Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (24): 6247-6258.doi: 10.12307/2026.165
Previous Articles Next Articles
Role and mechanism of ABL1 in myocardial necroptosis and cardiac ischemia/reperfusion injury
Yuan Min, Han Yu, Liu Jinhong, Zhang Jingyu, Cao Jimin, Sun Teng
- Key Laboratory of Cellular Physiology at Shanxi Medical University, Ministry of Education, Key Laboratory of Cellular Physiology of Shanxi Province, and the Department of Physiology, School of Basic Medicine, Shanxi Medical University, Taiyuan 030001, Shanxi Province, China
-
Received:2025-03-26Revised:2025-09-03Online:2026-08-28Published:2026-01-30 -
Contact:Sun Teng, PhD, Associate professor, Doctoral supervisor, Key Laboratory of Cellular Physiology at Shanxi Medical University, Ministry of Education, Key Laboratory of Cellular Physiology of Shanxi Province, and the Department of Physiology, School of Basic Medicine, Shanxi Medical University, Taiyuan 030001, Shanxi Province, China -
About author:Yuan Min, MS candidate, Key Laboratory of Cellular Physiology at Shanxi Medical University, Ministry of Education, Key Laboratory of Cellular Physiology of Shanxi Province, and the Department of Physiology, School of Basic Medicine, Shanxi Medical University, Taiyuan 030001, Shanxi Province, China -
Supported by:National Natural Science Foundation of China (General Program), No. 82170294 (to ST); National Natural Science Foundation of China (Youth Program), No. 81800268 (to ST); National Natural Science Foundation of China (General Program), No. 82170523 (to CJM); The Central Leading Local Science and Technology Development Fund Project, No. YDZJSX2022A061 (to ST); Shanxi Province Higher Education “Billion Project” Science and Technology Guidance Project, No. BYJL006 (to ST)
CLC Number:
Cite this article
Yuan Min, Han Yu, Liu Jinhong, Zhang Jingyu, Cao Jimin, Sun Teng . Role and mechanism of ABL1 in myocardial necroptosis and cardiac ischemia/reperfusion injury[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6247-6258.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
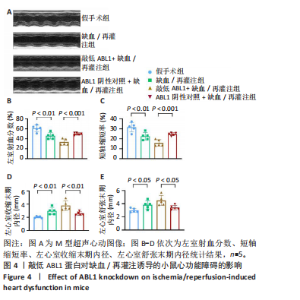
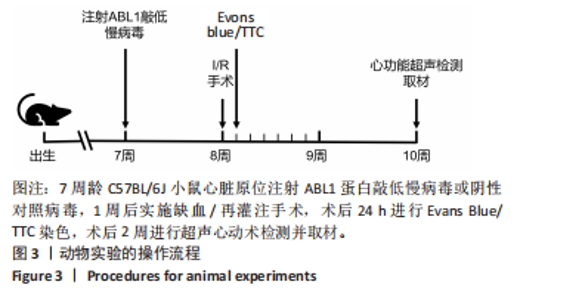
