Chinese Journal of Tissue Engineering Research ›› 2016, Vol. 20 ›› Issue (20): 2940-2948.doi: 10.3969/j.issn.2095-4344.2016.20.008
Previous Articles Next Articles
Atorvastatin promotes implant osseointegration via the activation of Wnt/β-catenin signal pathway in osteoporotic rats
Liang Yao-zhong1, Chen Shu2, Yang Yu-hao1, Lan Chun-hai1, Zhang Guo-wei1, Ji Zhi-sheng1, Lin Hong-sheng1
- 1 Department of Orthopedics, the First Affiliated Hospital of Jinan University, Guangzhou 510630, Guangdong Province, China
2 Department of Gynaecology and Obstetrics, the First Affiliated Hospital of Jinan University, Guangzhou 510630, Guangdong Province, China
-
Received:2016-04-06Online:2016-05-13Published:2016-05-13 -
Contact:Lin Hong-sheng, M.D., Chief physician, Department of Orthopedics, the First Affiliated Hospital of Jinan University, Guangzhou 510630, Guangdong Province, China -
About author:Liang Yao-zhong, Master, Attending physician, Department of Orthopedics, the First Affiliated Hospital of Jinan University, Guangzhou 510630, Guangdong Province, China -
Supported by:the Natural Science Foundation of Guangdong Province, China, No. 2014A030313357
CLC Number:
Cite this article
Liang Yao-zhong1, Chen Shu2, Yang Yu-hao1, Lan Chun-hai1, Zhang Guo-wei1, Ji Zhi-sheng1, . Atorvastatin promotes implant osseointegration via the activation of Wnt/β-catenin signal pathway in osteoporotic rats[J]. Chinese Journal of Tissue Engineering Research, 2016, 20(20): 2940-2948.
share this article
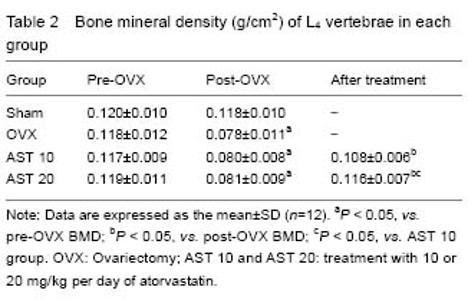
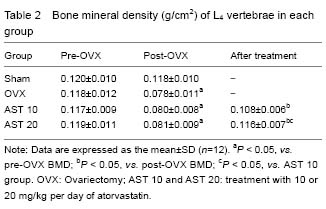
Effect of atorvastatin on BMD of osteoporotic rats BMD of L4 vertebrae in the Sham, ovariectomy, AST 10 and AST 20 groups at different time points are showed in Table 2. BMD of rats receiving ovariectomy was decreased. BMD of rats in AST 20 group was significantly increased compared with AST 10 group (P < 0.05). There was no significant difference in BMD between AST 20 and Sham groups (P > 0.05)."
