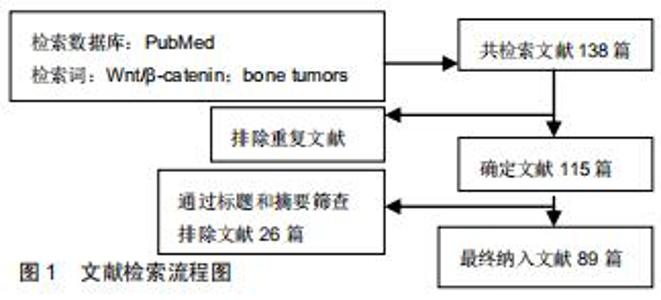
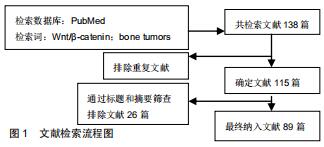
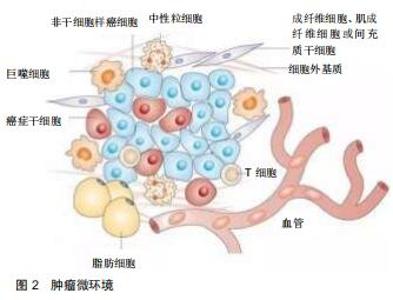
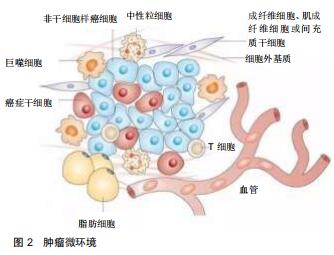
Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (32): 5213-5220.doi: 10.3969/j.issn.2095-4344.2871
Previous Articles Next Articles
Wnt/beta-catenin signaling pathway: multiple roles in osteosarcoma, bone remodeling and angiogenesis
Liu Chang, Song Keguan
- First Affiliated Hospital of Harbin Medical University, Harbin 150001, Heilongjiang Province, China
-
Received:2020-01-07Revised:2020-01-10Accepted:2020-03-04Online:2020-11-18Published:2020-09-26 -
Contact:Song Keguan, Chief physician, Professor, First Affiliated Hospital of Harbin Medical University, Harbin 150001, Heilongjiang Province, China -
About author:Liu Chang, Master, Physician, First Affiliated Hospital of Harbin Medical University, Harbin 150001, Heilongjiang Province, China -
Supported by:the National Key Research and Development Program of China, No. 2017YFB0702600; 2017YFB0702604
CLC Number:
Cite this article
Liu Chang, Song Keguan. Wnt/beta-catenin signaling pathway: multiple roles in osteosarcoma, bone remodeling and angiogenesis[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(32): 5213-5220.
share this article
|
[1] STRAUSS SJ, WHELAN JS. Current questions in bone sarcomas. Curr Opin Oncol.2018;30:252-259.
[2] LU P, WEAVER VM, WERB Z. The extracellular matrix: A dynamic niche in cancer progression. J Cell Biol.2012; 196:395-406.
[3] CRENN V, BITEAU K, AMIAUD J, et al.Bone microenvironment has an influence on the histological response of osteosarcoma to chemotherapy: Retrospective analysis and preclinical modeling. Am J Cancer Res.2017;7:2333-2349.
[4] ALFRANCA A, MARTINEZ-CRUZADO L, TORNIN J, et al.Bone microenvironment signals in osteosarcoma development.Cell Mol Life Sci.2015;72:3097-3113.
[5] JIN H, BARROTT JJ, CABLE MG, et al. The Impact of Microenvironment on the Synovial Sarcoma Transcriptome.Cancer Microenviron.2017;10:1-7.
[6] GOLDSTEIN SD, HAYASHI M, ALBERT CM, et al. An orthotopic xenograft model with survival hindlimb amputation allows investigation of the effect of tumor microenvironment on sarcoma metastasis.Clin Exp Metastasis.2015;32:703-715.
[7] RIEMANN A, SCHNEIDER B, GÜNDEL D, et al .Acidosis Promotes Metastasis Formation by Enhancing Tumor Cell Motility. Adv Exp Med Biol.2016;876:215-220.