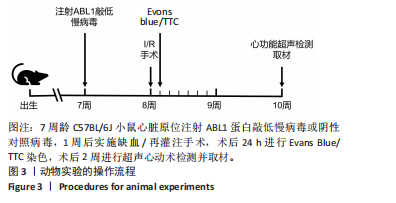
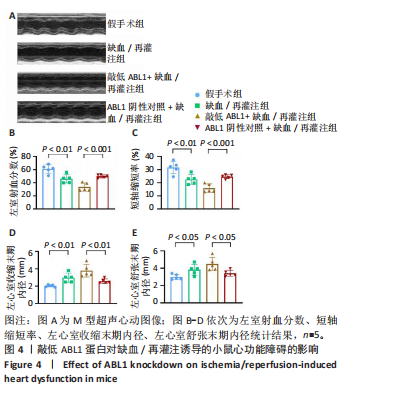
左心室短轴视图下小鼠心脏的M型超声图像显示,与假手术组相比,缺血/再灌注组小鼠心脏搏动明显减弱,这一现象被ABL1蛋白的敲低加重,见图4A。通过对多个心脏功能参数进行统计分析后发现,与假手术组相比,缺血/再灌注组小鼠心脏的左室射血分数和短轴缩短率显著减小(均P < 0.01),左心室收缩末期内径(P < 0.01)和左心室舒张末期内径(P < 0.05)显著增大,见图4B-E。在心脏原位注射ABL1蛋白敲低慢病毒后,与阴性对照+缺血/再灌注组相比,ABL1蛋白的敲低加剧了缺血/再灌注后心脏功能紊乱,表现为心脏左室射血分数和短轴缩短率进一步降低(均 P < 0.001),左心室收缩末期内径(P < 0.01)和左心室舒张末期内径(P < 0.05)进一步增大,见图4B-E。上述结果表明,ABL1蛋白调控了缺血/再灌注诱导的心功能障碍。"
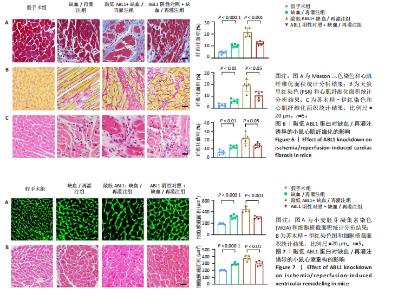
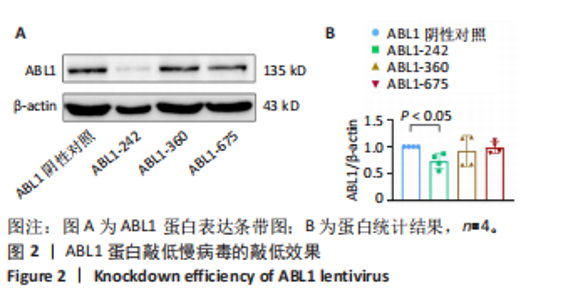
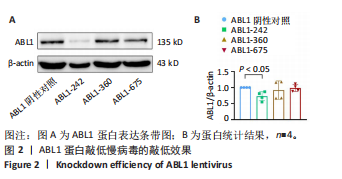
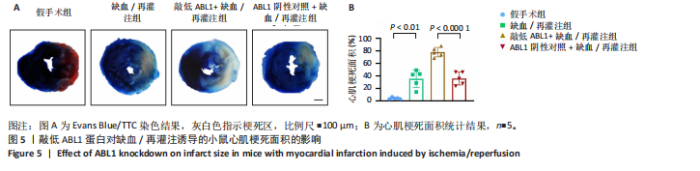
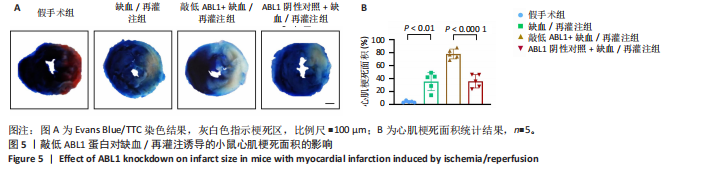
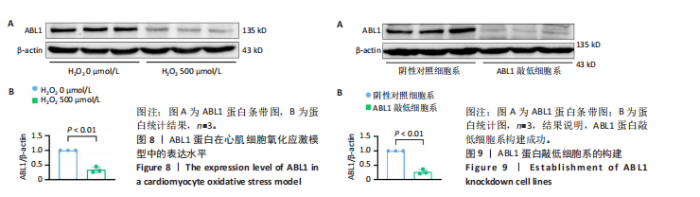
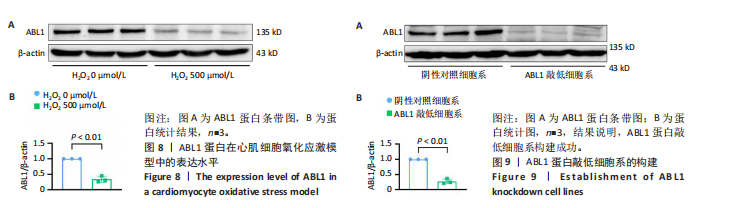
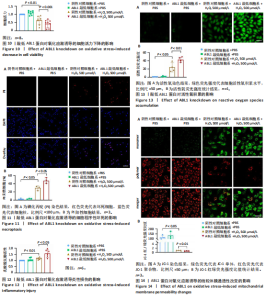
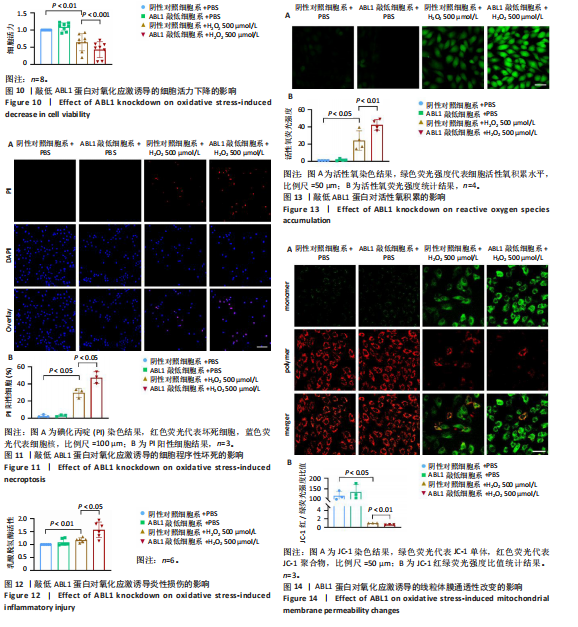
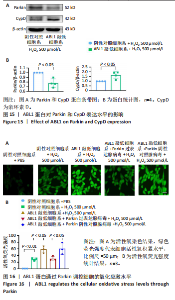
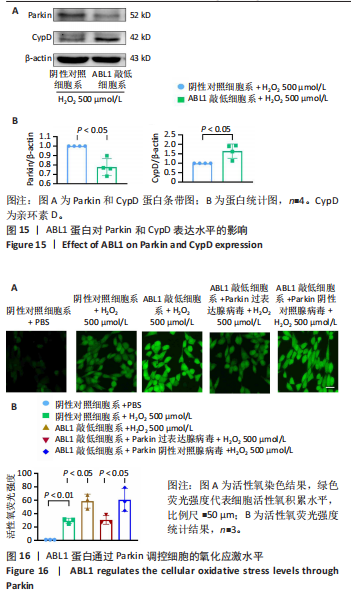
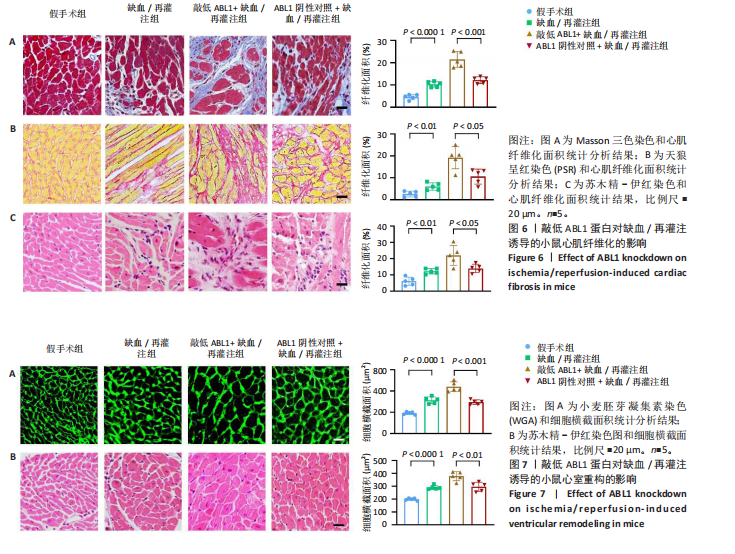
2.6 敲低ABL1蛋白加剧缺血/再灌注诱导的心肌纤维化 通过Masson三色染色、天狼星红染色(PSR)和苏木精-伊红染色评估敲低ABL1蛋白对缺血/再灌注后心肌纤维化的作用。结果显示,与假手术组相比,缺血/再灌注组小鼠心肌发生纤维化(Pmasson < 0.000 1,PPSR < 0.01,P苏木精-伊红 < 0.01),见图6A-C。在心脏原位注射ABL1蛋白敲低慢病毒后,与阴性对照组+缺血/再灌注组相比,ABL1蛋白敲低后小鼠心肌的纤维化面积进一步增大(Pmasson < 0.001,PPSR < 0.05,P苏木精-伊红 < 0.05),见图6A-C。上述结果说明,敲低ABL1蛋白显著加重缺血/再灌注诱导的心肌纤维化。 2.7 敲低ABL1蛋白加重缺血/再灌注诱导的小鼠心室重构 通过小麦胚芽凝集素(WGA)染色和苏木精-伊红染色来检测敲低ABL1蛋白对小鼠心肌细胞肥大的影响。与假手术组相比,缺血/再灌注组小鼠的心肌细胞表面积显著增大(PWGA < 0.000 1, P苏木精-伊红 < 0.000 1),这一现象被ABL1蛋白的敲低进一步加剧(PWGA < 0.001,P苏木精-伊红 < 0.01),见图7A-B。结果表明, ABL1蛋白调控缺血/再灌注诱导的心室重构。 2.8 ABL1蛋白在心肌细胞氧化应激模型中表达水平显著下调 为探究ABL1蛋白在心肌细胞氧化应激模型中的作用,使用500 μmol/L H2O2处理H9c2细胞12 h,通过蛋白质印迹法检测ABL1蛋白的表达水平,结果显示,与对照组相比,H2O2处理的心肌细胞中ABL1蛋白的表达水平显著下调(P < 0.01),见图8。 2.9 敲低ABL1蛋白加剧氧化应激诱导的细胞活力下降 2.9.1 蛋白质印迹法检测 为了研究ABL1蛋白在H2O2诱导的心肌细胞氧化应激模型中的作用,使用Crispr/Cas9技术构建了ABL1蛋白敲低心肌细胞系。与感染对照慢病毒的细胞系(阴性对照细胞系)相比,感染ABL1敲低慢病毒的细胞系(ABL1敲低细胞系)ABL1蛋白表达水平显著降低(P < 0.01),见图9。 2.9.2 CCK8法检测 氧化应激可抑制细胞活力[17],因此检测了敲低ABL1蛋白对氧化应激诱导的细胞活力下降的影响,用500 μmol/L H2O2分别处理阴性对照细胞系和ABL1蛋白敲低细胞系细胞12 h,结果显示,与用PBS处理的阴性对照细胞系细胞相比,H2O2处理后的阴性对照细胞系细胞的细胞活力显著下降(P < 0.01),而ABL1蛋白敲低细胞系细胞再经H2O2处理,其细胞活力进一步下降(P < 0.001),见图10。 上述结果说明,敲低ABL1蛋白加剧氧化应激诱导的细胞活力下降。 2.10 敲低ABL1蛋白加重氧化应激诱导的细胞程序性坏死 通过碘化丙啶(PI)染色和乳酸脱氢酶活性来评估细胞坏死情况。