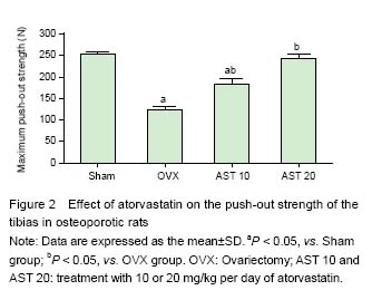
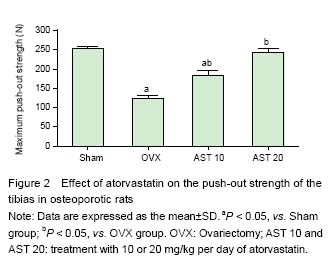
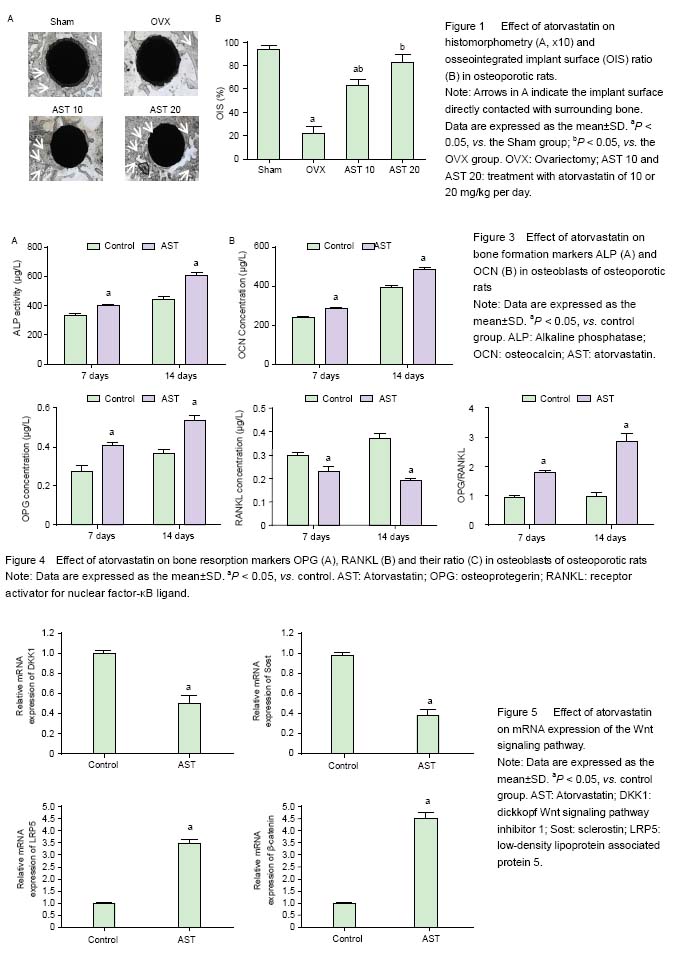
Effect of atorvastatin on histomorphometry and OIS ratio of implants in osteoporotic rats Bone mass around the implant and OIS were obviously decreased in the VOX group compared with those in the Sham group, while it was markedly increased after atorvastatin administration, especially in AST 20 group, the bone mass was similar to that in the Sham group (Figure 1A). Moreover, the administration of atorvastatin signficantly increased OIS compared with the OVX group (P < 0.05). More bone mass around the implant and higher OIS were observed in the OVX 20 group compared with the OVX group (P < 0.05), while no significant differences were found between the AST 20 and sham-surgery groups (Figure 1B). Effect of atorvastatin on the push-out strength of the tibias in osteoporotic rats As presented in Figure 2, it was found that the maximum push-out force of the tibias in the OVX group was signi?cantly decreased compared with that of the Sham group (P < 0.05). Oral administration of atorvastatin (AST 10 and AST 20) signi?cantly increased the maximum push-out force of the tibias (P < 0.05). Moreover, higher dosage of AST showed more pronounced effect on the increase of maximum push-out force of the tibias (P < 0.05). No difference was found between the Sham and AST 20 groups (P > 0.05). Effect of atorvastatin on bone formation markers in osteoblasts As displayed in Figure 3A, the ALP activity in the AST group was significantly increased compared with that in the control group on day 7 (P < 0.05). Further increase in the ALP activity in both groups was observed on day 14, especially in the AST group. It was also found that the OCN concentration was significantly increased in the AST group compared with the control group (P < 0.05), and the OCN concentration in both groups were increased with time (Figure 3B). Effect of atorvastatin on bone resorption markers in osteoblasts It was found that the OPG level in AST group was significantly increased compared with that in control group after 7- and 14-day of cell culture (Figure 4A). However, the RANKL concentration was significantly decreased in the osteoblasts treated with atorvastatin for 7 or 14 days (Figure 4B). The ratio between OPG and RANKL also increased with time in the AST group (Figure 4C). Effect of atorvastatin on mRNA expression of the Wnt signaling pathway mRNA expression of the Wnt signaling pathway including DKK1, Sost, LRP5, and β-catenin were detected by RT-PCR, and the results were presented in Figure 5. Atorvastatin treatment significantly decreased the mRNA expression of DKK-1 to 0.5-fold (P < 0.05), Sost to 0.4-fold (P < 0.05), respectively; and significantly increased LRP5 up to 3.5-fold (P < 0.05), β-catenin to 4.5-fold (P < 0.05), compared with those of the control group, suggesting atorvastatin activates the Wnt signaling pathway."
