Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (25): 6554-6565.doi: 10.12307/2026.416
Previous Articles Next Articles
Mechanism by which astragalus-peach kernel alleviates renal fibrosis in chronic kidney disease rats
Liu Jiayong1, Yao Jingjing2, Liu Shiyu1, Tang Yi1, Dong Jianing1, Zhang Xin1, Hou Lanwei1, Kang Jianying3, Zhao Yirui3
- 1The Third Clinical College, Shanxi University of Traditional Chinese Medicine, Jinzhong 030619, Shanxi Province, China; 2Taihang Materia Medica Research Institute, Shanxi University of Traditional Chinese Medicine, Jinzhong 030619, Shanxi Province, China; 3Shanxi Provincial Hospital of Integrated Traditional Chinese and Western Medicine, Taiyuan 030013, Shanxi Province, China
-
Received:2025-10-09Revised:2025-12-31Online:2026-09-08Published:2026-04-22 -
Contact:Zhao Yirui, Chief physician, Shanxi Provincial Hospital of Integrated Traditional Chinese and Western Medicine, Taiyuan 030013, Shanxi Province, China -
About author:Liu Jiayong, MS candidate, The Third Clinical College, Shanxi University of Traditional Chinese Medicine, Jinzhong 030619, Shanxi Province, China -
Supported by:Shanxi Province Traditional Chinese Medicine Science and Technology Innovation Project, No. 14000023218T200000289 (to ZYR); Shanxi Province Key Medical Research Project, No. 2022XM09 (to ZYR); 2024 Science and Technology Innovation Capacity Building Program—“National Natural Science Foundation Cultivation Project,” No. 2024PY-NS-008 (to ZYR); Shanxi Provincial Administration of Traditional Chinese Medicine Research Project, No. 2024ZYYC028 (to KJY)
CLC Number:
Cite this article
Liu Jiayong, Yao Jingjing, Liu Shiyu, Tang Yi, Dong Jianing, Zhang Xin, Hou Lanwei, Kang Jianying, Zhao Yirui. Mechanism by which astragalus-peach kernel alleviates renal fibrosis in chronic kidney disease rats[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(25): 6554-6565.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