结果显示,与阴性对照组相比,H2O2处理后发生坏死的细胞显著增多(P < 0.05),见图11,乳酸脱氢酶活性显著升高(P < 0.01),见图12。ABL1蛋白敲低细胞系细胞再经H2O2处理,细胞坏死情况进一步加剧(P < 0.05),见图11,乳酸脱氢酶活性进一步升高(P < 0.05),见图12。以上结果表明,ABL1蛋白调控了氧化应激诱导的心肌细胞程序性坏死。 2.11 敲低ABL1蛋白增强心肌细胞活性氧积累 通过测定细胞中活性氧的积累来探究ABL1蛋白对心肌细胞氧化应激损伤的影响。结果显示,与阴性对照细胞系+PBS组相比,阴性对照细胞系+H2O2 500 μmol/L组细胞活性氧生成显著增多(P < 0.05),而这一现象被敲低ABL1蛋白显著加强(P < 0.01),见图13。 2.12 ABL1蛋白调控心肌细胞线粒体膜通透性 实验结果显示,H2O2处理的心肌细胞线粒体膜电位水平显著降低(P < 0.05),而敲低ABL1蛋白则加剧了这一现象(P < 0.01),见图14。以上结果表明,ABL1蛋白调控了氧化应激诱导的线粒体膜通透性改变。 2.13 ABL1蛋白可能靶向Parkin-CypD通路介导氧化应激诱导的心肌细胞程序性坏死 Parkin/亲环素D信号轴是介导mPTP开放和线粒体途径程序性坏死的关键通路[27],于是,探究了ABL1蛋白是否靶向Parkin/亲环素D通路。结果显示,在H2O2处理的心肌细胞中,相较于阴性对照组,敲低ABL1蛋白显著下调Parkin表达水平(P < 0.05),且显著上调亲环素D表达水平(P < 0.05),见图15。与阴性对照组相比,敲低ABL1蛋白显著加重了H2O2诱导的活性氧积累(P < 0.05),而这一现象被过表达Parkin显著减弱了(P < 0.05),见图16。以上结果表明,在氧化应激模型中,ABL1蛋白调控Parkin和亲环素D表达,并通过Parkin调控细胞活性氧水平。"
| [1] HEUSCH G. Myocardial ischemia/reperfusion: Translational pathophysiology of ischemic heart disease. Med. 2024;5(1):10-31. [2] CHANG AJ, LIANG Y, HAMILTON SA, et al. Medical Decision-Making and Revascularization in Ischemic Cardiomyopathy. Med Clin North Am. 2024; 108(3):553-566. [3] MONTONE RA, CAMILLI M, CALVIERI C, et al. Exposome in ischaemic heart disease: beyond traditional risk factors. Eur Heart J. 2024;45(6):419-438. [4] XIAO H, ZHANG M, WU H, et al. CIRKIL Exacerbates Cardiac Ischemia/Reperfusion Injury by Interacting With Ku70. Circ Res. 2022;130(5):e3-e17. [5] WELT FGP, BATCHELOR W, SPEARS JR, et al. Reperfusion Injury in Patients With Acute Myocardial Infarction: JACC Scientific Statement.J Am Coll Cardiol. 2024;83(22):2196-2213. [6] BAWASKAR P, THOMAS N, ISMAIL K, et al. Nonischemic or Dual Cardiomyopathy in Patients With Coronary Artery Disease. Circulation. 2024;149(11):807-821. [7] PERERA D, CLAYTON T, O’KANE PD, et al. Percutaneous Revascularization for Ischemic Left Ventricular Dysfunction. N Engl J Med. 2022;387(15): 1351-1360. [8] PANG H, HUANG G, XIE Z, et al. The role of regulated necrosis in diabetes and its complications. J Mol Med (Berl). 2024;102(4):495-505. [9] WANG K, LI FH, ZHOU LY, et al. HNEAP Regulates Necroptosis of Cardiomyocytes by Suppressing the m(5) C Methylation of Atf7 mRNA. Adv Sci (Weinh). 