Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (28): 7388-7395.doi: 10.12307/2026.768
Previous Articles Next Articles
Sarcopenia and cognitive impairment: a data analysis based on European population databases
- 1School of Physical Education, Yunnan Normal University, Kunming 650500, Yunnan Province, China; 2The Third Affiliated Hospital of Yunnan University of Chinese Medicine (Rehabilitation Department, Kunming Hospital of Traditional Chinese Medicine), Kunming 650011, Yunnan Province, China
-
Received:2025-08-18Revised:2025-10-21Online:2026-10-08Published:2026-02-24 -
Contact:Chen Qigang, Chief physician, Professor, Master’s supervisor, The Third Affiliated Hospital of Yunnan University of Chinese Medicine (Rehabilitation Department, Kunming Hospital of Traditional Chinese Medicine), Kunming 650011, Yunnan Province, China Co-corresponding author: Shen Zhen, PhD, The Third Affiliated Hospital of Yunnan University of Chinese Medicine (Rehabilitation Department, Kunming Hospital of Traditional Chinese Medicine), Kunming 650011, Yunnan Province, China -
About author:Yin Xingxiao, MS candidate, School of Physical Education, Yunnan Normal University, Kunming 650500, Yunnan Province, China -
Supported by:the National Natural Science Foundation of China, No. 82360943 (to SZ); Young Talent Special Project of Yunnan Province “Xing Dian Talent Support Program,” No. XDYC-QNRC-2022-0609 (to SZ); Chinese Medicine Joint Project of Yunnan Provincial Department of Science and Technology, Nos. 202101AZ070001-257 and 202101AZ070001-123 (to SZ); Basic Research Special Project of Yunnan Provincial Department of Science and Technology, No. 202201AU070120 (to SZ)
CLC Number:
Cite this article
Yin Xingxiao, Peng Hao, Song Yanping, Yao Na, Shen Zhen, Jiang Yang, Chen Hongbo, Huang Li, Song Yueyu, Li Yanqi, Chen Qigang. Sarcopenia and cognitive impairment: a data analysis based on European population databases[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7388-7395.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
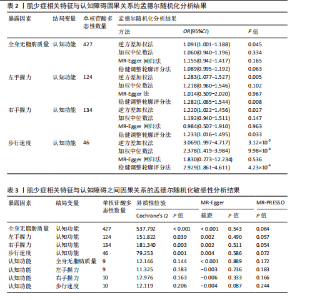
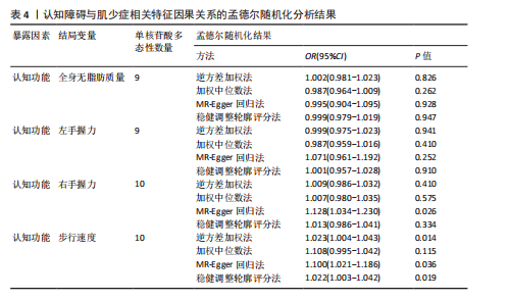
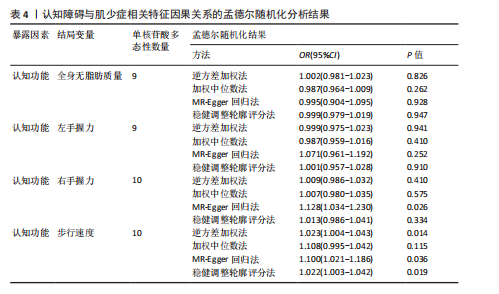
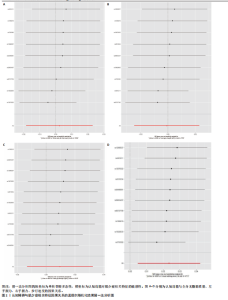
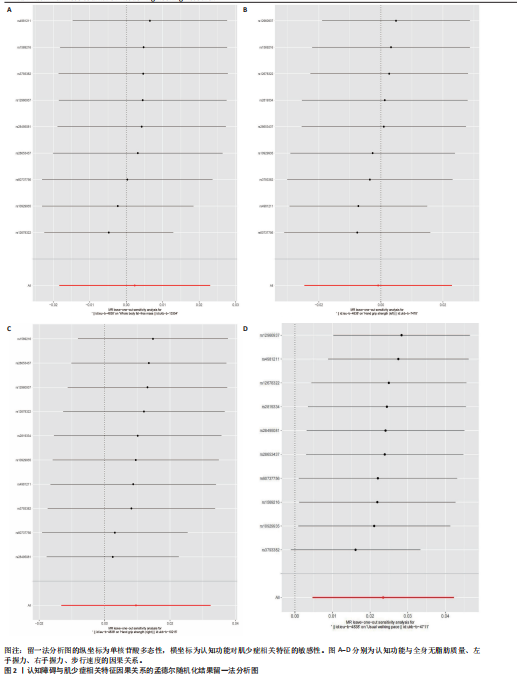
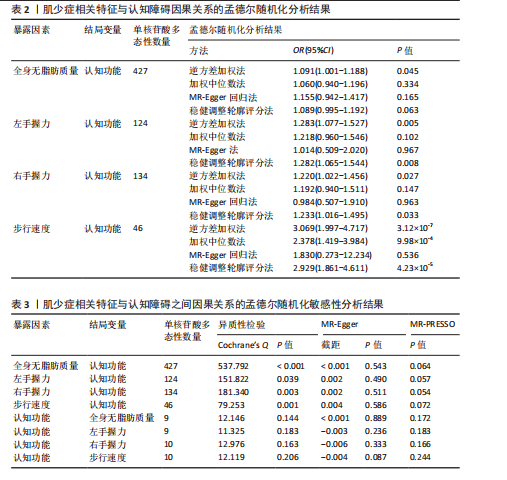
