Chinese Journal of Tissue Engineering Research ›› 2016, Vol. 20 ›› Issue (43): 6451-6457.doi: 10.3969/j.issn.2095-4344.2016.43.010
Previous Articles Next Articles
A plasma-modified polyurethane-collagen-chondroitin sulfate composite material: preparation and performance
- 1Department of Pharmacy, Qingdao University of Science and Technology, Qingdao 266000, Shandong Province, China; 2Qingdao ChungHao Tissue Engineering Company, Qingdao 266000, Shandong Province, China
-
Received:2016-08-04Online:2016-10-21Published:2016-10-21 -
About author:Xuan Guang-shan, Doctor, Professor, Department of Pharmacy, Qingdao University of Science and Technology, Qingdao 266000, Shandong Province, China
CLC Number:
Cite this article
Xuan Guang-shan, Mu Lan-lan, Sun Tong, Li Qing.
share this article
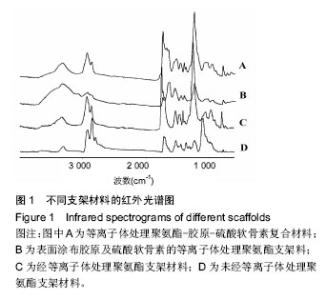
2.1 红外光谱分析结果 图1为4种支架材料的红外光谱图,其中位于3 303 cm-1特征吸收峰为N-H伸缩振动峰,位于2 930 cm-1和2 850 cm-1的特征吸收峰分别为亚甲基中C-H的对称和不对称伸缩振动吸收峰,位于 1 715 cm-1的特征峰为聚氨酯软段聚碳酸酯二醇的C=O伸缩振动峰,O=C-O的面外弯曲特征峰位于图中 790 cm-1。C组在CO2和NH3等离子体处理后,位于 2 850 cm-1的特征吸收峰明显变小,而位于3 303 cm-1特征吸收峰明显增宽增大,为伯胺伸缩振动特征峰,说明经氨气等离子体处理后聚氨酯表面接枝大量-NH2;位于3 600-3 200 cm-1的-OH特征峰出现,并且C=O伸缩振动峰及O=C-O的面外弯曲特征峰明显增大,表明CO2在激发转为等离子态后,接枝在聚氨酯的上形成羧基; 1 250 cm-1处出现C-O-C的不对称伸缩振动强峰,表示活化的CO2在聚氨酯表面形成酯基。表面涂布胶原及硫酸软骨素时,聚氨酯表面其他特征峰被掩盖,且由于胶原和硫酸软骨素含有大量-OH,红外光谱出现3 600- 3 200 cm-1较宽的峰。A组经EDC和NHS交联,-NH2和-COOH反应生成仲酰胺键(RCONHR')。较等离子体处理聚氨酯材料及表面涂布胶原及硫酸软骨素的等离子体处理聚氨酯材料,等离子体处理聚氨酯-胶原-硫酸软骨素复合材料红外光谱图中1 680 cm-1处出现仲酰胺C=O伸缩振动的强吸收峰,即酰胺Ⅰ带;1 530 cm-1处和1 305 cm-1处分别出现由于N-H变形振动和C-N伸缩振动偶合产生的酰胺Ⅱ带和酰胺Ⅲ带,表明存在仲酰胺键,可能是交联生成的新的仲酰胺键。"


2.2 扫描电镜观察支架材料表面形貌 由图2扫描电镜照片可见,该静电纺丝条件制备的聚氨酯支架形貌良好,纤维平滑直径分布均匀,平均直径为1 μm左右,纤维较粗,这是由于与电压、溶液的流速、距离相比,溶剂是影响静电纺丝形貌的一个比较重要因素。实验选用的是N,N-二甲基甲酰胺和四氢呋喃摩尔比为2∶1的混合溶剂,四氢呋喃的表面张力低且易挥发,使射流纤维外层快速挥发,限制了纤维的进一步拉伸,使纤维直径较粗;N,N-二甲基甲酰胺沸点高,不易挥发,可使纤维在电场中有效拉伸。因此,适当增加N,N-二甲基甲酰胺比例,可一定程度减小纤维直径,但当N,N-二甲基甲酰胺量过多时,由于溶剂挥发过慢易形成连珠。等离子体处理聚氨酯表面稍有毛糙,这是由于等离子体处理对聚氨酯纤维有一定的刻蚀作用,但整体表观影响不大。等离子体处理聚氨酯-胶原-硫酸软骨素复合材料纤维表面被胶原包裹填充,经化学交联复合材料整体出现皱缩现象,纤维有弯曲收缩现象;未经等离子体处理聚氨酯支架材料表面物理涂布胶原硫酸软骨素涂层,纤维直径稍有变粗。 2.3 亲水性检测结果 生物材料的亲疏水性是限制其应用的主要因素。就皮肤支架而言,适宜亲水性的皮肤修复材料可使伤口保持湿润,有利于细胞黏附生长,促进伤口愈合。对各种支架材料接触角进行分析发现,未经等离子体处理的聚氨酯支架材料接触角为110°,材料不发生浸润;经等离子体处理后,材料的亲水性显著增加,水滴滴至材料后被迅速吸收,无法测出接触角,涂布和接枝胶原硫酸软骨素的材料也迅速将水滴吸收。聚氨酯有大量非极性基团如酯基,有较高的疏水性,离子体处理可在表面接枝亲水基团,虽然等离子体只在接触面反应,但由于静电纺丝纤维之间空隙大,材料比表面积大,增加了处理效率,使亲水性显著增加;胶原蛋白中含有大量-NH2、-OH,硫酸软骨素含有-OH、-COOH、-SO3H亲水基团,也有很好的亲水性。故将聚氨酯进行等离子体处理并与胶原蛋白及硫酸软骨素偶联处理,可得亲水性良好的支架材料。"

