Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (18): 2935-2942.doi: 10.12307/2023.299
Previous Articles Next Articles
Immune complex expression and platelet-rich plasma repairing mechanism for lumbar discogenic neuralgia
Li Tusheng1, 2, Ding Yu1, 2, 3, Jiang Qiang3, Zhang Hanshuo3
- 1Second Clinical Medical School, Southern Medical University, Guangzhou 510515, Guangdong Province, China; 2Orthopedics Section of TCM Senior Department, Sixth Medical Center of PLA General Hospital, Second Clinical Medical School, Southern Medical University, Beijing 100048, China; 3Orthopedics Section of TCM Senior Department, Sixth Medical Center of PLA General Hospital, Beijing 100048, China
-
Received:2022-03-05Accepted:2022-05-19Online:2023-06-28Published:2022-09-19 -
Contact:Ding Yu, Professor, Chief physician, Doctoral supervisor, Second Clinical Medical School, Southern Medical University, Guangzhou 510515, Guangdong Province, China; Orthopedics Section of TCM Senior Department, Sixth Medical Center of PLA General Hospital, Second Clinical Medical School, Southern Medical University, Beijing 100048, China; Orthopedics Section of TCM Senior Department, Sixth Medical Center of PLA General Hospital, Beijing 100048, China -
About author:Li Tusheng, Master, Second Clinical Medical School, Southern Medical University, Guangzhou 510515, Guangdong Province, China; Orthopedics Section of TCM Senior Department, Sixth Medical Center of PLA General Hospital, Second Clinical Medical School, Southern Medical University, Beijing 100048, China -
Supported by:Research and Demonstration Application of Clinical Diagnosis and Treatment Technology in Beijing, No. Z191100006619028 (to DY)
CLC Number:
Cite this article
Li Tusheng, Ding Yu, Jiang Qiang, Zhang Hanshuo. Immune complex expression and platelet-rich plasma repairing mechanism for lumbar discogenic neuralgia[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(18): 2935-2942.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

