Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (19): 4825-4835.doi: 10.12307/2026.245
Acanthopanax exosome-like nanovesicles promote osteogenic differentiation of human bone marrow mesenchymal stem cells
Zhong Zhuolan1, Peng Zhina1, Tian Xiaohong2, Han Cuifei3, Zhang Zihan3, Chu Jiaqi4
- 1First Clinical Medical College of Guangdong Medical University, Zhanjiang 524001, Guangdong Province, China; 2Hebei PetroChina Central Hospital, Langfang 065000, Hebei Province, China; 3Guangdong Medical University, Zhanjiang 524003, Guangdong Province, China; 4Affiliated Hospital of Guangdong Medical University, Zhanjiang 524001, Guangdong Province, China
-
Received:2025-08-08Accepted:2025-12-10Online:2026-07-08Published:2026-02-13 -
Contact:Chu Jiaqi, Associate researcher, Affiliated Hospital of Guangdong Medical University, Zhanjiang 524001, Guangdong Province, China -
About author:Zhong Zhuolan, MS candidate, First Clinical Medical College of Guangdong Medical University, Zhanjiang 524001, Guangdong Province, China -
Supported by:Guangdong Medical University Affiliated Hospital "Clinical Medicine" Science and Technology Co-construction Platform Project, No. CLP2021A001 (to CJQ)
CLC Number:
Cite this article
Zhong Zhuolan, Peng Zhina, Tian Xiaohong, Han Cuifei, Zhang Zihan, Chu Jiaqi. Acanthopanax exosome-like nanovesicles promote osteogenic differentiation of human bone marrow mesenchymal stem cells[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 4825-4835.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
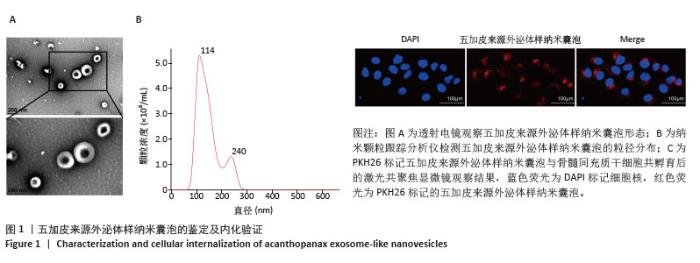
2.1 五加皮来源外泌体样纳米囊泡的鉴定及内化验证 透射电镜观察显示,成功提取到的五加皮来源外泌体样纳米囊泡呈现典型的杯托状或圆盘状结构,囊泡外形完整,轮廓清晰,大小较为均一,符合植物来源外泌体样囊泡的形态特征(图1A)。纳米颗粒跟踪分析仪测定五加皮来源外泌体样纳米囊泡的浓度约为4.13×1010颗粒/mL,粒径范围主要分布在102.8-200 nm之间,平均粒径为114 nm。粒径分布曲线基本呈正态分布,表明样品均一性较高(图1B)。为验证五加皮来源外泌体样纳米囊泡是否能被骨髓间充质干细胞有效摄取,采用PKH26荧光染料标记五加皮来源外泌体样纳米囊泡,随后与骨髓间充质干细胞共孵育,通过激光共聚焦显微镜观察结果显示,骨髓间充质干细胞胞质中出现明显红色荧光信号,荧光主要分布于细胞质区域,且未见于细胞核,提示五加皮来源外泌体样纳米囊泡成功被细胞内化(图1C)。以上结果表明,成功分离并获得了形态典型、粒径适宜的五加皮来源外泌体样纳米囊泡,且能够有效被骨髓间充质干细胞摄取,为后续功能实验奠定了基础。"