2.1 单核苷酸多态性的选择和验证 研究基于质量控制标准筛选单核苷酸多态性位点,并通过暴露因素与结局变量的匹配分析进行遗传变异选择。以F统计量> 10作为判断标准,确保所选单核苷酸多态性具有足够的强度以有效降低潜在偏倚的影响。 正向分析结果显示:在全身无脂肪质量与认知功能的关联性分析中,经过P值筛选和去除连锁不平衡处理后,共纳入556个单核苷酸多态性位点;经暴露因素与结局变量的效应等位基因对齐后保留498个单核苷酸多态性;进一步剔除回文单核苷酸多态性后剩余431个位点;最终通过MR-PRESSO方法检验并排除异常值,确定427(29.874 < F < 779.566)个单核苷酸多态性位点纳入最终分析。在左手握力与认知功能的关联性分析中,经过P值筛选和去除连锁不平衡处理后,共纳入157个单核苷酸多态性位点;经暴露因素与结局变量的效应等位基因对齐后保留143个单核苷酸多态性;进一步剔除回文单核苷酸多态性后剩余126个位点;最终通过MR-PRESSO方法检验并排除异常值,确定124(29.778 < F < 191.541)个单核苷酸多态性位点纳入最终分析。在右手握力与认知功能的关联性分析中,经过P值筛选和去除连锁不平衡处理后,共纳入176个单核苷酸多态性位点;经暴露因素与结局变量的效应等位基因对齐后保留157个单核苷酸多态性;进一步剔除回文单核苷酸多态性后剩余136个位点;最终通过MR-PRESSO方法检验并排除异常值,确定134(29.896 < F < 231.736)个单核苷酸多态性位点纳入最终分析。在步行速度与认知功能的关联性分析中,经过P值筛选和去除连锁不平衡处理后,共纳入57个单核苷酸多态性位点;经暴露因素与结局变量的效应等位基因对齐后保留49个单核苷酸多态性;进一步剔除回文单核苷酸多态性后剩余46个位点;最终通过MR-PRESSO方法检验并排除异常值,确定46(29.791 < F < 101.566)个单核苷酸多态性位点纳入最终分析。 反向分析结果表明:在认知功能与全身无脂肪质量的关联性分析中,经过P值筛选和去除连锁不平衡处理后,共纳入12个单核苷酸多态性位点;经暴露因素与结局变量的效应等位基因对齐后保留12个单核苷酸多态性;进一步剔除回文单核苷酸多态性后剩余10个位点;最终通过MR-PRESSO方法检验并排除异常值,确定9(24.637 < F < 29.915)个单核苷酸多态性位点纳入最终分析。在认知功能与左手握力的关联性分析中,经过P值筛选和去除连锁不平衡处理后,共纳入12个单核苷酸多态性位点;经暴露因素与结局变量的效应等位基因对齐后保留12个单核苷酸多态性;进一步剔除回文单核苷酸多态性后剩余10个位点;最终通过MR-PRESSO方法检验并排除异常值,确定9(24.637 < F < 29.915)个单核苷酸多态性位点纳入最终分析。在认知功能与右手握力的关联性分析中,经过P值筛选和去除连锁不平衡处理后,共纳入12个单核苷酸多态性位点;经暴露因素与结局变量的效应等位基因对齐后保留12个单核苷酸多态性;进一步剔除回文单核苷酸多态性后剩余12个位点;最终通过MR-PRESSO方法检验并排除异常值,确定10(24.637 < F < 31.143)个单核苷酸多态性位点纳入最终分析。在认知功能与步行速度的关联性分析中,经过P值筛选和去除连锁不平衡处理后,共纳入12个单核苷酸多态性位点;经暴露因素与结局变量的效应等位基因对齐后保留12个单核苷酸多态性;进一步剔除回文单核苷酸多态性后剩余12个位点;最终通过MR-PRESSO方法检验并排除异常值,确定10(24.637 < F < 31.143)个单核苷酸多态性位点纳入最终分析。 2.2 肌少症相关特征与认知障碍之间的因果关系 逆方差加权法结果显示,全身无脂肪质量(OR=1.091,95%CI:1.001-1.188,P=0.045)、左手握力OR=1.283,95%CI:1.077-1.527,P=0.005)、右手握力(OR=1.220,95%CI:1.022-1.456,P=0.027)和步行速度(OR=3.069,95%CI:1.997-4.717,P < 0.001)均与认知功能呈显著正相关。为进一步验证结果的可靠性,研究采用多种补充分析方法进行稳健性检验。在全身无脂肪质量与认知功能的关联分析中,虽然MR-Egger回归法、加权中位数法和稳健调整轮廓评分法未达到统计学显著性水平(P > 0.05),但3种方法所得效应值的方向与逆方差加权法一致,提示该结果具有稳健性。在握力与认知功能的关联分析中,MR-Egger回归法和加权中位数法未发现显著关联,但稳健调整轮廓评分法的分析结果与逆方差加权法一致(P < 0.05)。对于步行速度与认知功能的关联,虽然MR-Egger回归法未得出显著结果,但加权中位数法和稳健调整轮廓评分法的分析均支持这一正向关联(P < 0.05),进一步验证了研究结论的可靠性,见表2。敏感性分析结果显示:Cochran’s Q检验提示存在异质性(全身无脂肪质量:Q=537.792,P < 0.001;左手握力:Q=151.822,P=0.039;右手握力:Q=181.340,P=0.003;步行速度:Q=79.253,P=0.001),故采用随机效应模型解释这一结果。MR-Egger截距检验和MR-PRESSO全局检验显示不存在多效性(P > 0.05),见表3。 此外,留一法敏感性分析显示,肌少症相关特征与认知功能之间的因果关联不受单个单核苷酸多态性的影响,见图1。 2.3 认知障碍与肌少症相关特征之间的因果关系 为了探究认知障碍与肌少症的因果关系,研究进行了反向孟德尔随机化分析。逆方差加权法结果表明,认知功能与步行速度之间存在显著的正向因果关系(OR=1.023,95%CI:1.004-1.043,P=0.014),该结果得到加权中位数法和稳健调整轮廓评分法的一致性验证(P均< 0.05)。然而,认知功能与全身无脂肪质量、左右手握力之间未发现统计学意义的因果关联,见表4。在敏感性分析方面,Cochran’s Q检验结果显示研究不存在异质性(全身无脂肪质量:Q=12.146,P=0.144;左手握力:Q=11.325,P=0.183;右手握力:Q=12.976,P=0.163;步行速度:Q=12.119,P=0.206);MR-Egger截距检验和MR-PRESSO全局检验均未发现水平多效性证据( P > 0.05),见表3。此外,通过留一法敏感性分析证实,研究结果不受单个单核苷酸多态性的影响,表明因果关系具有稳健性,见图2。"
