Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (30): 4783-4789.doi: 10.3969/j.issn.2095-4344.2014.30.005
Previous Articles Next Articles
Effect of glutaraldehyde cross-linking on the properties of chitosan/hydroxyapatite- gentamicin delayed materials
Zhao Hong-xia
- School of Chemical Engineering and Light Industry, Guangdong University of Technology, Guangzhou 510006, Guangdong Province, China
-
Revised:2014-05-30Online:2014-07-16Published:2014-08-08 -
About author:Zhao Hong-xia, M.D., Lecturer, School of Chemical Engineering and Light Industry, Guangdong University of Technology, Guangzhou 510006, Guangdong Province, China -
Supported by:the National Natural Science Foundation of China, No. 31300804, 31301480
CLC Number:
Cite this article
Zhao Hong-xia. Effect of glutaraldehyde cross-linking on the properties of chitosan/hydroxyapatite- gentamicin delayed materials[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(30): 4783-4789.
share this article
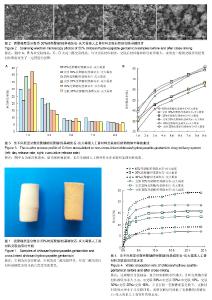
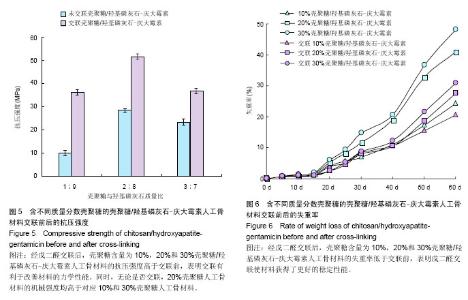
2.1 材料的表面形貌 图1是交联前后30%CS/HA-G缓释材料经冷等静压成型后的照片,从外观上观察,经戊二醛交联后材料的颜色由原来的白色变为淡黄色。从交联30% CS/HA-G的扫描电镜照片(图2)中可以看到,CS/HA-G粉体材料为球状和片状的混合体。与非交联材料相比,交联CS/HA-G其粉体粒径有所增大,说明戊二醛的交联作用使得材料颗粒发生了一定程度的团聚。 2.2 载药材料的体外药物释放 载药材料的药物突释现象一直以来都是研究者急待解决的一个难题。制剂在进入体内的第1天前后会迅速、大量地释放药物,这种现象被称为突释。给药初期的突释有可能导致血药浓度接近或超过中毒水平,从而产生明显的不良反应[31]。从图3可看出,未经交联的CS/HA-G存在着较明显的突释现象,第1天时间内10%CS/HA-G、20%CS/HA-G和30% CS/HA-G的药物释放量分别达到29.9%,39.0%和42.2%;经戊二醛交联后各种材料第1天内庆大霉素的释放量分别下降至22.3%,30.1%和33.6%。药物缓释体系中药物突释的原因,一般从以下两个方面来解释,首先是因为药物分子和药物载体分子之间的相互作用太弱,导致药物很容易从载体进入释放介质中;其次,药物在药物缓释体系中的分布导致了突释,药物疏松地吸附于材料表层或被包埋在材料表层。在释放初期,药物从材料的孔洞和缝隙中释放出来[31]。 CS/HA-G中壳聚糖/羟基磷灰石载体对药物的控释主要包括:壳聚糖能对药物进行有效包埋,壳聚糖形成的三维网络结构对药物释放有较强的阻滞作用;壳聚糖分子链上的活性基团可与药物成键;纳米羟基磷灰石对药物的有效吸附。以戊二醛作为交联剂对壳聚糖/羟基磷灰石进行交联,主要是对材料中壳聚糖的化学作用。经戊二醛交联后,壳聚糖分子内及分子间的交联度增大,形成的多孔结构变得更加致密,对药物释放能起到更大的阻滞作用,从而降低了药物的突释。药物载体是药物释放体系的重要组成部分,其亲水性也影响了药物的缓释速度。一般多聚物的亲水性越强,突释越大,释放速度越快[32]。交联反应使得壳聚糖的亲水基团氨基参与了成键,减少了材料的亲水基团数目,使材料的吸水性降低,从而降低了药物的突释。体外药物释放实验结果表明交联作用能改善CS/HA-G缓释材料的突释现象,从而减小不良反应,延长释药时间,能达到更好的治疗目的。 缓释体系中药物的释药原理主要有溶出、扩散、溶蚀、渗透压及离子交换作用,其中溶出和扩散释药机制远远超过了其他过程。