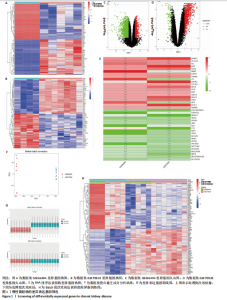
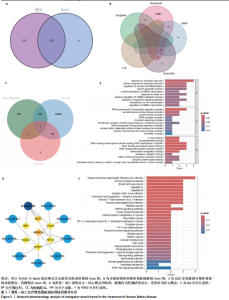
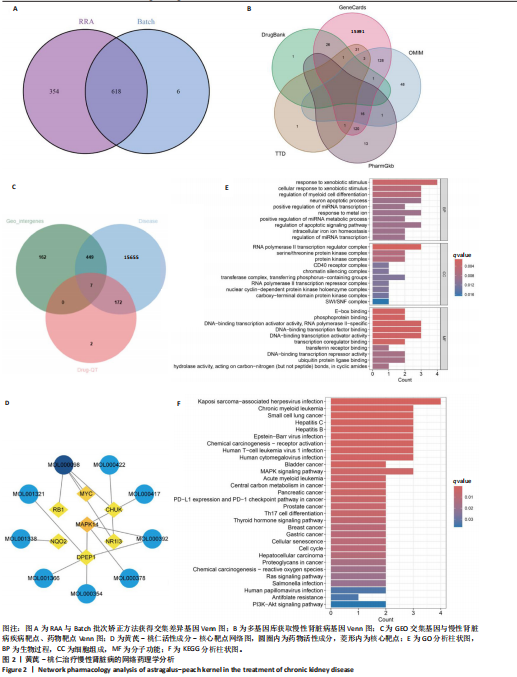
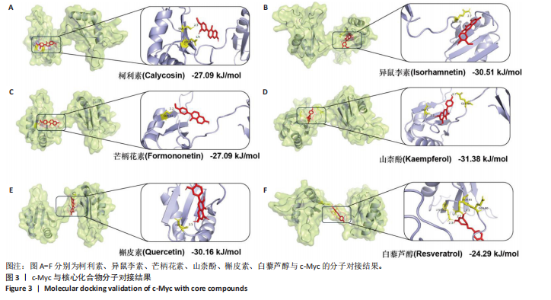
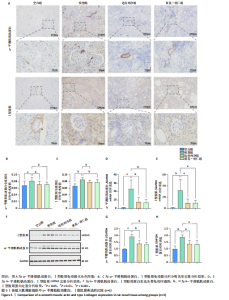
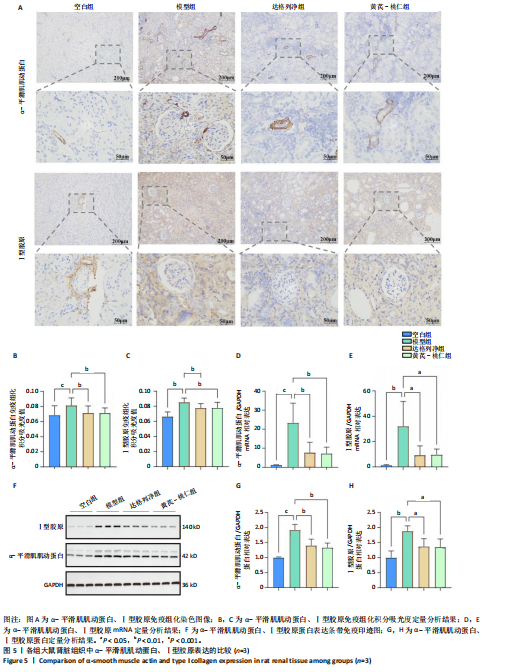
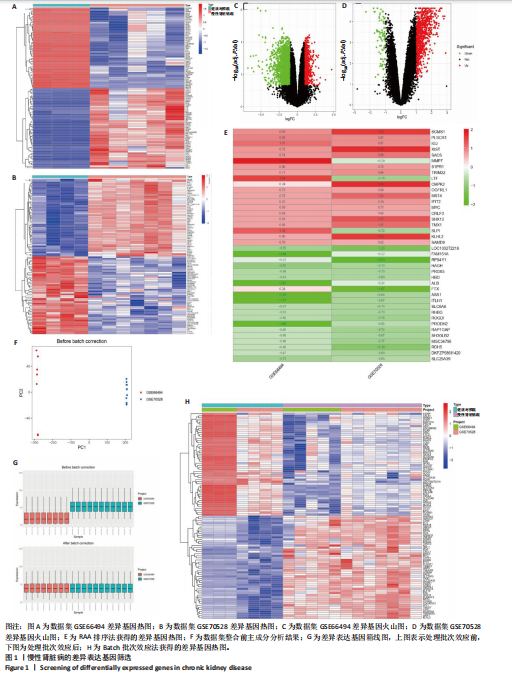
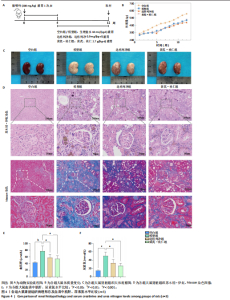
2.1 GEO数据库结合网络药理学分析结果 2.1.1 数据集的合并及差异基因的筛选 将GEO数据库中的两组数据集以|logFC|=1与P=0.05为条件获取单个数据的差异基因,并将上调和下调最显著的50个差异基因进行可视化,绘制热图和火山图(图1A-D)。使用RRA方法将2个数据集获得的差异基因进行排序,结果共得到972个差异基因,其中包含下调差异基因536个、上调差异基因436个,绘制热图(图1E)。主成分分析结果显示两组数据集有显著差异(图1F)。将数据进行归一化处理后应用Batch批次矫正法,依据|logFC|> 1及P < 0.05为阈值进行筛选,显示预处理后的合并基因表达矩阵共含有624个差异表达基因,其中上调基因数量为327个、下调基因数量为297个,绘制火山图、热图和箱线图(图1G,H)。 2.1.2 黄芪-桃仁活性成分-疾病核心靶点模型构建 在TCMSP数据库中依据生物利用度及类药性条件进行筛选,得到黄芪有效成分16个、潜在靶标159个,桃仁有效成分40个、潜在靶标63个。将黄芪桃仁有效成分、靶点合并去重后共得到有效成分56种,潜在病理靶标182个,见图2。在GeneCards、Pharmgkb、TTD和DrugBank与OMIM数据库中,通过以“Chronic kidney disease”为关键词,去重整理后共得到16 282个疾病靶点(图2B),将其与RRA和Batch法分析获得的交集差异基因(图2A)以及芪桃药对获得的潜在靶点进行合并,最终获得7个最具潜力的靶点基因(图2C),分别为c-Myc、RB1、CHUK、MAPK14、DPEP1、NR1I3、NQO2。基于已筛选出的靶点基因,利用Cytoscape进一步构建芪桃成分-疾病网络图(图2D),该网络包括9个药物活性成分和7个核心靶点,共有16个节点、13条边。拓扑分析提示了生物网络中双度值中位数是1,核心活性成分为槲皮素(MOL000098)。 2.1.3 GO、KEGG富集分析 如图2E所示,GO通路富集分析主要包括生物过程、细胞组成、分子功能3个方面。在生物过程中,主要富集条目涉及免疫和细胞分化调控、微小RNA代谢和转录的正向调控等;细胞组成相关富集条目有RNA聚合酶Ⅱ转录调控复合体、蛋白激酶复合体、染色质沉默复合体等;分子功能的富集条目有DNA结合转录激活和抑制活性、蛋白质配体酶连接等。GO通路富集分析提示了黄芪-桃仁可能通过调节应激反应、细胞凋亡、影响转录调控复合体的功能等过程来影响慢性肾病的进展。 根据KEGG富集分析结果,将最显著的30个信号通路进行可视化,结果包括MAPK信号通路、PI3K-Akt信号通路、细胞周期、细胞衰老、Ras信号通路等(图2F)。KEGG富集分析结果提示,Ras信号通路和MAPK信号通路与慢性肾脏病密切相关。Ras-MAPK信号通路可以通过提高上皮-间质转化有关转录因子(SNAIL和ZEB1)的表达[21],驱动细胞外基质的重塑;另一方面,RAS-MAPK信号通路可以增强转化生长因子β信号传导,激活肌成纤维细胞,加剧肾纤维化进展[22]。富集在MAPK信号通路中的相关基因有c-Myc、CHUK、RASA1,这些基因功能涉及炎症、细胞周期、及细胞的的凋亡分化。结合文献发现,c-Myc不但是MAPK下游效应因子[23],更是调控转化生长因子β活化的重要通路之一,因此作者推测黄芪-桃仁治疗慢性肾脏病可能与c-Myc有关。 2.1.4 分子对接验证 在黄芪-桃仁活性成分-核心靶点的分析中共获得9种活性成分,去除未能找到分子结构的3种化合物(MOL001366、MOL001338和MOL001321),将其余6种活性成与c-Myc进行分子对接,结果显示结合效果较好,能量均低于 0 kJ/mol。使用Pymol将结果进行可视化,见图3。 2.2 动物实验验证结果 2.2.1 实验动物数量分析 40只大鼠全部进入结果分析。 