2.1 LDN的免疫致病机制及免疫表达研究进展 2.1.1 椎间盘自身免疫特性及退变分析 腰椎作为受力最为集中、活动范围最大的脊柱节段,易发生腰椎退行性改变,其中椎间盘又是变形最大的弹性结构,传导椎体间应力并完成前屈后伸、侧弯旋转等活动,因而极易发生椎间盘退变而引起腰椎疾病[8]。正常来说,一个健康的椎间盘是人体中最大的无血管器官,由终板、纤维环与髓核组成,髓核主要由水、蛋白聚糖、Ⅱ型胶原组成,因其含水量丰富而起着缓冲应力的作用,而位于椎间盘周边部的纤维环则包裹着内在的髓核使之与宿主的免疫血液系统隔离开,构成了血液-髓核屏障,使髓核组织能够成为自身隐蔽抗原[4]。当椎间盘发生退变时,髓核细胞合成、分解代谢失衡,蛋白聚糖和Ⅱ型胶原合成减少而降解增加,Ⅰ型胶原合成增加[9-10];同时,髓核组织含水量不断下降,髓核组织逐渐脱水并纤维化,导致椎间盘受力不均及纤维环理化性质发生改变,纤维环弹性减弱,易出现纤维环松弛甚至破裂的现象[11]。 2.1.2 LDN的免疫反应 当退变或创伤发生造成纤维环破裂时,在压力的作用下髓核组织易从破裂的纤维环突出,当作为自身抗原的髓核与血液系统接触,将会刺激免疫系统生成相关致敏淋巴细胞和特异性抗体,通过抗原与特异性抗体、免疫细胞发生相互作用,从而导致机体产生细胞免疫反应和体液免疫反应[12]。髓核自身免疫反应是椎间盘突出导致神经根性疼痛的关键递质,当抗原抗体免疫复合物沉积于突出椎间盘时,在神经根可见各种类型的活化免疫细胞和炎症因子,免疫细胞包括巨噬细胞、NK细胞、肥大细胞、T细胞和B细胞等,炎症因子包括白三烯、磷脂酶A2、肿瘤坏死因子α、白细胞介素家族(如白细胞介素1β、白细胞介素6、白细胞介素8)、基质金属蛋白酶(如基质金属蛋白酶3、基质金属蛋白酶7)、NO、物质P等炎症因子,同时还浸润成纤维细胞生长因子、单核细胞化学吸引蛋白1、血管内皮生长因子及神经生长因子等[13-15],这些因素构成了一个复杂的区域,炎症因子可直接作用于神经的痛觉感受器,导致受损神经电生理变化,使其敏感化,在轻微的机械压力或牵拉刺激下即可导致神经病理性疼痛的发生。其中,肿瘤坏死因子α和白细胞介素1β是突出间盘组织中产生的最早炎性递质,减慢受损神经的传导速度的同时放大炎症级联反应[16],导致神经功能障碍及神经病理性疼痛,被认为是导致椎间盘退变的关键因素。肿瘤坏死因子α与白细胞介素1β导致LDN的机制:①加速细胞外基质的降解。肿瘤坏死因子α与白细胞介素1β可通过激活基质金属蛋白酶、解聚蛋白样金属蛋白酶(ADAMTS)等降解酶而促进基质降解[17]。②放大炎症级联反应。肿瘤坏死因子α、白细胞介素1β能促进白细胞介素6、白细胞介素17、物质P、NO等促炎物质分泌,同时还可加强CCL3、CCL5 等趋化因子表达[18-19]。③诱发椎间盘细胞凋亡。XIE等[20]发现,应用肿瘤坏死因子α、白细胞介素1处理椎间盘细胞后,细胞凋亡率明显上升。④肿瘤坏死因子α、白细胞介素1β可促进新生神经及血管长入退变的椎间盘组织,进而诱发疼痛[21]。 椎间盘微环境的免疫失衡,一方面是由于自身免疫反应导致自身抗体产生和细胞毒性T细胞活化而对椎间盘进行损害,另一方面是由于凋亡相关因子配体(FasL)表达降低削弱了髓核细胞对浸润的Fas阳性活化的细胞毒性T淋巴细胞的清除能力,FasL隶属肿瘤坏死因子家族,存在于正常髓核细胞中,一旦与其受体Fas结合,将可在短时间内杀死浸润含Fas的免疫细胞,从而维持椎间盘的免疫平衡,但FasL在退行性椎间盘中表达显著减少,无法保持免疫平衡而发生免疫炎症反应[22]。MALCANGIO等[23]研究发现,在慢性疼痛患者脑中神经胶质细胞被显著激活,表明免疫细胞可通过与神经元交流来传播和维持神经性疼痛。在免疫炎症反应致神经损伤后,神经元被激活使得单核细胞、巨噬细胞募集到神经损伤部位,激活小胶质细胞释放相关炎症递质,致使神经元敏感性提高[24],最终产生LDN。 正常神经受压时引起短暂的神经异常放电,由于放电时间太短只会导致感觉异常和麻木,而不会导致神经疼痛症状。MURPHY[25]认为无免疫炎症浸润的脊神经受压迫时并不会产生神经疼痛症状,但在有免疫炎症浸润时必然会引起神经疼痛。当椎间盘突出压迫脊神经时,脊髓主要组织相容性复合体Ⅱ类(MHCII)、抗原决定簇决定因子4、细胞间黏附分子1(intercellular cell adhesion molecule-1,ICAM-1)和血小板内皮细胞黏附分子1(Platelet endothelial cell adhesion molecule-1,PECAM-1)等免疫递质显著增加,同时,椎间盘内神经丝、S-100蛋白等神经胶质和神经损伤标记物水平显著升高[26]。综上所述,可知免疫炎症在神经病理性疼痛的产生及维持中有着重要作用。 2.1.3 椎间盘免疫表达研究进展 关于自身免疫学说在椎间盘退变的作用其实早在20世纪60年代已经被学者理论化了,并表明神经根性疼痛主要是由于突出的髓核组织诱发的相关化学反应和自身免疫反应引起[15]。BOBECHKO等[27]通过将髓核细胞植入兔耳研究支持髓核的自身抗原性并进一步支持椎间盘自身免疫作用。 1975年,NAYLAR等[28]首次提出了椎间盘自身免疫学说,指出椎间盘与人体血液系统隔离而具有自身抗原性,一旦纤维环出现破裂,髓核作为自身抗原将会被机体血液系统所识别,进而产生免疫反应,同时指出自身免疫影响椎间盘细胞的通透性和物质交换,导致椎间盘退变。1994年,SPILIOPOULOU等[29]研究发现,腰椎间盘突出症患者血清和脑脊液中IgG、IgM的含量显著增高,表明了椎间盘突出可引起免疫反应。