| [1] PIKE KE, CAVUOTO MG, LI L, et al. Subjective Cognitive Decline: Level of Risk for Future Dementia and Mild Cognitive Impairment, a Meta-Analysis of Longitudinal Studies. Neuropsychol Rev. 2022;32(4):703-735. [2] CHEN X, CAO M, LIU M, et al. Association between sarcopenia and cognitive impairment in the older people: a meta-analysis. Eur Geriatr Med. 2022;13(4):771-787. [3] CHOWDHARY N, BARBUI C, ANSTEY KJ, et al. Reducing the Risk of Cognitive Decline and Dementia: WHO Recommendations. Front Neurol. 2022;12:765584. [4] SEBASTIAN MJ, KHAN SK, PAPPACHAN JM, et al. Diabetes and cognitive function: An evidence-based current perspective. World J Diabetes. 2023;14(2):92-109. [5] ALDRICH L, ISPOGLOU T, PROKOPIDIS K, et al. Acute Sarcopenia: Systematic Review and Meta-Analysis on Its Incidence and Muscle Parameter Shifts During Hospitalisation. J Cachexia Sarcopenia Muscle. 2025;16(1):e13662. [6] ALVES FM, AYTON S, BUSH AI, et al. Age-Related Changes in Skeletal Muscle Iron Homeostasis. J Gerontol A Biol Sci Med Sci. 2023;78(1):16-24. [7] SAYER AA, COOPER R, ARAI H, et al. Sarcopenia. Nat Rev Dis Primers. 2024;10(1):68. [8] LEE DY. Prevalence and Associated Factors of Dynapenia, Pre-Sarcopenia, and Sarcopenia in Korean Adults: A Cross-Sectional Epidemiological Study. Medicina (Kaunas). 2025;61(4):575. [9] AMINI N, IBN HACH M, LAPAUW L, et al. Meta-analysis on the interrelationship between sarcopenia and mild cognitive impairment, Alzheimer’s disease and other forms of dementia. J Cachexia Sarcopenia Muscle. 2024;15(4):1240-1253. [10] YANG Y, XIAO M, LENG L, et al. A systematic review and meta-analysis of the prevalence and correlation of mild cognitive impairment in sarcopenia. J Cachexia Sarcopenia Muscle. 2023;14(1):45-56. [11] HUANG J, LI M, LUO Q, et al. The association of sarcopenia, possible sarcopenia and cognitive impairment: A systematic review and meta-analysis. PLoS One. 2025;20(5):e0324258. [12] WANG X, XIAO R, LI H, et al. Correlation between Mild Cognitive Impairment and Sarcopenia: The Prospective Role of Lipids and Basal Metabolic Rate in the Link. Nutrients. 2022;14(24):5321. [13] YUAN S, LARSSON SC. Epidemiology of sarcopenia: Prevalence, risk factors, and consequences. Metabolism. 2023;144:155533. [14] SEVERINSEN MCK, PEDERSEN BK. Muscle-Organ Crosstalk: The Emerging Roles of Myokines. Endocr Rev. 2020;41(4):594-609. [15] HU Y, PENG W, REN R, et al. Sarcopenia and mild cognitive impairment among elderly adults: The first longitudinal evidence from CHARLS. J Cachexia Sarcopenia Muscle. 2022;13(6):2944-2952. [16] CIPOLLI GC, DE ASSUMPÇÃO D, BORIM FSA, et al. Cognitive Impairment Predicts Sarcopenia 9 Years Later among Older Adults. J Am Med Dir Assoc. 