2.2.2 黄芪-桃仁改善慢性肾脏病大鼠肾功能和组织病理变化、减轻纤维化与炎症反应 动物实验流程见图4A。药物治疗过程中,空白组大鼠动作灵活,好斗,皮毛光滑,饮食与尿便尚可;模型组大鼠安静少动,皮毛枯暗无华,饮食减退,体质量减轻;与模型组相比,达格列净组、黄芪-桃仁组大鼠饮食、毛发色泽、活动状态均有好转。各组大鼠体质量变化见图4B。 大体观察肾脏组织外观可见,空白组肾脏组织表面光滑,呈红褐色,质地紧实;与空白组相比,模型组大鼠肾脏组织表面粗糙,凹凸不平,有纤维化触感,颜色呈苍白色,肾脏触感明显变硬;与模型组相比,达格列净组和黄芪-桃仁组肾脏组织外观均有明显改善,见图4C。 苏木精-伊红染色结果显示,空白组大鼠肾脏组织形态正常,肾小球、肾小管结构清晰,无病理改变;与空白组相比,模型组大鼠肾脏组织已有明显病理改变,肾小球数量减少、结构异常,出现萎缩、硬化,肾小管有大量炎性细胞浸润,空泡样改变,肾间质大量纤维化;与模型组相比,达格列净组和黄芪-桃仁组肾脏组织形态更趋完整,空泡改变减少,肾小球萎缩情况改善,肾间质纤维化水平下降,炎性细胞浸润明显降低,见图4D。 Masson染色结果显示,空白组大鼠肾脏组织中肾小球、肾小管未见明显胶原纤维沉积,蓝色信号稀少;与空白组相比,模型组大鼠肾脏组织中出现大量成纤维细胞,肾间质中有大量胶原纤维募集;与模型组相比,达格列净组和黄芪-桃仁组大鼠肾脏组织成纤维细胞减少,肾间质中胶原纤维募集程度减轻,见图4D。 为明确黄芪-桃仁对SD大鼠肾功能的影响,对肾功能相关血清指标进行了检测。结果显示,与空白组比较,模型组大鼠血清中肌酐、尿素氮水平分别升高0.8,2.3倍(P < 0.01,P < 0.001);与模型组比较,达格列净组大鼠血清中肌酐、尿素氮水平降低(P < 0.05),黄芪-桃仁组大鼠血清中肌酐、尿素氮水平降低(P < 0.05),达格列净组与黄芪-桃仁组大鼠血清中肌酐、尿素氮水平比较差异无显著性意义(P > 0.05),见图4E,F。 以上结果表明,黄芪-桃仁可以改善慢性肾脏病SD大鼠一般生理状态、肾脏组织结构,降低肾功能损伤,减轻纤维化和炎症反应,在慢性肾脏病中发挥了显著治疗作用。 2.2.3 黄芪-桃仁通过抑制α-平滑肌肌动蛋白和Ⅰ型胶原表达发挥抗纤维化作用 为验证黄芪-桃仁对肾纤维化的疗效,此次实验对肾纤维化的关键指标α-平滑肌肌动蛋白、Ⅰ型胶原进行验证。免疫组化染色结果显示,与空白组相比,模型组大鼠肾脏组织中α-平滑肌肌动蛋白、Ⅰ型胶原表达明显增加,差异有显著性意义;与模型组相比,达格列净组与黄芪-桃仁组大鼠肾脏组织中α-平滑肌肌动蛋白、Ⅰ型胶原表达均减少,差异有显著性意义;达格列净组与黄芪-桃仁组大鼠肾脏组织中α-平"
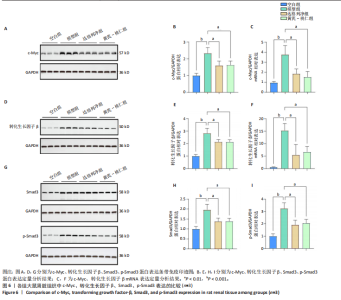
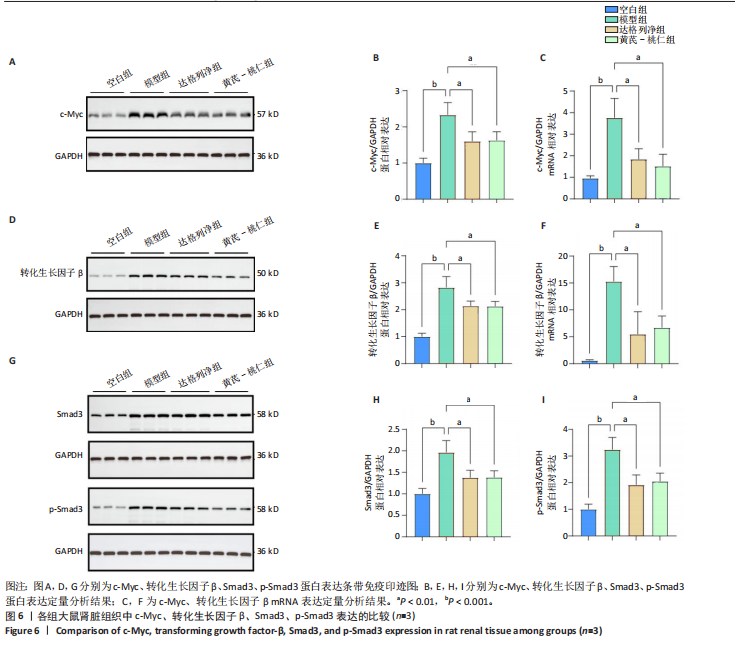
滑肌肌动蛋白、Ⅰ型胶原表达比较差异无显著性意义(P > 0.05),见图5A-C。 RT-qRCR 检测结果显示,与空白组相比,模型组大鼠肾脏组织中α-平滑肌肌动蛋白、Ⅰ型胶原mRNA表达升高(P < 0.01,P < 0.001);与模型组相比,达格列净组大鼠肾脏组织中α-平滑肌肌动蛋白、Ⅰ型胶原mRNA表达降低(P < 0.01),黄芪-桃仁组大鼠肾脏组织中α-平滑肌肌动蛋白、Ⅰ型胶原mRNA表达降低(P < 0.01);达格列净组与黄芪-桃仁组大鼠肾脏组织中α-平滑肌肌动蛋白、Ⅰ型胶原mRNA表达比较差异无显著性意义(P > 0.05),见图5D,E。 Western blot检测结果显示,与空白组相比,模型组大鼠肾脏组织中α-平滑肌肌动蛋白、Ⅰ型胶原蛋白表达升高(P < 0.001,P < 0.01);与模型组相比,达格列净组大鼠肾脏组织中α-平滑肌肌动蛋白、Ⅰ型胶原蛋白表达降低(P < 0.01,P < 0.05),黄芪-桃仁组大鼠肾脏组织中α-平滑肌肌动蛋白、Ⅰ型胶原蛋白表达降低(P < 0.01,P < 0.05),达格列净组与黄芪-桃仁组大鼠肾脏组织中α-平滑肌肌动蛋白、Ⅰ型胶原蛋白表达比较差异无显著性意义(P > 0.05),见图5F-H。 以上结果说明,黄芪-桃仁可能通过抑制α-平滑肌肌动蛋白、Ⅰ型胶原的表达发挥了抗纤维化作用。 2.2.4 黄芪-桃仁通过抑制c-Myc和转化生长因子β/Smad3信号通路减轻肾组织纤维化 为证实GEO数据库结合网络药理学分析预测的关键靶点c-Myc在慢性肾脏病中的作用,此次实验对c-Myc及其相关通路的基因与蛋白表达进行了检测。