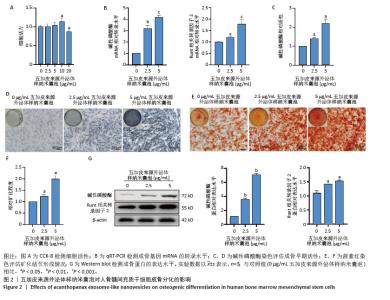
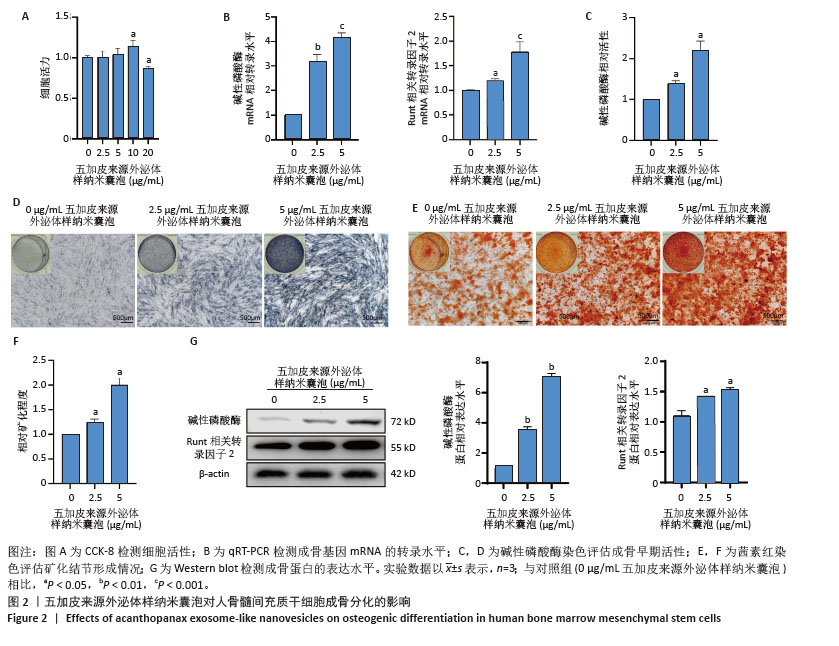
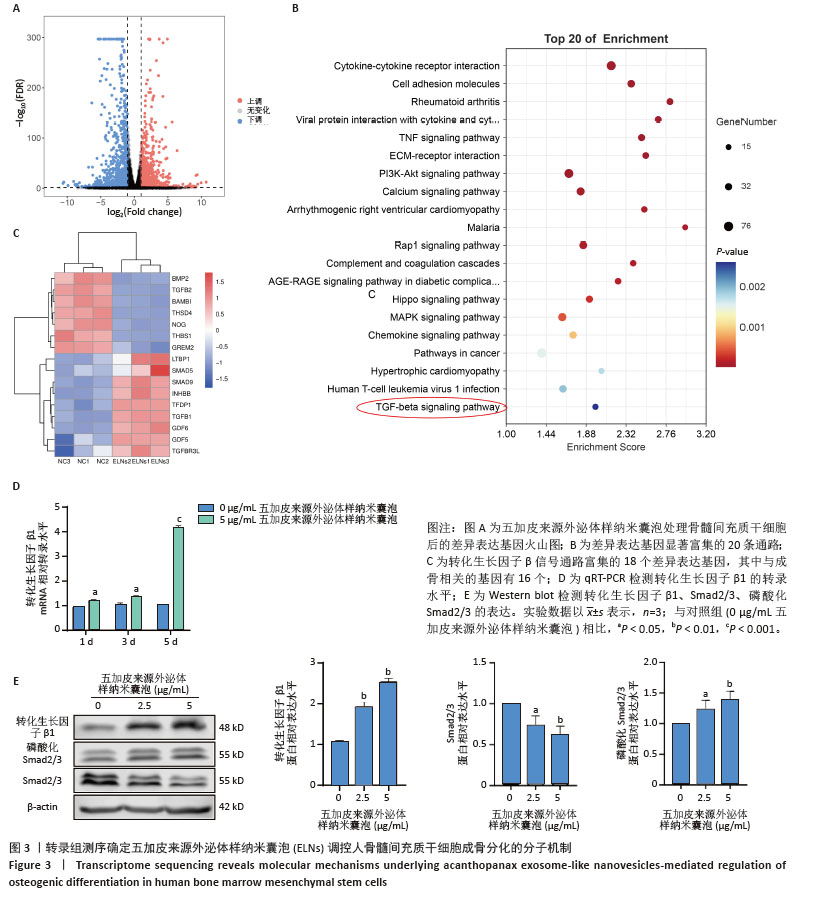
2.2 五加皮来源外泌体样纳米囊泡促进骨髓间充质干细胞成骨分化 CCK-8检测结果显示,五加皮来源外泌体样纳米囊泡在2.5-10 μg/mL质量浓度范围内对骨髓间充质干细胞的增殖无明显抑制作用(图2A)。qRT-PCR检测显示,五加皮来源外泌体样纳米囊泡处理可显著上调骨髓间充质干细胞中成骨相关基因Runt相关转录因子2和碱性磷酸酶的表达水平(P < 0.05,图2B)。成骨诱导第6天行碱性磷酸酶染色,结果显示五加皮来源外泌体样纳米囊泡处理后碱性磷酸酶活性显著增强,碱性磷酸酶染色面积和染色强度均高于对照组,且随外泌体样纳米囊泡质量浓度增加而增强(P < 0.05,图2C,D)。成骨诱导第12天行茜素红S染色,结果显示五加皮来源外泌体样纳米囊泡处理明显促进矿化结节形成(P < 0.05,图2E,F)。Western blot分析进一步证实,五加皮来源外泌体样纳米囊泡组细胞中Runt相关转录因子2、碱性磷酸酶蛋白表达量显著增加(P < 0.05,图2G)。综上,五加皮来源外泌体样纳米囊泡能够显著促进骨髓间充质干细胞的成骨分化,且质量浓度为5 μg/mL以下对细胞无明显的毒性作用。"
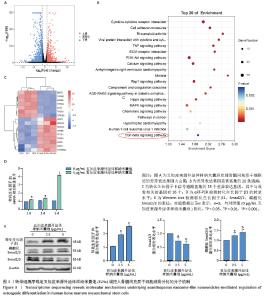
2.3 转录组测序确定五加皮来源外泌体样纳米囊泡调控骨髓间充质干细胞成骨分化的分子机制 用含或不含五加皮来源外泌体样纳米囊泡(5 μg/mL)的成骨诱导培养基处理骨髓间充质干细胞,全转录组测序显示两组细胞在基因转录水平存在显著差异,火山图分析显示共有964个基因显著上调,992个基因显著下调,差异有显著性意义(图3A)。基因显著富集于20条KEGG通路(图3B),其中转化生长因子β信号通路富集了18个差异基因,与成骨相关的基因有16个(图3C),提示该通路可能是五加皮来源外泌体样纳米囊泡调控骨髓间充质干细胞成骨分化的信号通路之一。 qRT-PCR结果显示,与对照组相比,五加皮来源外泌体样纳米囊泡组的转化生长因子β1 mRNA转录水平呈时间依赖性显著上调(P < 0.05),见图3D。Western blot结果显示,与对照组相比,五加皮来源外泌体样纳米囊泡组(2.5,5 μg/mL)的转化生长因子β1、磷酸化Smad2/3蛋白表达均显著上调(P < 0.05),见图3E。上述结果表明,五加皮来源外泌体样纳米囊泡可能通过激活转化生长因子β1引起下游Smads蛋白发生级联反应,即通过转化生长因子β1/Smad2/3信号转导通路实现对骨髓间充质干细胞成骨分化的调控作用。"
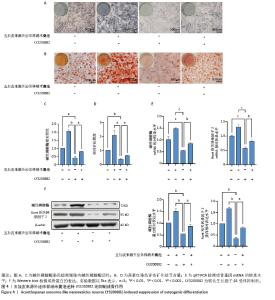
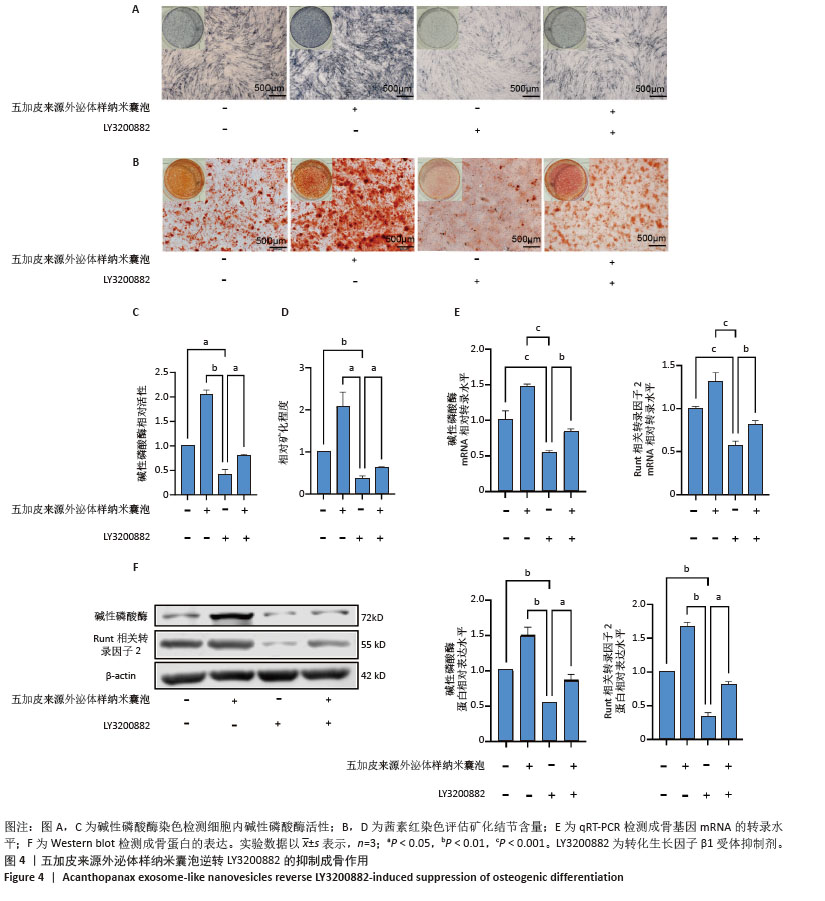
2.4 五加皮来源外泌体样纳米囊泡逆转LY3200882的抑制成骨作用 为进一步验证转化生长因子β1/Smad2/3通路在五加皮来源外泌体样纳米囊泡促成骨分化中的作用,采用转化生长因子β1受体抑制剂LY3200882进行功能干预实验。碱性磷酸酶染色结果显示,LY3200882组骨髓间充质干细胞的碱性磷酸酶活性显著下降,而在LY3200882基础上联合五加皮来源外泌体样纳米囊泡处理,可部分逆转抑制效应,表现为碱性磷酸酶活性水平上升(P < 0.05,图4A,C)。茜素红S染色显示,LY3200882处理显著抑制矿化结节形成,而联合五加皮来源外泌体样纳米囊泡处理后,矿化结节面积明显增多,部分恢复到对照水平(P < 0.05,图4B,D)。qRT-PCR结果显示,LY3200882联合五加皮来源外泌体样纳米囊泡处理后,碱性磷酸酶和Runt相关转录因子2 mRNA转录水平显著高于单用LY3200882组(P < 0.05,图4E)。Western blot检测进一步证实,LY3200882组成骨相关蛋白碱性磷酸酶、Runt相关转录因子2表达量显著低于对照组,而LY3200882联合五加皮来源外泌体样纳米囊泡处理后碱性磷酸酶和Runt相关转录因子2蛋白表达水平较LY3200882组明显升高(P < 0.05,图4F)。上述结果进一步验证了五加皮来源外泌体样纳米囊泡通过激活转化生长因子β1信号通路促进骨髓间充质干细胞成骨分化,且转化生长因子β1可能为五加皮来源外泌体样纳米囊泡介导成骨效应的重要分子靶点。 "
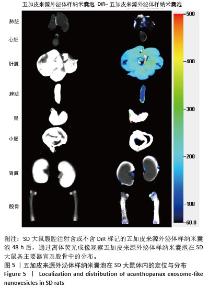
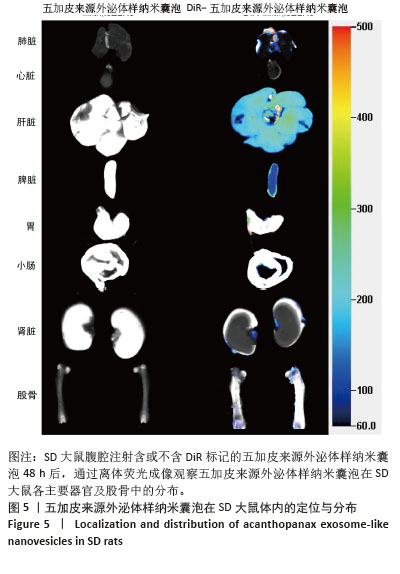