在骨架型药物缓释体系中,药物分散在不溶性骨架材料中,药物释放是通过骨架中弯曲的孔道扩散进行的,药物释放速度取决于药物在骨架材料中的扩散速率[33-34]。缓释体系在长时间的浸泡过程中,材料会因吸水而出现膨胀,膨胀会使得材料的孔道变大,更利于药物的扩散溶出。在CS/HA-G缓释体系中,交联后材料的吸水率下降,因吸水而造成材料膨胀及药物扩散就会比未交联材料更迟缓。因此在长期浸泡过程中,非交联材料的膨胀表现为速率快而时间短地达到膨胀极值,而交联材料则表现为速率慢而时间长地达到膨胀极值。因此,在后期浸泡过程中,当非交联材料已无明显膨胀时,交联材料还会继续膨胀,此时交联材料因膨胀引起的药物扩散就能大于非交联材料。由于有机高分子壳聚糖在CS/HA-G材料的膨胀中占主导地位,因此壳聚糖含量越大,上述这种交联及非交联材料的膨胀行为差异就会越大。因此第9天时,出现了交联30%CS/HA-G的单次释放率高于非交联30%CS/HA-G的现象。而从总释放量来看,各种壳聚糖含量的交联CS/HA-G总释放量均低于相同壳聚糖含量的CS/HA-G,表明交联CS/HA-G将具备更长的有效药物浓度释放时间。 2.3 材料的亲水性能 交联对CS/HA-G载药材料的吸水性能有较大影响,从图4可看出交联CS/HA-G的吸水率比未交联CS/HA-G低。这主要有两个原因:首先,交联反应减少了材料的亲水基团。材料中有机相壳聚糖分子侧链上有亲水基团氨基和羟基,戊二醛对材料的交联作用主要在于戊二醛与壳聚糖的氨基形成-C=N-键,从而使材料的亲水基团数目减少,直接导致了材料的亲水性能减弱。其次,吸水率也与材料的孔隙率及孔径有关,材料的孔隙率和孔径越大,平衡时能容纳的水越多,从而吸水率也越大。交联反应使壳聚糖形成了更为致密的网络结构,材料的孔隙率下降,从而降低了材料的吸水率。从图4还可看出,3种壳聚糖含量材料的吸水率大小为:30%CS/HA-G> 20%CS/HA-G>10%CS/HA-G,交联30%CS/HA-G>交联20%CS/HA-G>交联10%CS/HA-G。这说明材料中无机相与有机相的含量配比也直接影响了材料的吸水率,CS/HA-G复合材料中无机相羟基磷灰石对水分子的吸附及容纳能力远远小于有机相壳聚糖,因此壳聚糖的含量越高,复合材料的吸水性越大。"
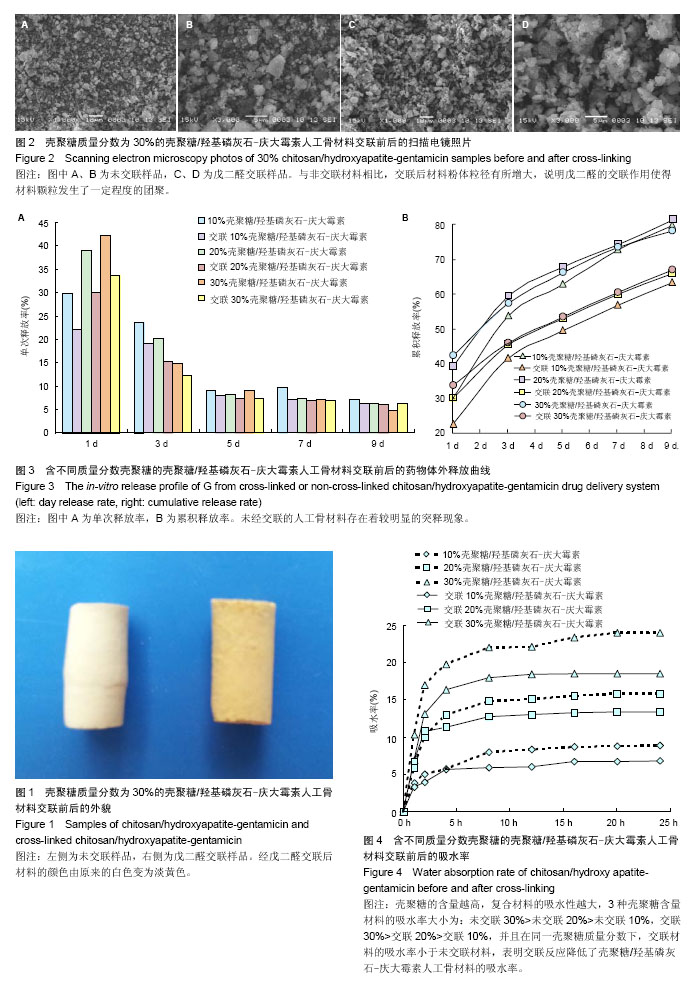
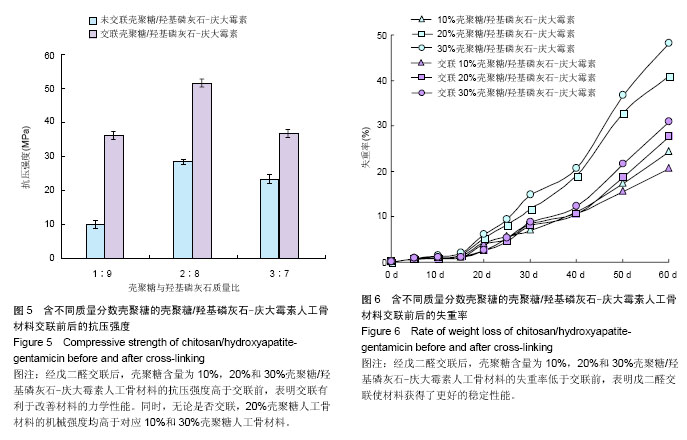
2.4 载药材料的机械性能 良好的力学性能是组织工程人工骨材料的基本要求。从图5可看出,交联使CS/HA-G的机械强度得到显著提高。壳聚糖含量为10%,20%和30%的CS/HA-G抗压强度分别为(10.16±1.17),(28.40±0.64),(23.28±1.30) MPa,经戊二醛交联后材料的机械强度都得到了提高,其抗压强度分别达到(36.30± 1.20),(51.60±2.08),(36.90±3.22) MPa,这一结果表明,交联有利于改善CS/HA-G人工骨材料的力学性能。同时,从图5还可以看出,无论是否交联,20%CS/HA-G的机械强度均高于对应的10%和30%CS/HA-G。单纯的羟基磷灰石脆性较大、强度较低,研究者常采用有机高分子与羟基磷灰石复合来增加材料的弹性和韧性,从而提高材料的力学性能。壳聚糖与羟基磷灰石的复合有效改善了单纯羟基磷灰石的力学性能。随着壳聚糖含量的增大,复合材料的机械强度也随之增大,因此20%CS/HA-G的抗压强度高于10%CS/HA-G。但胶原、壳聚糖、丝素蛋白等这些天然高分子材料在提取制备过程中已经失去了其在生物组织中特有的三维结构,导致其在生物组织中特有的高机械性能也随之丧失了。所以,壳聚糖含量的进一步增大并不能带给复合材料更高的强度。相反,由于壳聚糖本身力学强度不高,复合物中壳聚糖含量过高反而会导致材料的机械强度降低,导致30%CS/HA-G的抗压强度反低于20%CS/HA-G。因此若单从CS/HA-G人工骨材料的机械强度这一角度考虑,复合材料以20%壳聚糖含量为最佳。 2.5 载药材料的体外降解 图6为CS/HA-G在交联前后的体外降解情况,从图中可看到,交联反应降低了材料的降解速率。这主要有3个原因:首先,交联反应减少了材料的亲水基团数目,使材料的吸水性降低,这将减缓材料内部液态环境的物质交换,导致材料的累积失重率降低。其次,戊二醛使壳聚糖发生了分子间或分子内交联,使得材料在降解时需要断裂更多的化学键,从而使降解变得更困难。第三,交联反应使壳聚糖形成了更为致密的网络结构,使与壳聚糖复合的羟基磷灰石更难溶解或扩散到浸泡液中。在浸泡60 d时,未经交联10%,20%,30%CS/HA-G的降解失重率分别为24.2%,40.8%,48.0%,而交联CS/HA-G的失重率降低至20.5%,27.5%,30.8%,这一实验结果表明戊二醛交联使材料获得了更好的稳定性能。"