2023;24(8):1207-1212. [17] RAMOO K, HAIRI NN, YAHYA A, et al. Longitudinal Association between Sarcopenia and Cognitive Impairment among Older Adults in Rural Malaysia. Int J Environ Res Public Health. 2022;19(8):4723. [18] SHARMA N, SRIVASTAV AK, SAMUEL AJ, et al. Randomized clinical trial: gold standard of experimental designs-importance, advantages, disadvantages and prejudices. Revista Pesquisa em Fisioterapia. 2020;10(3):512-519. [19] LARSSON SC, BUTTERWORTH AS, BURGESS S. Mendelian randomization for cardiovascular diseases: principles and applications. Eur Heart J. 2023;44(47):4913-4924. [20] DENG MG, LIU F, LIANG Y, et al. Association between frailty and depression: A bidirectional Mendelian randomization study. Sci Adv. 2023; 9(38):eadi3902. [21] PEI YF, LIU YZ, YANG XL, et al. The genetic architecture of appendicular lean mass characterized by association analysis in the UK Biobank study. Commun Biol. 2020; 3(1):608. [22] JONES G, TRAJANOSKA K, SANTANASTO AJ, et al. Genome-wide meta-analysis of muscle weakness identifies 15 susceptibility loci in older men and women. Nat Commun. 2021;12(1):654. [23] HU MJ, TAN JS, GAO XJ, et al. Effect of Cheese Intake on Cardiovascular Diseases and Cardiovascular Biomarkers. Nutrients. 2022; 14(14):2936. [24] YANG H, CHEN L, LIU Y. Novel Causal Plasma Proteins for Hypothyroidism: A Large-scale Plasma Proteome Mendelian Randomization Analysis. J Clin Endocrinol Metab. 2023;108(2):433-442. [25] LIU C, LIU N, XIA Y, et al. Osteoporosis and sarcopenia-related traits: A bi-directional Mendelian randomization study. Front Endocrinol (Lausanne). 2022;13:975647. [26] HE J, HUANG M, LI N, et al. Genetic Association and Potential Mediators between Sarcopenia and Coronary Heart Disease: A Bidirectional Two-Sample, Two-Step Mendelian Randomization Study. Nutrients. 2023;15(13):3013. [27] DU Y, XIE B, WANG M, et al. Roles of sex hormones in mediating the causal effect of vitamin D on osteoporosis: A two-step Mendelian randomization study. Front Endocrinol (Lausanne). 2023;14:1159241. [28] SU Q, JIN C, YANG Y, et al. Association Between Autoimmune Diseases and Sarcopenia: A Two-Sample Mendelian Randomization Study. Clin Epidemiol. 2023;15:901-910. [29] ZHOU S, TAO B, GUO Y, et al. Integrating plasma protein-centric multi-omics to identify potential therapeutic targets for pancreatic cancer. J Transl Med. 2024;22(1):557. [30] NI JJ, XU Q, YAN SS, et al. Gut Microbiota and Psychiatric Disorders: A Two-Sample Mendelian Randomization Study. Front Microbiol. 