[8] CHATTOPADHYAY S, CHAKLADER M, CHATTERJEE R,et al. Differential expression of mitotic regulators and tumor microenvironment influences the regional growth pattern of solid sarcoma along the cranio-caudal axis.Exp Cell Res. 2016;340:91-101.
[9] LAMOUREUX F, RICHARD P, WITTRANT Y, et al. Therapeutic relevance of osteoprotegerin gene therapy in osteosarcoma: Blockade of the vicious cycle between tumor cell proliferation and bone resorption. Cancer Res.2007;67:7308-7318.
[10] DASS CR, CHOONG PF. Zoledronic acid inhibits osteosarcoma growth in an orthotopic model.Mol Cancer Ther.2007;6: 3263-3270.
[11] HAN Y, WU C, WANG J, et al. CXCR7 maintains osteosarcoma invasion after CXCR4 suppression in bone marrow microenvironment. Tumour Biol. 2017;39:101042831770163.
[12] CORTINI M, MASSA A, AVNET S, et al. Tumor-Activated Mesenchymal Stromal Cells Promote Osteosarcoma Stemness and Migratory Potential via IL-6 Secretion.PLoS ONE.2016;11: e0166500.
[13] QI J, ZHOU Y, JIAO Z, et al. Exosomes Derived from Human Bone Marrow Mesenchymal Stem Cells Promote Tumor Growth Through Hedgehog Signaling Pathway.Cell Physiol Biochem. 2017;42:2242-2254.
[14] WANG Y, CHU Y, YUE B, et al. Adipose-derived mesenchymal stem cells promote osteosarcoma proliferation and metastasis by activating the STAT3 pathway.Oncotarget.2017;8:23803-23816.
[15] HEYMANN MF, LÉZOT F, HEYMANN D. The contribution of immune infiltrates and the local microenvironment in the pathogenesis of osteosarcoma.Cell Immunol. 2019;343:103711.
[16] NOY R, POLLARD JW.Tumor-associated macrophages: From mechanisms to therapy.Immunity.2014;41:49-61.
[17] GOMEZ-BROUCHET A, ILLAC C, GILHODES J, et al. CD163-positive tumor-associated macrophages and CD8-positive cytotoxic lymphocytes are powerful diagnostic markers for the therapeutic stratification of osteosarcoma patients: An immunohistochemical analysis of the biopsies fromthe French OS2006 phase 3 trial.Oncoimmunology.2017;6:e1331193.
[18] DUMARS C, NGYUEN JM, GAULTIER A, et al.Dysregulation of macrophage polarization is associated with the metastatic process in osteosarcoma.Oncotarget.2016;7:78343-78354.
[19] ZHOU Q, XIAN M, XIANG S, et al.All Trans Retinoic Acid Prevents Osteosarcoma Metastasis by Inhibiting M2 Polarization of Tumor- Associated Macrophages.Cancer Immunol Res.2017;5:547-559.
[20] HAN Q, SHI H, LIU F. CD163(+) M2-type tumor-associated macrophage support the suppression of tumor-infiltrating T cells in osteosarcoma. Int Immunopharmacol. 2016;34:101-106.
[21] CHANG CH, QIU J, O’SULLIVAN D, et al.Metabolic Competition in the Tumor Microenvironment Is a Driver of Cancer Progression. Cell.2015;162:1229-1241.
[22] LEWIS DM, PRUITT H, JAIN N, et al.A Feedback Loop between Hypoxia and Matrix Stress Relaxation Increases Oxygen-Axis Migration and Metastasis in Sarcoma.Cancer Res. 2019;79: 1981-1995.
[23] ITOH H, KADOMATSU T, TANOUE H, et al. TET2-dependent IL-6 induction mediated by the tumor microenvironment promotes tumor metastasis in osteosarcoma.Oncogene. 2018;37: 2903-2920.
[24] PIPERNO-NEUMANN S, LE DELEY MC, RÉDINI F, et al. Zoledronate in combination with chemotherapy and surgery to treat osteosarcoma (OS2006): A randomised, multicentre, open-label, phase 3 trial.Lancet Oncol.2016;17:1070-1080.