2023;10(34):e2304329. [10] GUO R, JIA X, DING Z, et al. Loss of MLKL ameliorates liver fibrosis by inhibiting hepatocyte necroptosis and hepatic stellate cell activation.Theranostics. 2022;12(11):5220-5236. [11] MEIER P, LEGRAND AJ, ADAM D, et al. Immunogenic cell death in cancer: targeting necroptosis to induce antitumour immunity. Nat Rev Cancer. 2024;24(5):299-315. [12] VRINGER E, TAIT SWG. Mitochondria and cell death-associated inflammation. Cell Death Differ. 2023;30(2):304-312. [13] YIN H, GUO X, CHEN Y, et al. TAB2 deficiency induces dilated cardiomyopathy by promoting RIPK1-dependent apoptosis and necroptosis. J Clin Invest. 2022;132(4):e152297. [14] GAO P, CAO M, JIANG X, et al. Cannabinoid Receptor 2-Centric Molecular Feedback Loop Drives Necroptosis in Diabetic Heart Injuries.Circulation. 2023;147(2):158-174. [15] SONG Z, SONG H, LIU D, et al. Overexpression of MFN2 alleviates sorafenib-induced cardiomyocyte necroptosis via the MAM-CaMKIIδ pathway in vitro and in vivo.Theranostics. 2022;12(3):1267-1285. [16] ZHU H, TAN Y, DU W, et al. Phosphoglycerate mutase 5 exacerbates cardiac ischemia-reperfusion injury through disrupting mitochondrial quality control.Redox Biol. 2021;38:101777. [17] LI Z, DAI R, CHEN M, et al. p55γ degrades RIP3 via MG53 to suppress ischaemia-induced myocardial necroptosis and mediates cardioprotection of preconditioning. Cardiovasc Res. 2023;119(14):2421-2440. [18] SONG C, DONG Q, YAO Y, et al. Nonreceptor tyrosine kinase ABL1 regulates lysosomal acidification by phosphorylating the ATP6V1B2 subunit of the vacuolar-type H(+)-ATPase. Autophagy. 2025;11:1-20. [19] MALNASSY G, KEATING CR, GAD S, et al. Inhibition of Abelson Tyrosine-Protein Kinase 2 Suppresses the Development of Alcohol-Associated Liver Disease by Decreasing PPARgamma Expression. Cell Mol Gastroenterol Hepatol. 2023;16(5):685-709. [20] BLAKES AJM, GAUL E, LAM W, et al. Pathogenic variants causing ABL1 malformation syndrome cluster in a myristoyl-binding pocket and increase tyrosine kinase activity. Eur J Hum Genet. 2021;29(4):593-603. [21] WANG F, HOU W, CHITSIKE L, et al. ABL1, Overexpressed in Hepatocellular Carcinomas, Regulates Expression of NOTCH1 and Promotes Development of Liver Tumors in Mice. Gastroenterology. 