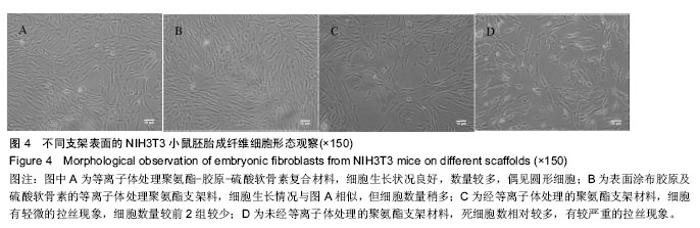
2.4 机械性能测试结果 复合材料的应用是需要良好的机械性能。4种支架材料的应力-应变曲线见图3,等离子体处理前后聚氨酯支架都呈明显的黏弹性力学性质,初始模量小,断裂伸长率达550%左右,拉伸强度可至30 MPa,4种支架材料机械性没有明显差异。这是由于等离子体只在材料表面接枝,对材料内部结构无影响,不破坏材料整体的机械性能,表面接枝的胶原蛋白及硫酸软骨素后支架材料的机械强度也没有明显变化。 2.5 体外细胞毒性实验结果 由于聚氨酯是用N,N-二甲基甲酰胺和四氢呋喃溶解后进行静电纺丝,并与胶原蛋白及硫酸软骨素进行化学交联,各支架材料虽经乙醇梯度清洗,仍可能有微量残留,对细胞有一定的毒性。由图4可见A组等离子体处理聚氨酯经胶原和硫酸软骨素修饰后细胞生长状况良好,数量较多,偶见圆形细胞。B组细胞生长情况与A组相似,但细胞数量较A组稍多,可能由于表面涂布胶原及硫酸软骨素为细胞生长提供了营养物质;C组细胞有轻微的拉丝现象,细胞数量较前2组较少;D组死细胞数相对较多,有较严重的拉丝现象。A-D组的细胞相对增殖率分别为105.5%、106.6%、98%、91.8%,4种材料细胞相对增殖率均大于90%,即4种材料的细胞毒性为0-1级,符合国家标准对于医疗器械的生物学要求[22-23]。 "

| [1]Mansbridge J. Skin tissue engineering.J Biomat Sci Polym E.2008;19(8):955-968. [2]董丽,王旭昇,马绍英,等.组织工程皮肤的构建及组织形态学观察[J].中国组织工程研究,2011,15(41):7631-7634. [3]Cao H,Chen MM,Liu Y,et al.Fish collagen-based scaffold containing PLGA microspheres for controlled growth factor delivery in skin tissue engineering. Colloids Surf B Biointerfaces. 2015;136:1098-1106. [4]Wang X,You C,Hu X,et al.The roles of knitted mesh-reinforced collagen–chitosan hybrid scaffold in the one-step repair of full-thickness skin defects in rats.Acta Biomaterialia.2013; 9(8):7822-7832. [5]Lee CH,Chang SH,Chen WJ,et al.Augmentation of diabetic wound healing and enhancement of collagen content using nanofibrous glucophage-loaded collagen/PLGA scaffold membranes.J Colloid Interface Sci.2015;439:88-97. [6]Sundaramurthi D,Vasanthan KS,Kuppan P,et al.Electrospun nanostructured chitosan–poly (vinyl alcohol) scaffolds: a biomimetic extracellular matrix as dermal substitute.Biomed Mater.2012; 7(4):045005. [7]Gholipour-Kanani A,Bahrami SH, Samadi-Kochaksaraie A,et al.Effect of tissue-engineered chitosan-poly (vinyl alcohol) nanofibrous scaffolds on healing of burn wounds of rat skin.IET Nanobiotechnol.2012;6(4):129-135. [8]Prasad T,Shabeena EA,Vinod D,et al.Characterization and in vitro evaluation of electrospun chitosan/polycaprolactone blend fibrous mat for skin tissue engineering. J Mater Sci Mater Med.2015;26(1):1-13. [9]Zhang J,Li G,Gao S,et al.Monocyte chemoattractant protein-1 released from polycaprolactone/chitosan hybrid membrane to promote angiogenesis in vivo.J Bioact Compat Pol.2014:0883911514554146. [10]Shalumon KT,Anulekha KH,Chennazhi KP,et al. Fabrication of chitosan/poly (caprolactone) nanofibrous scaffold for bone and skin tissue engineering.Int J Biol Macromol.2011;48(4):571-576. [11]Por?ba R,Špírková M,Bro?ová L,et