Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (24): 6345-6353.doi: 10.12307/2026.237
Previous Articles Next Articles
Effects of antioxidant pretreatment on skeletal muscle damage and oxidative stress following acute high-intensity exercise: a meta-analysis
Xia Caigui, Li Wei, Su Yuying, Shi Yu, Yang Zhonghe
- Beijing Sport University, Beijing 100084, China
-
Received:2025-07-29Revised:2025-10-19Online:2026-08-28Published:2026-02-04 -
Contact:Li Wei, PhD, Professor, Beijing Sport University, Beijing 100084, China -
About author:Xia Caigui, MS candidate, Beijing Sport University, Beijing 100084, China
CLC Number:
Cite this article
Xia Caigui, Li Wei, Su Yuying, Shi Yu, Yang Zhonghe. Effects of antioxidant pretreatment on skeletal muscle damage and oxidative stress following acute high-intensity exercise: a meta-analysis[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6345-6353.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
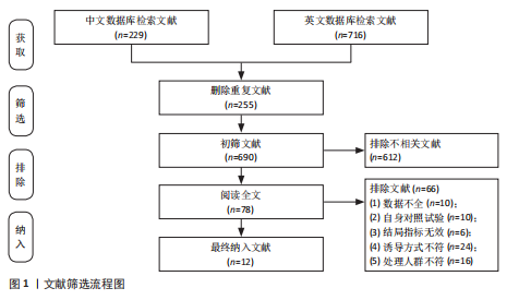
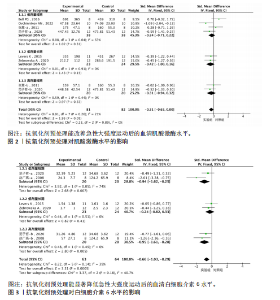
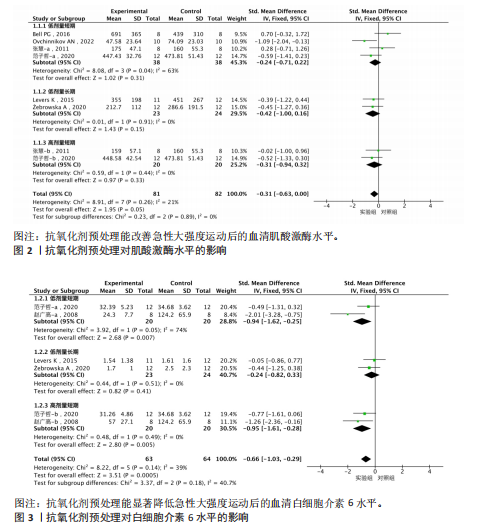
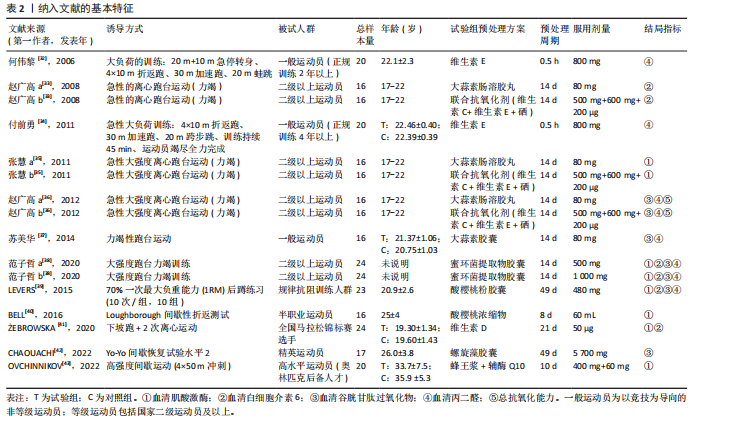
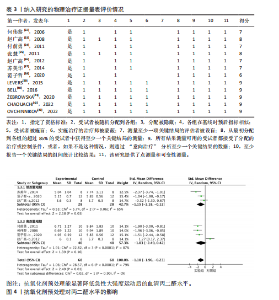
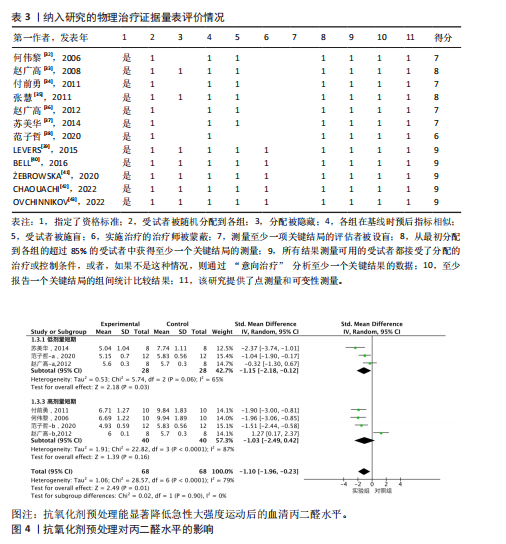
2.1 文献筛选结果 初步从各数据库共检索文献945篇,经过Endnote 20软件去重后得到690篇文献,排除不相关文献612篇后,对所剩的78篇文献全文阅读,最终剩余12篇[32-43]。文献纳入的具体流程见图1。 2.2 纳入文献的基本特征 最终剩余12篇文献,总计16份研究报告纳入此次Meta分析。其中从赵广高等(2008)[33]、张慧等(2011)[35]、赵广高等(2012)[36]及范子哲等(2020)[38]的文献中提取研究2份。16份研究报告共计264名受试者,11篇文献为运动员,1篇文献为规律运动人群,年龄为17-42岁;试验组采用抗氧化剂预处理,对照组采用安慰剂预处理。预处理时间为0.5 h-49 d,以血清肌酸激酶、血清白细胞介素6、血清谷胱甘肽过氧化物、血清丙二醛、总抗氧化能力为结局指标。详细的基本特征见表2。 2.3 纳入研究的质量评价 使用物理治疗证据量表对纳入的12篇文献进行规范的质量性评价[32-43],其中较高质量文献有7篇[32-38],高质量文献5篇[39-43]。研究被试纳入条件明确,均能够按照随机分配的方式完成实验方案,测量报告完整。英文文献均使用分配隐藏与双盲方式干预,中文文献基本采用单盲形式,较少使用分配隐藏。整体上,纳入的所有文献得分均不少于6分,表明方法学的质量较高(表3)。 2.4 Meta分析结果 此次Meta分析中,将抗氧化剂大于500 mg归类为高剂量,低剂量为不超过500 mg;短期预处理为不超过14 d,长期为大于14 d预处理。故将各个研究报告分为低剂量短期、低剂量长期、高剂量短期以及高剂量长期预处理亚组。 2.4.1 血清肌酸激酶 共有8个研究报告对血清肌酸激酶进行分析[35,38-41,43],I2=21%,P=0.26,同质性较好,因此采用固定效应模型进行Meta分析。结果显示,抗氧化剂预处理组的肌酸激酶值低于对照组[SMD=-0.31,95%CI(-0.63,0.00),P=0.05],其中低剂量短期抗氧化剂预处理[SMD=-0.24,95%CI(-0.71,0.22),P=0.31]、低剂量长期预处理[SMD=-0.42,95%CI(-1.00,0.16),P=0.15]、高剂量短期预处理[SMD=-0.31,95%CI(-0.94,0.32),P=0.33]的肌酸激酶含量与对照组相比差异无显著性意义,但低剂量长期预处理降低肌酸激酶含量的效果最好(图2)。 2.4.2 血清白细胞介素6 共有6个研究报告对血清白细胞介素6进行分析[33,38-39,41],I2=39%,P=0.14,存在轻度异质性,故采用固定效应模型进行Meta分析。结果显示,抗氧化剂预处理组的白细胞介素6含量显著低于对照[SMD=-0.66,95%CI(-1.03,-0.29),P=0.000 5],其中低剂量短期[SMD=-0.94,95%CI(-1.62,-0.25),P=0.007]与高剂量短期[SMD=-0.95,95%CI(-1.61,-0.28),P=0.005]预处理抗氧化剂的白细胞介素6含量与对照组相比有显著性差异,且高剂量短期预处理抗氧化剂更能有效降低白细胞介素6水平。低剂量长期组的白细胞介素6水平与对照组相比差异无显著性意义[SMD=-0.24,95%CI(-0.82,0.33),P=0.41](图3)。 2.4.3 血清丙二醛 共有7个研究报告对血清丙二醛水平进行分析[32,34,36-38],I2=79%,P < 0.000 01,存在高度异质性,因此采用随机效应模型进行Meta分析。结果显示:抗氧化剂预处理组的丙二醛含量显著低于对照组[SMD=-1.10,95%CI(-1.96,-0.23),P=0.01]。其中低剂量短期组[SMD=-1.15,95%CI(-2.18,-0.12),P=0.03]的丙二醛含量与对照组相比有显著性差异;高剂量短期预处理组[SMD=-1.03,"
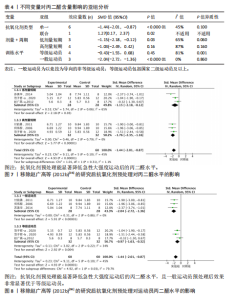
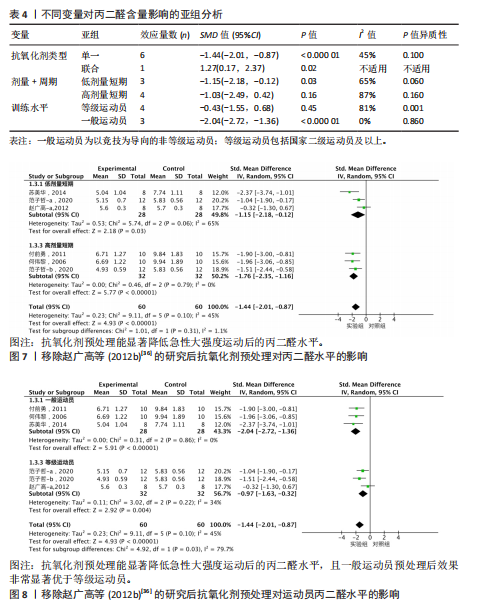
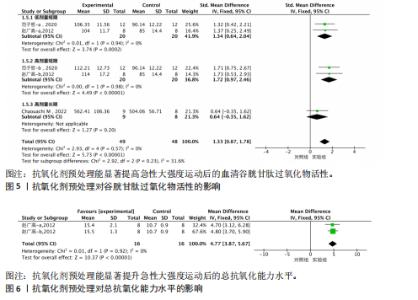
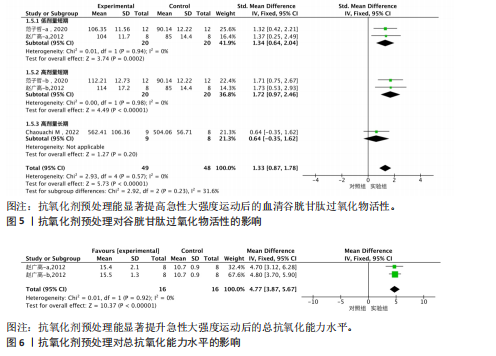