2020;159(1):289-305.e16. [22] SOURBIER C, RICKETTS C J, MATSUMOTO S, et al. Targeting ABL1-mediated oxidative stress adaptation in fumarate hydratase-deficient cancer. Cancer cell. 2014;26(6):840-850. [23] KERKELÄ R, GRAZETTE L, YACOBI R, et al. Cardiotoxicity of the cancer therapeutic agent imatinib mesylate. Nature medicine. 2006;12(8):908-916. [24] MAHARSY W, ARIES A, MANSOUR O, et al. Ageing is a risk factor in imatinib mesylate cardiotoxicity. Eur J Heart Fail. 2014;16(4):367-376. [25] YE Y, YANG K, LIU H, et al. SIRT2 counteracts primate cardiac aging via deacetylation of STAT3 that silences CDKN2B. Nat Aging. 2023; 3(10):1269-1287. [26] LIU Y, ZHONG C, CHEN S, et al. Circulating exosomal mir-16-2-3p is associated with coronary microvascular dysfunction in diabetes through regulating the fatty acid degradation of endothelial cells. Cardiovasc Diabetol. 2024;23(1):60. [27] SUN T, DING W, XU T, et al. Parkin Regulates Programmed Necrosis and Myocardial Ischemia/Reperfusion Injury by Targeting Cyclophilin-D.Antioxid Redox Signal. 2019;31(16):1177-1193. [28] ALGOET M, JANSSENS S, HIMMELREICH U, et al. Myocardial ischemia-reperfusion injury and the influence of inflammation. Trends Cardiovasc Med. 2023;33(6):357-366. [29] ZHANG M, LIU Q, MENG H, et al. Ischemia-reperfusion injury: molecular mechanisms and therapeutic targets. Signal Transduct Target Ther. 2024; 9(1):12. [30] WU F, HUANG W, TAN Q, et al. ZFP36L2 regulates myocardial ischemia/reperfusion injury and attenuates mitochondrial fusion and fission by LncRNA PVT1. Cell Death Dis. 2021;12(6):614. [31] SÁNCHEZ-PÉREZ P, MATA A, TORP MK, et al. Energy substrate metabolism, mitochondrial structure and oxidative stress after cardiac ischemia-reperfusion in mice lacking UCP3. Free Radic Biol Med. 2023;205:244-261. [32] LINDSEY ML, BRUNT KR, KIRK JA, et al. Guidelines for in vivo mouse models of myocardial infarction. Am J Physiol Heart Circ Physiol. 