Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (43): 6914-6921.doi: 10.3969/j.issn.2095-4344.2014.43.005
Previous Articles Next Articles
High-performance porous beta-tricalcium phosphate bone tissue engineering scaffolds using 3D printing
Yuan Jing1, 2, Zhen Ping1, Zhao Hong-bin1
- 1Department of Orthopedics, General Hospital of Lanzhou Military Region, Lanzhou 730050, Gansu Province, China; 2Graduate School, Gansu University of Traditional Chinese Medicine, Lanzhou 730030, Gansu Province, China
-
Received:2014-09-20Online:2014-10-15Published:2014-10-15 -
Contact:Zhen Ping, M.D., Professor, Master’s supervisor, Department of Orthopedics, Lanzhou General Hospital of Lanzhou Military Area Command of Chinese PLA, Lanzhou 730050, Gansu Province, China -
About author:Yuan Jing, Studying for master’s degree, Department of Orthopedics, Lanzhou General Hospital of Lanzhou Military Area Command of Chinese PLA, Lanzhou 730050, Gansu Province, China; Graduate School, Gansu University of Traditional Chinese Medicine, Lanzhou 730030, Gansu Province, China -
Supported by:the National Natural Science Foundation of China, No. 81371983
CLC Number:
Cite this article
Yuan Jing, Zhen Ping, Zhao Hong-bin . High-performance porous beta-tricalcium phosphate bone tissue engineering scaffolds using 3D printing[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(43): 6914-6921.
share this article

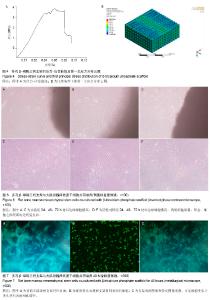
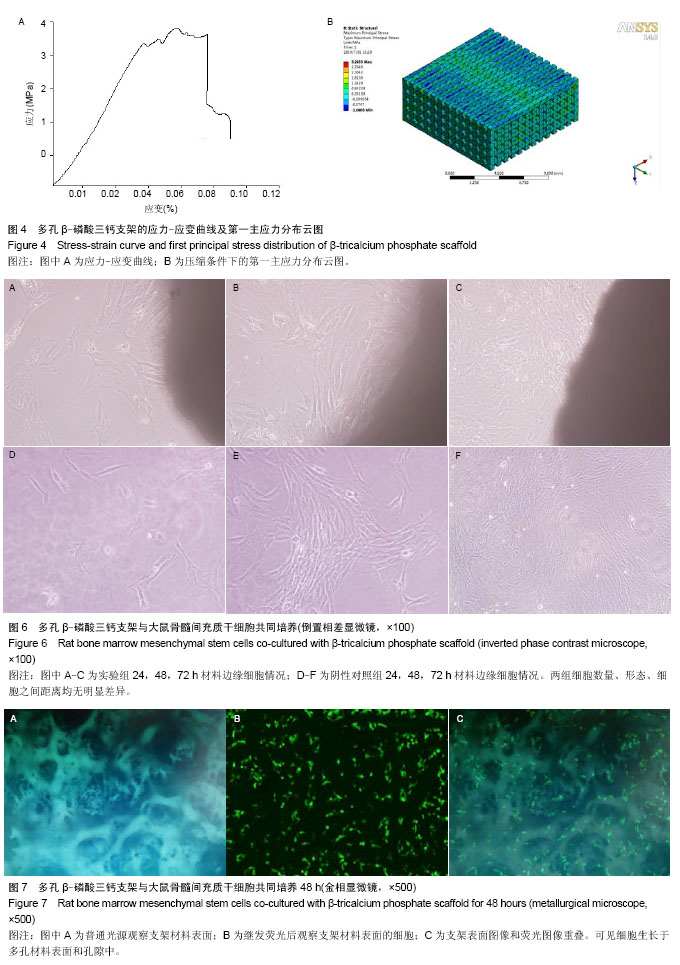
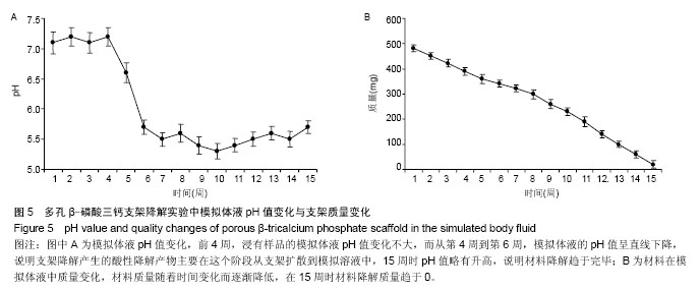
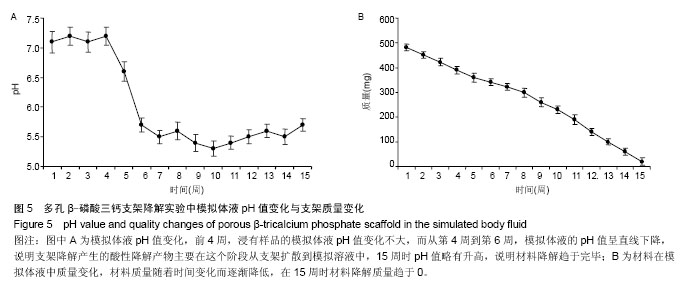
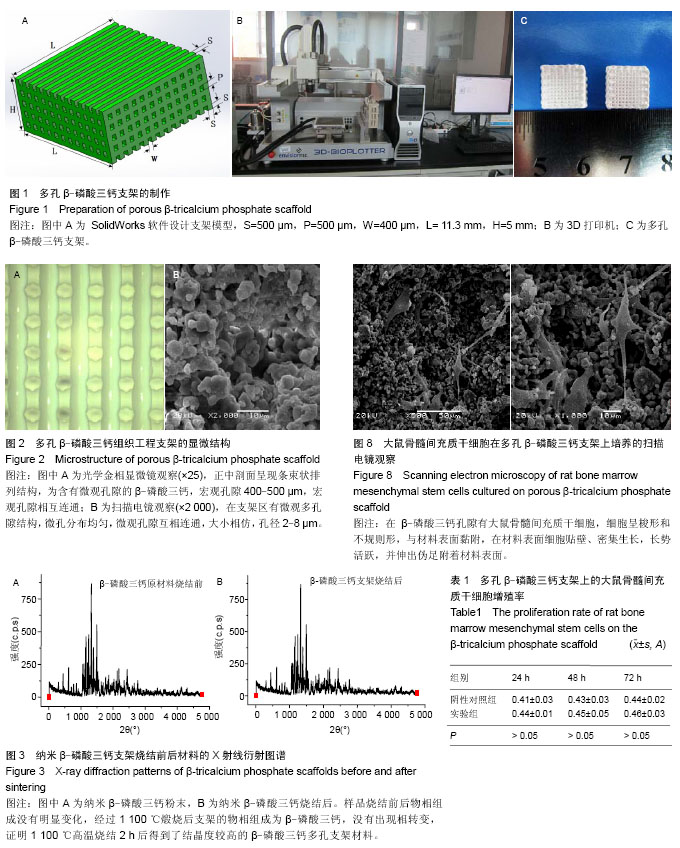
2.1 β-磷酸三钙骨组织工程支架的外观结构、表面显微结构及孔隙率 制备的β-磷酸三钙立体支架截面结构如图2A所示,正中剖面呈现条束状排列结构,为含有微观孔隙的β-磷酸三钙,宏观孔隙400-500 μm,宏观孔隙相互连通。环境扫描电镜观察显示该材料在支架区有微观多孔隙结构,微孔分布均匀,微观孔隙互相连通,大小相仿,孔径2-8 μm,如图2B所示。经检测,三维多孔β-磷酸三钙支架总孔隙率检测为(61.76±2.53)%,其中宏观孔隙率约为42.5%,微观孔隙率为19.26%。 2.2 烧结后多孔β-磷酸三钙支架材料的物相分析 烧结后的β-磷酸三钙支架X射线衍射分析图谱见图3B,纳米β-磷酸三钙粉末用同样方法获得X射线衍射分析图谱见图3A。经过对2个图谱主要波峰对比可知,样品烧结前后物相组成没有明显变化,经过1 100 ℃煅烧后支架的物相组成为β-磷酸三钙,没有出现相转变,证明1 100 ℃高温烧结2 h后得到了结晶度较高的β-磷酸三钙多孔支架材料。 2.3 多孔β-磷酸三钙支架抗压强度及有限元分析 取烧结后的β-磷酸三钙支架通过抗压强度测试,最大压缩强度为(3.31±0.64)MPa,力学性能可以达到松质骨的抗压强度2-12 MPa[19]。随机抽取1支架的压缩应力-应变曲线见图4A,曲线上的第一个峰为支架的抗压强度。β-磷酸三钙支架第一主应力分布云图显示,当第一主应力作用时受力最大的部位在材料中部,见图4B。 