2022;12: 737197. [31] KARAGEORGIOU V, TYRRELL J, MCKINLEY TJ, et al. Weak and pleiotropy robust sex-stratified Mendelian randomization in the one sample and two sample settings. Genet Epidemiol. 2023; 47(2):135-151. [32] DING M, ZHANG Z, CHEN Z, et al. Association between periodontitis and breast cancer: two-sample Mendelian randomization study. Clin Oral Investig. 2023;27(6):2843-2849. [33] KJAERGAARD AD, TEUMER A, WITTE DR, et al. Obesity and Kidney Function: A Two-Sample Mendelian Randomization Study. Clin Chem. 2022;68(3):461-472. [34] PENG TC, CHEN WL, WU LW, et al. Sarcopenia and cognitive impairment: A systematic review and meta-analysis. Clin Nutr. 2020;39(9):2695-2701. [35] XIN X, LIU Q, JIA S, et al. Correlation of muscle strength, information processing speed and cognitive function in the elderly with cognitive impairment-evidence from EEG. Front Aging Neurosci. 2025;17: 1496725. [36] KIM K, KIM H. Handgrip Strength and Cognitive Function among Elderly Koreans: Insights from the Korean Longitudinal Study of Ageing. Int J Environ Res Public Health. 2022;19(9):5262. [37] CHEN KK, LEE SY, PANG BWJ, et al. Associations of low handgrip strength and hand laterality with cognitive function and functional mobility - the Yishun Study. BMC Geriatr. 2022;22(1):677. [38] AUYEUNG TW, KWOK T, LEE J, et al. Functional decline in cognitive impairment--the relationship between physical and cognitive function. Neuroepidemiology. 2008;31(3):167-173. [39] XIE X, LI D, ZHOU M, et al. Effects of Hand Strength and Walking Speed Combined and in Isolation on the Prediction of Cognitive Decline and Dementia in Middle-Aged and Older Adults: A Systematic Review and Meta-Analysis. J Am Med Dir Assoc. 2025;26(6):105576. [40] LI H, ZHANG J, ZOU X, et al. The Bidirectional Association Between Cognitive Function and Gait Speed in Chinese Older Adults: Longitudinal Observational Study. JMIR Public Health Surveill. 2023;9:e44274. [41] SKILLBÄCK T, BLENNOW K, ZETTERBERG H, et al. Slowing gait speed precedes cognitive decline by several years. Alzheimers Dement. 2022;18(9): 1667-1676. [42] TESSIER AJ, WING SS, RAHME E, et al. Association of Low Muscle Mass With Cognitive Function During a 3-Year Follow-up Among Adults Aged 65 to 86 Years in the Canadian Longitudinal Study on Aging. JAMA Netw Open. 2022;5(7):e2219926. [43] MERCHANT RA, SEETHARAMAN S, AU L, et al. Relationship of Fat Mass Index and Fat Free Mass Index With Body Mass Index and Association With Function, Cognition and Sarcopenia in Pre-Frail Older Adults. Front Endocrinol (Lausanne). 