[25] DUCHARTRE Y, KIM YM, KAHN M. The Wnt signaling pathway in cancer. Crit Rev Oncol Hematol.2016;99: 141-149.
[26] ZHAN T, RINDTORFF N, BOUTROS M. Wnt signaling in cancer. Oncogene.2017;36:1461-1473.
[27] HARB J, LIN PJ, HAO J. Recent Development of Wnt Signaling Pathway Inhibitors for Cancer Therapeutics. Curr Oncol Rep. 2019;21:12.
[28] KAHN M. Can we safely target the WNT pathway? Nat Rev Drug Discov. 2014;13:513-532.
[29] CHEN C, ZHAO M, TIAN A, et al.Aberrant activation of Wnt/β-catenin signaling drives proliferation of bone sarcoma cells.Oncotarget. 2015;6:17570-17583.
[30] LU Y, GUAN GF, CHEN J,e t al.Aberrant CXCR4 and β-catenin expression in osteosarcoma correlates with patient survival.Oncol Lett.2015;10:2123–2129.
[31] CAI Y, MOHSENY AB, KARPERIEN M, et al. Inactive Wnt/beta- catenin pathway in conventional high-grade osteosarcoma.J Pathol. 2010;220:24-33.
[32] SHIMOZAKI S,YAMAMOTO N, DOMOTO T, et al. Efficacy of glycogen synthase kinase-3β targeting against osteosarcoma via activation of β-catenin.Oncotarget.2016;7:77038-77051.
[33] JIE XX, ZHANG XY, XU CJ. Epithelial-to-mesenchymal transition, circulating tumor cells and cancer metastasis: Mechanisms and clinical applications. Oncotarget 2017;8:81558-81571.
[34] YANG G, YUAN J, LI K. EMT transcription factors: Implication in osteosarcoma.Med Oncol.2013;30: 697.
[35] VERRECCHIA F, RÉDINI F. Transforming Growth Factor-β Signaling Plays a Pivotal Role in the Interplay Between Osteosarcoma Cells and Their Microenvironment.Front Oncol. 2018;8:133.
[36] FUXE J, VINCENT T, DE GARCIA HERREROS A. Transcriptional crosstalk between TGF-β and stem cell pathways in tumor cell invasion: Role of EMT promoting Smad complexes. Cell Cycle. 2010;9:2363-2374.
[37] TIAN H, ZHOU T, CHEN H, et al. Bone morphogenetic protein-2 promotes osteosarcoma growth by promoting epithelial-mesenchymal transition (EMT) through the Wnt/β-catenin signaling pathway.J Orthop Res.2019;37: 1638-1648.
[38] WANG S, ZHANG D, HAN S, et al.Fibulin-3 promotes osteosarcoma invasion and metastasis by inducing epithelial to mesenchymal transition and activating the Wnt/β-catenin signaling pathway.Sci Rep.2017; 7:6215.
[39] FAN S, GAO X, CHEN P, et al. Carboxypeptidase E-ΔN promotes migration, invasiveness, and epithelial mesenchymal transition of human osteosarcoma cells via the Wnt-β-catenin pathway. Biochem. Cell Biol.2018:1-8.
[40] ZENG Q, LI Z, ZHAO X, et al. Ubiquitin-specific protease 7 promotes osteosarcoma cell metastasis by inducing epithelial-mesenchymal transition. Oncol Rep.2019;41:543-551.
[41] CAI Z, CAO Y, LUO Y, et al. Signalling mechanism(s) of epithelial-mesenchymal transition and cancer stem cells in tumour therapeutic resistance.Clin Chim Acta.2018;483:156-163.
[42] TAKEBE N, MIELE L, HARRIS PJ, et al. Targeting Notch, Hedgehog, and Wnt pathways in cancer stem cells: Clinical update. Nat Rev Clin Oncol.2015;12:445-464.
[43] ADHIKARI AS, AGARWAL N, WOOD BM, et al. CD117 and Stro-1 identify osteosarcoma tumor-initiating cells associated with metastasis and drug resistance.Cancer Res. 2010;70:4602-4612.
