Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (12): 1824-1831.doi: 10.12307/2023.082
Previous Articles Next Articles
Biomimetic microenvironment constructed from gelatin methacrylamide/platelet-rich plasma hydrogel promotes the function of insulinoma cell line MIN6 in mice
Zhu Biwen1, 2, 3, Wang Dongzhi1, 2, 3, Wu Di1, 2, 3, Gong Tiancheng2, 3, Pan Haopeng2, Lu Yuhua1, 2, 3, Guo Yibing2, 3, Wang Zhiwei1, 2, 3, Huang Yan1, 2, 3
- 1First Department of Hepatic and Splenic Surgery, 2Research Center of Clinical Medicine, Affiliated Hospital of Nantong University, Nantong 226001, Jiangsu Province, China; 3Medical School of Nantong University, Nantong 226001, Jiangsu Province, China
-
Received:2021-12-24Accepted:2022-02-18Online:2023-04-28Published:2022-07-30 -
Contact:Huang Yan, MD, Attending physician, First Department of Hepatic and Splenic Surgery, and Research Center of Clinical Medicine, Affiliated Hospital of Nantong University, Nantong 226001, Jiangsu Province, China; Medical School of Nantong University, Nantong 226001, Jiangsu Province, China Wang Zhiwei, Professor, Chief physician, Doctoral supervisor, First Department of Hepatic and Splenic Surgery, and Research Center of Clinical Medicine, Affiliated Hospital of Nantong University, Nantong 226001, Jiangsu Province, China; Medical School of Nantong University, Nantong 226001, Jiangsu Province, China -
About author:Zhu Biwen, Master candidate, First Department of Hepatic and Splenic Surgery, and Research Center of Clinical Medicine, Affiliated Hospital of Nantong University, Nantong 226001, Jiangsu Province, China; Medical School of Nantong University, Nantong 226001, Jiangsu Province, China -
Supported by:the National Natural Science Foundation of China, No. 82001977 (to HY); the Natural Science Foundation of Jiangsu Province, No. BK20201445 (to HY)
CLC Number:
Cite this article
Zhu Biwen, Wang Dongzhi, Wu Di, Gong Tiancheng, Pan Haopeng, Lu Yuhua, Guo Yibing, Wang Zhiwei, Huang Yan. Biomimetic microenvironment constructed from gelatin methacrylamide/platelet-rich plasma hydrogel promotes the function of insulinoma cell line MIN6 in mice[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1824-1831.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
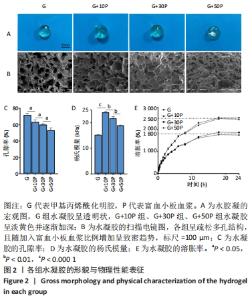
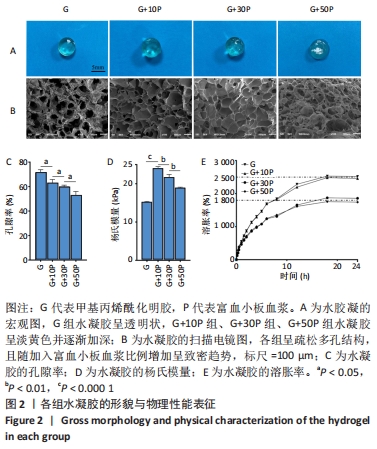
2.1 各组水凝胶的形貌 纯甲基丙烯酰化明胶水凝胶呈透明状,随着富血小板血浆比例的增加,复合成分水凝胶呈淡黄色并逐渐加深,见图2A;扫描电镜显示各组呈疏松多孔结构,且随加入富血小板血浆比例增加呈致密趋势,G组、G+10P组、G+30P组及G+50P组水凝胶孔径大小分别为(172.1±25.7),(152.5±12.6),(135.2±33.5),(84.4±31.7) μm,见图2B。G组水凝胶孔隙率为(71.5±2.3)%,随加入富血小板血浆比例升高,复合成分水凝胶孔隙率呈下降趋势,G+10P组、G+30P组及G+50P组水凝胶孔隙率分别为(63.3±2.8)%,(60.6±1.6)%及(53.5±3.1)%,见图2C,与扫描电镜观察结果一致。 2.2 各组水凝胶的杨氏模量与溶胀性能 G组、G+10P组、G+30P组、G+50P组水凝胶的杨氏模量分别为(15.2±0.1),(24.0±0.5),(21.7±0.8),(19.0±0.2) kPa,复合水凝胶的杨氏模量均大于单一成分水凝胶(P < 0.05),随着加入富血小板血浆比例的增加,复合水凝胶的杨氏模量降低,见图2D。各组水凝胶具有良好的吸水性能,G组、G+10P组、G+30P组、G+50P组水凝胶的溶胀率分别为(2 473.7±213.7)%,(2 507.7±296.6)%,(1 866.0±37.7)%,(1 758.3±134.1)%,随着富血小板血浆比例的升高,复合水凝胶的溶胀率呈下降趋势(P < 0.05),各组均在18 h后达溶胀平衡,见图2E。"
