Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (30): 4771-4776.doi: 10.3969/j.issn.2095-4344.2014.30.003
Previous Articles Next Articles
Preparation of tissue-engineered esophageal scaffolds using electrospinning technology
Yu Xue-chan, Shen Qiu-xia, Lu Zhen-zhen, Zhang Chen, Deng Ling, Hu Pin, Zhu Ya-bin
- Medical School of Ningbo University, Ningbo 315211, Zhejiang Province, China
-
Revised:2014-06-14Online:2014-07-16Published:2014-08-08 -
Contact:Zhu Ya-bin, Professor, Medical School of Ningbo University, Ningbo 315211, Zhejiang Province, China -
About author:Yu Xue-chan, Medical School of Ningbo University, Ningbo 315211, Zhejiang Province, China -
Supported by:the National Natural Science Foundation of China, 81171476; the Innovative Team Program of Ningbo City, No. 2011B82014; the Social Development Project of Ningbo City, No. 2013C50031
CLC Number:
Cite this article
Yu Xue-chan, Shen Qiu-xia, Lu Zhen-zhen, Zhang Chen, Deng Ling, Hu Pin, Zhu Ya-bin. Preparation of tissue-engineered esophageal scaffolds using electrospinning technology[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(30): 4771-4776.
share this article
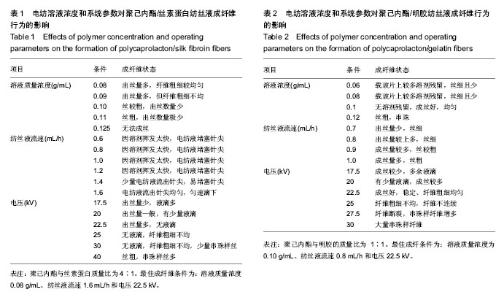
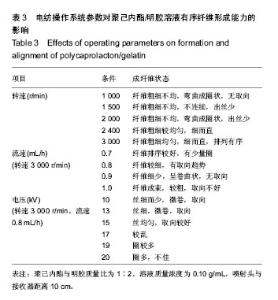
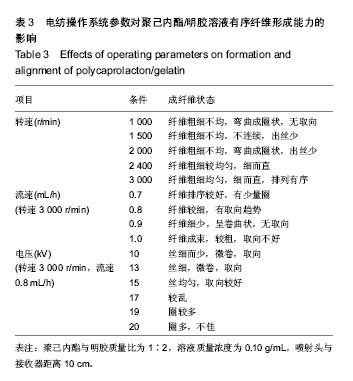
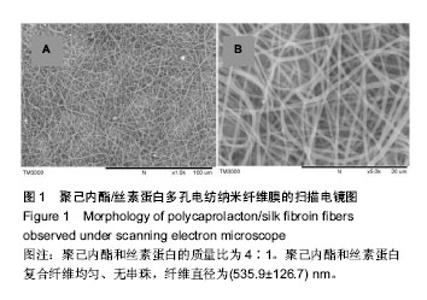
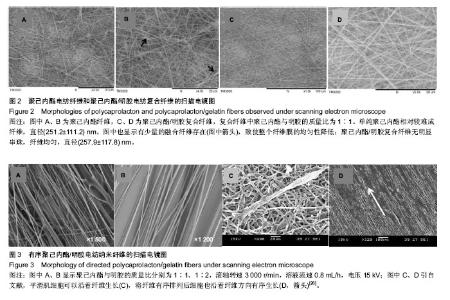
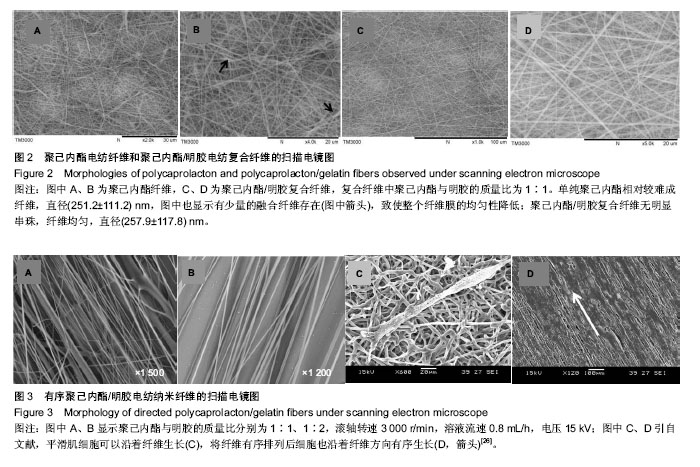
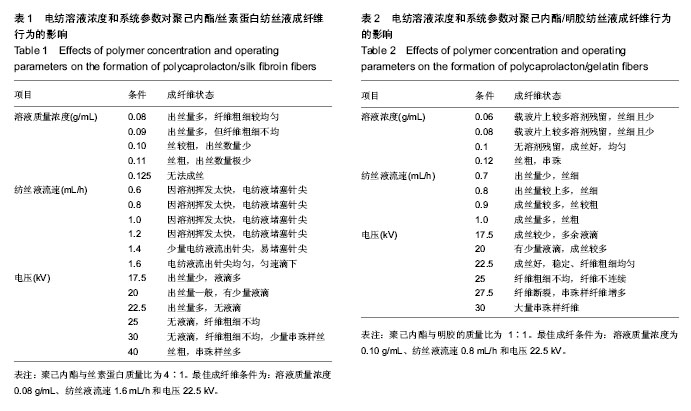
运用电纺丝技术可以制备纤维直径介于5 nm至5 µm、多种微观结构、高孔隙率的膜片状或管状支架,并依据目标支架的结构性能要求进行设计和调整,使这一技术在生物工程领域如制备仿生化和功能化的组织工程支架方面得到了广泛研究和应用[23-24]。根据食管组织的结构特点,作者分别设计了无规和有序排列的纳米级多孔纤维,为构建食管中的功能成分——黏膜层和肌层提供支架。理论上,聚合物的溶液性质,即纺丝液性质、电纺丝系统中所采用的电压、喷射速度或流量、喷射口与收集板距离,以及接收滚轴转速等都会对最后制备的纤维膜结构产生影响。在前期工作的基础上,选用具有良好生物相容性并已被美国FDA论证可用于体内的聚己内酯,以及价格低廉、原料易得、性能良好的明胶和丝素蛋白为基材,运用本实验室自行研制的电纺丝系统和技术,探索了溶液性质和系统参数对聚合物成纤性能的影响,以制备符合食管组织工程需要的仿生支架。 2.1 聚己内酯/丝素蛋白复合纤维 浙江省是蚕丝之乡,蚕丝来源丰富易得。从蚕丝或蚕茧中提取得到的丝素蛋白是一种天然结构性蛋白,具有高度重复的丙氨酸-甘氨酸-丝氨酸序列,具有其他高分子材料所不可比拟的优点,如无毒、无刺激、无污染、独特的机械性能及优良的透气透湿性和生物相容性,对机体细胞黏附力强,支持组织细胞的生长与增殖[25-27],实验选它作为基材是非常有优势的。根据前期关于聚己内酯与丝素蛋白混合电纺的实验,认为在两者4∶1的比例时制备的电纺丝溶液成纤维性能良好[28],因此本实验仍采用此比例探讨系统参数对纺丝液成纤行为的影响,见表1所示。在溶液质量浓度0.8-0.125 g/mL、供给速率(纺丝液流速)0.6-1.6 mL/h和电压17-30 kV的探索范围内,得到最佳成纤维条件为:溶液质量浓度0.08 g/mL、纺丝液流速1.6 mL/h和电压 22.5 kV的条件下,制得了均匀、无串珠、纤维直径为(535.9±126.7) nm的多孔纳米纤维膜(图1)。 2.2 聚己内酯/明胶复合纤维 明胶虽是变性的胶原,但它仍然维持了胶原蛋白所具有的优良生物和组织相容性,而且价格更低廉,原料易得,已在生物材料领域得到广泛应用。作者曾对明胶促进血管内皮细胞、食管上皮细胞、成纤维细胞和平滑肌细胞生长的情况进行了研究并得到正向结果,但它的缺点是成纤维能力较差[29-31]。