| [1] Kanis JA. Assessment of fracture risk and its application to screening for postmenopausal osteoporosis: synopsis of a WHO report. WHO Study Group. Osteoporos Int. 1994;4(6):368-381.[2] Ray NF, Chan JK, Thamer M, et al. Medical expenditures for the treatment of osteoporotic fractures in the United States in 1995: report from the National Osteoporosis Foundation. J Bone Miner Res. 1997;12(1):24-35.[3] Kwak B, Mulhaupt F, Myit S, et al. Statins as a newly recognized type of immunomodulator. Nat Med. 2000; 6(12):1399-1402.[4] Kobashigawa JA. Statins as immunosuppressive agents. Liver Transpl. 2001;7(6):559-561.[5] Gomberg-Maitland M, Fuster V, Fayad ZA, et al. Statins and plaque stability. J Cardiovasc Risk. 2003;10(3):161-167.[6] Skaletz-Rorowski A, Walsh K. Statin therapy and angiogenesis. Curr Opin Lipidol. 2003;14(6):599-603.[7] Dimmeler S, Aicher A, Vasa M, et al. HMG-CoA reductase inhibitors (statins) increase endothelial progenitor cells via the PI 3-kinase/Akt pathway. J Clin Invest. 2001;108(3):391-397.[8] Lu D, Goussev A, Chen J, et al. Atorvastatin reduces neurological deficit and increases synaptogenesis, angiogenesis, and neuronal survival in rats subjected to traumatic brain injury. J Neurotrauma. 2004;21(1):21-32.[9] Edwards CJ, Hart DJ, Spector TD. Oral statins and increased bone-mineral density in postmenopausal women. Lancet. 2000;355(9222):2218-2219.[10] Chan KA, Andrade SE, Boles M, et al. Inhibitors of hydroxymethylglutaryl-coenzyme A reductase and risk of fracture among older women. Lancet. 2000;355 (9222): 2185-2188.[11] Meier CR, Schlienger RG, Kraenzlin ME, et al. HMG-CoA reductase inhibitors and the risk of fractures. JAMA. 2000;283(24):3205-3210.[12] Wang PS, Solomon DH, Mogun H, et al. HMG-CoA reductase inhibitors and the risk of hip fractures in elderly patients. JAMA. 2000;283(24):3211-3216.[13] Schoofs MW, Sturkenboom MC, van der Klift M, et al. HMG-CoA reductase inhibitors and the risk of vertebral fracture. J Bone Miner Res. 2004;19(9):1525-1530.[14] Bauer DC, Mundy GR, Jamal SA, et al. Use of statins and fracture. Results of 4 prospective studies and cumulative meta-analysis of observational studies and controlled trials. Arch Int Med. 2004;164(2):146-152.[15] Ruiz-Gaspa S, Nogues X, Enjuanes A, et al. Simvastatin and atorvastatin enhance gene expression of collagen type 1 and osteocalcin in primary human osteoblasts and MG-63 cultures. J Cell Biochem. 2007;101(6):1430-1438.[16] Kim JY, Lee EY, Lee EB, et al. Atorvastatin inhibits osteoclastogenesis by decreasing the expression of RANKL in the synoviocytes of rheumatoid arthritis. Arthritis Res Ther. 2012;14(4):R187.[17] Viereck V, Grundker C, Blaschke S, et al. Atorvastatin stimulates the production of osteoprotegerin by human osteoblasts. J Cell Biochem. 2005;96(6):1244-1253.[18] Gradosova I, Zivna H, Palicka V, et al. Protective effect of atorvastatin on bone tissue in orchidectomised male albino Wistar rats. Eur J Pharmacol. 2012;679(1-3):144-150.[19] Lima CEVD, Calixto JC, Anbinder AL. Influence of the association between simvastatin and demineralized bovine bone matrix on bone repair in rats. Braz Oral Res. 2011;25(1):42-48.[20] Alvarez K, Fukuda M, Yamamoto O. Titanium Implants after alkali heating treatment with a [Zn(OH)4]2 complex: analysis of interfacial bond strength using push-out tests. Clin Implant Dent Relat Res. 2010;12 Suppl 1:e114-e125.[21] NIH Consensus Development Panel on Osteoporosis Prevention, Diagnosis, and Therapy. Osteoporosis prevention, diagnosis, and therapy. JAMA. 2001;285(6): 785-795.