| [1] Netz DJ,Sepulveda P,Pandolfelli VC,et al.Potential use of gelcasting hydroxyapatite porous ceramic as an implantable drug delivery system.Int J Pharm.2001;213(1-2):117-125. [2] Santos C,Rovath CF,Franke RP,et al.Spray-dried hydroxyapatite-5-Fluorouracil granules as a chemotherapeutic delivery system.Ceram Int. 2009;35(1): 509-513. [3] 邹琴,张利,左奕,等.载黄芪多糖骨水泥的理化性能及体外细胞相容性研究[J].功能材料, 2008,39(9):1515-1521. [4] Ginebra MP,Traykova T,Planell JA.Calcium phosphate cements as bone drug delivery systems: A review.J Control Release.2006;113(2):102-110. [5] Gao C,Cai Y,Kong X,et al.Development and characterization of injectable chitosan-based hydrogels containing dexamethasone/rhBMP-2 loaded hydroxyapatite nanoparticles. Mat Lett.2013;93(1):312-315. [6] 蓝琳,智伟,黄晶,等.丹酚酸B缓释羟基磷灰石颗粒与成骨细胞的活性[J].中国组织工程研究与临床康复,2010,14(51): 9501-9506. [7] Teng SH,Lee EJ,Wang P,et al.Functionally gradient chitosan/hydroxyapatite composite scaffolds for controlled drug release.J Biomed Mater Res B Appl Biomater. 2009; 90(1):275-282. [8] Mahmoodi S,Sorkhi L,Farrokhi-Rad M,et al.Electrophoretic deposition of hydroxyapatite–chitosan nanocomposite coatings in different alcohols. Surf Coat Technol. 2013;216: 106114. [9] 王新,刘玲蓉,张其清.纳米羟基磷灰石-壳聚糖骨组织工程支架的研究[J].中国修复重建外科杂志,2007,21(2):120-124. [10] Kong L,Gao Y,Cao W,et al.Preparation and characterization of nano-hydroxyapatite/chitosan composite with enhanced compressive strength by urease-catalyzed method.J Biomed Mater Res A.2005;75(2):275-295. [11] Baskar D,Balu R,Kumar TS.Mineralization of pristine chitosan film through biomimetic process.Int J Biol Macromol. 2011; 49(3):385-389. [12] 杨洪,傅山岗.尿素共沉淀法制备纤维状羟基磷灰石/壳聚糖复合粉料[J].应用化学, 2005,22(1):87-90. [13] Liu C,Wang W,Shen W,et al.Evaluation of the biocompatibility of a nonceramic hydroxyapatite.J. Endodontics. 1997;23(8): 490-493. [14] Lawton DM,Lamaletie MDJ,Gardner DL.Biocompatibility of hydroxyapatite ceramic: response of chondrocytes in a test system using low temperature scanning electron microscopy. J Dent.1989;17(1):21-27. [15] Craig GT,Brook IM,Lamb DJ.Tissue response to subperiosteal implantation of dense hydroxyapatite: case