1999年,SATOH等[30]对8例腰椎间盘突出症患者进行研究后发现,抗原-抗体免疫复合物广泛存在于突出的椎间盘组织中,表明突出的椎间盘存在免疫反应。2007年,GEISS等[31]研究发现,椎间盘自身抗原一旦与血液循环接触即可发生免疫应答,产生细胞免疫和体液免疫反应,从而导致椎间盘基质降解及椎间盘组织完整性结构的损伤。2013年,曹涌等[32]研究发现髓核作为自身抗原,可诱导Th细胞向Th17细胞分化并促进其分泌白细胞介素17,进而通过增加NK细胞的表达而加重椎间盘的突出及进展。2014年,CAPOSSELA等[33]在退化椎间盘样本中研究发现,突出的髓核细胞暴露于机体免疫系统,产生拮抗胶原纤维和蛋白聚糖的IgG抗体,表明了椎间盘的自身免疫反应。2014年,欧阳冰等[34]研究发现,相对于纤维环完整的椎间盘,破裂纤维环的椎间盘中白细胞介素6、白细胞介素10及单核细胞趋化蛋白1表达明显升高,认为这些炎症因子在椎间盘退变过程中起着重要作用,并且与椎间盘突出的程度有关。2015年,STICH等[35]研究发现,免疫细胞在3D培养物中的增殖速度高于在2D培养物中,并且退化严重的椎间盘细胞的免疫细胞反应呈上升趋势,表明自身免疫应答可能因不同的细胞培养物和椎间盘细胞退变程度而异。2016年,刘振刚等[36]在对70例椎间盘组织的研究中,通过免疫组化对CD68阳性巨噬细胞和CD25+T细胞进行检测,发现腰椎间盘突出症患者的髓核周围有明显自身免疫炎症反应。2017年,MONCHAUX等[37]研究发现,在狗的椎间盘模型中单核细胞和巨噬细胞广泛存在于突出的椎间盘组织中,表明了各种免疫细胞参与髓核的自身免疫反应。2018年,WANG等[38]研究发现,椎间盘退变患者的外周血血清中白细胞介素10和白细胞介素17表达明显升高。2019年,HASVIK等[39]研究发现,介导肿瘤坏死因子产生增加和巨噬细胞活性的循环microRNA-17表达上调与椎间盘突出后的腰神经根疼痛有关。2019年,SILVA等[40]通过在椎间盘培养模型中发现,人类巨噬细胞通过退化的椎间盘组织可以转化为更加促炎的表型,并通过白细胞介素1β而下调蛋白聚糖和Ⅱ胶原基因表达来干扰细胞外基质的重塑。2020年,LEE等[41]通过对穿刺小鼠椎间盘模型的组织学分析中发现,巨噬细胞大量浸润受伤的椎间盘并导致病理性神经支配。综上可知,暴露的椎间盘组织作为自身抗原可以诱导免疫炎症反应,刺激免疫细胞活化和炎症因子浸润,从而导致一系列的神经病理性疼痛。 LDN的免疫表达研究进展脉络图,见图4。 "

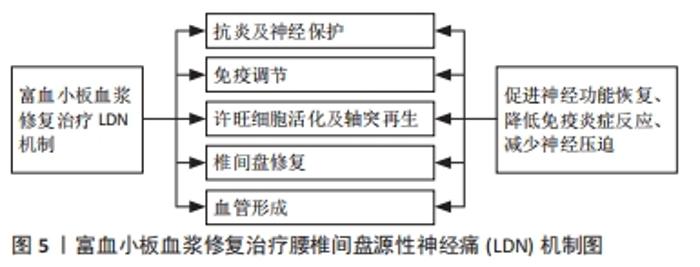
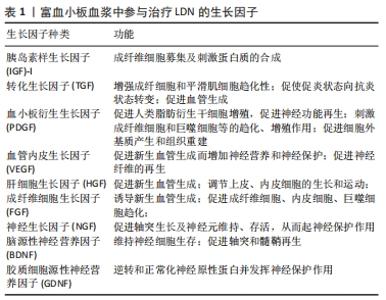
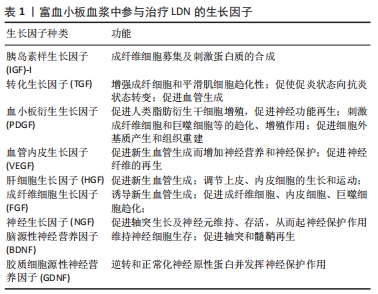
2.2.2 富血小板血浆的制备 起初,富血小板血浆仅用作纤维蛋白组织黏合剂,用于支持更强的纤维蛋白聚合以改善组织封闭。随着时间的发展,现在富血小板血浆已广泛应用于多个学科领域。目前,根据产生方式的不同,富血小板血浆的制备可分为密度梯度离心法和血浆置换法,其中前者因操作相对简单,并且可制备出足够有效浓度的富血小板血浆,一直受到众多学者的青睐。同时,根据离心次数可分为一次离心、二次离心、三次离心法,临床上主要应用的是Landesberg的二次离心法制备富血小板血浆。富血小板血浆制备大致可分为3步:①抽取适量患者的外周静脉血,将加入枸橼酸钠抗凝剂的全血进行初次离心后试管内的血液分为3层,上层为贫血小板血浆(PPP),中间为白膜层(富含大量白细胞和血小板),底层为红细胞层;②抽取中上层血液至新的试管,再次离心后吸弃管内上层 3/4上清液,将剩余液体摇匀即为未激活的富血小板血浆;③将未激活的富血小板血浆与激活剂按10∶1比例混合,待血凝块充分收缩凝固后,离心10 min吸取上清液即为活化的富血小板血浆。 2.3 富血小板血浆修复治疗LDN机制研究 目前,对于腰椎退行性疾病的有效治疗主要有保守及手术治疗,保守治疗包括非类固醇类抗炎治疗、物理康复治疗、硬膜外类固醇和射频消融术等,主要目的是降低疼痛,手术治疗包括脊柱微创、开放手术,如椎间盘摘除术、腰椎融合术等。但是,这些治疗仅能减轻症状,并不能从根本上减缓或逆转椎间盘退变进展[48]。近年来,随着对腰椎退行性疾病发病机制深入的研究,对于其治疗有一个重要的共识,那就是生物学疗法[49]。其中,富血小板血浆技术作为一种新型治疗技术,无免疫排斥等风险,同时具有广阔的临床应用前景,已经广泛应用于治疗LDN等腰椎疾病。富血小板血浆对于LDN的治疗主要基于其丰富的生长因子,在修复过程中主要涉及了抗炎及神经保护、免疫调节、促进许旺细胞活化及轴突再生、促进椎间盘退变的修复及血管形成等方面作用。见图5。"
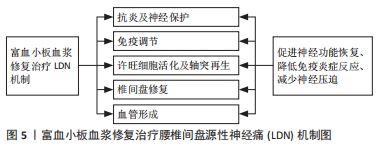