[22] Osteoporosis prevention, diagnosis, and therapy. NIH Consens Statement. 2000;17(1):1-45.[23] Sato M, Bryant HU, Iversen P, et al. Advantages of raloxifene over alendronate or estrogen on nonreproductive and reproductive tissues in the long-term dosing of ovariectomized rats. J Pharmacol Exp Ther. 1996;279(1):298-305.[24] Zheng X, Wu G, Nie Y, et al. Electroacupuncture at the governor vessel and bladder meridian acupoints improves postmenopausal osteoporosis through osteoprotegerin/ RANKL/RANK and Wnt/beta-catenin signaling pathways. Exp Ther Med. 2015;10(2):541-548.[25] Ray S, Thormann U, Sommer U, et al. Effects of macroporous, strontium loaded xerogel-scaffolds on new bone formation in critical-size metaphyseal fracture defects in ovariectomized rats. Injury. 2016;47 Suppl 1:S52-S61.[26] Mundy G, Garrett R, Harris S, et al. Stimulation of bone formation in vitro and in rodents by statins. Science. 1999;286(5446):1946-1949.[27] Chen PY, Sun JS, Tsuang YH, et al. Simvastatin promotes osteoblast viability and differentiation via Ras/Smad/Erk/BMP-2 signaling pathway. Nutr Res. 2010; 30(3):191-199.[28] Chen S, Yang JY, Zhang SY, et al. Effects of simvastatin gel on bone regeneration in alveolar defects in miniature pigs. Chin Med J (Engl). 2011;124(23):3953-3958.[29] Kajinami K, Takekoshi N, Matsui S, et al. Effect of pretreatment vitamin D levels on in vivo effects of atorvastatin on bone metabolism in patients with heterozygous familial hypercholesterolemia. Am J Cardiol. 2003;92(9):1113-1116.[30] Liu C, Wu Z, Sun HC. The effect of simvastatin on mRNA expression of transforming growth factor-beta1, bone morphogenetic protein-2 and vascular endothelial growth factor in tooth extraction socket. Int J Oral Sci. 2009;1(2):90-98.[31] Zhang M, Zhou SH, Li XP, et al. Atorvastatin downregulates BMP-2 expression induced by oxidized low-density lipoprotein in human umbilical vein endothelial cells. Circ J. 2008;72(5):807-812.[32] Savopoulos C, Dokos C, Kaiafa G, et al. Adipogenesis and osteoblastogenesis: trans-differentiation in the pathophysiology of bone disorders. Hippokratia. 2011;15(1):18-21.[33] Song C, Guo Z, Ma Q, et al. Simvastatin induces osteoblastic differentiation and inhibits adipocytic differentiation in mouse bone marrow stromal cells. Biochem Biophys Res Commun. 2003;308(3):458-462.[34] Cruz AC, Gruber BL. Statins and osteoporosis: can these lipid-lowering drugs also bolster bones? Cleve Clin J Med. 2002;69(4):277-278.[35] Lacey DL, Timms E, Tan HL, et al. Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell. 1998;93(2):165-176.[36] Maritz FJ, Conradie MM, Hulley PA, et al. Effect of statins on bone mineral density and bone histomorphometry in rodents. Arterioscler Thromb Vasc Biol. 2001;21(10):1636-1641.[37] Golabek K, Ostrowska Z, Ziora K, et al. Association between omentin-1, bone metabolism markers, and cytokines of the RANKL/RANK/OPG system in girls with anorexia nervosa. Endokrynologia Polska. 2015;66(6): 514-520.[38] Goes P, Lima AP, Melo IM, et al. Effect of Atorvastatin in radiographic density on alveolar bone loss in wistar rats. Braz Dent J. 2010;21(3):193-198.[39] Marie PJ,Kassem M. Osteoblasts in osteoporosis: past, emerging, and future anabolic targets. Eur J Endocrinol. 2011;165(1):1-10.[40] Rabbani SA, Arakelian A, Farookhi R. LRP5 knockdown: effect on prostate cancer invasion growth and skeletal metastasis in vitro and in vivo. Cancer Med. 2013;2(5): 625-635.[41] Chu TQ, Teng JJ, Jiang LY, et al. Lung cancer-derived Dickkopf1 is associated with bone metastasis and the mechanism involves the inhibition of osteoblast differentiation. Biochem Biophys Res Commun. 