2.4 模拟体液pH值变化和组织工程支架的质量变化 前4周,浸有样品的模拟体液pH值变化不大,pH值在7.4左右;而从第4周到第6周,模拟体液的pH值呈直线下降,pH值在5.5左右,说明支架降解产生的酸性降解产物主要在这个阶段从支架扩散到模拟溶液中;15周时,pH值略有升高,说明材料降解趋于完毕,出现了pH有所回升的现象,变化情况见图5A。在体外降解过程中,材料质量随着时间变化而逐渐降低,在15周时材料降解质量趋于0,降解曲线见图5B。 2.5 活细胞增殖率与成骨活性测定结果 实验组与阴性对照组细胞碱性磷酸酶活性分别为(63.4±3.4),(143.3±1.4)金氏单位/L,组间比较差异有显著性意义(P < 0.05)。MTT 检测结果见表1。实验组细胞24,48,72 h的细胞相对增殖率分别为107%,104%,104%,对应的细胞毒性级别均为0。实验组材料上生长良好,有少量细胞长入孔隙,实验组与阴性对照组相比无差异。 2.6 多孔β-磷酸三钙支架上细胞黏附与生长形态观察 显微镜观察材料边缘细胞情况观察:实验组24 h后,见材料边缘有大鼠骨髓间充质干细胞,生长态势良好,细胞较稀疏,三四个细胞簇拥在一起,见图6A;培养48 h后,见材料边缘细胞数量显著增加,布满整个视野,细胞之间紧挨,生长旺盛,并在材料边缘有向上生长,有的细胞进入支架内部,见图6B;72 h后,见细胞有重叠生长,细胞向材料空间内部生长,细胞生长旺盛,在材料边界细胞生长密集,见图6C。通过单位面积计数,发现实验组24,48,72 h细胞数量和阴性对照组无明显差异,两组细胞形态、细胞之间距离均无明显差异。 金相显微镜观察支架材料表面细胞:共同培养48 h取出材料后,荧光染料CFSE对支架材料进行染色,见图7,可见细胞生长于多孔材料表面和孔隙中。 扫描电镜观察材料表面:共同培养48 h,可见在β-磷酸三钙孔隙有大鼠骨髓间充质干细胞,细胞呈梭形和不规则形,与材料表面黏附,在材料表面细胞贴壁、密集生长,长势活跃,并伸出伪足附着材料表面,见图8。"
| [1] Schliephake H,Weich HA,Dullin C,et al.Mandibular bone repair by implantation of rhBMP-2 in a slow release carrier of polylactic acid—an experimental study in rats. Biomaterials. 2008;29(1): 103-110. [2] Jang JH,Castano O,Kim HW.Electrospun materials as potential platforms for bone tissue engineering.Adv Drug Deliver Rev. 2009;61(12):1065-1083. [3] Guarino V,Ambrosio L.Temperature-driven processing techniques for manufacturing fully interconnected porous scaffolds in bone tissue engineering.Proc Inst Mech Eng H. 2010;224(12):1389-1400. [4] 毛恩亮,戴红莲,雷军,等.热致相分离/粒子滤出法制备多孔支架的研究[J].武汉理工大学学报,2010,32(12):15-18. [5] 赵娜如,康海峰,魏煊,等.原位凝固成型法制备可控孔隙结构的多孔β-磷酸三钙陶瓷[J].硅酸盐学报,2010,38(12):2314-2318. [6] Olsson M.Chemical stability of grain boundariesin β-tricalcium phosphate ceramics: β-TCP as bone substitute material. Studentuppsats (Examensarbete).2012:11-30. [7] Tagil M.Bone Substitutes,Grafts and Cement.Distal Radius Fractures. Springer.2014:233-239. [8] Bohner M,Galea L,Doebelin N.Calcium phosphate bone graft substitutes: Failures and hopes.J Eur Ceram Soc.2012; 32(11): 2663-2671. [9] Bohner M.Design of ceramic-based cements and putties for bone graft substitution. Eur Cell Mater.2010;20(1):3-10. [10] Sariibrahimoglu K,Wolke JG,Leeuwenburgh SC,et al. Characterization of α/β-TCP Based Injectable Calcium Phosphate Cement as a Potential Bone Substitute.Key Eng Mater.2013;529:157-160. [11] Sugawara A,Asaoka K,Ding SJ.Calcium phosphate-based cements: clinical needs and recent progress.J Mater Chem. 2013;1(8):1081-1089. [12] Butscher A,Bohner M,Hofmann S,et al.Structural and material approaches to bone tissue engineering in powder-based three-dimensional printing .Acta Biomaterialia. 