另一方面,聚己内酯是生物可降解的合成聚合物,其良好的生物相容性使其在组织工程领域中得到了广泛关注和应用,将明胶和聚己内酯混合明显提高了二者的成纤维能力,改变了各自都不容易被电纺的事实:在溶液质量浓度为0.10 g/mL的条件下,聚己内酯与明胶质量比为2∶1质量比时有少量丝形成,粗细不均;在1∶1、1∶2时成丝量多。明胶的加入提高了纺丝液的成纤维能力,如聚己内酯与明胶质量比为1∶1时,溶液浓度和操作参数对成纤维行为的影响见表2所示。在溶液质量浓度 0.06-0.12 g/mL、纺丝液流速0.7-1.0 mL/h、电压17-30 kV的探索范围内,得到最佳成纤维条件为:溶液质量浓度为 0.10 g/mL、纺丝液流速0.8 mL/h和电压22.5 kV的条件下,制得了无明显串珠、纤维直径为(257.9±117.8) nm的多孔纳米纤维膜(图2),其扫描电镜下的形貌见图2C、D所示。而单纯聚己内酯(图2A、B)相对较难成纤维,图中也显示有少量 的融合纤维存在(图中箭头),致使整个纤维膜的均匀性降低。在相似条件下,聚己内酯与明胶在1∶2时更易成纤维,纤维直径为(255.5±90.6) nm,纤维尺寸更均匀(标准偏差更小)。 2.3 有序聚己内酯/明胶复合纤维 根据上述聚己内酯/明胶成纤能力及纤维性能的探索结果,考虑到明胶具有良好的细胞和组织相容性,以聚己内酯/明胶(1∶2)的纺丝溶液制备取向的多孔纤维支架。将平板接收器改为滚轴接收器,研究了系统参数如滚轴转速、流速和电压对纤维有序性的影响,列于表3。此时喷射头与接收板距离由原来的20 cm改为10 cm,缩短纤维从喷射头行至接收板的距离其目的是为了减少纤维发生卷曲的概率,使纤维的取向能力提高。 从表3中的参数可见,滚轴转速越高纤维的取向性更好,这是合理的,因为旋转的接收器对喷射头中射出的纤维产生一种惯性拉力,转速越高拉力越大,随着滚轴以合适的步进前移,纤维在滚轴表面可以一条接一条有序的排列。相对于平板接收,滚轴接收所要求的电场压力较低,这也是由于旋转给予了纤维移动的动力,只需要较低的压力即可使射流从喷射头到达接收器。图3显示了纤维的形貌,在相同的操作参数下,聚己内酯∶明胶(1∶2)(图3B)比聚己内酯∶明胶(1∶1)的纤维排序更理想,纤维也更均匀。在前期实验中发现纤维可以引导平滑肌细胞沿着纤维生长(图3C)[21],因此,本实验所得的有序纤维膜也有望使食管平滑肌细胞沿着取向的纤维有序生长,这将对维持平滑肌的功能是非常有利的。"
| [1] D'Journo XB,Martin J,Fermro P,et al.The esophageal remnant after gastric interposition.Dis Esophagus.2008;1:377-388. [2] Ferahkose Z,Bedirli A,Kerem M,et al.Comparison of free jejunal graft with gastric pull-up reconstruction after resection of hypopharyngeal and cervical esophageal carcinoma.Dis Esophagus.2008;21:340-345. [3] Doki Y, Okada K, Miyata H,et al.Long-term and short-term evaluation of esophageal reconstruction using the colon or the jejunum in esophageal cancer patients after gastrectomy. Dis Esophagus.2008;21(2):132-138. [4] Londono R,Jobe BA,Hoppo T,et al.Esophagus and regenerative medicine.World J Gastroenterol. 2012;18(47): 6894-6899. [5] 谢远财,张本固,彭品贤,等.应用钛镍合金-硅橡胶组合式人工食管重建食管缺损[J].中国组织工程研究与临床康复, 2009, 13(34):6675-6679. [6] Takimoto Y,Nakamura T,Yamamoto Y,et al.The experimental replacement of a cervical esophageal segment with an artificial prosthesis with the use of collagen matrix and a silicone stent.J Thorac Cardiovasc Surg.1998;116:98-106. [7] 谭波,魏人前,杨志明,等.口腔黏膜上皮细胞与猪小肠黏膜下层体外复合培养的实验研究[J].华西口腔医学杂志,2010,28(1): 76-80. [8] Barnes CA,Brison J,Michel R,et al.The surface molecular functionality of decellularized extracellular matrices. Biomaterials.2011;32(1):137-143. [9] Wolf MT, Daly KA,Brennan-Pierce EP,et al.A hydrogel derived from decellularized dermal extracellular matrix. Biomaterials. 