2021; 21(6):H1056-H1073. [33] WANG L, QIU S, LI X, et al. Myocardial-Targeting Tannic Cerium Nanocatalyst Attenuates Ischemia/Reperfusion Injury. Angew Chem Int Ed Engl. 2023; 62(39):e202305576. [34] ZHANG L, LIU Y, LI JY, et al. Protective Effect of Rosamultin against H(2)O(2)-Induced Oxidative Stress and Apoptosis in H9c2 Cardiomyocytes. Oxid Med Cell Longev. 2018;2018:8415610. [35] YU C, KIM B S, KIM E. FAF1 mediates regulated necrosis through PARP1 activation upon oxidative stress leading to dopaminergic neurodegeneration. Cell Death Differ. 2016;23(11):1873-1885. [36] JABBOUR E, KANTARJIAN H. Chronic myeloid leukemia: 2025 update on diagnosis, therapy, and monitoring. Am J Hematol. 2024;99(11):2191-2212. [37] LEE H, SEO J, SHIN S, et al. Development and validation of sensitive BCR::ABL1 fusion gene quantitation using next-generation sequencing.Cancer Cell Int. 2023;23(1):106. [38] HEFNI E, MENON D, MA T, et al. Angiopoietin-like 4 induces head and neck squamous cell carcinoma cell migration through the NRP1/ABL1/PXN pathway.Cell Signal. 2023;108:110697. [39] JULIAN L, OLSON MF. Rho-associated coiled-coil containing kinases (ROCK): structure, regulation, and functions.Small GTPases. 2014;5:e29846. [40] BALAJI RAGUNATHRAO VA, ANWAR M, AKHTER MZ, et al. Sphingosine-1-Phosphate Receptor 1 Activity Promotes Tumor Growth by Amplifying VEGF-VEGFR2 Angiogenic Signaling. Cell Rep. 2019;29(11):3472-3487. [41] ZHOU X, CHENG J, CHEN Z, et al. Role of c-Abl in Ang II-induced aortic dissection formation: Potential regulatory efficacy on phenotypic transformation and apoptosis of VSMCs. Life Sci. 2020;256:117882. [42] YAMANI A, WU D, WAGGONER L, et al. The vascular endothelial specific IL-4 receptor alpha-ABL1 kinase signaling axis regulates the severity of IgE-mediated anaphylactic reactions. J Allergy Clin Immunol. 2018;142(4):1159-1172.e5. [43] QIU Z, CANG Y, GOFF SP. c-Abl tyrosine kinase regulates cardiac growth and development. Proc Natl Acad Sci U S A. 2010;107(3):1136-1141. [44] GUO X, YIN H, LI L, et al. Cardioprotective Role of Tumor Necrosis Factor Receptor-Associated Factor 2 by Suppressing Apoptosis and Necroptosis. Circulation. 