2.3.1 富血小板血浆抗炎及神经保护作用 髓核突出压迫神经可吸引中性粒细胞、肥大细胞和巨噬细胞等多种炎性细胞浸润,释放大量的免疫化学物质,从而使神经细胞敏感,减弱神经再生能力[46]。富血小板血浆中肝细胞生长因子(HGF)可通过对E-选择素表达的特异性抑制以及诱导抗炎因子生成而发挥抗炎作用[50]。同时,富血小板血浆能够促进受损组织从促炎转为抗炎状态,下调肿瘤坏死因子α、白细胞介素1β、环氧合酶2(COX-2)等促炎因子释放,上调可溶性肿瘤坏死因子受体(sTNF-R)、白细胞介素-受体拮抗剂(IL-RA)等抗炎细胞因子释放[51]。此外,富血小板血浆中胰岛素样生长因子1、肝细胞生长因子可通过阻止IκBα活化,对核因子κB级联及相关靶基因活化的抑制以降低瘤坏死因子α、白细胞介素1β基因的转录[52]。 富血小板血浆中脑源性神经营养因子(BDNF)、胶质细胞源性神经营养因子(GDNF)、转化生长因子β(TGF-β)、血管内皮生长因子(VEGF)、胰岛素样生长因子1(IGF-1)等生长因子对受损神经元有营养活性作用,促进神经细胞的增殖分化、成熟等生理变化。脑源性神经营养因子能够促进受损神经细胞的增殖分化,同时有效阻止神经细胞的炎症性死亡[53]。DEMBO等[54]研究发现,当敲除模型小鼠脑源性神经营养因子基因后小鼠不能存活,若条件性敲除感觉神经元的脑源性神经营养因子也会炎症影响其生长发育。胶质细胞源性神经营养因子来源于神经胶质细胞,参与神经元的功能维持和神经损伤的修复,具有神经保护和神经再生的双重神经营养功能[55]。在细胞凋亡过程中,Bcl-2家族成员起着至关重要的作用,转化生长因子β可通过Bcl-2及Bcl-xl的mRNA表达,显著降低凋亡相关基因caspase-3和bad的活性表达,并诱导纤溶酶原激活物抑制剂1(PAI-1)的表达,从而发挥保护神经细胞作用[56]。AYADI等[57]研究发现,胰岛素样生长因子1作为一种神经调质,在成年人的神经系统中参与突触可塑性、突触密度和神经传递等过程的调节,促进细胞外基质合成和神经元损伤的修复。TEYMUR等[58]研究发现,应用自体神经移植物联合富血小板血浆治疗受损大鼠坐骨神经时,发现富血小板血浆能促进大鼠坐骨神经功能恢复及有效保护损伤的神经纤维,减少神经纤维化和轴突退化。因此,富血小板血浆可通过下调炎症因子、促进神经损伤修复与功能维持等发挥抗炎及神经保护作用,从而有效缓解神经疼痛症状。 2.3.2 富血小板血浆的免疫调节作用 富血小板血浆作为血小板的浓缩物,富含大量生长因子及血小板等活性物质,对神经病理性疼痛起着免疫调节作用[46]。血小板和白细胞在先天和适应性免疫应答中有着至关重要的作用[59],血小板可通过表达并激活免疫刺激分子而将先天和适应性免疫应答连接起来,同时血小板及其本身释放的免疫炎症递质也可参与自身免疫性疾病的生理病理过程。 血小板主要通过与B细胞、T细胞等免疫细胞和抗原提呈细胞的相互作用来调节机体的适应性免疫应答。中性粒细胞、单核细胞和树突状细胞是最常见的先天免疫细胞,而淋巴细胞及其亚群则在适应性免疫系统中起着关键作用。树突状细胞作为淋巴抗原呈递细胞是启动T细胞免疫反应重要环节,具有摄取和处理抗原的能力,起着连接先天和适应性免疫系统的桥梁作用[60],主要表达主要组织相容性复合物(MHC)和共刺激分子(CD40、CD70和CD80等),从而激活细胞间的免疫应答反应[61-62]。 免疫炎症反应是导致髓核组织突出压迫神经后继发的神经病理性疼痛的关键因素,可见大量炎症因子及免疫细胞浸润[15]。血小板有着免疫抑制和抗炎作用,可通过对免疫调节因子、生长因子和营养因子等调节及分泌作用,降低免疫炎症反应对受损神经的损害[63],血小板可通过释放多种生长因子和黏附分子介导各种免疫细胞相互趋化、黏附等,刺激细胞增殖和促进神经营养因子的合成而调节免疫反应活性。活化血小板可通过跨膜蛋白CD40配体(CD40L)、血小板特异性Toll样受体、血小板因子4(PF4)、血小板糖蛋白GPⅡb/Ⅲa、CD63、P-选择素、趋化因子RANTES等免疫调节受体的表达,募集和激活免疫细胞[46,59,64],从而下调受损神经周围炎症因子表达,降低神经病理性疼痛症状。其中,血小板分泌的CD40L是适应性免疫应答的关键调节分子[46],CD40主要表达于树突状细胞、单核细胞及巨噬细胞表面,活化的血小板可通过表达CD40L结合并促进树突状细胞及单核细胞活化和成熟,进而促进抗原向抗原提呈细胞呈递,增强机体适应性免疫应答。P-选择素可通过P-选凝素糖蛋白配基1(P-selectin glycoprotein ligand-1,PSGL-1)与白细胞结合,进而促进血小板、内皮细胞及白细胞间的黏附[65]。GPⅡb/Ⅲa可通过介导血小板间及血小板与其他细胞间相互作用[66]。同时,血小板中三磷酸腺苷(ATP)、二磷酸腺苷(ADP)、5-羟色胺(5-HT)等致密颗粒成分也具有显著的免疫调节作用[67],如谷氨酸盐和血清素致密颗粒能够诱导T细胞迁移,增加单核细胞向树突状细胞的分化,ADP可通过与树突状细胞上的P2Y12ADP受体结合,诱导抗原内吞作用而起到保护神经细胞作用。因此,富血小板血浆可通过细胞间黏附、免疫调节因子的激活与释放、诱导抗原内吞等作用减少神经周围的炎症反应,降低神经病理性疼痛的发生。 2.3.3 富血小板血浆促进许旺细胞活化及轴突再生作用 富血小板血浆中转化生长因子β1、血小板衍生生长因子(PDGF)、成纤维细胞生长因子(FGF)、神经生长因子(NGF)等多种生长因子能够协同促进许旺细胞的增殖,其中,血小板衍生生长因子能够促进许旺细胞的增殖分化,对轴突迁移和神经再生有着引导和延伸作用。