2.5 五加皮来源外泌体样纳米囊泡在体内的分布特征 为研究五加皮来源外泌体样纳米囊泡在体内的分布情况,采用DiR荧光染料标记五加皮来源外泌体样纳米囊泡后经腹腔注射至大鼠体内。注射48 h后,取主要脏器(心、肝、脾、肺、肾)及右侧股骨,离体荧光成像结果显示,DiR信号在肝脏、脾脏、肺脏和肾脏等高血供器官中显著富集,表明五加皮来源外泌体样纳米囊泡能够通过血液循环系统广泛分布于多个组织器官(图5)。此外,在股骨大血管分布区域也观察到明显荧光信号积累,提示五加皮来源外泌体样纳米囊泡可通过血液循环富集至骨组织。以上结果表明,腹腔注射五加皮来源外泌体样纳米囊泡后,能够有效进入体循环,并具有一定的骨组织靶向性。"
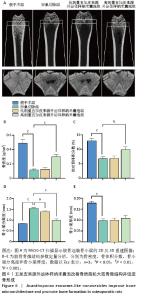
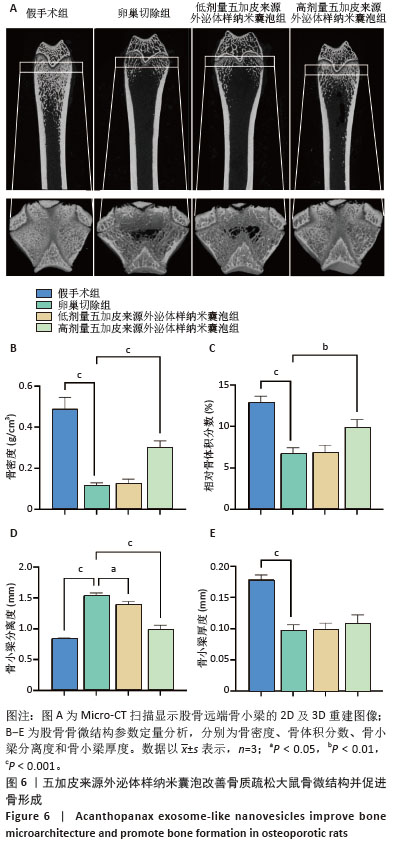
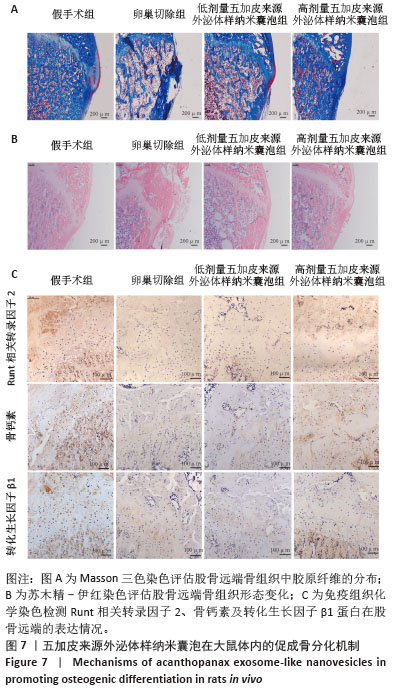
2.6 五加皮来源外泌体样纳米囊泡改善骨质疏松大鼠骨微结构并促进骨形成 Micro-CT扫描及三维重建结果显示,假手术组大鼠股骨远端骨小梁排列致密、连续,骨小梁厚度均匀,整体结构完整,呈典型正常骨微结构特征(图6A)。卵巢切除组则表现出明显骨质疏松表型,骨小梁数量减少,连续性中断,厚度变薄,密度降低。经高剂量五加皮来源外泌体样纳米囊泡干预后,骨小梁数量显著增加,排列更为致密,连续性改善,骨微结构较卵巢切除组明显恢复(图6A)。Micro-CT定量分析进一步证实,与假手术组相比,卵巢切除组大鼠骨密度、骨体积分数、骨小梁厚度均显著下降,骨小梁分离度显著升高(P < 0.05,图6B-E)。高剂量五加皮来源外泌体样纳米囊泡干预后,骨密度显著高于卵巢切除组(P < 0.05,图6B);骨体积分数亦显著回升(P < 0.05,图6C);骨小梁分离度下降(P < 0.05,图6D);而骨小梁厚度虽有上升趋势,但与卵巢切除组差异无显著性意义(P > 0.05,图6E)。综上所述,五加皮来源外泌体样纳米囊泡可有效改善卵巢切除大鼠的骨质疏松状态,重建骨小梁微结构,提升骨密度,具有体内促进骨形成的作用。 "
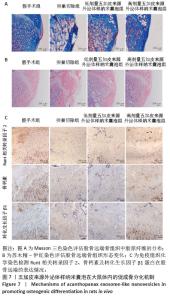
2.7 五加皮来源外泌体样纳米囊泡在体内促进成骨分化的机制 为进一步探讨五加皮来源外泌体样纳米囊泡在体内促进成骨分化的分子机制,采用Masson三色染色评估胶原纤维的生长情况,免疫组织化学染色检测成骨相关蛋白的表达水平。Masson染色结果显示,假手术组大鼠股骨远端骨小梁区域胶原纤维排列致密,分布均匀,呈现典型正常骨组织结构特征(图7A)。卵巢切除组大鼠骨组织中胶原纤维密度明显降低,排列松散且连续性中断,提示卵巢切除成功诱导形成骨质疏松模型。与卵巢切除组相比,高剂量五加皮来源外泌体样纳米囊泡组骨组织中胶原纤维数量显著增加,排列较为致密且连续性恢复,提示五加皮来源外泌体样纳米囊泡干预可有效促进骨组织中胶原纤维的生成与重建(图7A)。苏木精-伊红染色结果显示,假手术组股骨远端骨小梁结构完整、排列密集且连续,呈典型正常骨微结构特征;卵巢切除组则表现出明显骨质疏松表型,骨小梁数量显著减少,骨小梁明显变细、断裂,骨小梁间距异常增宽;经高剂量五加皮来源外泌体样纳米囊泡干预后,骨小梁数量显著增加,连续性部分恢复,骨微结构较卵巢切除组明显恢复(图7B)。免疫组织化学染色进一步验证了成骨相关蛋白表达的变化(图7C)。与卵巢切除组相比,高剂量五加皮来源外泌体样纳米囊泡组股骨组织中Runt相关转录因子2、晚期成骨标志蛋白骨钙素以及信号通路关键因子转化生长因子β1的表达水平均明显上调。综合以上结果,提示五加皮来源外泌体样纳米囊泡可能通过上调转化生长因子β1表达,进一步促进骨组织中胶原纤维沉积及成骨相关蛋白表达,进而增强骨形成能力。"
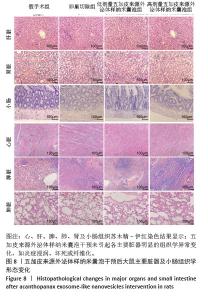
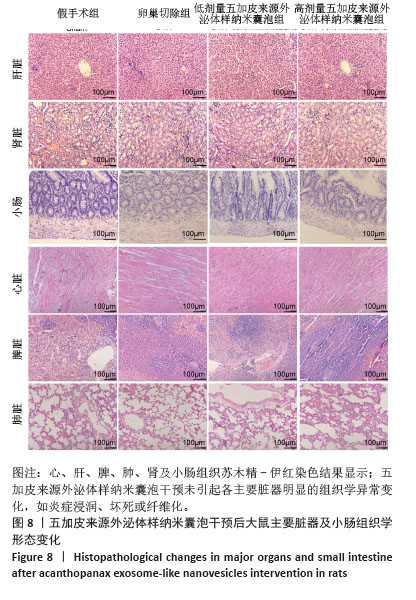
2.8 五加皮来源外泌体样纳米囊泡在体内的安全性评估 为评估五加皮来源外泌体样纳米囊泡在体内的生物安全性,采用苏木精-伊红染色对干预结束后大鼠主要脏器(心脏、肝脏、脾脏、肺脏、肾脏、小肠)进行组织病理学观察(图8),结果显示,五加皮来源外泌体样纳米囊泡组大鼠心脏组织中心肌纤维排列规则,结构完整,未见炎症细胞浸润及坏死灶;肝脏组织肝细胞索排列整齐,肝窦结构清晰,无脂肪变性、坏死或纤维化改变;脾脏白髓与红髓结构完整,边界清晰,未见异常增生或出血灶;肺组织肺泡结构规则,间质无明显水肿或炎症细胞浸润;肾脏中肾小球结构正常,肾小管排列紧密,无硬化、萎缩或坏死现象;小肠绒毛排列整齐,未见明显脱落、萎缩或水肿,腺体结构完整,黏膜层无明显炎症细胞浸润或坏死。与卵巢切除组和假手术组相比,五加皮来源外泌体样纳米囊泡组在各脏器组织学结构上均无明显异常改变,提示在此研究设定的剂量及给药方式下,五加皮来源外泌体样纳米囊泡具有良好的体内生物安全性。"