RT-qRCR与Western blot检测结果显示,模型组大鼠肾脏组织中c-Myc、转化生长因子β的mRNA和蛋白表达均高于空白组,差异均有显著性意义,其中c-Myc、转化生长因子β mRNA表达分别上调约1倍和24.88倍,c-Myc、转化生长因子β蛋白表达分别增加约1.3倍和1.8倍;与模型组相比,达格列净组和黄芪-桃仁组肾脏组织中c-Myc、转化生长因子β的mRNA和蛋白表达均降低,差异均有显著性意义(P < 0.05),见图6。 RT-qRCR与Western blot检测结果显示,与空白组相比较,模型组大鼠肾脏组织中Smad3、p-Smad3蛋白表达分别上调约0.9倍和1.2倍,差异有显著性意义;与模型组比较,达格列净组与黄芪-桃仁组大鼠肾脏组织中Smad3、p-Smad3蛋白表达均降低,差异有显著性意义;达格列净组与黄芪-桃仁组大鼠肾脏组织c-Myc、转化生长因子β的mRNA与蛋白表达以及Smad3、p-Smad3蛋白表达比较差异均无显著性意义(P > 0.05),见图6。 以上结果说明,黄芪-桃仁可以通过抑制c-Myc信号通路下调转化生长因子β/Smad3的表达,减轻肾组织的纤维化程度,从而发挥治疗慢性肾脏病的作用。"

| [1] RUIZ-ORTEGA M, RAYEGO-MATEOS S, LAMAS S, et al. Targeting the progression of chronic kidney disease. Nat Rev Nephrol. 2020;16(5):269-288. [2] KUPPE C, IBRAHIM MM, KRANZ J, et al. Decoding myofibroblast origins in human kidney fibrosis. Nature. 2021;589(7841):281-286. [3] ZHOU S, AI Z, LI W, et al. Deciphering the Pharmacological Mechanisms of Taohe-Chengqi Decoction Extract Against Renal Fibrosis Through Integrating Network Pharmacology and Experimental Validation In Vitro and In Vivo. Front Pharmacol. 2020;11:425. [4] AUGUST P. Chronic Kidney Disease — Another Step Forward. N Engl J Med. 2023;388(2):179-180. [5] RUIZ-ORTEGA M, RAYEGO-MATEOS S, LAMAS S, et al. Targeting the progression of chronic kidney disease. Nat Rev Nephrol. 2020;16(5):269-288. [6] LI L, FU H, LIU Y. The fibrogenic niche in kidney fibrosis: components and mechanisms. Nat Revi Nephrol. 2022;18(9):545-557. [7] 郭诗韵,邓志灏,李艳,等.中药复方配伍科学内涵阐释研究的新理论[J/OL].中国实验方剂学杂志,1-8[2025-06-12].https://doi.org/10.13422/j.cnki.syfjx.20241645. [8] 韩阳,孙岚云,徐英,等.补肾活血法治疗肾脏病研究述评[J].中华中医药杂志,2022,37(2):629-632. [9] ZHANG J, WU C, GAO L, et al. Astragaloside IV derived from Astragalus membranaceus: A research review on the pharmacological effects. Adv Pharmacol. 2020;87:89-112. [10] ZHENG W, HUANG T, TANG QZ, et al. Astragalus Polysaccharide Reduces Blood Pressure, Renal Damage, and Dysfunction Through the TGF-β1-ILK Pathway. Front Pharmacol. 2021;12:706617. [11] ZHENG W, HUANG T, TANG QZ, et al. Astragalus Polysaccharide Reduces Blood Pressure, Renal Damage, and Dysfunction Through the TGF-β1-ILK Pathway. Front Pharmacol. 