神经受损后,受损的轴突再生并延伸至重新支配其原始组织,而轴突这种再生主要由许旺细胞通过c-Jun转录因子介导释放神经生长因子、脑源性神经营养因子和胶质细胞源性神经营养因子等神经营养因子及其细胞外基质来促进,同时还促进了再生轴突重新有髓化,恢复神经再通功能[68-69]。同时,血小板衍生生长因子可通过ERK1/2、PI3K/Akt和JNK信号通路促进人类脂肪衍生干细胞增殖,刺激神经的再生[70]。OYA等[71]研究发现,在神经损伤的周围血小板衍生生长因子β的mRNA转录表达明显活跃,血小板衍生生长因子β的浓度在损伤神经处显著升高,并随着受损神经再生轴突数量的增加而减少,表明了血小板衍生生长因子β在促进许旺细胞活化及轴突再生作用发挥了重要作用。此外,脑源性神经营养因子可诱导N-甲基-D-天冬氨酸受体(NMDAR)激活,调节钙调蛋白激酶(CAMKII)表达,激活丝裂原激活蛋白激酶(mitogen-activated protein kinases,MAPK)一系列级联反应,进而诱导突触可塑性相关基因表达。ZHENG等[72]研究发现富血小板血浆通过提高许旺细胞的神经生长因子和胶质细胞源性神经营养因子分泌量,能显著促进大鼠许旺细胞的增殖和迁移。AL-MASSRI等[73]研究发现,神经生长因子可通过促进并维持轴突及神经元的生长及活性而起到保护神经作用。 富血小板血浆可直接生成为纤维蛋白基质,不仅可刺激新生血管的形成,也可促进许旺细胞黏附而形成 Bungner带,促进轴突再生并为再生轴突提供细胞外基质[74],促进神经损伤的修复治疗。转化生长因子β1通过促进β1结合素的表达而调控许旺细胞的延伸,并且β1结合素可通过识别受损神经细胞的微环境变化,进而引起神经损伤后级联反应的发生。血管内皮生长因子可通过与神经细胞结合而激活细胞内信号通路而促进轴突生长,胰岛素样生长因子1可通过为神经元发育提供营养,激活CaMKII和MAPK信号通路,调节突触素Ⅰ的释放,改善突触传递功能并进而促进许旺细胞增殖和神经再生修复[75]。 CASTRO等[76]发现,富血小板血浆应用于背根神经损伤的大鼠模型中时可通过转化生长因子、血管内皮生长因子、脑源性神经营养因子、神经生长因子、胶质细胞源性神经营养因子等生长因子表达,从而恢复大鼠部分缩爪退缩反射,促进背根神经损伤术后感觉运动的恢复。因此,富血小板血浆可通过促进许旺细胞活化与轴突再生的作用,有效修复及保护神经,促进神经功能恢复。 2.3.4 富血小板血浆促进椎间盘退变的修复作用 LDN的发生主要由机械压迫和继发的神经免疫炎症反应产生,这些机制通过复杂的相互作用来维持神经痛症状。在退变的椎间盘中,髓核脱水导致椎间隙高度降低减少了脊神经的活动空间,同时纤维环破裂导致突出的髓核组织机械压迫脊神经,从而导致受损神经缺血水肿及高度敏感,最终导致严重的神经痛症状。 富血小板血浆治疗可通过促进蛋白聚糖和胶原蛋白的合成、增强纤维环细胞和椎间盘细胞的增殖分化以及抑制细胞外基质的降解,从而重塑椎间盘组织及招募外周细胞和营养物质,最终提高椎间盘细胞活性及恢复椎间隙高度,在一定程度上可消除突出椎间盘组织对邻近神经的机械压迫,从而缓解神经疼痛症状[2,51,77]。同时,富血小板血浆中大量的细胞因子和生长因子可通过相互协同作用促进新血管形成以及诱导巨噬细胞对突出椎间盘的吞噬吸收以解除脊神经的压迫[78]。血小板也可通过促进撕裂的椎间盘纤维环细胞相互愈合以封闭突出的髓核,起到隔离髓核与免疫炎症反应的屏障作用。ANITUA等[79]在兔退变椎间盘模型中研究发现,自体富血小板血浆相对于自体贫血小板血浆及PBS对维持原有椎间盘高度、MRI影像图T2加权像信号改善等具有明显的作用。杜薇等[80]在分析经皮内窥镜下椎间盘摘除联合富血小板血浆治疗腰椎间盘突出症中发现,末次随访时观察组椎间隙高度及椎间盘Pfirrmann退变分级较对照组均有明显改善,表明富血小板血浆能够延缓椎间隙高度下降及椎间盘退变。因此,富血小板血浆可通过恢复椎间隙高度及诱导吞噬细胞对突出间盘的吞噬吸收以解除对神经的压迫,从而能够缓解神经疼痛症状。 2.3.5 富血小板血浆促进血管形成 在神经损伤初期,轴浆逆向运输神经营养因子迅速减少,不能为神经细胞提供足够营养物质,长时间将会导致神经轴突坏死,随之是神经鞘模增生、髓鞘发生变性,最终形成无活性的神经碎屑[81]。富血小板血浆中释放的血管内皮生长因子、血小板衍生生长因子BB、成纤维细胞生长因子、转化生长因子β1和表皮细胞生长因子(EGF)等可直接刺激神经血管与滋养动脉的再生,为轴突再生和许旺细胞增殖提供氧分和营养物质,同时血管内皮细胞还可通过促进细胞因子分泌,刺激轴突生长,激活神经内源性恢复机制以达到神经再生的目的。血管内皮生长因子家族成员如VEGF-A、VEGF-B、VEGF-C、VEGF-D 及胎盘生长因子(PIGF)等可通过结合并激活3种不同类型的酪氨酸激酶受体,形成血管内皮生长因子与酪氨酸激酶二聚体,激发MAPK、Akt和磷脂酶C等一系列细胞内信号级联反应,进而促进新生血管形成和发挥神经保护作用[82]。TEYMUR等[58]在应用自体神经移植物联合富血小板血浆治疗受损大鼠坐骨神经模型中,发现富血小板血浆能促进损伤神经周围血管再生的作用,从而加速大鼠坐骨神经功能恢复。因此,富血小板血浆可通过促进新生血管形成以增加神经营养及神经保护作用,促进神经功能的恢复。 "

