985;232(3):891-896. [45] GREEN H, JONES AM. The microbiome and emerging pathogens in cystic fibrosis and non-cystic fibrosis bronchiectasis. Semin Respir Crit Care Med. 2015;36(2):225-235. [46] DEGAND N, LOTTE R, DECONDÉ LE BUTOR C, et al. Epidemic spread of Pandoraea pulmonicola in a cystic fibrosis center. BMC Infect Dis. 2015;15:583. [47] COSTELLO A, HERBERT G, FABUNMI L, et al. Virulence of an emerging respiratory pathogen, genus Pandoraea, in vivo and its interactions with lung epithelial cells. J Med Microbiol. 2011;60(Pt 3):289-299. [48] MA Z, ZOU X, LIN J, et al. Sepsis Due to Pandoraea sputorum Infection After Multiple Trauma in a Non-Cystic Fibrosis Patient: A Case Report from Southeast China. Infect Drug Resist. 2022;15:7043-7052. [49] KOLAHIAN S, FERNANDEZ IE, EICKELBERG O, et al. Immune Mechanisms in Pulmonary Fibrosis. Am J Respir Cell Mol Biol. 2016;55(3):309-322. [50] HEUKELS P, MOOR CC, VON DER THÜSEN JH, et al. Inflammation and immunity in IPF pathogenesis and treatment. Respir Med. 2019;147:79-91. [51] SHI X, PAN Z, CAI W, et al. Identification and immunological characterization of cuproptosis-related molecular clusters in idiopathic pulmonary fibrosis disease. Front Immunol. 2023;14:1171445. [52] CELADA LJ, KROPSKI JA, HERAZO-MAYA JD, et al. PD-1 up-regulation on CD4+ T cells promotes pulmonary fibrosis through STAT3-mediated IL-17A and TGF-β1 production. Sci Transl Med. 2018; 10(460):eaar8356. [53] GILANI SR, VUGA LJ, LINDELL KO, et al. CD28 down-regulation on circulating CD4 T-cells is associated with poor prognoses of patients with idiopathic pulmonary fibrosis. PLoS One. 2010;5(1):e8959. [54] BARDE F, LORENZON R, VICAUT E, et al. Induction of regulatory T cells and efficacy of low-dose interleukin-2 in systemic sclerosis: interventional open-label phase 1-phase 2a study. RMD Open. 2024;10(2):e003500. [55] KOTSIANIDIS I, NAKOU E, BOUCHLIOU I, et al. Global impairment of CD4+CD25+FOXP3+ regulatory T cells in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med. 2009;179(12):1121-1130. [56] BIRJANDI SZ, PALCHEVSKIY V, XUE YY, et al. CD4(+)CD25(hi)Foxp3(+) Cells Exacerbate Bleomycin-Induced Pulmonary Fibrosis. Am J Pathol. 2016; 186(8):2008-2020. [57] ANDUGULAPATI SB, GOURISHETTI K, TIRUNAVALLI SK, et al. Biochanin-A ameliorates pulmonary fibrosis by suppressing the TGF-β mediated EMT, myofibroblasts differentiation and collagen deposition in in vitro and in vivo systems. Phytomedicine. 2020;78:153298. [58] LI ZY, XIAO L, LIN G, et al. Contribution of tissue transglutaminase to the severity of hepatic fibrosis resulting from Schistosoma japonicum infection through the regulation of IL-33/ST2 expression. Parasit Vectors. 2019;12(1):302. [59] WANG QR, LIU SS, MIN JL, et al. CCL17 drives fibroblast activation in the progression of pulmonary fibrosis by enhancing the TGF-β/Smad signaling. Biochem Pharmacol. 2023;210:115475. [60] 王璐,徐婷贞,周林水,等.M2型巨噬细胞通过CCL22-CCR4信号促进肺泡上皮细胞EMT[J].中国病理生理杂志,2022,38(10):1848-1855. [61] 汪泽婷,蒋钰玉,王晓慧,等.巨噬细胞在肺纤维化中的作用研究进展[J].中国免疫学杂志, 2025,41(3):513-521. [62] CHANDA D, OTOUPALOVA E, SMITH SR, et al. Developmental pathways in the pathogenesis of lung fibrosis. Mol Aspects Med. 2019;65:56-69. [63] SHI X, CHEN Y, LIU Q, et al. LDLR dysfunction induces LDL accumulation and promotes pulmonary fibrosis. Clin Transl Med. 2022;12(1):e711. [64] CHU SG, VILLALBA JA, LIANG X, et al. Palmitic Acid-Rich High-Fat Diet Exacerbates Experimental Pulmonary Fibrosis by Modulating Endoplasmic Reticulum Stress. Am J Respir Cell Mol Biol. 2019; 61(6):737-746. [65] ROMERO F, HONG X, SHAH D, et al. Lipid Synthesis Is Required to Resolve Endoplasmic Reticulum Stress and Limit Fibrotic Responses in the Lung. Am J Respir Cell Mol Biol. 2018;59(2):225-236. [66] NEIGHBORS M, LI Q, ZHU SJ, et al. Bioactive lipid lysophosphatidic acid species are associated with