| [1] WANG J, SHU B, TANG DZ, et al. The prevalence of osteoporosis in China: a community-based cohort study. Front Public Health. 2023;11:1084005. [2] 邹顺一,易进,曾浩,等.绝经后骨质疏松症:相关信号通路的可视化分析[J].中国组织工程研究,2026,30(16):4229-4239. [3] LEBOFF MS, GREENSPAN SL, INSOGNA KL, et al. The clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int. 2022;33(10):2049-2102. [4] AIBAR-ALMAZÁN A, VOLTES-MARTÍNEZ A, CASTELLOTE-CABALLERO Y, et al. Current status of the diagnosis and management of osteoporosis. Int J Mol Sci. 2022;23(16):9465. [5] 王瑞.选择性雌激素受体调节剂治疗绝经后女性骨质疏松症的效果及骨代谢指标研究[J].医学理论与实践,2025,38(15):2603-2605. [6] IMAMUDEEN N, BASHEER A, IQBAL AM, et al. Management of osteoporosis and spinal fractures: contemporary guidelines and evolving paradigms. Clin Med Res. 2022;20(2):95-106. [7] 王露,刘伟兵,钟嘉伟,等.骨质疏松症药物治疗的现状和研究进展[J].中国现代医生,2024,62(27):124-128. [8] ENSRUD KE, CRANDALL CJ. Osteoporosis. Ann Intern Med. 2024;177(1):ITC1-ITC6. [9] REID IR, BILLINGTON EO. Drug therapy for osteoporosis in older adults. Lancet. 2022;399(10329):1080-1092. [10] XU H, XU J, CHEN F, et al. Acanthopanax senticosus aqueous extract ameliorates ovariectomy-induced bone loss by inhibiting NF-κB ligand-induced osteoclastogenesis. Food Funct. 2020;11(11):9696-9709. [11] HWANG YC, JEONG IK, AHN KJ, et al. Effects of Acanthopanax senticosus extract on bone turnover and bone mineral density in postmenopausal women. J Bone Miner Metab. 2009;27(5):584-590. [12] BOHN B, NEBE CT, BIRR C. Durchflußzytometrische Untersuchungen auf immunmodulatorische Wirkungen von Eleutherococcus senticosus-Extrakt. Arzneim Forsch. 1987;37:1193-1196. [13] LIM DW, KIM JG, LEE Y, et al. Preventive effects of Eleutherococcus senticosus bark extract in OVX-induced osteoporosis. Molecules. 2013;18(7):7998-8008. [14] LIU J, ZHANG Z, GUO Q, et al. Syringin prevents bone loss via TRAF6-mediated inhibition of NF-κB and activation of PI3K/AKT. Phytomedicine. 