| [1] 梁萌,陈治军.糖皮质激素与臭氧硬膜外腔注射治疗椎间盘源性神经痛的比较性研究[J].颈腰痛杂志,2018,39(5):660-661. [2] NAVANI A, MANCHIKANTI L, ALBERS SL, et al. Responsible, safe, and effective use of biologics in the management of low back pain: American society of interventional pain physicians (ASIPP) guidelines. Pain Physician. 2019;22(1S):S1-S74. [3] MEYER-FRIEßEM CH, EITNER LB, KAISLER M, et al. Perineural injection of botulinum toxin-A in painful peripheral nerve injury - a case series: pain relief, safety, sensory profile and sample size recommendation. Curr Med Res Opin. 2019;35(10):1793-1803. [4] WANG L, HE T, LIU J, et al. Revealing the immune infiltration landscape and identifying diagnostic biomarkers for lumbar disc herniation. Front Immunol. 2021;12:666355. [5] NAKAZAWA KR, WALTER BA, LAUDIER DM, et al. Accumulation and localization of macrophage phenotypes with human intervertebral disc degeneration. Spine J. 2018;18(2):343-356. [6] 陈开广,刘群会,陈绣娟,等.射频热凝并骶管注射对椎间盘源性腰神经痛的治疗作用[J].中国康复医学杂志,2006,21(8):736-737. [7] STEFFEN F, BERTOLO A, AFFENTRANGER R, et al. Treatment of naturally degenerated canine lumbosacral intervertebral discs with autologous mesenchymal stromal cells and collagen microcarriers: a prospective clinical study. Cell Transplant. 2019;28(2):201-211. [8] 庞胤,尹帅,赵长义,等.脊柱腰段三维有限元模型的构建与椎间盘应力分析[J].河北医科大学学报,2019,40(12):1368-1371. [9] WANG AM, CAO P, YEE A, et al. Detection of extracellular matrix degradation in intervertebral disc degeneration by diffusion magnetic resonance spectroscopy. Magn Reson Med. 2015;73(5):1703-1712. [10] KIM H, HONG JY, LEE J, et al. IL-1β promotes disc degeneration and inflammation through direct injection of intervertebral disc in a rat lumbar disc herniation model. Spine J. 2021;21(6):1031-1041. [11] 秦大平,张晓刚,聂文忠,等.不同运动状态下模拟人体腰椎结构特征变化的有限元分析[J].医用生物力学,2017,32(4):355-362. [12] 冯阳,严旭,王永魁,等.腰椎间盘退变性疾病与外周血淋巴细胞亚群的关系[J].中国组织工程研究,2020,24(17):2630-2635. [13] JOYCE K, MOHD ISA IL, KROUWELS A, et al. The role of altered glycosylation in human nucleus pulposus cells in inflammation and degeneration. Eur Cell Mater. 