2011;7(3): 907-920. [13] Bose S,Vahabzadeh S,Bandyopadhyay A.Bone tissue engineering using 3D printing. Mater Today. 2013;16(12): 496-504. [14] Ribeiro G,Trommer RM,dos Santos LA,et al.Novel method to produce β-TCP scaffolds.Mater Lett.2011;65(2):275-277. [15] Karageorgiou V,Kaplan D.Porosity of 3D biomaterial scaffolds and osteogenesis.Biomaterials.2005;26(27):5474-5491. [16] Miranda P,Pajares A,Saiz E,et al.Fracture modes under uniaxial compression in hydroxyapatite scaffolds fabricated by robocasting. J Biomed Mater Res A.2007; 83(3): 646-655. [17] Miranda P,Pajares A,Guiberteau F.Finite element modeling as a tool for predicting the fracture behavior of robocast scaffolds.Acta Biomaterialia.2008;4(6): 1715-1724. [18] Bandyopadhyay A,Petersen J,Fielding G,et al.ZnO, SiO2, and SrO doping in resorbable tricalcium phosphates: Influence on strength degradation, mechanical properties, and in vitro bone–cell material interactions.J Biomed Mater Res B.2012; 100(8):2203-2212. [19] Olszta MJ,Cheng X,Jee SS,et al.Bone structure and formation: A new perspective. Mater Sci Eng Rev.2007;58(3):77-116. [20] Carletti E,Motta A,Migliaresi C.Scaffolds for tissue engineering and 3D cell culture.3D cell culture.Springer.2011:17-39. [21] Warnke PH,Seitz H,Warnke F,et al.Ceramic scaffolds produced by computer‐assisted 3D printing and sintering: Characterization and biocompatibility investigations.J Biomed Mater Res B.2010;93(1):212-217. [22] Bose S,Tarafder S,Banerjee SS,et al.Understanding in vivo response and mechanical property variation in MgO, SrO and SiO2 doped β-TCP. Bone.2011;48(6): 1282-1290. [23] Santos CF,Silva AP,Lopes L,et al.Design and production of sintered β-tricalcium phosphate 3D scaffolds for bone tissue regeneration.Mater Sci Eng C.2012;32(5): 1293-1298. [24] Wu Q,Zhang X,Wu B,et al.Fabrication and characterization of porous HA/β-TCP scaffolds strengthened with micro-ribs structure. Mater Lett.2013;92:274-277. [25] Best S,Porter A,Thian E,et al.Bioceramics: past, present and for the future. J Eur Ceram Soc.2008;28(7):1319-1327. [26] 张永光,王志强.骨移植替代材料研究进展[J].