report.Biomaterials.1989;10(2):133-135. [16] Martin L,Søren O,Cody B,et al.Improved bone anchorage of hydroxyapatite coated implants compared with tricalcium-phosphate coated implants in trabecular bone in dogs.Biomaterials.1999;20(9):803-808. [17] Matsumoto T,Okazaki M,Inoue M,et al.Hydroxyapatite particles as a controlled release carrier of protein. Biomaterials.2004;25(17):3807-3812. [18] Ravi Kumar MNV.A review of chitin and chitosan applications. React Funct Polym.2000;46(1):1-27. [19] Britto DD,Campana-Filho SP.Kinetics of the thermal degradation of chitosan.Thermochimica Acta. 2007;465(1-2): 73-82. [20] Kong M,Chen XG,Xing K,et al.Antimicrobial properties of chitosan and mode of action: A state of the art review.Int J Food Microbiol. 2010;144(1):51-63. [21] Hong Y, Song H, Gong Y,et al.Covalently crosslinked chitosan hydrogel: Properties of in vitro degradation and chondrocyte encapsulation.Acta Biomaterialia.2007;3(1):23-31. [22] Klokkevold PR,Lew DS,Ellis DG,et al.Effect of chitosan on lingual hemostasis in rabbits.J Oral Maxillofac Surg. 1991; 49(8):858-863. [23] Kang PL,Chang SJ,Manousakas I,et al.Development and assessment of hemostasis chitosan dressings.Carbohydr Polym.2011;85(3): 565-570. [24] 陈黄琴,黄彬.复合蟾毒灵的壳聚糖/纳米羟基磷灰石人工骨对骨肉瘤细胞增殖的影响[J].现代中西医结合杂志, 2011,20(25): 3143-3146. [25] Liu TY,Chen SY,Li JH,et al.Study on drug release behavior of CDHA/chitosan nanocomposites—Effect of CDHA nanoparticles.J Control Release. 2006;112(1):88-95. [26] Sivakumar M,Panduranga Rao K.Preparation, characterization and in vitro release of gentamicin from coralline hydroxyapatite-gelatin composite microspheres. Biomaterials.2002;23(15):3175-3181. [27] 朱华跃,肖玲.戊二醛交联对壳聚糖/PVA共混膜结构和性能的影响[J].浙江海洋学院学报:自然科学版,2005,24(2):126-142. [28] Beppu MM,Vieira RS,Aimoli CG,et al.Crosslinking of chitosan membranes using glutaraldehyde: Effect on ion permeability and water absorption.J Membr Sci. 2007; 301(1-2):126-130. [29] Kikuchi M,Matsumoto HN,Yamada T,et al.Glutaraldehyde cross-linked hydroxyapatite/collagen self-organized nanocomposites. Biomaterials.2004;25(1):63-69. [30] 陈新,邵正中,黄郁芳,等.