2017;136(8):729-742. [45] GUPTA R, KUMARI S, TRIPATHI R, et al. Unwinding the modalities of necrosome activation and necroptosis machinery in neurological diseases. Ageing Res Rev. 2023;86:101855. [46] DEL RE DP, AMGALAN D, LINKERMANN A, et al. Fundamental Mechanisms of Regulated Cell Death and Implications for Heart Disease. Physiol Rev. 2019;99(4):1765-1817. [47] BONORA M, GIORGI C, PINTON P. Molecular mechanisms and consequences of mitochondrial permeability transition. Nat Rev Mol Cell Biol. 2022; 23(4):266-285. [48] MENDOZA A, PATEL P, ROBICHAUX D, et al. Inhibition of the mPTP and Lipid Peroxidation Is Additively Protective Against I/R Injury. Circ Res. 2024;134(10):1292-1305. [49] SAMANTA S, AKHTER F, ROY A, et al. New cyclophilin D inhibitor rescues mitochondrial and cognitive function in Alzheimer’s disease. Brain. 2024; 147(5):1710-1725. [50] LU A, XU Z, ZHAO Z, et al. Double Braking Effects of Nanomedicine on Mitochondrial Permeability Transition Pore for Treating Idiopathic Pulmonary Fibrosis. Adv Sci (Weinh). 2024;11(47):e2405406. [51] KUDO T, NAKAZAWA D, WATANABE-KUSUNOKI K, et al. Regulation of NETosis and Inflammation by Cyclophilin D in Myeloperoxidase-Positive Antineutrophil Cytoplasmic Antibody-Associated Vasculitis. Arthritis Rheumatol. 2023;75(1):71-83. [52] HALECKOVA A, BENEK O, ZEMANOVÁ L, et al. Small-molecule inhibitors of cyclophilin D as potential therapeutics in mitochondria-related diseases. Med Res Rev. 2022;42(5):1822-1855. [53] ZHOU C, KUANG M, TAO Y, et al. Nynrin preserves hematopoietic stem cell function by inhibiting the mitochondrial permeability transition pore opening. Cell Stem Cell. 2024;31(9):1359-1375.e8. [54] DIKALOVA A, FEHRENBACH D, MAYOROV V, et al. Mitochondrial CypD Acetylation Promotes Endothelial Dysfunction and Hypertension. Circ Res. 2024;134(11):1451-1464. [55] WANG K, AN T, ZHOU LY, et al. E2F1-regulated miR-30b suppresses Cyclophilin D and protects heart from ischemia/reperfusion injury and necrotic cell death.Cell Death Differ. 2015;22(5):743-754. |
| [1] | Yang Xuetao, Zhu Menghan, Zhang Chenxi, Sun Yimin, Ye Ling. Applications and limitations of antioxidant nanomaterials in oral cavity [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2044-2053. |
| [2] |
Dong Chunyang, Zhou Tianen, Mo Mengxue, Lyu Wenquan, Gao Ming, Zhu Ruikai, Gao Zhiwei.