2018;42:43-50. [15] JIN Z, NA J, LIN X, et al. Plant-derived exosome-like nanovesicles: a novel nanotool for disease therapy. Heliyon. 2024;10(9):e30630. [16] 韩菲,马小梅,石旭柳,等.柑橘属植物来源的外泌体样纳米颗粒及其疾病治疗研究进展[J].中草药,2024,55(19):6768-6778. [17] FENG H, YUE Y, ZHANG Y, et al. Plant-derived exosome-like nanoparticles: emerging nanosystems for enhanced tissue engineering. Int J Nanomedicine. 2024;19:1189-1204. [18] 秦启顺,王兴盛,徐世红,等.中药及其活性成分诱导骨髓间充质干细胞成骨分化相关信号通路的研究进展[J].生物医学工程与临床,2024,28(6): 884-892. [19] 药文鑫,王毅,蔡林,等.植物源外泌体样纳米囊泡刺激成骨细胞、抑制破骨细胞并促进成骨[J].中国组织工程研究,2025,29(31):6765-6771. [20] HWANG JH, PARK YS, KIM HS, et al. Yam-derived exosome-like nanovesicles stimulate osteoblast formation and prevent osteoporosis in mice. J Control Release. 2023;355:184-198. [21] ZHAN W, DENG M, HUANG X, et al. Pueraria lobata-derived exosome-like nanovesicles alleviate osteoporosis by enhancing autophagy. J Control Release. 2023;364:644-653. [22] ZHANG X, WANG G, WANG W, et al. Bone marrow mesenchymal stem cells paracrine TGF-β1 to mediate osteoblast activity in bone repair. Cytokine. 2023; 164:156139. [23] YIN B, YANG M, WANG B, et al. Total flavonoids isolated from Eucommia ulmoides can alleviate bone loss and regulate intestinal microbiota in ovariectomized rats. Front Pharmacol. 2025;16:1513863. [24] WAN Y, HU C, HOU Y, et al. OPG gene-modified adipose-derived stem cells improve bone formation around implants in osteoporotic rat maxillae. Heliyon. 2023;9(10):e19474. [25] 王丹,邵子芹,王雅雯,等.巴戟天-丹参对去卵巢大鼠骨质疏松影响的研究[J].中国骨质疏松杂志,2024,30(12):1751-1756. [26] 李敏,扎克艳•地力夏提,胡君萍,等.酒花提取物调控Wnt/β-catenin信号通路对去卵巢骨质疏松大鼠的作用及机制研究[J].中药新药与临床药理,2024,35(9):1337-1343. [27] YAKYMOVYCH I, YAKYMOVYCH M, HAMIDI A, et al. The type II TGF-β receptor phosphorylates Tyr182 in the type I receptor to activate downstream Src signaling. Sci Signal. 2022;15(760):eabp9521. [28] LIU H, CHEN YG. The interplay between TGF-β signaling and cell metabolism. Front Cell Dev Biol. 2022;10:846723. [29] FREGNANI A, SAGGIN L, GIANESIN K, et al. CK1α/RUNX2 axis in the bone marrow microenvironment: a novel therapeutic target in multiple myeloma. Cancers (Basel). 