不同交联剂含量对戊二醛交联壳聚糖膜结构与性能影响的研究[J].化学学报, 2000,58(12): 1654-1659. [31] 王襄平,梅兴国.多肽及蛋白类药物微球包封率和释放的研究进展[J].国外医学:药学分册,2006,33(3):219-223. [32] 王丹.多肽及蛋白类药物微球的载体材料、制备以及突释现象[J].中国组织工程研究与临床康复,2008,12(10):1931-1934. [33] 李向荣.药剂学[M].杭州:浙江大学出版社,2010:456-460. [34] 周建平.药剂学[M].北京:化学工业出版社,2004:216-218. [35] 赵宏霞,黄才欢,金花,等.壳聚糖/羟基磷灰石-庆大霉素缓释材料的抗菌性能研究及AFM观察[J].功能材料,2009,40(3): 429-435. [36] 赵宏霞,金花,蔡继业.庆大霉素对羟基磷灰石/壳聚糖复合材料性能的影响[J].无机化学学报,2010,26(1):106-111. [37] 吴国杰,吴炜亮,崔英德.聚醚一壳聚糖水凝胶合成工艺条件对性能的影响研究[J].材料导报,2005,19(11):128-130. [38] 杨旭东,吴国杰,林玩金.壳聚糖水凝胶的制备及性能研究[J].化工新型材料,2005,33(12):48-50. [39] 李雪洁,刘万顺,韩宝芹.戊二醛交联型羧甲基壳聚糖的制备和生物相容性的研究[J].中国海洋大学学报:自然科学版, 2007,37 (S2): 121-126. [40] 肖海军,侯春林,薛锋.羧甲基壳聚糖-羧甲基纤维素防粘连膜的制备及其理化特性[J].中国组织工程研究与临床康复, 2010,14 (38): 7069-7074. [41] 许勇,洪华,钱颖,等.医用壳聚糖膜的制备和性能研究[J].功能高分子学报,2004,17(1): 55-60. [42] 蒋柳云,李玉宝,张利,等.纳米羟基磷灰石/壳聚糖-羧甲基纤维素复合支架材料的研究[J].无机材料学报,2008,23(1): 135-140. [43] Zou B,Li X,Zhuang H,et al.Degradation behaviors of electrospun fibrous composites of hydroxyapatite and chemically modified poly(DL-lactide). Polym Degrad Stabil. 2011;96:114-122. [44] Li L,Li G,Jiang J,et al.Electrospun fibrous scaffold of hydroxyapatite/poly (ε-caprolactone) for bone regeneration.J Mater Sci Mater Med. 2012;23(2):547-554. [45] 宁思敏,王东,孙海钰,等.大鼠成骨细胞与壳聚糖/羟基磷灰石复合支架降解产物的生物相容性[J].中国组织工程研究, 2014,18 (12): 1846-1851. [46] 楼维维,董伊雯,金逸凡,等.掺镧羟基磷灰石涂层的合成和性能表征[J].中国组织工程研究,2014,18(8):1224-1230. [47] 王海霞,岳进.梯度掺锶羟基磷灰石骨水泥的体外细胞生物学性能[J].中国组织工程研究,2013,17(47):8149-8154. [48] 李斯日古楞,胡晓文,章超,等.珊瑚羟基磷灰石修复种植体周不同类型骨缺损的效果观察[J].中华口腔医学研究杂志(电子版), 2011,5(2):192-204. [49] Xia Y,Zhou P,Cheng X,et al.Selective laser sintering fabrication of nano-hydroxyapatite/poly-ε-caprolactone scaffolds for bone tissue engineering applications.Int J Nanomedicine.2013;8:4197-4213. [50] Wang H,Li Y,Zuo Y,et al.Biocompatibility and osteogenesis of biomimetic nano-hydroxyapatite/polyamide composite scaffolds for bone tissue engineering. Biomaterials. 2007; 28(22):3338-3348. [51] Heo SJ, Kim SE, Wei J,et al.Fabrication and characterization of novel nano- and micro-HA/PCL composite scaffolds using a modified rapid prototyping process.J Biomed Mater Res A. 2009;89(1):108-116. [52] Fu S,Ni P,Wang B,et al.In vivo biocompatibility and osteogenesis of electrospun poly(ε-caprolactone)-poly (ethylene glycol)-poly (ε-caprolactone)/nano-hydroxyapatite composite scaffold.Biomaterials. 2012;33(33):8363-8371. |