2012;33(29):7028-7038. [10] Badylak SF,Weiss DJ,Caplan A,et al.Engineered whole organs and complex tissues.Lancet.2012;379(9819):943-952. [11] Nieponice A,Ciotola F,NachmanF,et al.Patch Esophagoplasty: Esophageal Reconstruction Using Biologic Scaffolds.Ann Thorac Surg.2014;97(1):283-288. [12] Urita Y,Komuro H,Chen G. Regeneration of the esophagus using gastric acellular matrix: An experimental study in a rat mode1. Pediatr Surg Int. 2007;23:21-26. [13] 谭波,魏人前,杨志明,等.食管黏膜上皮细胞与SIS复合培养及生物学特性研究[J].中国修复重建外科杂志,2008,22(6):742-746. [14] Saxena AK,Komann C,Ainoedhofer H,et al.Esophagus tissue engineering: hybrid approach with esophageal epithelium and unidirectional smooth muscle tissue component generation in vitro. J Gastrointest Surg.2009;13:1037-1043. [15] 谭波解慧琪.组织工程在食管修复重建外科中的应用[J].生物物理学报,2011,27(6):475-482. [16] Lei YN,Zhu YB,Gong CF,et al.Synthesis, characterization and cytocompatibility of a degradable polymer using ferric catalyst for esophageal tissue engineering.J Mater Sci Mater Med. 2014;25(2):273-282. [17] Zhu Y,Leong MF,Ong WF,etal.Esophageal epithelium regeneration on fibronectin grafted poly (L-lactide-co-caprolactone) (PLLC) nanofiber scaffold. Biomaterials. 2007;28(5):861-868. [18] Gong CF,Hou L,Zhu YB,et al.In vitro constitution of esophageal muscle tissue with endocyclic and exo- longitudinal patterns.ACS Appl Mater Interfaces.2013;5(14): 6549-6555. [19] Tan JY,Chua CK,Leong KF,et al.Esophageal tissue engineering: an in-depth review on scaffold design.Biotech Bioeng.2011;109:1-15. [20] Li YY,Zhu YB,Yu HW,et al.Topographic characterization and protein quantification of esophageal basement membrane for scaffold design reference in tissue engineering.J Biomed Mater Res Part B: Appl Biomater.2012;100B:265-273. [21] Zhu YB,Cao Y,Pan J,et al.Macro-alignment of electrospun fibers for vascular tissue engineering.J Biomed Mater Res. 2010;92B:508-516. [22] 陈玲,竺亚斌,李媛媛,等.丝素蛋白在电纺丝法构建组织工程支架中的应用进展[J].生物工程学报,2011,27(6):831−837. [23] Pham QP,Sharma U,Mikos AG.Electrospinning of polymeric nanofibers for tissue engineering applications: a review. Tissue Eng.2006;12:1197-1211. [24] Lannuttia J,Renekerb D,Mac T,et al.Electrospinning for tissue engineering scaffolds. Mater SciEngC. 2007;27(3):504-509. [25] Shao ZZ, Vollrath F, et al. Materials: Surprising strength of silkworm silk. Nature. 2002;418:741. [26] Altman GH,Diaz F,Jakuba C,et al.Silk-based biomaterials. Biomaterials.2003;24(3):401-416. [27] Park SY,Ki CS,Park YH,et al.Electrospun Silk Fibroin Scaffolds with Macropores for Bone Regeneration: An In Vitro and In Vivo Study.Tissue Eng A.2010;16(4):1271-1279. [28] 陈玲,吕静静,於学婵,等.