[44] MARTINS-NEVES SR, CORVER WE, PAIVA-OLIVEIRA DI, et al. Osteosarcoma Stem Cells Have Active Wnt/β- catenin and Overexpress SOX2 and KLF4.J Cell Physiol.2016;231:876-886.
[45] LIU Y, WANG Y, YANG H, et al. MicroRNA-873 targets HOXA9 to inhibit the aggressive phenotype of osteosarcoma by deactivating the Wnt/β-catenin pathway.Int J Oncol.2019;54:1809-1820.
[46] CAI W, XU Y, YIN J, et al. miR-552-5p facilitates osteosarcoma cell proliferation and metastasis by targeting WIF1. Exp Ther Med.2019;17:3781-3788.
[47] LIU Y, BAO Z, TIAN W, et al. miR-885-5p suppresses osteosarcoma proliferation, migration and invasion through regulation of β-catenin.Oncol Lett.2019;17:1996-2004.
[48] REN J, YANG M, XU F, et al.microRNA-758 inhibits the malignant phenotype of osteosarcoma cells by directly targeting HMGA1 and deactivating the Wnt/β-catenin pathway.Am J Cancer Res. 2019;9: 36-52.
[49] XIA P, GU R, ZHANG W, et al.MicroRNA-377 exerts a potent suppressive role in osteosarcoma through the involvement of the histone acetyltransferase 1-mediated Wnt axis.J Cell Physiol. 2019;234(12):22787-22798.
[50] LI C, WANG F, WEI B, et al. LncRNA AWPPH promotes osteosarcoma progression via activation of Wnt/β-catenin pathway through modulating miR-93-3p/FZD7 axis. Biochem. Biophys. Res Commun.2019;514: 1017-1022.
[51] BROWN HK, SCHIAVONE K, GOUIN F, et al.Biology of Bone Sarcomas and New Therapeutic Developments. Calcif Tissue Int. 2018;102:174-195.
[52] PEDERSON L, RUAN M, WESTENDORF JJ, et al.Regulation of bone formation by osteoclasts involves Wnt/BMP signaling and the chemokine sphingosine-1-phosphate. Proc Natl Acad Sci.USA 2008;105: 20764-20769.
[53] WEIVODA MM, RUAN M, HACHFELD CM, et al.Wnt Signaling Inhibits Osteoclast Differentiation by Activating Canonical and Noncanonical cAMP/PKA Pathways.J Bone Miner Res.2016; 31:65-75.
[54] SADANANDAM A, FUTAKUCHI M, LYSSIOTIS CA, et al.A cross-species analysis of a mouse model of breast cancer-specific osteolysis and human bone metastases using gene expression profiling. BMC Cancer.2011; 11:304.
[55] BU G, LU W,LIU CC, et al.Breast cancer-derived Dickkopf1 inhibits osteoblast differentiation and osteoprotegerin expression: Implication for breast cancer osteolytic bone metastases.Int J Cancer.2008; 123:1034-1042.
[56] BJØRNLAND K, FLATMARK K, PETTERSEN S,et al.Matrix metalloproteinases participate in osteosarcoma invasion.J Surg Res.2005;127:151-156.
[57] KUNZ P, SÄHR H, LEHNER B, et al. Elevated ratio of MMP2/MMP9 activity is associated with poor response to chemotherapy in osteosarcoma.BMC Cancer.2016;16:223.
[58] GUO Y, ZI X, KOONTZ Z, et al.Blocking Wnt/LRP5 signaling by a soluble receptor modulates the epithelial to mesenchymal transition and suppresses met and metalloproteinases in osteosarcoma Saos-2 cells.J Orthop Res.2007;25:964-971.
[59] LIU B, LI G, WANG X, et al.A furin inhibitor downregulates osteosarcoma cell migration by downregulating the expression levels of MT1-MMP via the Wnt signaling pathway.Oncol Lett. 2014;7: 1033-1038.
[60] OLSEN JJ, POHL SÖ, DESHMUKH A, et al.The Role of Wnt Signalling in Angiogenesis. Clin Biochem Rev. 2017;38;131-142.