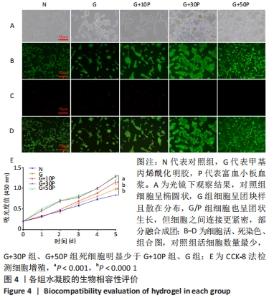
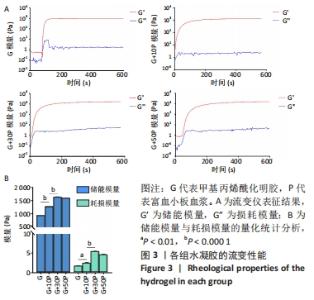
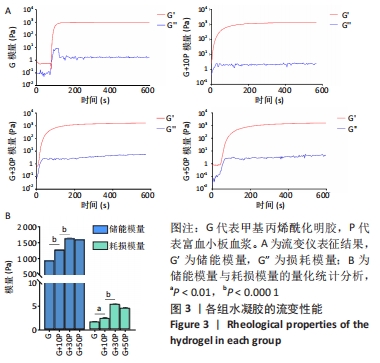
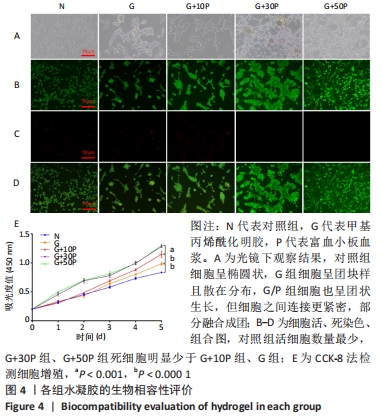
各组水凝胶展现出良好的流变行为,G组、G+10P组、G+30P组、G+50P组水凝胶的储能模量分别为(943.2±0.2),(1 282.2±0.4),(1 644.6±2.4),(1 605.6±2.2) Pa,损耗模量分别为(1.7±0.1),(2.4±0.2),(5.4±0.2),(4.6±0.2) Pa,各组水凝胶的储能模量G’均远高于损耗模量G”(P < 0.001);并且随加入富血小板血浆比例的增加,水凝胶的储能模量和损耗模量均呈先上升后降低的趋势。 2.4 各组水凝胶的生物相容性 光镜下可见,对照组细胞呈椭圆状,G组细胞呈团块样且散在分布,G/P组细胞也呈团状生长,但细胞之间连接更紧密,部分融合成团,见图4A。细胞活死染色显示,对照组活细胞数量最少,G+30P组、G+50P组死细胞明显少于G+10P组、G组和对照组,见图4B-D。细胞增殖性量化分析曲线显示,各组细胞数量都在随着时间的推移而增加,G+30P组、G+50P组细胞增殖能力相近且增殖吸光度值始终大于G+10P组、G组和对照组(P < 0.05);前3 d时,对照组、G组和G+10P组增殖吸光度值相近;第5天时,细胞增殖吸光度值大小为:G+10P组>G组>对照组(P < 0.05),见图4E。以上结果说明,单一成分与复合成分水凝胶均具有良好的生物相容性。 "
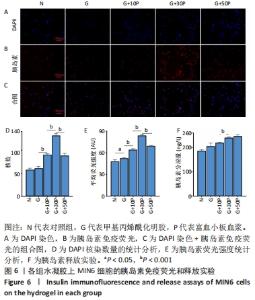
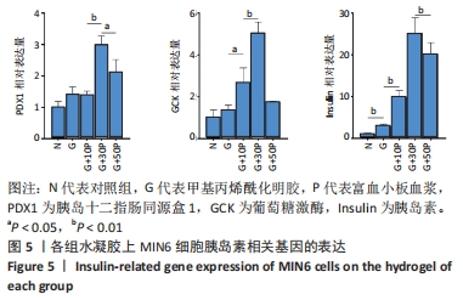
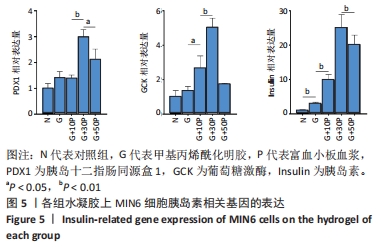
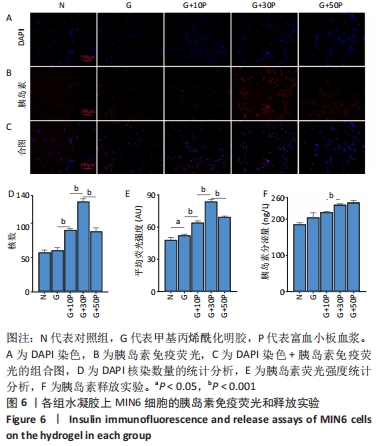
各组水凝胶上MIN6细胞的DAPI染色、胰岛素免疫荧光及组合图,见图6A-C。DAPI染色统计结果显示,G/P组核数量多于G组、对照组(P < 0.001),G+30P组核数量多于G+10P组、G+50P组(P < 0.001),见图6D。胰岛素免疫荧光统计结果显示,G组平均荧光强度大于对照组(P < 0.05),G/P组平均荧光强度大于G组(P < 0.05),G+30P组平均荧光强度最大(P < 0.001),见图6E。胰岛素释放实验显示,对照组、G组、G+10P组、G+30P组、G+50P组释放胰岛素量分别为(186.5±0.4),(205.6±1.2),(220.1±0.2),(240.6±0.2),(246.6±0.5) ng/L,胰岛素释放量随加入富血小板血浆比例的升高呈增加趋势,G+30P组释放量高于G+10P组(P < 0.05),见图6F。 "
| [1] VANTYGHEM MC, DE KONING EJP, PATTOU F, et al. Advances in β-cell replacement therapy for the treatment of type 1 diabetes. Lancet. 2019;394(10205):1274-1285. [2] SCHASCHKOW A, SIGRIST S, MURA C, et al. Glycaemic control in diabetic rats treated with islet transplantation using plasma combined with hydroxypropylmethyl cellulose hydrogel. Acta Biomater. 2020; 102(259-272. [3] SHAPIRO AM, POKRYWCZYNSKA M, RICORDI C. Clinical pancreatic islet transplantation. Nat Rev Endocrinol. 2017;13(5):268-277. [4] LLACUA LA, FAAS MM, DE VOS P. Extracellular matrix molecules and their potential contribution to the function of transplanted pancreatic islets. Diabetologia. 2018;61(6):1261-1272. [5] SMINK AM, DE VOS P. Therapeutic Strategies for Modulating the Extracellular Matrix to Improve Pancreatic Islet Function and Survival After Transplantation. Curr Diab Rep. 2018;18(7):39. [6] ZHANG F, ZHENG L, CHENG S, et al. Comparison of the Interactions of Different Growth Factors and Glycosaminoglycans. Molecules. 2019; 24(18):3360. [7] LI D, ZHANG W, CHEN X, et al. Proteomic profiling of MIN6 cell-derived exosomes. J Proteomics. 