95%CI(-2.49,0.42),P=0.16]的丙二醛含量与对照组相比差异无显著性意义(图4)。 2.4.4 血清谷胱甘肽过氧化物 共5个研究报告对血清谷胱甘肽过氧化物活性进行分析[36,38,42],I2=0%,P=0.57,结果间不存在异质性,故采用固定效应模型进行Meta分析。结果显示,抗氧化剂预处理组的谷胱甘肽过氧化物活性相比对照组有显著性差异[SMD=1.33,95%CI(0.87,1.78),P < 0.000 01]。因高剂量长期预处理组的研究仅1份,故只对低剂量长期组和高剂量长期组进行组间比较,发现低剂量短期预处理组[SMD=1.34,95%CI(0.64,2.04),P=0.000 2]和高剂量短期预处理组[SMD=1.72,95%CI(0.97,2.46),P < 0.000 01]的谷胱甘肽过氧化物活性相比对照组均有显著性差异,但高剂量短期预处理组更能有效地提高谷胱甘肽过氧化物活性(图5)。 2.4.5 总抗氧化能力 共2个研究报告对总抗氧化能力进行分析[36],I2=0%,P=0.92,结果间不存在异质性,因此采用固定效应模型进行Meta分析。结果显示,抗氧化剂预处理组的总抗氧化能力相比对照组有显著性差异[MD=4.77,95%CI(3.87,5.67),P < 0.000 01](图6)。 2.5 敏感性分析 针对丙二醛含量指标存在的显著异质性(I2=79%),此次研究通过亚组分析系统考察了抗氧化剂预处理类型、周期、剂量及受试者训练水平等潜在影响因素。如表4所示,经分层解析发现,赵广高等(2012b)[36]采用的联合抗氧化剂预处理方案(维生素C+维生素E+硒,总剂量1 100.2 mg/d)与其他研究的单一抗氧化剂干预模式存在显著差异,这可能是构成异质性的主要来源。敏感性分析证实,剔除该研究后整体异质性从79%显"
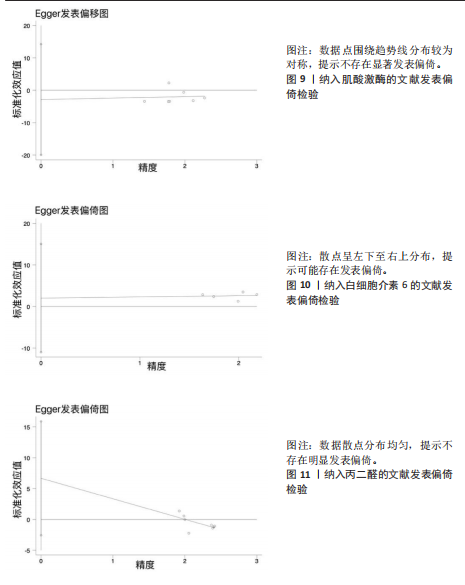
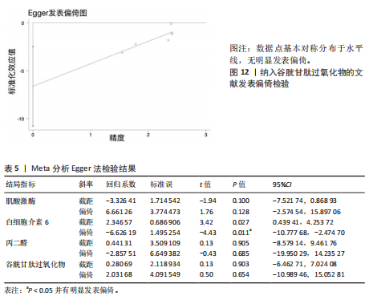
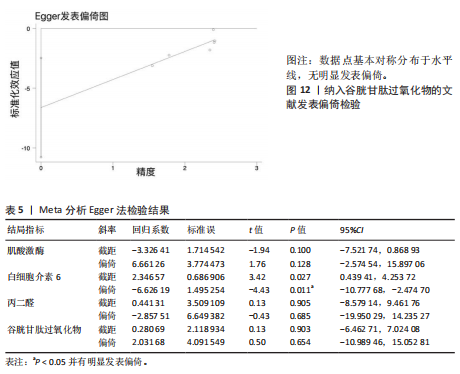
著下降至45%,表明该研究对异质性贡献率较大。进一步分析该研究后发现,其研究对象为24名二级及以上水平运动员,实施周期14 d的联合抗氧化剂预处理(维生素C 500 mg+维生素E 600 mg+硒200 μg/d)。这些参数与其他研究的单一抗氧化剂补充方案(如单纯补充大蒜素或维生素E)形成显著方法学差异。当排除该研究后,合并效应量由原-1.10(95%CI:-1.96,-0.23)提升至-1.44(95%CI:-2.01,-0.87),且统计学显著性进一步增强(P < 0.000 01),见图7。因此,尽管该研究对整体异质性产生显著影响,但考虑到其研究人群(等级运动员)的代表性及干预方案的实践价值,经综合评估仍建议保留该研究进行合并分析。 当剔除赵广高等(2012b)[36]研究报告后,丙二醛含量指标的异质性为45%(P=0.10),因而此次研究根据周期、剂量及训练水平为分组依据进行进一步亚组分析。结果显示:以训练水平为依据的亚组分析后,两组的异质性明显降低,其中等级运动员组I2=34%(P > 0.1),一般运动员组I2=0%(P > 0.1),亚组分析显示显著的组间异质性(I2=79.7%)。这提示训练水平可能是导致异质性较高的主要原因。另外,从图8可见,抗氧化剂预处理均能显著降低一般运动员(P < 0.000 01)和等级运动员(P=0.004)在急性剧烈运动后的丙二醛水平,且一般运动员预处理后效果显著优于等级运动员(P < 0.05)。 2.6 发表偏倚分析 当纳入Meta分析的文献少于10篇时,不建议使用漏斗图进行发表偏倚分析[44],运用Egger检验后,由于总抗氧化能力分析仅纳入2篇文献,故不对总抗氧化能力含量进行Egger检验。如图9-12所示,对肌酸激酶、白细胞介素6、丙二醛水平以及谷胱甘肽过氧化物活性进行Egger检验后发现,纳入肌酸激酶、白细胞介素6、谷胱甘肽过氧化物指标的文献均无明显的发表偏倚(P > 0.1),纳入白细胞介素6指标的文献有明显的发表偏倚(表5)。但对纳入白细胞介素6指标的文献进行Trim and fill检验后,结果表明,无研究剪补,合并效应量为-0.658(约-0.66),异质性低,结果较为稳健。"
| [1] HAYASHI G, CORTOPASSI G. Oxidative stress in inherited mitochondrial diseases. Free Radic Biol Med. 2015;88(Pt A):10-17. [2] DILLARD CJ, LITOV RE, SAVIN WM, et al. Effects of exercise, vitamin E, and ozone on pulmonary function and lipid peroxidation. J Appl Physiol Respir Environ Exerc Physiol. 1978;45(6):927-932. [3] 刘静, 龙建纲, 刘健康. 运动与氧化还原信号调控[J]. 生理科学进展,2014,45(4):263-266. [4] POWERS SK, NELSON WB, HUDSON MB. Exercise-induced oxidative stress in humans: cause and consequences. Free Radic Biol Med. 2011;51(5):942-950. [5] POWERS SK, DEMINICE R, OZDEMIR M, et al. Exercise-induced oxidative stress: Friend or foe? J Sport Health Sci. 2020;9(5):415-425. [6] 孙学军,康志敏,刘文武. H2生物学[M]. 上海:第二军医大学出版社,2013. [7] FORMAN HJ, ZHANG H. Author Correction: Targeting oxidative stress in disease: promise and limitations of antioxidant therapy. Nat Rev Drug Discov. 2021;20(8): 652. [8] CHEN X, ZHU X, WEI A, et al. Nrf2 epigenetic derepression induced by running exercise protects against osteoporosis. Bone Res. 2021;9(1):15. [9] FASIPE B, LI S, LAHER I. Harnessing the cardiovascular benefits of exercise: Are Nrf2 activators useful?. Sports Med Health Sci. 2021;3(2):70-79. [10] DONE AJ, GAGE MJ, NIETO NC, et al. Exercise-induced Nrf2-signaling is impaired in aging. Free Radic Biol Med. 2016;96:130-138. [11] CHENG AJ, YAMADA T, RASSIER DE, et al. Reactive oxygen/nitrogen species and contractile function in skeletal muscle during fatigue and recovery. J Physiol. 