中国修复重建外科杂志,2008,22(10): 1264-1268. [27] Duarte Campos DF,Vogt M,Lindner M,et al.Two-photon laser scanning microscopy as a useful tool for imaging and evaluating macrophage-, IL-4 activated macrophage-and osteoclast-based In Vitro degradation of beta-tricalcium phosphate bone substitute material.Microsc Res Tech. 2014;77(2):143-152. [28] Bose S,Roy M,Bandyopadhyay A.Recent advances in bone tissue engineering scaffolds.Trends Biotechnol. 2012;30(10): 546-554. [29] Kapoor S,Batra U,Kohli S.Sintering Effects on Morphology, Thermal Stability and Surface Area of Sol‐Gel Derived Nano‐Hydroxyapatite Powder [C].Proceedings of the INTERNATIONAL CONFERENCE ON ADVANCES IN CONDENSED AND NANO MATERIALS (ICACNM‐2011), AIP Publishing, 2011:375-376. [30] Feng P,Wei P,Shuai C,et al.Characterization of mechanical and biological properties of 3-D scaffolds reinforced with zinc oxide for bone tissue engineering. PloS One.2014;9(1):1-13. [31] Fielding GA,Bandyopadhyay A,Bose S.Effects of silica and zinc oxide doping on mechanical and biological properties of 3D printed tricalcium phosphate tissue engineering scaffolds.Dent Mater.2012;28(2):113-122. [32] Tarafder S,Balla VK,Davies NM,et al.Microwave‐sintered 3D printed tricalcium phosphate scaffolds for bone tissue engineering.J Tissue Eng Regen Med.2013;7(8):631-641. [33] Shie MY,Ding SJ,Chang HC.The role of silicon in osteoblast-like cell proliferation and apoptosis.Acta Biomaterialia. 2011; 7(6): 2604-2614. [34] Woodard JR,Hilldore AJ,Lan SK,et al.The mechanical properties and osteoconductivity of hydroxyapatite bone scaffolds with multi-scale porosity. Biomaterials.2007;28(1):45-54. [35] Lu L,Zhang Q,Wootton D,et al.Biocompatibility and biodegradation studies of PCL/β-TCP bone tissue scaffold fabricated by structural porogen method.J Mater Sci Mater Med. 2012;23(9):2217-2226. [36] Miyaji H,Yokoyama H,Kosen Y,et al.Bone augmentation in rat by highly porous β-TCP scaffolds with different open-cell sizes in combination with fibroblast growth factor-2.J Oral Tissue Eng 2013;10(3):172-181. [37] Bernstein A,Suedkamp N,Mayr HO,et al.Microporous ß-Tricalcium Phosphate (TCP)-A Delivery Vehicle of Growth Factors and Drugs. Key Eng Mater.2014;587:93-96. [38] Kang Y,Kim S,Bishop J,et al.The osteogenic differentiation of human bone