2014; 443(3):962-968.[42] Morvan F, Boulukos K, Clement-Lacroix P, et al. Deletion of a single allele of the Dkk1 gene leads to an increase in bone formation and bone mass. J Bone Miner Res. 2006;21(6):934-945.[43] Li J, Sarosi I, Cattley RC, et al. Dkk1-mediated inhibition of Wnt signaling in bone results in osteopenia. Bone. 2006;39(4):754-766.[44] Lozano D, Sanchez-Salcedo S, Portal-Nunez S, et al. Parathyroid hormone-related protein (107-111) improves the bone regeneration potential of gelatin-glutaraldehyde biopolymer-coated hydroxyapatite. Acta Biomater. 2014;10(7):3307-3316.[45] Li XD, Ominsky MS, Niu QT, et al. Targeted deletion of the sclerostin gene in mice results in increased bone formation and bone strength. J Bone Miner Res. 2008; 23(6):860-869.[46] Winkler DG, Sutherland MK, Geoghegan JC, et al. Osteocyte control of bone formation via sclerostin, a novel BMP antagonist. EMBO J. 2003;22(23):6267-6276.[47] Rajamannan NM, Subramaniam M, Caira F, et al. Atorvastatin inhibits hypercholesterolemia-induced calcification in the aortic valves via the Lrp5 receptor pathway. Circulation. 2005;112(9 Suppl):1229-1234. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Tang Hui, Yao Zhihao, Luo Daowen, Peng Shuanglin, Yang Shuanglin, Wang Lang, Xiao Jingang. High fat and high sugar diet combined with streptozotocin to establish a rat model of type 2 diabetic osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1207-1211. |
| [3] | Li Zhongfeng, Chen Minghai, Fan Yinuo, Wei Qiushi, He Wei, Chen Zhenqiu. Mechanism of Yougui Yin for steroid-induced femoral head necrosis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1256-1263. |
| [4] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [5] | Hou Guangyuan, Zhang Jixue, Zhang Zhijun, Meng Xianghui, Duan Wen, Gao Weilu. Bone cement pedicle screw fixation and fusion in the treatment of degenerative spinal disease with osteoporosis: one-year follow-up [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 878-883. |
| [6] | Li Shibin, Lai Yu, Zhou Yi, Liao Jianzhao, Zhang Xiaoyun, Zhang Xuan. Pathogenesis of hormonal osteonecrosis of the femoral head and the target effect of related signaling pathways [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 935-941. |
| [7] | Xiao Fangjun, Chen Shudong, Luan Jiyao, Hou Yu, He Kun, Lin Dingkun. An insight into the mechanism of Salvia miltiorrhiza intervention on osteoporosis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 772-778. |
| [8] | Liu Bo, Chen Xianghe, Yang Kang, Yu Huilin, Lu Pengcheng. Mechanism of DNA methylation in exercise intervention for osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 791-797. |
| [9] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [10] | Li Chenjie, Lü Linwei, Song Yang, Liu Jingna, Zhang Chunqiu. Measurement and statistical analysis of trabecular morphological parameters of titanium alloy peri-prosthesis under preload [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 516-520. |
| [11] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [12] | Shi Xiaoxiu, Mao Shilong, Liu Yang, Ma Xingshuang, Luo Yanfeng. Comparison of tantalum and titanium (alloy) as orthopedic materials: physical and chemical indexes, antibacterial and osteogenic ability [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 593-599. |
| [13] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [14] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [15] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||