2016;594(18):5149-5160. [12] HOSTRUP M, BANGSBO J. Limitations in intense exercise performance of athletes - effect of speed endurance training on ion handling and fatigue development. J Physiol. 2017;595(9):2897-2913. [13] KOCH AJ, PEREIRA R, MACHADO M. The creatine kinase response to resistance exercise. J Musculoskelet Neuronal Interact. 2014;14(1):68-77. [14] WU RE, HUANG WC, LIAO CC, et al. Resveratrol protects against physical fatigue and improves exercise performance in mice. Molecules. 2013; 18(4):4689-4702. [15] SEN CK. Antioxidants in exercise nutrition. Sports Med. 2001;31(13):891-908. [16] RANCHORDAS MK, ROGERSON D, SOLTANI H, et al. Antioxidants for preventing and reducing muscle soreness after exercise: a Cochrane systematic review. Br J Sports Med. 2020;54(2):74-78. [17] GOLDFARB AH, PATRICK SW, BRYER S, et al. Vitamin C supplementation affects oxidative-stress blood markers in response to a 30-minute run at 75% VO2max. Int J Sport Nutr Exerc Metab. 2005;15(3):279-290. [18] BRYER SC, GOLDFARB AH. Effect of high dose vitamin C supplementation on muscle soreness, damage, function, and oxidative stress to eccentric exercise. Int J Sport Nutr Exerc Metab. 2006;16(3):270-280. [19] MERRY TL, RISTOW M. Do antioxidant supplements interfere with skeletal muscle adaptation to exercise training? J Physiol. 2016;594(18):5135-5147. [20] RISTOW M, ZARSE K, OBERBACH A, et al. Antioxidants prevent health-promoting effects of physical exercise in humans. Proc Natl Acad Sci U S A. 2009;106(21):8665-8670. [21] GOMEZ-CABRERA MC, DOMENECH E, ROMAGNOLI M, et al. Oral administration of vitamin C decreases muscle mitochondrial biogenesis and hampers training-induced adaptations in endurance performance. Am J Clin Nutr. 2008;87(1):142-149. [22] 胡国鹏, 王人卫. 体育运动中抗氧化补剂的研究现状与思考[J]. 首都体育学院学报,2012, 24(6):556-564+568. [23] POWERS SK, RADAK Z, JI LL, et al. Reactive oxygen species promote endurance exercise-induced adaptations in skeletal muscles. J Sport Health Sci. 2024;13(6):780-792. [24] KALAFATI M, JAMURTAS AZ, NIKOLAIDIS MG, et al. Ergogenic and antioxidant effects of spirulina supplementation in humans. Med Sci Sports Exerc. 2010;42(1):142-151. [25] SLATTERY KM, DASCOMBE B, WALLACE LK, et al. Effect of N-acetylcysteine on cycling performance after intensified training. Med Sci Sports Exerc. 2014;46(6):1114-1123. [26] WILLEMS ME, BLACKER SD, MONTANARI S, et al. Anthocyanin-Rich Blackcurrant Supplementation as a Nutraceutical Ergogenic Aid for Exercise Performance and Recovery: A Narrative Review. Curr Dev Nutr. 2025;9(1):104523. [27] BOWTELL J, KELLY V. Fruit-Derived Polyphenol Supplementation for Athlete Recovery and Performance. Sports Med. 2019;49(Suppl 1):3-23. [28] PAGE MJ, MCKENZIE JE, BOSSUYT PM, et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. J Clin Epidemiol. 