2021;12:706617. [12] 李想,刘庆,高晨,等.苦杏仁苷对肾纤维化大鼠的保护作用及其机制[J].医药导报,2022,41(9):1282-1289. [13] ZHAO J, WANG L, CAO AL, et al. HuangQi Decoction Ameliorates Renal Fibrosis via TGF-β/Smad Signaling Pathway In Vivo and In Vitro. Cell Physiol Biochem. 2016;38(5):1761-1774. [14] SHI H, ZHENG G, CHEN N. Amygdalin improves the renal function via anti-oxidation and anti-fibrosis in rats with chronic renal failure. Panminerva Med. 2020. doi: 10.23736/S0031-0808.20.03977-4. [15] BALUAPURI A, WOLF E, EILERS M. Target gene-independent functions of MYC oncoproteins. Nat Rev Mol Cell Biol. 2020;21(5):255-267. [16] THOMPSON EB. The many roles of c-Myc in apoptosis. Annu Rev Physiol. 1998; 60(1):575-600. [17] SHEN Y, MIAO N, WANG B, et al. c-Myc promotes renal fibrosis by inducing integrin αv-mediated transforming growth factor-β signaling. Kidney Int. 2017; 92(4):888-899. [18] SU J, MORGANI SM, DAVID CJ, et al. TGF-β orchestrates fibrogenic and developmental EMTs via the RAS effector RREB1. Nature. 2020;577(7791):566-571. [19] 李雨婷,林钐,王克邪.达格列净对慢性肾脏病大鼠的肾脏保护作用及对炎症细胞因子的影响[J].现代中西医结合杂志,2024,33(17):2379-2383. [20] 陈俊蓉,陈利国,谢林林.关于腺嘌呤慢性肾衰实验模型的思考[J].实验动物科学,2013,30(2):65-67. [21] YUAN Q, TANG B, ZHANG C. Signaling pathways of chronic kidney diseases, implications for therapeutics. Signal Transduct Target Ther. 2022;7(1):182. [22] LIU J. Anillin contributes to prostate cancer progression through the regulation of IGF2BP1 to promote c-Myc and MAPK signaling. Am J Cancer Res. 2024; 14(2):490-506. [23] YANG Q, SU S, LUO N, et al. Adenine-induced animal model of chronic kidney disease: current applications and future perspectives. Ren Fail. 2024;46(1): 2336128. [24] WU W, WANG X, YU X, et al. Smad3 Signatures in Renal Inflammation and Fibrosis. Int J Biol Sci. 2022;18(7):2795-2806. [25] RAYEGO-MATEOS S, VALDIVIELSO JM. New therapeutic targets in chronic kidney disease progression and renal fibrosis. Expert Opin Ther Targets. 