2022;14(17):4173. [30] LIU Y, XIONG W, LI J, et al. Application of dental pulp stem cells for bone regeneration. Front Med (Lausanne). 2024;11:1339573. [31] GAO M, ZHANG Z, SUN J, et al. The roles of circRNA-miRNA-mRNA networks in the development and treatment of osteoporosis. Front Endocrinol (Lausanne). 2022;13:945310. [32] NAM B, PARK H, LEE YL, et al. TGFβ1 suppressed matrix mineralization of osteoblasts differentiation by regulating SMURF1-C/EBPβ-DKK1 axis. Int J Mol Sci. 2020;21:9771. [33] ZHANG XF, WANG ZX, ZHANG BW, et al. TGF-β1-triggered BMI1 and SMAD2 cooperatively regulate miR-191 to modulate bone formation. Mol Ther Nucleic Acids. 2024;35(2):102164. [34] LU X, LI W, WANG H, et al. The role of the Smad2/3/4 signaling pathway in osteogenic differentiation regulation by ClC-3 chloride channels in MC3T3-E1 cells. J Orthop Surg Res. 2022;17(1):338. [35] LAMORA A, TALBOT J, MULLARD M, et al. TGF-β signaling in bone remodeling and osteosarcoma progression. J Clin Med. 2016;5(11):96. [36] CHEN Y, MEI L, QIAN Y, et al. Integrated bioinformatic analysis of protein landscape in gingival crevicular fluid unveils sequential bioprocess in orthodontic tooth movement. Prog Orthod. 2024;25(1):37. [37] INOUE M, NAGAI-YOSHIOKA Y, YAMASAKI R, et al. Mechanisms involved in suppression of osteoclast supportive activity by TGF-β1 via the ubiquitin-proteasome system. PLoS One. 2022;17(2):e0262612. [38] WU Z, LI W, JIANG K, et al. Regulation of bone homeostasis: signaling pathways and therapeutic targets. MedComm (2020). 2024;5(8):e657. [39] XIA Y, INOUE K, DU Y, et al. TGFβ reprograms TNF stimulation of macrophages towards a non-canonical pathway driving inflammatory osteoclastogenesis. Nat Commun. 2022;13(1):3920. [40] 朱文潇, 王向阳, 刘永辉, 等.平乐三七接骨丸对桡骨骨折兔模型转化生长因子-β/骨形态发生蛋白信号通路表达的影响[J]. 陕西中医,2022,43(9): 1175-1179. [41] XIA C, GE Q, FANG L, et al. TGF-β/Smad2 signalling regulates enchondral bone formation of Gli1+ periosteal cells during fracture healing. Cell Prolif. 2020;53(11):e12904. [42] GELIBTER S, MAROSTICA G, MANDELLI A, et al. The impact of storage on extracellular vesicles: a systematic study. J Extracell Vesicles. 2022;11(2):e12162. |