2021;12:765415. [44] SUI SX, WILLIAMS LJ, HOLLOWAY-KEW KL, et al. Skeletal Muscle Health and Cognitive Function: A Narrative Review. Int J Mol Sci. 2020;22(1):255. [45] AUYEUNG TW, LEE JS, KWOK T, et al. Physical frailty predicts future cognitive decline - a four-year prospective study in 2737 cognitively normal older adults. J Nutr Health Aging. 2011;15(8):690-694. [46] YE JY, CHANG LF, CHEN R, et al. Prevalence of sarcopenia in middle-aged and older adults with cognitive impairment: a meta-analysis. Age Ageing. 2025;54(5):afaf114. [47] ZENG D, MIZUTANI K, QI X, et al. The association of hemopexin, muscle quality, and sarcopenia in Japanese older adults with cognitive impairment: a cross-sectional study. BMC Geriatr. 2025;25(1):332. [48] OUDBIER SJ, GOH J, LOOIJAARD SMLM, et al. Pathophysiological Mechanisms Explaining the Association Between Low Skeletal Muscle Mass and Cognitive Function. J Gerontol A Biol Sci Med Sci. 2022;77(10):1959-1968. [49] VAN DAM R, VAN ANCUM JM, VERLAAN S, et al. Lower Cognitive Function in Older Patients with Lower Muscle Strength and Muscle Mass. Dement Geriatr Cogn Disord. 2018;45(3-4):243-250. [50] OGAWA Y, KANEKO Y, SATO T, et al. Sarcopenia and Muscle Functions at Various Stages of Alzheimer Disease. Front Neurol. 2018;9:710. [51] INYUSHKIN AN, POLETAEV VS, INYUSHKINA EM, et al. Irisin/BDNF signaling in the muscle-brain axis and circadian system: A review. J Biomed Res. 2023;38(1):1-16. [52] WANG Y, ZHANG Y, WANG X, et al. Sesamol Mitigates Chronic Iron Overload-Induced Cognitive Impairment and Systemic Inflammation via IL-6 and DMT1 Regulation. Mol Nutr Food Res. 2023;67(17):e2300012. [53] TANG L, XIE D, WANG S, et al. Piezo1 Knockout Improves Post-Stroke Cognitive Dysfunction by Inhibiting the Interleukin-6 (IL-6)/Glutathione Peroxidase 4 (GPX4) Pathway. J Inflamm Res. 2024;17:2257-2270. [54] TEGELER C, O’SULLIVAN JL, BUCHOLTZ N, et al. The inflammatory markers CRP, IL-6, and IL-10 are associated with cognitive function--data from the Berlin Aging Study II. Neurobiol Aging. 2016;38:112-117. [55] KERKIS I, DA SILVA ÁP, ARALDI RP. The impact of interleukin-6 (IL-6) and mesenchymal stem cell-derived IL-6 on neurological conditions. Front Immunol. 2024;15:1400533. [56] LIU PZ, NUSSLOCK R. Exercise-Mediated Neurogenesis in the Hippocampus via BDNF. Front Neurosci. 