2020; 24(7):655-670. [26] KLINKHAMMER BM, BOOR P. Kidney fibrosis: Emerging diagnostic and therapeutic strategies. Mol Aspects Med. 2023;93:101206 [27] ZHOU R, LIAO J, CAI D, et al. Nupr1 mediates renal fibrosis via activating fibroblast and promoting epithelial‐mesenchymal transition. FASEB J. 2021;35(3):e21381. [28] NIE W, LI M, LIU B, et al. A circular RNA, circPTPN14, increases MYC transcription by interacting with FUBP1 and exacerbates renal fibrosis. Cell Mol Life Sci. 2022; 79(12):595. [29] HUANG H, WENG H, ZHOU H, et al. Attacking c-Myc: Targeted and Combined Therapies for Cancer. Curr Pharm Des. 2014;20(42):6543-6554. [30] OGAGAYERE LO, NAIHO AO, EMOJEVWE V, et al. Quercetin flavonoid and vitamin C recuperate kidney functions in potassium bromate-induced renal dysfunction in Wistar rats. Naunyn-Schmiedeberg’s Arch Pharmacol. 2023;396(12):3789-3796. [31] LI R, SHI C, WEI C, et al. Fufang Shenhua tablet inhibits renal fibrosis by inhibiting PI3K/AKT. Phytomedicine. 2023;116:154873 [32] CHEN YQ, CHEN HY, TANG QQ, et al. Protective effect of quercetin on kidney diseases: From chemistry to herbal medicine. Front Pharmacol. 2022;13:968226. [33] WIDOWATI W, PRAHASTUTI S, TJOKROPRANOTO R, et al. Quercetin prevents chronic kidney disease on mesangial cells model by regulating inflammation, oxidative stress, and TGF-β1/SMADs pathway. PeerJ. 2022;10:e13257. [34] SÁNCHEZ-JARAMILLO EA, GASCA-LOZANO LE, VERA-CRUZ JM, et al. Nanoparticles Formulation Improves the Antifibrogenic Effect of Quercetin on an Adenine-Induced Model of Chronic Kidney Disease. Int J Mol Sci. 2022;23(10):5392. [35] GRANATO M, RIZZELLO C, ROMEO MA, et al. Concomitant reduction of c-Myc expression and PI3K/AKT/mTOR signaling by quercetin induces a strong cytotoxic effect against Burkitt’s lymphoma. Int J Biochem Cell Biol. 2016;79:393-400. |