2020;224:103841. [8] YIN H, YAN H, QIN C, et al. Protective effect of fermented Diospyros lotus L. extracts against the high glucose-induced apoptosis of MIN6 cells. J Food Biochem. 2021;45(4):e13685. [9] HU S, KUWABARA R, DE HAAN B, et al. Acetate and Butyrate Improve β-cell Metabolism and Mitochondrial Respiration under Oxidative Stress. Int J Mol Sci. 2020;21(4):1542. [10] BEENKEN-ROTHKOPF L, KARFELD-SULZER L, DAVIS N, et al. The incorporation of extracellular matrix proteins in protein polymer hydrogels to improve encapsulated beta-cell function. Ann Clin Lab Sci. 2013;43(2):111-121. [11] LIM D, ANTIPENKO S, VINES J, et al. Improved MIN6 β-cell function on self-assembled peptide amphiphile nanomatrix inscribed with extracellular matrix-derived cell adhesive ligands. Macromol Biosci. 2013;13(10):1404-1412. [12] 潘钰.富血小板血浆治疗安全,有效[J].江苏卫生保健,2019(4):1. [13] 吕敏,裴国献,刘勇,等.富血小板血浆的制备现状及研究进展[J].现代生物医学进展,2013,13(27):5. [14] ANITUA E, PRADO R, ORIVE G. Allogeneic Platelet-Rich Plasma: At the Dawn of an Off-the-Shelf Therapy? Trends Biotechnol. 2017;35(2):91-93. [15] BARIA M, VASILEFF W, MILLER M, et al. Cellular Components and Growth Factor Content of Platelet-Rich Plasma With a Customizable Commercial System. Am J Sports Med. 2019;47(5):1216-1222. [16] OUDELAAR B, PEERBOOMS J, HUIS IN ‘T VELD R , et al. Concentrations of Blood Components in Commercial Platelet-Rich Plasma Separation Systems: A Review of the Literature. Am J Sports Med. 2019;47(2): 479-487. [17] EPPLEY B, WOODELL J, HIGGINS JJP, et al. Platelet quantification and growth factor analysis from platelet-rich plasma: implications for wound healing. Plast Reconstr Surg. 2004;114(6):1502-1508. [18] PULCINI S, MEROLLE L, MARRACCINI C, et al. Apheresis Platelet Rich-Plasma for Regenerative Medicine: An In Vitro Study on Osteogenic Potential. Int J Mol Sci. 2021;22(16):8764. [19] WEI S, XU P, YAO Z, et al. A composite hydrogel with co-delivery of antimicrobial peptides and platelet-rich plasma to enhance healing of infected wounds in diabetes. Acta Biomater. 2021;124:205-218. [20] SAMBERG M, STONE R, NATESAN S, et al. Platelet rich plasma hydrogels promote in vitro and in vivo angiogenic potential of adipose-derived stem cells. Acta Biomater. 2019;87:76-87. [21] DE ANGELIS B, D’AUTILIO M, ORLANDI F, et al. Wound Healing: In Vitro and In Vivo Evaluation of a Bio-Functionalized Scaffold Based on Hyaluronic Acid and Platelet-Rich Plasma in Chronic Ulcers. J Clin Med. 2019;8(9):1486. [22] BELLIS SJB. Advantages of RGD peptides for directing cell association with biomaterials. Biomaterials. 2011;32(18):4205-4210. [23] ROSE J, PACELLI S, HAJ A, et al. Gelatin-Based Materials in Ocular Tissue Engineering. Materials(Basel). 2014;7(4):3106-3135. [24] YUE K, TRUJILLO-DE SANTIAGO G, ALVAREZ MM, et al. Synthesis, properties, and biomedical applications of gelatin methacryloyl (GelMA) hydrogels. Biomaterials. 