2018;12:52. [57] CAMUSO S, LA ROSA P, FIORENZA MT, et al. Pleiotropic effects of BDNF on the cerebellum and hippocampus: Implications for neurodevelopmental disorders. Neurobiol Dis. 2022;163:105606. [58] RUIZ-GONZÁLEZ D, HERNÁNDEZ-MARTÍNEZ A, VALENZUELA PL, et al. Effects of physical exercise on plasma brain-derived neurotrophic factor in neurodegenerative disorders: A systematic review and meta-analysis of randomized controlled trials. Neurosci Biobehav Rev. 2021;128:394-405. [59] LEGER C, QUIRIÉ A, MÉLOUX A, et al. Impact of Exercise Intensity on Cerebral BDNF Levels: Role of FNDC5/Irisin. Int J Mol Sci. 2024;25(2):1213. [60] BAEK JY, JANG IY, JUNG HW, et al. Serum irisin level is independent of sarcopenia and related muscle parameters in older adults. Exp Gerontol. 2022;162:111744. [61] GULLETT JM, CHEN Z, O’SHEA A, et al. MicroRNA predicts cognitive performance in healthy older adults. Neurobiol Aging. 2020;95:186-194. [62] ALCHUS LAIFEROVÁ N, VERNEROVÁ L, NEMEC M, et al. Muscle-specific miRNAs in plasma and skeletal muscle of patients with idiopathic inflammatory myopathy are modulated by disease and training. Rheumatology (Oxford). 2025;64(7):4347-4356. [63] BIESEMANN N, RIED JS, DING-PFENNIGDORFF D, et al. High throughput screening of mitochondrial bioenergetics in human differentiated myotubes identifies novel enhancers of muscle performance in aged mice. Sci Rep. 2018;8(1):9408. |
| [1] | Yang Zhijie, Zhao Rui, Yang Haolin, Li Xiaoyun, Li Yangbo, Huang Jiachun, Lin Yanping, Wan Lei, HuangHongxing. Postmenopausal osteoporosis: predictive values of muscle mass, grip strength, and appendicular skeletal muscle index [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1073-1080. |
| [2] | Zhou Jian, Zhang Tao, Zhou Weili, Zhao Xingcheng, Wang Jun, Shen Jie, Qian Li, Lu Ming. Effects of resistance training on quadriceps mass and knee joint function in patients with osteoporosis and sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1081-1088. |
| [3] | Liu Yu, Lei Senlin, Zhou Jintao, Liu Hui, Li Xianhui. Mechanisms by which aerobic and resistance exercises improve obesity-related cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1171-1183. |
| [4] | Liu Fengzhi, Dong Yuna, Tian Wenyi, Wang Chunlei, Liang Xiaodong, Bao Lin. Gene-predicted associations between 731 immune cell phenotypes and rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1311-1319. |
| [5] | Sun Jiahe, Shi Jipeng, Zhu Tianrui, Quan Helong, Xu Hongqi. Effect of exercise intervention in elderly individuals with sarcopenia and its comorbidities: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 997-1007. |