Action mechanism of metformin combined with Eomecon chionantha Hance dressing in treatment of deep second-degree burn wounds#br#
#br#
[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2001-2013.
|
| [3] | Liu Anting, Lu Jiangtao, Zhang Wenjie, He Ling, Tang Zongsheng, Chen Xiaoling. Regulation of AMP-activated protein kinase by platelet lysate inhibits cadmium-induced neuronal apoptosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1800-1807. |
| [4] | Liu Huan, Zeng Shaopeng, Chen Jun, He Linqian, Yang Ying, Zhang Jing. Aging-related dysregulation of glucose metabolism: crossroads of cancer and neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1527-1538. |
| [5] | Leng Xiaoxuan, Zhao Yuxin, Liu Xihua. Effects of different neuromodulatory stimulation modalities on non-motor symptoms in Parkinson’s patients: a network meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1282-1293. |
| [6] | Yang Xiao, Bai Yuehui, Zhao Tiantian, Wang Donghao, Zhao Chen, Yuan Shuo. Cartilage degeneration in temporomandibular joint osteoarthritis: mechanisms and regenerative challenges [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 926-935. |
| [7] | Zhang Xiaoxu, Tian Zhenli, Xie Tingting. Roles of pregnane X receptor in sodium arsenite-induced oxidative stress and inflammatory injury in human normal hepatocytes [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6259-6266. |
| [8] | Li Huaying, Li Hao, Peng Wuxun, Dong Wentao. Mechanism of cuproptosis in the diagnosis and treatment of orthopedic-related diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6275-6281. |
| [9] | Xia Caigui, Li Wei, Su Yuying, Shi Yu, Yang Zhonghe. Effects of antioxidant pretreatment on skeletal muscle damage and oxidative stress following acute high-intensity exercise: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6345-6353. |
| [10] | Huang Yushan, Wang Rongrong, Li Xiangmiao, Bai Jinzhu. Prostaglandin E1 pretreatment inhibits ferroptosis in endothelial cells in a rat model of spinal cord ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(22): 5716-5727. |
| [11] | Yang Zijiang, Guo Chenggen, Deng Ziao, Xue Xinxuan. Postbiotic targeting muscle aging: mechanistic insights and application prospects of urolithin A [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(22): 5804-5813. |
| [12] | Ji Long, Gong Guopan, Kong Xiangkui, Jin Pan, Chen Ziyang, Pu Rui. The role of exercise-regulated mitophagy in cardiovascular diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(22): 5832-5843. |
| [13] | Dong Chao, Zhao Mohan, Liu Yunan, Yang Zeli, Chen Leqin, Wang Lanfang. Effects of magnetic nano-drug carriers on exercise-induced muscle injury and inflammatory response in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(2): 345-353. |
| [14] | Wang Siwei, Yao Xiaosheng, Qi Xiaonan, Wang Yu, Cui Haijian, Zhao Jiaxuan. Matrix metalloproteinase 9 mediates mitophagy to regulate osteogenesis and myogenesis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(18): 4557-4567. |
| [15] | Liao Xingzhuan, Li Guangdi, Wu Yabin, Liu Xingyu, Wan Jiajia. Molecular mechanisms underlying non-coding RNA regulation of ferroptosis in osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(18): 4713-4725. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||