静电纺丝法构建食道上皮组织[J].生物工程学报, 2013, 29(12): 1796-1807. [29] Zhu YB,Gao CY,Shen JC.Surface modification of polycaprolactone with poly(methacrylic acid) and gelatin covalent immobilization for promoting its cytocompatibility. Biomaterials.2002;23 (24):4889-4895. [30] Zhu YB,Gao CY,He T,et al.Endothelium regeneration on luminal surface of polyurethane vascular scaffold modified with diamine and covalently grafted with gelatin. Biomaterials. 2004;25(3):423-430. [31] Shen QX,Shi PN,Gao MN,et al.Progress on materials and scaffold fabrications applied to esophageal tissue engineering. MaterSci Eng C.2013;33(4):1860-1866. [32] LvJ,ChenL,ZhuY, et al.Promoting Epithelium Regeneration for Esophageal Tissue Engineering through Basement Membrane Reconstitution.ACS Appl Mater Interfaces. 2014; 6(7):4954-4964. [33] Shen ZS,KangC,Chen JJ,et al.Surface modification of polyurethane towards promoting the ex vivo cytocompatibility and in vivo biocompatibility for hypopharyngeal tissue engineering. J Biomater Appl.2013;28(4):607-616. |
| [1] | Li Xiaoyin, Yang Xiaoqing, Chen Shulian, Li Zhengchao, Wang Ziqi, Song Zhen, Zhu Daren, Chen Xuyi. Collagen/silk fibroin scaffold combined with neural stem cells in the treatment of traumatic spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 890-896. |
| [2] | Sun Kexin, Zeng Jinshi, Li Jia, Jiang Haiyue, Liu Xia. Mechanical stimulation enhances matrix formation of three-dimensional bioprinted cartilage constructs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(21): 3293-3299. |
| [3] | Ye Xuwen, Gu Yong, Chen Liang. Curcumin loaded injectable microspheres retard progression of intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1884-1891. |
| [4] | Zhu Biwen, Wang Dongzhi, Wu Di, Gong Tiancheng, Pan Haopeng, Lu Yuhua, Guo Yibing, Wang Zhiwei, Huang Yan. Biomimetic microenvironment constructed from gelatin methacrylamide/platelet-rich plasma hydrogel promotes the function of insulinoma cell line MIN6 in mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1824-1831. |
| [5] | Tan Guozhong, Tu Xinran, Guo Liyang, Zhong Jialin, Zhang Yang, Jiang Qianzhou. Biosafety evaluation of three-dimensional printed gelatin/sodium alginate/58S bioactive glass scaffolds for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 521-527. |
| [6] | He Guanyu, Xu Baoshan, Du Lilong, Zhang Tongxing, Huo Zhenxin, Shen Li. Biomimetic orientated microchannel annulus fibrosus scaffold constructed by silk fibroin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 560-566. |