2021;134:178-189. [29] SCHULZ KF, CHALMERS I, HAYES RJ, et al. Empirical evidence of bias. Dimensions of methodological quality associated with estimates of treatment effects in controlled trials. Jama. 1995;273(5):408-412. [30] DE MORTON NA. The PEDro scale is a valid measure of the methodological quality of clinical trials: a demographic study. Aust J Physiother. 2009;55(2):129-133. [31] MAHER CG, SHERRINGTON C, HERBERT RD, et al. Reliability of the PEDro scale for rating quality of randomized controlled trials. Phys Ther. 2003; 83(8):713-721. [32] 何伟黎, 阎洪亮, 徐海兴,等. VitE对足球运动员无氧训练血浆自由基水平和抗氧化能力的影响[J]. 北京体育大学学报,2006,29(2):204-205. [33] 赵广高, 苏全生, 张慧,等. 大蒜素与联合抗氧化剂干预对运动员DOMS和血浆IL-6水平变化的影响[J]. 体育科学,2008,28(7):66-71. [34] 付前勇. 大负荷无氧训练前服用VitE对篮球运动员血浆自由基和抗氧化能力的影响[J]. 武汉船舶职业技术学院学报,2011,10(3):47-49. [35] 张慧, 时震宇, 赵广高,等. 大蒜素与联合抗氧化剂干预对运动员DOMS和CK-MM、CK等的影响[J]. 武汉体育学院学报,2011,45(3):59-63. [36] 赵广高, 苏全生, 李新建,等. 大蒜素与联合抗氧化剂对一次大强度离心运动后运动员血清T-AOC等水平的影响[J]. 首都体育学院学报, 2012,24(2):189-192. [37] 苏美华, 杨多多, 许庆忠. 大蒜素抗运动性氧应激和DNA损伤的作用研究[J]. 山东体育学院学报,2014,30(6):87-91. [38] 范子哲, 孙宇岸. 蜜环菌提取物对运动性损伤的改善作用[J]. 中国食用菌,2020,39(8): 210-214. [39] LEVERS K, DALTON R, GALVAN E, et al. Effects of powdered Montmorency tart cherry supplementation on an acute bout of intense lower body strength exercise in resistance trained males. J Int Soc Sports Nutr. 2015;12:41. [40] BELL PG, STEVENSON E, DAVISON GW, et al. The Effects of Montmorency Tart Cherry Concentrate Supplementation on Recovery Following Prolonged, Intermittent Exercise. Nutrients. 2016;8(7):441. [41] ŻEBROWSKA A, SADOWSKA-KRĘPA E, STANULA A, et al. The effect of vitamin D supplementation on serum total 25(OH) levels and biochemical markers of skeletal muscles in runners. J Int Soc Sports Nutr. 2020;17(1):18. [42] CHAOUACHI M, GAUTIER S, CARNOT Y, et al. Spirulina supplementation prevents exercise-induced lipid peroxidation, inflammation and skeletal muscle damage in elite rugby players. J Hum Nutr Diet. 2022;35(6):1151-1163. [43] OVCHINNIKOV AN, PAOLI A, SELEZNEV VV, et al. Royal jelly plus coenzyme Q10 supplementation improves high-intensity interval exercise performance via changes in plasmatic and salivary biomarkers of oxidative stress and muscle damage in swimmers: a randomized, double-blind, placebo-controlled pilot trial. J Int Soc Sports Nutr. 2022;19(1):239-257. [44] 刘壮, 高奉, 朱婷,等. 减重训练对前交叉韧带重建术后患者临床疗效的系统评价与Meta分析[J]. 体育科学,2023,43(3):78-88+97. [45] SILVA LA, PINHO CA, SILVEIRA PC, et al. Vitamin E supplementation decreases muscular and oxidative damage but not inflammatory response induced by eccentric contraction. J Physiol Sci. 2010;60(1):51-57. [46] KOURIE JI. Interaction of reactive oxygen species with ion transport mechanisms. Am J Physiol. 1998;275(1):C1-24. [47] ICHIMURA M, KATO S, TSUNEYAMA K, et al. Phycocyanin prevents hypertension and low serum adiponectin level in a rat model of metabolic syndrome. Nutr Res. 2013;33(5):397-405. [48] BRITO AF, SILVA AS, DE SOUZA AA, et al. Aortic Response to Strength Training and Spirulina platensis Dependent on Nitric Oxide and Antioxidants. Front Physiol. 