2015;73:254-271. [25] SHIN SR, ZIHLMANN C, AKBARI M, et al. Reduced Graphene Oxide-GelMA Hybrid Hydrogels as Scaffolds for Cardiac Tissue Engineering. Small. 2016;12(27):3677-3689. [26] MODARESIFAR K, HADJIZADEH A, NIKNEJAD HJAC, et al. Design and fabrication of GelMA/chitosan nanoparticles composite hydrogel for angiogenic growth factor delivery. Artif Cells Nanomed Biotechnol. 2018;46(8):1799-1808. [27] WU S, WANG L, FANG Y, et al. Advances in Encapsulation and Delivery Strategies for Islet Transplantation. Adv Healthc Mater. 2021;10(20): e2100965. [28] TERAMURA Y, OOMMEN O, OLERUD J, et al. Microencapsulation of cells, including islets, within stable ultra-thin membranes of maleimide-conjugated PEG-lipid with multifunctional crosslinkers. Biomaterials. 2013;34(11):2683-2693. [29] URBANCZYK M, ZBINDEN A, LAYLAND S, et al. Controlled Heterotypic Pseudo-Islet Assembly of Human β-Cells and Human Umbilical Vein Endothelial Cells Using Magnetic Levitation. Tissue Eng Part. 2020;26: 387-399. [30] ZHU Q, LU C, JIANG X, et al. Using Recombinant Human Collagen With Basic Fibroblast Growth Factor to Provide a Simulated Extracellular Matrix Microenvironment for the Revascularization and Attachment of Islets to the Transplantation Region. Front Pharmacol. 2019;10:1536. [31] ZHONG Z, BALYAN A, TIAN J, et al. Bioprinting of dual ECM scaffolds encapsulating limbal stem/progenitor cells in active and quiescent statuses. Biofabrication. 2021;13(4):044101. [32] JIN R, CUI Y, CHEN H, et al. Three-dimensional bioprinting of a full-thickness functional skin model using acellular dermal matrix and gelatin methacrylamide bioink. Acta Biomater. 2021;131:248-261. [33] VAN DEN BULCKE A, BOGDANOV B, DE ROOZE N, et al. Structural and rheological properties of methacrylamide modified gelatin hydrogels. Biomacromolecules. 2000;1(1):31-38. [34] SUN X, MA Z, ZHAO X, et al. Three-dimensional bioprinting of multicell-laden scaffolds containing bone morphogenic protein-4 for promoting M2 macrophage polarization and accelerating bone defect repair in diabetes mellitus. Bioact Mater. 2020;6(3):757-769. [35] EVERTS P, ONISHI K, JAYARAM P, et al. Platelet-Rich Plasma: New Performance Understandings and Therapeutic Considerations in 2020. Int J Mol Sci. 2020;21(20):7794. [36] FADADU PP, MAZZOLA AJ, HUNTER CW, et al. Review of concentration yields in commercially available platelet-rich plasma (PRP) systems: a call for PRP standardization. Reg Anesth Pain Med. 2019 :rapm-2018-100356. [37] KLEIN M, KäMMERER P, SCHOLZ T, et al. Modulation of platelet activation and initial cytokine release by alloplastic bone substitute materials. Clin Oral Implants