| [1] | Chen Huiting, Zeng Weiquan, Zhou Jianhong, Wang Jie, Zhuang Congying, Chen Peiyou, Liang Zeqian, Deng Weiming. Tail anchoring technique of vertebroplasty in treatment of osteoporotic vertebral compression fractures with intravertebral cleft: a finite element analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2145-2152. |
| [2] | Zeng Xuan, Weng Rui, Ye Shicheng, Tang Jiadong, Mo Ling, Li Wenchao. Two lumbar rotary manipulation techniques in treating lumbar disc herniation: a finite element analysis of biomechanical differences [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2153-2161. |
| [3] | Cheng Qisheng, Julaiti·Maitirouzi, Xiao Yang, Zhang Chenwei, Paerhati·Rexiti. Finite element analysis of novel variable-diameter screws in modified cortical bone trajectory of lumbar vertebrae [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2162-2171. |
| [4] | Liu Wenlong, Dong Lei, Xiao Zhengzheng, Nie Yu. Finite element analysis of tibial prosthesis loosening after fixed-bearing unicompartmental knee arthroplasty for osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2191-2198. |
| [5] | Chen Long, Wang Xiaozhen, Xi Jintao, Lu Qilin. Biomechanical performance of short-segment screw fixation combined with expandable polyetheretherketone vertebral body replacement in osteoporotic vertebrae [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2226-2235. |
| [6] | Wu Yanting, Li Yu, Liao Jinfeng. Magnesium oxide nanoparticles regulate osteogenesis- and angiogenesis-related gene expressions to promote bone defect healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1885-1895. |
| [7] | Hu Xiongke, Liu Shaohua, Tan Qian, Liu Kun, Zhu Guanghui. Shikonin intervention with bone marrow mesenchymal stem cells improves microstructure of femur in aged mice [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1609-1615. |