2018;9:1522. [49] CALELLA P, CERULLO G, DI DIO M, et al. Antioxidant, anti-inflammatory and immunomodulatory effects of spirulina in exercise and sport: A systematic review. Front Nutr. 2022; 9:1048258. [50] FRANCA GAM SA, COSTA MJC. Spirulina Does Not Decrease Muscle Damage nor Oxdidative Stress in Cycling Athletes with Adequate Nutritional Status. Biol Sport. 2010;27:249-253. [51] 葛泓丽. 抗氧化补剂短期与长期干预对运动表现的影响探究[J]. 当代体育科技,2025,15(4):5-8. [52] CROISIER JL, CAMUS G, VENNEMAN I, et al. Effects of training on exercise-induced muscle damage and interleukin 6 production. Muscle Nerve. 1999;22(2):208-212. [53] FISCHER CP, HISCOCK NJ, PENKOWA M, et al. Supplementation with vitamins C and E inhibits the release of interleukin-6 from contracting human skeletal muscle. J Physiol. 2004;558(Pt 2): 633-645. [54] 苏全生, 田野, 王东辉,等. 大蒜素对大鼠离心运动后血IL-6、CK、CK-MM和Ca2+变化的影响[J]. 体育科学,2006,26(3):73-76. [55] TENÓRIO M, GRACILIANO NG, MOURA FA, et al. N-Acetylcysteine (NAC): Impacts on Human Health. Antioxidants (Basel). 2021;10(6):967. [56] DE ANDRADE KQ, MOURA FA, DOS SANTOS JM, et al. Oxidative Stress and Inflammation in Hepatic Diseases: Therapeutic Possibilities of N-Acetylcysteine. Int J Mol Sci. 2015;16(12): 30269-30308. [57] AGUIAR SS, SOUSA CV, DEUS LA, et al. Oxidative stress, inflammatory cytokines and body composition of master athletes: The interplay. Exp Gerontol. 2020;130:110806. [58] FERNANDES MSS, FIDELIS D, AIDAR FJ, et al. Coenzyme Q10 Supplementation in Athletes: A Systematic Review. Nutrients. 2023;15(18):3990. [59] NGO V, DUENNWALD ML. Nrf2 and Oxidative Stress: A General Overview of Mechanisms and Implications in Human Disease. Antioxidants (Basel). 2022;11(12):2345. [60] TKACZENKO H, KURHALUK N. Antioxidant-Rich Functional Foods and Exercise: Unlocking Metabolic Health Through Nrf2 and Related Pathways. Int J Mol Sci. 2025;26(3):1098. [61] DROBNIC F, LIZARRAGA MA, CABALLERO-GARCÍA A, et al. Coenzyme Q(10) Supplementation and Its Impact on Exercise and Sport Performance in Humans: A Recovery or a Performance-Enhancing Molecule? Nutrients. 2022;14(9):1811. [62] FERNÁNDEZ-LÁZARO D, MIELGO-AYUSO J, SECO CALVO J, et al. Modulation of Exercise-Induced Muscle Damage, Inflammation, and Oxidative Markers by Curcumin Supplementation in a Physically Active Population: A Systematic Review. Nutrients. 2020;12(2):501. [63] FERNÁNDEZ-LÁZARO D, DOMÍNGUEZ-ORTEGA C, BUSTO N, et al. Influence of N-Acetylcysteine Supplementation on Physical Performance and Laboratory Biomarkers in Adult Males: A Systematic Review of Controlled Trials. Nutrients. 2023;15(11):2463. [64] LARSEN S. Acute antioxidant supplementation and performance - Should this be considered. Free Radic Biol Med. 2024;224:301-309. [65] MORRISON D, HUGHES J, DELLA GATTA PA, et al. Vitamin C and E supplementation prevents some of the cellular adaptations to endurance-training in humans. Free Radic Biol Med. 2015; 89:852-862. [66] YANG CT, WENG PW, CHIEN LH, et al. Effects of vitamin D(3) supplementation on oxidative stress and antioxidant enzyme after strenuous endurance exercise in healthy young men: a double-blind, placebo-controlled trial. Biol Sport. 