| [1] | Zhong Zhuolan, Peng Zhina, Tian Xiaohong, Han Cuifei, Zhang Zihan, Chu Jiaqi. Acanthopanax exosome-like nanovesicles promote osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 4825-4835. |
| [2] | Hong Runyang, Zhou Qiyue, Fan Zhencheng, Shi Yujie, Chen Hao, Pan Chun. Impact and mechanism of low-dose hexafluoropropylene oxide dimer acid exposure during pregnancy on renal toxicity in offspring mice [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(11): 2752-2763. |
| [3] | Wang Wanchun, , Yi Jun, Yan Zhangren, Yang Yue, Dong Degang, Li Yumei. 717 Jiedu Decoction remodels homeostasis of extracellular matrix and promotes repair of local injured tissues in rats after Agkistrodon halys bite [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(30): 6457-6465. |
| [4] | Peng Zehong, Zhu Xi, Wen Jianglong, Zhu Wenzhuo, Liu Chao, Tang Jianwei, Cao Ziyue, Zhu Lili. Causal relationship between 39 plasma coagulation factors and chronic kidney disease based on samples from the GWAS Catalog database [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(24): 5272-5280. |
| [5] | Wang Lian, Xie Na, Zhao Peiling, Chen Hao, Li Duyou, Wang Yuping. Effects of cannabidiol on hepatic stellate cell activation and hepatic fibrosis induced by transforming growth factor beta1 [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(23): 4965-4974. |
| [6] | Chen Jiahui, Dai Xiaoqi, Xu Yangang, Li Yuanchao, Huang Mei, Zhan Yifei, Du Yuxuan, Li Liuqiang, Guo Yaochuan, Bian Jun, Lai Dehui. Isolation, culture and differentiation of human urine-derived stem cells into smooth muscle cells [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(19): 4076-4082. |
| [7] | Yu Yangyi , Song Zhuoyue, Lian Qiang, Ding Kang, Li Guangheng . AAV-mediated expression of p65shRNA and bone morphogenetic protein 4 synergistically enhances chondrocyte regeneration [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(17): 3537-3547. |