[61] CARMELIET P, JAIN RK. Principles and mechanisms of vessel normalization for cancer and other angiogenic diseases.Nat Rev Drug Discov.2011;10:417-427.
[62] MAVROGENIS AF, VOTTIS CT, MEGALOIKONOMOS PD, et al. Neovascularization in Ewing’s sarcoma. Neoplasma.2018;65: 317-325.
[63] CHEN D, ZHANG YJ, ZHU K, et al. A systematic review of vascular endothelial growth factor expression as a biomarker of prognosis in patients with osteosarcoma.Tumour Biol.2013;34: 1895-1899.
[64] YANG J, YANG D, SUN Y, et al. Genetic amplification of the vascular endothelial growth factor (VEGF) pathway genes, including VEGFA,in human osteosarcoma.Cancer.2011;117: 4925-4938.
[65] EASWARAN V, LEE SH, INGE L, et al.beta-Catenin regulates vascular endothelial growth factor expression in colon cancer. Cancer Res.2003;63:3145-3153.
[66] KAWANO Y, KYPTA R. Secreted antagonists of the Wnt signalling pathway.J Cell Sci.2003;116: 2627-2634.
[67] DUFOURCQ P, LEROUX L, EZAN J, et al. Regulation of endothelial cell cytoskeletal reorganization by a secreted frizzled- related protein-1 and frizzled 4- and frizzled 7-dependent pathway: Role in neovessel formation.Am J Pathol.2008;172:37-49.
[68] MULEY A, MAJUMDER S, KOLLURU GK, et al. Secreted frizzled-related protein 4: An angiogenesis inhibitor. Am J Pathol. 2010;176:1505-1516.
[69] ZHAO S, KURENBEKOVA L, GAO Y, et al.NKD2, a negative regulator of Wnt signaling, suppresses tumor growth and metastasis in osteosarcoma.Oncogene.2015;34:5069.
[70] LIAO D, JOHNSON RS. Hypoxia: A key regulator of angiogenesis in cancer.Cancer Metastasis Rev. 2007; 26;281-290.
[71] KROCK BL, SKULI N, SIMON MC. Hypoxia-induced angiogenesis: Good and evil.Genes Cancer.2011;2: 1117-1133.
[72] ZHANG XD, WU Q, YANG SH.Effects of siRNA-mediated HIF-1α gene silencing on angiogenesis in osteosarcoma. Pak J Med Sci. 2017;33:341-346.
[73] ZHANG B, LI YL, ZHAO JL, et al.Hypoxia-inducible factor-1 promotes cancer progression through activating AKT/Cyclin D1 signaling pathway in osteosarcoma. Biomed Pharmacother.2018; 105:1-9.
[74] LI Y, ZHANG W, LI S, et al. Prognosis value of Hypoxia-inducible factor-1α expression in patients with bone and soft tissue sarcoma: A meta-analysis.Springerplus.2016;5:1370.
[75] DEMIR R, DIMMLER A, NASCHBERGER E, et al. Malignant progression of invasive tumour cells seen in hypoxia present an accumulation of beta-catenin in the nucleus at the tumour front. Exp Mol Pathol. 2009;87:109-116.
[76] KAIDI A, WILLIAMS AC, PARASKEVA C. Interaction between beta-catenin and HIF-1 promotes cellular adaptation to hypoxia. Nat Cell Biol.2007;9:210-217.
[77] SCHOLTEN DJ 2ND, TIMMER CM, PEACOCK JD, et al.Down regulation of Wnt signaling mitigates hypoxia-induced chemoresistance in human osteosarcoma cells.PLoS ONE. 2014; 9:e111431.
[78] QIAN BZ, POLLARD JW. Macrophage diversity enhances tumor progression and metastasis.Cell.2010; 141:39-51.
[79] YANG Y, YE YC, CHEN Y, et al.Crosstalk between hepatic tumor cells and macrophages via Wnt/β-catenin signaling promotes M2-like macrophage polarization and reinforces tumor malignant behaviors.Cell Death Dis.2018;9:793.
[80] LIU T, FANG XC, DING Z, et al.Pre-operative lymphocyte-to- monocyte ratio as a predictor of overall survival in patients suffering from osteosarcoma. FEBS Open Bio.2015;5:682-687.