marrow MSCs on HUVEC-derived ECM and β-TCP scaffold.Biomaterials.2012;33(29):6998-7007. [39] Hao W,Pang L,Jiang M,et al.Skeletal repair in rabbits using a novel biomimetic composite based on adipose-derived stem cells encapsulated in collagen I gel with PLGA-β-TCP scaffold.J Orthop Res.2010;28(2):252-257 |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Chen Xinmin, Li Wenbiao, Xiong Kaikai, Xiong Xiaoyan, Zheng Liqin, Li Musheng, Zheng Yongze, Lin Ziling. Type A3.3 femoral intertrochanteric fracture with augmented proximal femoral nail anti-rotation in the elderly: finite element analysis of the optimal amount of bone cement [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1404-1409. |
| [3] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [4] | Cai Qunbin, Zou Xia, Hu Jiantao, Chen Xinmin, Zheng Liqin, Huang Peizhen, Lin Ziling, Jiang Ziwei. Relationship between tip-apex distance and stability of intertrochanteric femoral fractures with proximal femoral anti-rotation nail: a finite element analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 831-836. |
| [5] | Song Chengjie, Chang Hengrui, Shi Mingxin, Meng Xianzhong. Research progress in biomechanical stability of lateral lumbar interbody fusion [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 923-928. |
| [6] | Liu Zhao, Xu Xilin, Shen Yiwei, Zhang Xiaofeng, Lü Hang, Zhao Jun, Wang Zhengchun, Liu Xuzhuo, Wang Haitao. Guiding role and prospect of staging and classification combined collapse prediction method for osteonecrosis of femoral head [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 929-934. |
| [7] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [8] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [9] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [10] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [11] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [12] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [13] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [14] | Chen Lu, Zhang Jianguang, Deng Changgong, Yan Caiping, Zhang Wei, Zhang Yuan. Finite element analysis of locking screw assisted acetabular cup fixation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 356-361. |
| [15] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||