| [1] | Zhong Yizheng, Huang Peizhen, Cai Qunbin, Zheng Liqin, He Xingpeng, Dong Hang. Microstructural indexes that determine the trabecular bone maximum stress of micro-finite element models [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1313-1318. |
| [2] | Wu Taoguang, Nie Shaobo, Chen Hua, Zhu Zhengguo, Qi Lin, Tang Peifu. Biomechanical characteristics of a new multi-dimensional cross locking plate in the treatment of subtrochanteric nonunion [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1330-1334. |
| [3] | Peng Zhixin, Yan Wengang, Wang Kun, Zhang Zhenjiang. Finite element analysis and structural optimization design of 3D printed forearm braces [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1340-1345. |
| [4] | Liu Jinyu, Zhang Hanshuo, Cui Hongpeng, Pan Lingzhi, Zhao Boran, Li Fei, Ding Yu. Finite element biomechanical analysis of minimally invasive treatment of cervical spondylotic myelopathy and accurate exercise rehabilitation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1359-1364. |
| [5] | Wen Xinghua, Ding Huanwen, Cheng Kai, Yan Xiaonan, Peng Yuanhao, Wang Yuning, Liu Kang, Zhang Huiwu. Three-dimensional finite element model analysis of intramedullary nailing fixation design for large femoral defects in Beagle dogs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1371-1376. |
| [6] | Liu Jiaxin, Jia Peng, Men Yutao, Liu Lu, Wang Yeming, Ye Jinduo. Design and optimization of bone trabecular structure with triply periodic minimal surfaces [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 992-997. |
| [7] | Li Huiqin, Wang Chunjuan, Wang Yu, Wang Weifeng, Chen Dinggen, Li Na. Clear aligner orthodontic therapy of rotated mandibular teeth with different shapes: a three-dimensional finite element analysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1050-1054. |