disease progression in idiopathic pulmonary fibrosis. J Lipid Res. 2023;64(6):100375. [67] EL AGHA E, MOISEENKO A, KHEIROLLAHI V, et al. Two-Way Conversion between Lipogenic and Myogenic Fibroblastic Phenotypes Marks the Progression and Resolution of Lung Fibrosis. Cell Stem Cell. 2017;20(2):261-273.e3. [68] ONAL G, KUTLU O, GOZUACIK D, et al. Lipid Droplets in Health and Disease. Lipids Health Dis. 2017;16(1):128. [69] WANG P, DENG Y, FU X. MiR-509-5p suppresses the proliferation, migration, and invasion of non-small cell lung cancer by targeting YWHAG. Biochem Biophys Res Commun. 2017;482(4):935-941. [70] WANG J, PAN X, LI J, et al. TXNDC9 knockdown inhibits lung adenocarcinoma progression by targeting YWHAG. Mol Med Rep. 2022;25(6):203. [71] YOO JO, KWAK SY, AN HJ, et al. miR-181b-3p promotes epithelial-mesenchymal transition in breast cancer cells through Snail stabilization by directly targeting YWHAG. Biochim Biophys Acta. 2016;1863(7 Pt A):1601-1611. [72] FUJITA H, NAGAKAWA K, KOBUCHI H, et al. Phytoestrogen Suppresses Efflux of the Diagnostic Marker Protoporphyrin IX in Lung Carcinoma. Cancer Res. 2016;76(7):1837-1846. [73] HYUN DS, KIM HT, JHEON SH, et al. A preliminary study of protoporphyrin-IX as a potential candidate for identification of lung cancer cells using fluorescence microscopy. Photodiagnosis Photodyn Ther. 2009;6(3-4):221-226. [74] SHU Y, MA M, PAN X, et al. Cobalt protoporphyrin-induced nano-self-assembly for CT imaging, magnetic-guidance, and antioxidative protection of stem cells in pulmonary fibrosis treatment. Bioact Mater. 2022;21:129-141. [75] MINGONE CJ, GUPTE SA, CHOW JL, et al. Protoporphyrin IX generation from delta-aminolevulinic acid elicits pulmonary artery relaxation and soluble guanylate cyclase activation. Am J Physiol Lung Cell Mol Physiol. 2006;291(3):L337-L344. [76] ALZAHRANI AR, MOHAMED DI, ABO NAHAS HH, et al. Trimetazidine Alleviates Bleomycin-Induced Pulmonary Fibrosis by Targeting the Long Noncoding RNA CBR3-AS1-Mediated miRNA-29 and Resistin-Like Molecule alpha 1: Deciphering a Novel Trifecta Role of LncRNA CBR3-AS1/miRNA-29/FIZZ1 Axis in Lung Fibrosis. Drug Des Devel Ther. 2024;18:3959-3986. [77] HOU M, WU N, YAO L. LncRNA CBR3-AS1 potentiates Wnt/β-catenin signaling to regulate lung adenocarcinoma cells proliferation, migration and invasion. Cancer Cell Int. 2021;21(1):36. [78] EBERT B, KISIELA M, MALÁTKOVÁ P, et al. Regulation of human carbonyl reductase 3 (CBR3; SDR21C2) expression by Nrf2 in cultured cancer cells. Biochemistry. 2010;49(39):8499-8511. [79] JIE X, FONG WP, ZHOU R, et al. USP9X-mediated KDM4C deubiquitination promotes lung cancer radioresistance by epigenetically inducing TGF-β2 transcription. Cell Death Differ. 2021;28(7):2095-2111. [80] JIE X, CHEN Y, ZHAO Y, et al. Targeting KDM4C enhances CD8+ T cell mediated antitumor immunity by activating chemokine CXCL10 transcription in lung cancer. J Immunother Cancer. 2022;10(2):e003716. [81] SALMINEN A, KAARNIRANTA K, KAUPPINEN A. Hypoxia-Inducible Histone Lysine Demethylases: Impact on the Aging Process and Age-Related Diseases. Aging Dis. 2016;7(2):180-200. [82] PUHL AC, FERNANDES RS, GODOY AS, et al. The protein disulfide isomerase inhibitor 3-methyltoxoflavin inhibits Chikungunya virus. Bioorg Med Chem. 2023;83:117239. |
| [1] | Rong Xiangbin, , Zheng Haibo, Mo Xueshen, Hou Kun, Zeng Ping, . Plasma metabolites, immune cells, and hip osteoarthritis: causal inference based on GWAS data from European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1028-1035. |
| [2] | Gu Fucheng, Yang Meixin, Wu Weixin, Cai Weijun, Qin Yangyi, Sun Mingyi, Sun Jian, Geng Qiudong, Li Nan. Effects of Guilu Erxian Glue on gut microbiota in rats with knee osteoarthritis: machine learning and 16S rDNA analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1058-1072. |