| [7] | Kang Kunlong, Wang Xintao. Research hotspot of biological scaffold materials promoting osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 597-603. |
| [8] | Lu Dongdong, Zhu Tianfeng, Zhang Yijian, Zhao Zhijian, Liu Yang, Shen Xu, Zhu Xuesong. 3D bio-printing methylacrylated gelatin hydrogel scaffolds promote the repair of subchondral bone defects [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(34): 5454-5460. |
| [9] | Jiang Haoran, Gao Jianming, Lin Wancheng, Li Ting, Li Huo, Wang Peng, Feng Jing, Meng Haoye, Peng Jiang, Ding Lixiang. Gelatin-alginate composite microspheres and gels for cartilage damage repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(28): 4452-4457. |
| [10] | Liu Gang, Gao Lilan, Shi Feifei, Wang Shixin, Luo Lailong, Li Ruixin, Zhang Chunqiu. Preparation and mechanical properties of collagen type II-silk fibroin-hyaluronic acid composite scaffold [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(28): 4519-4524. |
| [11] | Jiang Chaorui, Xu Yan, Xiong Ying, Zhang Xujing. Preparation and properties of graphene oxide/silk fibroin/rifampicin drug-loading microspheres [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(22): 3467-3473. |
| [12] | Liu Yue, Jiang Ziyi, Li Jingjing, Meng Kai, Zhao Huijing. Cell co-culture and in vivo biocompatibility of poly(L-lactic caprolactone)/silk fibroin small-diameter artificial blood vessels [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(22): 3505-3513. |
| [13] | Meng Lulu, Liu Hao, Liu Han, Zhang Jun, Li Ruixin, Gao Lilan. Mechanical properties of silk fibroin/type I collagen/hydroxyapatite scaffolds based on low-temperature 3D printing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(22): 3550-3555. |
| [14] | Guo Xiaopeng, Liu Yingsong, Shang Hui. Silk fibroin/nano hydroxyapatite composite combined with icariin can promote the proliferation and differentiation of bone marrow mesenchymal stem cells into nucleus pulposus like cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(22): 3528-3534. |
| [15] | Li Xiuya, Sheng Yang, Wang Shengyi, Li Yue, Zhang Ruzhi, Pan Yan, Sun Yixin, Deng Linhong, Mark Bradley, Zhang Rong. Preparation of protein chips for screening matrix for Muse cell culture based on fibrinogen and gelatin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(21): 3312-3318. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||