2025;42(1):137-144. [67] TONG TK, LIN H, LIPPI G, et al. Serum oxidant and antioxidant status in adolescents undergoing professional endurance sports training. Oxid Med Cell Longev. 2012;2012:741239. [68] 海春旭. 自由基医学[M]. 西安: 第四军医大学出版社,2006:180-191. [69] PINGITORE A, LIMA GP, MASTORCI F, et al. Exercise and oxidative stress: potential effects of antioxidant dietary strategies in sports. Nutrition. 2015;31(7-8):916-922. [70] NIKI E. Action of ascorbic acid as a scavenger of active and stable oxygen radicals. Am J Clin Nutr. 1991;54(6 Suppl):1119s-1124s. [71] MORTENSEN A, LYKKESFELDT J. Does vitamin C enhance nitric oxide bioavailability in a tetrahydrobiopterin-dependent manner? In vitro, in vivo and clinical studies. Nitric Oxide. 2014;36:51-57. [72] MASON SA, TREWIN AJ, PARKER L, et al. Antioxidant supplements and endurance exercise: Current evidence and mechanistic insights. Redox Biol. 2020;35:101471. [73] MICHAILIDIS Y, KARAGOUNIS LG, TERZIS G, et al. Thiol-based antioxidant supplementation alters human skeletal muscle signaling and attenuates its inflammatory response and recovery after intense eccentric exercise. Am J Clin Nutr. 2013;98(1):233-245. [74] MCKENNA MJ, MEDVED I, GOODMAN CA, et al. N-acetylcysteine attenuates the decline in muscle Na+,K+-pump activity and delays fatigue during prolonged exercise in humans. J Physiol. 2006;576(Pt 1):279-288. [75] BENTINGER M, TEKLE M, DALLNER G. Coenzyme Q--biosynthesis and functions. Biochem Biophys Res Commun. 2010;396(1):74-79. [76] SCHMELZER C, LINDNER I, RIMBACH G, et al. Functions of coenzyme Q10 in inflammation and gene expression. Biofactors. 2008;32(1-4): 179-183. [77] SINGH RB, NIAZ MA, RASTOGI SS, et al. Effect of hydrosoluble coenzyme Q10 on blood pressures and insulin resistance in hypertensive patients with coronary artery disease. J Hum Hypertens. 1999;13(3):203-208. [78] VITALE KC, HUEGLIN S, BROAD E. Tart Cherry Juice in Athletes: A Literature Review and Commentary. Curr Sports Med Rep. 2017;16(4): 230-239. [79] BOREK C. Antioxidant health effects of aged garlic extract. J Nutr. 2001;131(3s): 1010s-1015s. [80] COLÍN-GONZÁLEZ AL, SANTANA RA, SILVA-ISLAS CA, et al. The antioxidant mechanisms underlying the aged garlic extract- and S-allylcysteine-induced protection. Oxid Med Cell Longev. 2012; 2012:907162. [81] KIM KM, CHUN SB, KOO MS, et al. Differential regulation of NO availability from macrophages and endothelial cells by the garlic component S-allyl cysteine. Free Radic Biol Med. 2001;30(7): 747-756. [82] KALAYARASAN S, SRIRAM N, SURESHKUMAR A, et al. Chromium (VI)-induced oxidative stress and apoptosis is reduced by garlic and its derivative S-allylcysteine through the activation of Nrf2 in the hepatocytes of Wistar rats. J Appl Toxicol. 2008;28(7):908-919. [83] IDE N, LAU BH. Garlic compounds minimize intracellular oxidative stress and inhibit nuclear factor-kappa b activation. J Nutr. 2001; 131(3s):1020s-1026s. [84] FINAMORE A, PALMERY M, BENSEHAILA S, et al. Antioxidant, Immunomodulating, and Microbial-Modulating Activities of the Sustainable and Ecofriendly Spirulina. Oxid Med Cell Longev. 