| [3] | Liu Chu, Qiu Boyuan, Tong Siwen, He Linyuwei, Chen Haobo, Ou Zhixue. A genetic perspective reveals the relationship between blood metabolites and osteonecrosis: an analysis of information from the FinnGen database in Finland [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(3): 785-794. |
| [4] | Wang Wei, Chen Jun, Jia Shaohui, Xue Xinxuan, Dong Kunwei. Mechanisms by which exercise regulates gut microbiota in the prevention and treatment of non-alcoholic fatty liver disease [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6328-6336. |
| [5] | Zhang Zheng, Zhang Yibo, Xu Bin, Yan Shichao, Guo Hui. Sarcopenia and non-alcoholic fatty liver disease: analysis of the gut microbiota [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 6000-6009. |
| [6] | Li Yiguang, Guo Haonan, Ding Xiaotao, Yuan Mengyao, Jiang Lijin, Fan Xinfeng, Feng Yan. Visual analysis of research hotspots in the field of gut microbiota in the elderly at home and abroad [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 6071-6080. |
| [7] | Chai Jinlian, Liang Xuezhen, Sun Tiefeng, Li Shudong, Li Wei, Li Guangzheng, Yu Huayun, Wang Ping. Mechanistic insights into how Cervi Cornus Colla regulates the intestinal flora-bile acid metabolic pathway to alleviate steroid-induced osteonecrosis of the femoral head in a rat model [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(18): 4568-4581. |
| [8] | Wu Yilin, Tian Hongying, Sun Jiale, Jiao Jiajia, Zhao Zihan, Shao Jinhuan, Zhao Kaiyue, Zhou Min, Li Qian, Li Zexin, Yue Changwu. Intervention effect and mechanism of Compound Herba Gueldenstaedtiae in a mouse model of breast hyperplasia [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(17): 4377-4389. |
| [9] | Zou Yuxiong, Liu Xiaomeng, Liu Ying, Zhu Yue, Li Shuming, Guo Fangyang, Yu Xinyu, Nie Heyun, Liu Qian, Ao Meiying. Cerebral palsy decoction improves cerebral palsy in male and female young rats: mechanisms based on the “gut-brain-muscle” axis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(16): 4054-4066. |
| [10] | Sun Long, Wu Haiyang, Tong Linjian, Liu Rui, Yang Weiguang, Xiao Jian, Liu Lice, Sun Zhiming. Regulatory mechanism of leptin in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(12): 3100-3108. |
| [11] | Wang Tao, Min Youjiang, Wang Min, Wang Shunpu, Li Le, Zhang Chen, Xiao Weiping, Yu Yiping. Causal relationship between gut microbiota and amyotrophic lateral sclerosis: sample analysis from the IEU Open GWAS Database [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(12): 3182-3189. |
| [12] | Wu Fangjia, Lei Senlin, Li Xianhui, Yang Yang. Aerobic and resistance exercise interventions in a mouse model of nonalcoholic fatty liver disease: correlation between gut microbiota and irisin [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(12): 3029-3043. |
| [13] | Qiu Xueli, Cui Hao, Wu Chenyang, Tao Lide, Yao Yuqian, Tian Bo, Bai Jinyu, Zhang Yingzi. Gut microbiota tryptophan metabolite indole-3-propionic acid alleviates inflammatory bowel disease-related osteoporosis in a mouse model [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(10): 2413-2421. |
| [14] | Qi Xiang, Cao Shan, Chen Jian, Zhang Yijia, Liu Keke, Xu Zifu, Liu Wang, Fu Xiaoxiao, Yin Xiaolei. Screening of genes related to mitochondrial dysfunction and ferroptosis in atherosclerosis and target prediction of regulatory traditional Chinese medicine [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(10): 2641-2652. |
| [15] | Li Jiagen, Chen Yueping, Huang Keqi, Chen Shangtong, Huang Chuanhong. The construction and validation of a prediction model based on multiple machine learning algorithms and the immunomodulatory analysis of rheumatoid arthritis from the perspective of mitophagy [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(在线): 1-15. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||