| [8] | Li Yaping, Liu Hong, Gao Zhen, Chen Xiaolin, Huang Wujie, Jiang Zheng. Three-dimensional motion analysis of lower limb biomechanical performance in Tai Chi practitioners accompanied by knee joint pain [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 520-526. |
| [9] | Li Shihao, Li Qi, Li Zhen, Zhang Yuanyuan, Liu Miaomiao, Ouyang Yi, Xu Weiguo. Plantar pressure and gait analysis in patients with anterior cruciate ligament injury and reconstruction [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 626-631. |
| [10] | Zhan Yi, Wang Biao, Ma Yuli, He Simin, Sun Honghui, Hao Dingjun. Biomechanical comparison between a novel bone cement screw system and common surgical methods for the treatment of Kummell’s disease [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 385-390. |
| [11] | Lu Hui, Wu Qimei, Liu Rong. Finite element analysis and application of unilateral and bilateral bone-filling mesh container in treatment of osteoporotic vertebral compression fracture [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 391-397. |
| [12] | Li Rui, Liu Zhen, Guo Zige, Lu Ruijie, Wang Chen. Aspirin-loaded chitosan nanoparticles and polydopamine modified titanium sheets improve osteogenic differentiation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 374-379. |
| [13] | Wu Lihao, Shao Anliang, Xu Lin, Ren Kang, Wang Hongjian, Chen Liang, Xu Ling. Evaluation of immunotoxicity of the absorbable macroporous polysaccharides composite hemostatic material [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 329-334. |
| [14] | Zong Mingrui, Liu Haiyan, Li Bing, Wu Xiuping. Application of carboxymethyl chitosan in tissue engineering of stomatology [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 447-452. |
| [15] | Lu Yu, Xiang Junyi, Yin Benjing, Bao Chaoyu, Bi Heng, Li Jizheng, Chen Shuai, He Guangxiong, Li Jubao. Finite element analysis of oblique-pulling manipulation in the treatment of lumbar synovial incarceration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(27): 4271-4276. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||