2017;2017:3247528. [85] LU HK, HSIEH CC, HSU JJ, et al. Preventive effects of Spirulina platensis on skeletal muscle damage under exercise-induced oxidative stress. Eur J Appl Physiol. 2006;98(2):220-226. [86] BERMEJO-BESCÓS P, PIÑERO-ESTRADA E, VILLAR DEL FRESNO AM. Neuroprotection by Spirulina platensis protean extract and phycocyanin against iron-induced toxicity in SH-SY5Y neuroblastoma cells. Toxicol In Vitro. 2008;22(6):1496-1502. [87] SEPIDARKISH M, FARSI F, AKBARI-FAKHRABADI M, et al. The effect of vitamin D supplementation on oxidative stress parameters: A systematic review and meta-analysis of clinical trials. Pharmacol Res. 2019;139:141-152. [88] YIU YF, YIU KH, SIU CW, et al. Randomized controlled trial of vitamin D supplement on endothelial function in patients with type 2 diabetes. Atherosclerosis. 2013;227(1): 140-146. [89] 吴军,李君哲,黄聪聪,等. 蜜环菌多糖对肺癌患者SOD、GSH-Px、GSH和MDA水平的影响及原因分析[J]. 解放军预防医学杂志, 2019,37(8):115-116+119. [90] CLEMENTE-SUÁREZ VJ, BUSTAMANTE-SANCHEZ Á, MIELGO-AYUSO J, et al. Antioxidants and Sports Performance. Nutrients. 2023;15(10):2371. [91] FERNÁNDEZ-LÁZARO D, FERNANDEZ-LAZARO CI, MIELGO-AYUSO J, et al. The Role of Selenium Mineral Trace Element in Exercise: Antioxidant Defense System, Muscle Performance, Hormone Response, and Athletic Performance. A Systematic Review. Nutrients. 2020; 12(6):1790. [92] ZHANG H, SU QS. Effect of Compound Antioxidant to Athletes’ T-AOC, SOD, GSH-Px and MDA after One-off High-intensity Exercise. J Beijing Sport Univ. 2008;31(4):483-485. [93] PAULSEN G, CUMMING KT, HOLDEN G, et al. Vitamin C and E supplementation hampers cellular adaptation to endurance training in humans: a double-blind, randomised, controlled trial. J Physiol. 2014;592(8):1887-1901. [94] BLOOMER RJ, GOLDFARB AH, MCKENZIE MJ, et al. Effects of antioxidant therapy in women exposed to eccentric exercise. Int J Sport Nutr Exerc Metab. 2004;14(4):377-388. [95] CANNON JG, ORENCOLE SF, FIELDING RA, et al. Acute phase response in exercise: interaction of age and vitamin E on neutrophils and muscle enzyme release. Am J Physiol. 1990;259(6 Pt 2):R1214-1219. [96] KNEZ WL, JENKINS DG, COOMBES JS. Oxidative stress in half and full Ironman triathletes. Med Sci Sports Exerc. 2007;39(2):283-288. |
| [1] | Yang Xuetao, Zhu Menghan, Zhang Chenxi, Sun Yimin, Ye Ling. Applications and limitations of antioxidant nanomaterials in oral cavity [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2044-2053. |
| [2] |
Dong Chunyang, Zhou Tianen, Mo Mengxue, Lyu Wenquan, Gao Ming, Zhu Ruikai, Gao Zhiwei.
Action mechanism of metformin combined with Eomecon chionantha Hance dressing in treatment of deep second-degree burn wounds#br#
#br#
[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2001-2013.
|
| [3] | Liu Anting, Lu Jiangtao, Zhang Wenjie, He Ling, Tang Zongsheng, Chen Xiaoling. Regulation of AMP-activated protein kinase by platelet lysate inhibits cadmium-induced neuronal apoptosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1800-1807. |