| [6] | Ding Yu, Chen Jingwen, Chen Xiuyan, Shi Huimin, Yang Yudie, Zhou Meiqi, Cui Shuai, . Circulating inflammatory proteins and myocardial hypertrophy: large sample analysis of European populations from GWAS Catalog and FinnGen databases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1047-1057. |
| [7] | Zhang Zheng, Zhang Yibo, Xu Bin, Yan Shichao, Guo Hui. Sarcopenia and non-alcoholic fatty liver disease: analysis of the gut microbiota [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 6000-6009. |
| [8] | Yin Xingxiao, Jiang Yang, Song Yanping, Yao Na, Shen Zhen, Li Yanqi, Song Yueyu, Peng Hao, Chen Qigang. Association between sarcopenia and osteoporosis: a genome-wide data analysis in European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 6030-6039. |
| [9] | Yu Yueyue, Zhang Xu, Liu Yiwei, Meng Zihan, Hao Xinyue, Tian Chunyu, Li Ji’an, Zhang Yixin . Lipid types and knee osteoarthritis: a genome-wide association study in European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(18): 4763-4770. |
| [10] | Liu Enxu, Sun Yu, Duan Jiahao, Yang Lei, Jiang Haobo, Yang Shaofeng. Bidirectional causal interplay between Epstein-Barr virus and ankylosing spondylitis: data analysis based on the UK Biobank and FinnGen databases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(17): 4542-4547. |
| [11] | Wei Bingqi, Sun Jiahui, Chen Liu, Li Yijing, Wan Hejia, Qi Yifan, Wang Shangzeng. BTN3A2 is a key target for the development or prevention of new drugs for knee osteoarthritis: a randomization study based on drug targeting [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(16): 4021-4029. |
| [12] | Yan Wenjian, Li Yinghui, Zhang Yong. Daily diet and structural damage of the knee joint: a large-scale genetic analysis based on UK and FinnGen databases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(11): 2877-2885. |
| [13] | Huang Fengqin, Hu Yalin, Yang Boyin, Luo Xingmei. Constructing a risk prediction nomogram model for cognitive impairment in hypertensive intracerebral hemorrhage [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(10): 2466-2474. |
| [14] | Jiang Kai, Rong Yifa, Jia Haifeng, Li Hanzheng, Lu Bowen, Liang Xuezhen, Li Gang. Relationship between inflammatory factors and rheumatoid arthritis: a large-sample analysis based on the FinnGen R10 database and genome-wide association studies [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(10): 2629-2640. |
| [15] | Dong Tingting, Chen Tianxin, Li Yan, Zhang Sheng, Zhang Lei. Causal relationship between modifiable factors and joint sports injuries [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(9): 1953-1962. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||