Res. 2010;21(3):336-345. [38] HUBER S, CUNHA JúNIOR J, MONTALVãO S, et al. In vitro study of the role of thrombin in platelet rich plasma (PRP) preparation: utility for gel formation and impact in growth factors release. J Stem Cells Regen Med. 2016;12(1):2-9. [39] GAO X, GAO L, GROTH T, et al. Fabrication and properties of an injectable sodium alginate/PRP composite hydrogel as a potential cell carrier for cartilage repair. J Biomed Mater Res A. 2019;107(9):2076-2087. [40] ENDERAMI SE, MORTAZAVI Y, SOLEIMANI M, et al. Generation of Insulin-Producing Cells From Human-Induced Pluripotent Stem Cells Using a Stepwise Differentiation Protocol Optimized With Platelet-Rich Plasma. J Cell Physiol. 2017;232(10):2878-2886. [41] ANITUA E, DE LA SEN-CORCUERA B, ORIVE G, et al. Progress in the use of plasma rich in growth factors in ophthalmology: from ocular surface to ocular fundus. Expert Opin Biol Ther. 2022;22(1):31-45. [42] BYNIGERI RR, MITNALA S, TALUKDAR R, et al. Pancreatic stellate cell-potentiated insulin secretion from Min6 cells is independent of interleukin 6-mediated pathway. J Cell Biochem. 2020;121(1):840-855. [43] FURUKAWA A, TANAKA A, YAMAGUCHI S, et al. Using recombinase-mediated cassette exchange to engineer MIN6 insulin-secreting cells based on a newly identified safe harbor locus. J Diabetes Investig. 2021;12(12):2129-2140. [44] SCHULTZ J, WARKUS J, WOLKE C, et al. MiD51 Is Important for Maintaining Mitochondrial Health in Pancreatic Islet and MIN6 Cells. Front Endocrinol (Lausanne). 2020;11:232. [45] BLESIA V, PATEL VB, AL-OBAIDI H, et al. Excessive Iron Induces Oxidative Stress Promoting Cellular Perturbations and Insulin Secretory Dysfunction in MIN6 Beta Cells. Cells. 2021;10(5):1141. [46] 石红梅,张英兰,侯小红,等.外观异常的血清标本对生化结果的影响[J].实用医技杂志,2003,10(12):1495-1495. [47] SIBORO S, ANUGRAH D, RAMESH K, et al. Tunable porosity of covalently crosslinked alginate-based hydrogels and its significance in drug release behavior. Carbohydr Polym. 2021;260:117779. [48] 姜铧,张洹,李东升,等.PDX1与糖尿病[J].湖北医药学院学报, 2008,27(1):81-84. [49] 邹大进.血糖稳态调控的核心酶:葡萄糖激酶及其家族[J].中华糖尿病杂志,2019,11(10):3. |
| [1] | Zheng Hongrui, Zhang Wenjie, Wang Yunhua, He Bin, Shen Yajun, Fan Lei. Femoral neck system combined with platelet-rich plasma in the treatment of femoral neck fracture [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1390-1395. |
| [2] | Sun Jiajia, Zhu Haidi, Lu Yun, Zhang Kai. Comparison of bone metabolism markers between type 2 diabetes mellitus and non-type 2 diabetes mellitus patients with hip fracture [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1156-1160. |