[81] ASSAL A, KANER J, PENDURTI G, et al.Emerging targets in cancer immunotherapy: Beyond CTLA-4 and PD-1. Immunotherapy. 2015;7:1169-1186.
[82] KOIRALA P, ROTH ME, GILL J, et al. HHLA2, a member of the B7 family, is expressed in human osteosarcoma and is associated with metastases and worse survival.Sci Rep.2016;6:31154.
[83] KOIRALA P, ROTH ME, GILL J, et al. Immune infiltration and PD-L1 expression in the tumor microenvironment are prognostic in osteosarcoma.Sci Rep.2016;6:30093.
[84] WANG L, ZHANG Q, CHEN W, et al. B7-H3 is overexpressed in patients suffering osteosarcoma and associated with tumor aggressiveness and metastasis.PLoS ONE.2013;8:e70689.
[85] LUKE JJ, BAO R, SWEIS RF, et al. Wnt/β-catenin Pathway Activation Correlates with Immune Exclusion across Human Cancers. Clin Cancer Res.2019;25:3074-3083.
[86] SPRANGER S, BAO R, GAJEWSKI TF.Melanoma-intrinsic β-catenin signalling prevents anti-tumour immunity.Nature. 2015; 523:231-235.
[87] DE RUIZ GALARRETA M, BRESNAHAN E, MOLINA-SANCHEZ P, et al.β-catenin activation promotes immune escape and resistance to anti-PD-1 therapy in hepatocellular carcinoma. Cancer Discov. 2019;9(8):1124-1141.
[88] DHUPKAR P, GORDON N, STEWART J, et al.Anti-PD-1 therapy redirects macrophages from an M2 to an M1 phenotype inducing regression of OS lung metastases.Cancer Med.2018;7: 2654-2664.
[89] GALLUZZI L, SPRANGER S, FUCHS E, et al.WNT Signaling in Cancer Immunosurveillance.Trends Cell Biol. 2019;29:44-65. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Yuan Jiawei, Zhang Haitao, Jie Ke, Cao Houran, Zeng Yirong. Underlying targets and mechanism of Taohong Siwu Decoction in prosthetic joint infection on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1428-1433. |
| [3] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [4] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Multiple problems in the selection of implants for patellar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1440-1445. |
| [5] | Wang Debin, Bi Zhenggang. Related problems in anatomy mechanics, injury characteristics, fixed repair and three-dimensional technology application for olecranon fracture-dislocations [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1446-1451. |
| [6] | Gu Xia, Zhao Min, Wang Pingyi, Li Yimei, Li Wenhua. Relationship between hypoxia inducible factor 1 alpha and hypoxia signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1284-1289. |
| [7] | Ji Zhixiang, Lan Changgong. Polymorphism of urate transporter in gout and its correlation with gout treatment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1290-1298. |
| [8] | Yuan Mei, Zhang Xinxin, Guo Yisha, Bi Xia. Diagnostic potential of circulating microRNA in vascular cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1299-1304. |
| [9] | Chai Le, Lü Jianlan, Hu Jintao, Hu Huahui, Xu Qingjun, Yu Jinwei, Quan Renfu. Signal pathway variation after induction of inflammatory response in rats with acute spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1218-1223. |
| [10] | Geng Qiudong, Ge Haiya, Wang Heming, Li Nan. Role and mechanism of Guilu Erxianjiao in treatment of osteoarthritis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1229-1236. |
| [11] | Li Zhongfeng, Chen Minghai, Fan Yinuo, Wei Qiushi, He Wei, Chen Zhenqiu. Mechanism of Yougui Yin for steroid-induced femoral head necrosis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1256-1263. |
| [12] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [13] | Wan Ran, Shi Xu, Liu Jingsong, Wang Yansong. Research progress in the treatment of spinal cord injury with mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1088-1095. |
| [14] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [15] | Zhao Min, Feng Liuxiang, Chen Yao, Gu Xia, Wang Pingyi, Li Yimei, Li Wenhua. Exosomes as a disease marker under hypoxic conditions [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1104-1108. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||