2021;41:401-420. [14] XU Z, ZHOU X, CHEN G. Expression and mechanism of interleukin 1 (IL-1), interleukin 2 (IL-2), interleukin 8 (IL-8), BMP, fibroblast growth factor 1 (FGF1), and insulin-like growth factor (IGF-1) in lumbar disc herniation. Med Sci Monit. 2019;25:984-990. [15] SUN Z, LIU B, LUO ZJ. The immune privilege of the intervertebral disc: implications for intervertebral disc degeneration Treatment. Int J Med Sci. 2020;17(5):685-692. [16] SCHROEDER GD, MARKOVA DZ, KOERNER JD, et al. Are modic changes associated with intervertebral disc cytokine profiles? Spine J. 2017;17(1):129-134. [17] JOHNSON ZI, SCHOEPFLIN ZR, CHOI H, et al. Disc in flames: roles of TNF-α and IL-1β in intervertebral disc degeneration. Eur Cell Mater. 2015;30:104-117. [18] WANG C, YU X, YAN Y, et al. Tumor necrosis factor-α: a key contributor to intervertebral disc degeneration. Acta Biochim Biophys Sin. 2017; 49(1):1-13. [19] YANG W, YU XH, WANG C, et al. Interleukin-1β in intervertebral disk degeneration. Clin Chim Acta. 2015;450:262-272. [20] XIE J, LI B, YAO B, et al. Transforming growth factor-β1-regulated Fas/FasL pathway activation suppresses nucleus pulposus cell apoptosis in an inflammatory environment. Biosci Rep. 2020;40(2):BSR20191726. [21] LEE JM, SONG JY, BAEK M, et al. Interleukin-1β induces angiogenesis and innervation in human intervertebral disc degeneration. J Orthop Res. 2011;29(2):265-269. [22] SUN Z, ZHANG M, ZHAO XH, et al. Immune cascades in human intervertebral disc: the pros and cons. Int J Clin Exp Pathol. 2013;6(6): 1009-1014. [23] MALCANGIO M. Role of the immune system in neuropathic pain. Scand J Pain. 2019;20(1):33-37. [24] OZAKI-SAITOH H, TSUDA M. Microglia-neuron interactions in the models of neuropathic pain. Biochem Pharmacol. 2019;169:113614. [25] MURPHY RW. Nerve roots and spinal nerves in degenerative disk disease. Clin Orthop Relat Res. 1977;(129):46-60. [26] DOWER A, DAVIES MA, GHAHREMAN A. Pathologic basis of lumbar radicular pain. World Neurosurg. 