| [3] | Zhang Yan, He Ruibo, Wang Qingbo, Pi Yihua, Lu Chunmin, Xu Chuanyi, Ma Gang, Peng Peng. Effects of aerobic exercises with different load volumes on inflammatory response and insulin signaling pathway of skeletal muscle in obese rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1237-1244. |
| [4] | Lian Shilin, Zhang Yan, Jiang Qiang, Zhang Hanshuo, Li Tusheng, Ding Yu. Interventional effects of whole blood and platelet-rich plasma with different preparation methods on nucleus pulposus cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1199-1204. |
| [5] | Wang Ji, Zhang Min, Yang Zhongya, Zhang Long. A review of physical activity intervention in type 2 diabetes mellitus with sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1272-1277. |
| [6] | Liu Xiaolin, Mu Xinyue, Ma Ziyu, Liu Shutai, Wang Wenlong, Han Xiaoqian, Dong Zhiheng. Effect of hydrogel-loaded simvastatin microspheres on osteoblast proliferation and differentiation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 998-1003. |
| [7] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [8] | Li Yue, Lyu Yan, Feng Wanying, Song Yang, Yan Yu, Guan Yongge. Preparation of hyperoside nanoparticles to repair endometrial injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 360-366. |
| [9] | Ning Ziwen, Wang Xu, Shi Zhengliang, Qin Yihua, Wang Guoliang, Jia Di, Wang Yang, Li Yanlin. Meniscal injury repair methods for non-blood supply area [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 420-426. |
| [10] | Li Tusheng, Ding Yu, Jiang Qiang, Zhang Hanshuo. Immune complex expression and platelet-rich plasma repairing mechanism for lumbar discogenic neuralgia [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(18): 2935-2942. |
| [11] | Peng Zifu, Guo Xiangying, Fang Hongbo, He Yimin, Jiang Ning. Exercise intervention improves hippocampal cognitive function by regulating insulin-like growth factor 1 [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(17): 2729-2737. |
| [12] | Zhang Ziyu, Wang Yiming, Li Han, Wang Zhonghan, Lu Jialin, Xu Rui, Jin Hui. Platelet-rich plasma meets the therapeutic needs of different articular cartilage defects [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(17): 2772-2779. |
| [13] | Huang Shuangshuang, Xiang Xiaona, Yu Xi, He Jing. Mechanism and effect of platelet-rich plasma-derived exosomes for treating knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(15): 2420-2426. |
| [14] | Peng Kun. Properties of force growth factor E peptide bionic bone matrix with polyethylene glycol derivative hydrogel as a carrier [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1811-1816. |
| [15] | Ling Huajun, Cui Ruiwen, Wang Qiyou. 3D extracellular matrix hydrogel loaded with exosomes promotes wound repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1900-1905. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||