| [8] | Li Zhenyu, Zhang Siming, Bai Jiaxiang, Zhu Chen. Osthole improves osteogenic differentiation function of bone marrow mesenchymal stem cells under high-glucose conditions [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1641-1648. |
| [9] | Jin Dongsheng, Zhao Zhanghong, Zhu Ziyin, Zhang Sen, Sun Zuyan, Deng Jiang. Effects of icariin-loaded microsphere-three-dimensional scaffold on osteogenic differentiation of rabbit bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1658-1668. |
| [10] | Zou Yulian, Chen Chaopei, Huang Haixia, Lan Yuyan, Liu Min, Huang Ting. Resveratrol promotes osteogenic differentiation of bone marrow mesenchymal stem cells in an inflammatory microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1669-1678. |
| [11] | Wu Zhilin, , He Qin, Wang Pingxi, Shi Xian, Yuan Song, Zhang Jun, Wang Hao . DYRK2: a novel therapeutic target for rheumatoid arthritis combined with osteoporosis based on East Asian and European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1569-1579. |
| [12] | Zhang Haiwen, Zhang Xian, Xu Taichuan, Li Chao. Bibliometric and visual analysis of the research status and trends of senescence in osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1580-1591. |
| [13] | Wen Guangwei, Zhen Yinghao, Zheng Taikeng, Zhou Shuyi, Mo Guoye, Zhou Tengpeng, Li Haishan, Lai Yiyi. Effects and mechanisms of isoginkgetin on osteoclastogenesis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1348-1358. |
| [14] | Huang Jie, Zeng Hao, Wang Wenchi, Lyu Zhucheng, Cui Wei. Visualization analysis of literature on the effect of lipid metabolism on osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1558-1568. |
| [15] | Yang Zhijie, Zhao Rui, Yang Haolin, Li Xiaoyun, Li Yangbo, Huang Jiachun, Lin Yanping, Wan Lei, HuangHongxing. Postmenopausal osteoporosis: predictive values of muscle mass, grip strength, and appendicular skeletal muscle index [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1073-1080. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||