| [8] | Huang Chaolu, Huang Yi, Wu Changyan, Li Fangfei, Li Haiyan. Aerobic exercise mitigates liver fibrosis in db/db diabetes mice by regulating transforming growth factor beta/Smad pathway [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(14): 2951-2957. |
| [9] | Zhang Xingzhou, Wei Ming, Dong Guoqiang, Du Wei, Luo Yiwen, Zhang Nan . Mechanism of postoperative abdominal adhesion formation and therapeutic prospect of mesenchymal stem cell exosomes [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(1): 147-155. |
| [10] | Shen Jiangyong, He Xi, Tang Yuting, Wang Jianjun, Liu Jinyi, Chen Yuanyuan, Wang Xinyi, Liu Tong, Sun Haoyuan. RAS-selective lethal small molecule 3 inhibits the fibrosis of pathological scar fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1168-1173. |
| [11] | Ma Sicong, Chen Jing, Li Yunqing. Functions and roles of connective tissue growth factor in nervous systems [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 615-620. |
| [12] | Guo Lei, Qi Yansong, Niu Xiaobo. Regulatory role of transforming growth factor beta subfamily in osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(35): 5695-5701. |
| [13] | Chen Guanting, Zhang Linqi, Wang Xixi, Chen Xu. Autophagy, ferroptosis-related targets and renal function progression in patients with chronic kidney disease: bioinformatics analysis and experimental verification [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(32): 5122-5129. |
| [14] | Cao Jiawei, Ding Shaorui, Tie Hua, Xue Jing, Jia Yuanyuan, Liang Xueyun, Li Feng. Human placental mesenchymal stem cells inhibit occurrence of pulmonary fibrosis by regulating transforming growth factor-beta 1/Smad3 signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(31): 4970-4974. |
| [15] | Xiong Yang, Zhou Shibo, Yu Xing, Bi Lianyong, Yang Jizhou, Wang Fengxian, Qu Yi, Yang Yongdong, Zhao Dingyan, Zhao He, Qiu Ziye, Jiang Guozheng. Molecular biological mechanism of acquired heterotopic ossification [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(30): 4881-4888. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||