| [4] | Chen Qiang, Wu Wenjuan, Jiang Shuhua, Huang Da. Physical exercise improves physical function in burn patients: a systematic review and meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1269-1281. |
| [5] | Yang Xiao, Bai Yuehui, Zhao Tiantian, Wang Donghao, Zhao Chen, Yuan Shuo. Cartilage degeneration in temporomandibular joint osteoarthritis: mechanisms and regenerative challenges [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 926-935. |
| [6] | Fang Enhui, Guan Hui, Ma Lihong. Non-invasive brain stimulation for core symptoms in children with autism spectrum disorder: a network meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6373-6381. |
| [7] | Yuan Min, Han Yu, Liu Jinhong, Zhang Jingyu, Cao Jimin, Sun Teng . Role and mechanism of ABL1 in myocardial necroptosis and cardiac ischemia/reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6247-6258. |
| [8] | Zhang Xiaoxu, Tian Zhenli, Xie Tingting. Roles of pregnane X receptor in sodium arsenite-induced oxidative stress and inflammatory injury in human normal hepatocytes [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6259-6266. |
| [9] | Li Huaying, Li Hao, Peng Wuxun, Dong Wentao. Mechanism of cuproptosis in the diagnosis and treatment of orthopedic-related diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6275-6281. |
| [10] | Li Yanfeng, Zhang Yilin, Kong Hao, Zheng Hang, Liu Jiajun, Yin Mingyue, Qiu Bopeng, Huang Kongyun, Liu Hengxian, Zhong Yuming, Chen Jun, Xu Kai. Effect of blood flow restriction training on the magnitude and temporal characteristics of post-activation performance enhancement: a systematic review and meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 6150-6146. |
| [11] | Yang Zijiang, Guo Chenggen, Deng Ziao, Xue Xinxuan. Postbiotic targeting muscle aging: mechanistic insights and application prospects of urolithin A [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(22): 5804-5813. |
| [12] | Dong Chao, Zhao Mohan, Liu Yunan, Yang Zeli, Chen Leqin, Wang Lanfang. Effects of magnetic nano-drug carriers on exercise-induced muscle injury and inflammatory response in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(2): 345-353. |
| [13] | Liao Xingzhuan, Li Guangdi, Wu Yabin, Liu Xingyu, Wan Jiajia. Molecular mechanisms underlying non-coding RNA regulation of ferroptosis in osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(18): 4713-4725. |
| [14] | Zhang Shuli, Hou Chaowen, Yuan Shanshan, Ma Yuhua . Mechanism by which exercise regulates autophagy in different physiological systems [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(18): 4737-4748. |
| [15] | Huang Lei, Wang Xianghong, Zhang Xianxu, Li Shicheng, Luo Zhiqiang. Mechanism and therapeutic potential of nuclear factor E2-related factor 2 in regulating non-infectious spinal diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(15): 3971-3982. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||