al.Aliphatic polycarbonate‐based polyurethane elastomers and nanocomposites. II. Mechanical, thermal, and gas transport properties.J Appl Polym Sci.2013;127(1): 329-341. [12]Kojio K,Furukawa M,Shimada M,et al.Improvement of the Low-Temperature Property of Aliphatic Polycarbonate Glycols-Based Polyurethane Elastomers. SciAdv Mater.2015;7(5):934-939. [13]Zhou H,Wang H,Tian X,et al.Effect of 3-Aminopropyltriethoxysilane on polycarbonate based waterborne polyurethane transparent coating.Prog Org Coat.2014;77(6):1073-1078. [14]Por?ba R,Špírková M,Bro?ová L,et al.Aliphatic polycarbonate‐based polyurethane elastomers and nanocomposites. II.Mechanical, thermal, and gas transport properties.J Appl Polym Sci.2013;127(1): 329-341. [15]Kucinska-Lipka J,Gubanska I,Janik H,et al.Fabrication of polyurethane and polyurethane based composite fibres by the electrospinning technique for soft tissue engineering of cardiovascular system.Mater Sci Eng C.2015;46:166-176. [16]Sheikh FA,Macossay J,Cantu T,et al.Imaging, spectroscopy, mechanical, alignment and biocompatibility studies of electrospun medical grade polyurethane (Carbothane 3575A) nanofibers and composite nanofibers containing multiwalled carbon nanotubes.J Mech Behav Biomed Mater.2015;41: 189-198. [17]Shi C,Yuan W,Khan M,et al.Hydrophilic PCU scaffolds prepared by grafting PEGMA and immobilizing gelatin to enhance cell adhesion and proliferation.Mater Sci Eng C.2015;50:201-209. [18]Gauvin R,Berthod F.Collagen-based biomaterials for tissue engineering applications. Materials. 2010;3(3): 1863-1887. [19]Huang R,Li W,Lv X,et al.Biomimetic LBL structured nanofibrous matrices assembled by chitosan/collagen for promoting wound healing.Biomaterials. 2015;53: 58-75. [20]Wang W,Lin S,Xiao Y,et al.Acceleration of diabetic wound healing with chitosan-crosslinked collagen sponge containing recombinant human acidic fibroblast growth factor in healing-impaired STZ diabetic rats.Life Sci.2008;82(3):190-204. [21]Mikami T,Kitagawa H.Biosynthesis and function of chondroitin sulfate.Biochimica et Biophysica Acta. 2013;1830(10):4719-4733. [22]GB/T 16886.5-2003,医疗器械生物学评价第十二部分:样品制备与参照样品[S].中国标准出版社,2005. [23]GB/T 16886.5-2003,医疗器械生物学评价第五部分:体外细胞毒性试验[S].中国标准出版社,2005. [24]Yang J,Wan Y,Yang J,et al.Plasma-treated, collagen-anchored polylactone: Its cell affinity evaluation under shear or shear-free conditions.J Biomed Mater Res Part A.2003; 67(4):1139-1147. [25]Desmet T,Morent R,Geyter ND,et al.Nonthermal plasma technology as a versatile strategy for polymeric biomaterials surface modification: a review.Biomacromolecules.2009;10(9):2351-2378. [26]张燕搏,姜明,胡平,等.聚氨酯不同低温等离子体改性及生物相容性评价[J].中华实用诊断与治疗杂志, 2011, 25(10): 944-946. [27]张燕搏,王强.低温等离子体技术在生物医用材料改性中的应用[J].中华实用诊断与治疗杂志, 2012, 26(1): 1-3. [28]陈璐,杨庆,邵梅玲.PHBV/PCL静电纺纤维膜的低温等离子处理研究[J].合成纤维工业,2013,36(4): 30-33. [29]Sugiyama K,Okamura A,Kawazoe N,et al.Coating of collagen on a poly (l-lactic acid) sponge surface for tissue engineering.Mater Sci Eng C. 2012;32(2): 290-295. [30]Chen XX,Zhang XR,Li QS.Research advance of collagen protein in eye diseases.Guoji Yanke Zazhi (Int Eye Sci).2015;15(10):1745-1748. [31]Müller R,Abke J,Schnell E,et al.Surface engineering of stainless steel materials by covalent collagen immobilization to improve implant biocompatibility. Biomaterials.2005; 26(34): 6962-6972. [32]陈锐.静电纺制备胶原蛋白/聚氨酯心脏瓣膜组织工程支架材料的研究[D].东华大学,2010. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Shen Jinbo, Zhang Lin. Micro-injury of the Achilles tendon caused by acute exhaustive exercise in rats: ultrastructural changes and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1190-1195. |
| [3] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [4] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [5] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [6] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [7] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [8] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [9] | Liu Liu, Zhou Qingzhu, Gong Zhuo, Liu Boyan, Yang Bin, Zhao Xian. Characteristics and manufacturing techniques of collagen/inorganic materials for constructing tissue-engineered bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 607-613. |
| [10] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [11] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [12] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [13] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [14] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [15] | Xu Xiaoming, Chen Yan, Song Qian, Yuan Lu, Gu Jiaming, Zhang Lijuan, Geng Jie, Dong Jian. Human placenta derived mesenchymal stem cell gel promotes the healing of radiation skin damage in SD rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3976-3980. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