2019;128:114-121. [27] BOBECHKO WP, HIRSCH C. Auto-immune response to nucleus pulposus in the rabbit. J Bone Joint Surg Br. 1965;47:574-580. [28] NAYLOR A, HAPPEY F, TURNER RL, et al. Enzymic and immunological activity in the intervertebral disk. Orthop Clin North Am. 1975;6(1):51-58. [29] SPILIOPOULOU I, KOROVESSIS P, KONSTANTINOU D, et al. IgG and IgM concentration in the prolapsed human intervertebral disc and sciatica etiology. Spine. 1994;19(12):1320-1323. [30] SATOH K, KONNO S, NISHIYAMA K, et al. Presence and distribution of antigen-antibody complexes in the herniated nucleus pulposus. Spine. 1999;24:1980-1984. [31] GEISS A, LARSSON K, RYDEVIK B, et al. Autoimmune properties of nucleus pulposus: an experimental study in pigs. Spine. 2007;32:168-173. [32] 曹涌,姚羽,张烽.腰椎间盘突出症与Th细胞、NK细胞的相关性研究[J].中国脊柱脊髓杂志,2013,23(10):912-915. [33] CAPOSSELA S, SCHLAFLI P, BERTOLO A, et al. Degenerated human intervertebral discs contain autoantibodies against extracellular matrix proteins. Eur Cell Mater. 2014;27:251-263. [34] 欧阳冰,苏建成,曾月东,等.退变腰椎间盘组织中炎症因子IL-6、IL-10及MCP-1的表达变化及意义[J]. 海南医学院学报,2014, 20(3):381-383. [35] STICH S, STOLK M, GIROD PP, et al. Regenerative and immunogenic characteristics of cultured nucleus pulposus cells from human cervical intervertebral discs. PLoS One. 2015;10(5):e0126954. [36] 刘振刚,卢一生,施建东,等.腰椎间盘突出症和椎间盘源性疼痛的免疫病理学研究[J].颈腰痛杂志,2016,37(4):272-275. [37] MONCHAUX M, FORTERRE S, SPRENG D, et al. Inflammatory processes associated with canine intervertebral disc herniation. Front Immunol. 2017; 8:1681. [38] WANG S, WEI J, FAN Y, et al. Progranulin is positively associated with intervertebral disc degeneration by interaction with IL-10 and IL-17 through TNF pathways. Inflammation. 2018;41(5):1852-1863. [39] HASVIK E, SCHJØLBERG T, JACOBSEN DP, et al. Up-regulation of circulating microRNA-17 is associated with lumbar radicular pain following disc herniation. Arthritis Res Ther. 2019;21(1):186. [40] SILVA AJ, FERREIRA JR, CUNHA C, et al. Macrophages down-regulate gene expression of intervertebral disc degenerative markers under a pro-inflammatory microenvironment. Front Immunol. 2019;10:1508. [41] LEE S, MILLECAMPS M, FOSTER DZ, et al. Long-term histological analysis of innervation and macrophage infiltration in a mouse model of intervertebral disc injury-induced low back pain. J Orthop Res. 2020; 38(6):1238-1247. [42] CHENG J, SANTIAGO KA, NGUYEN JT, et al. Treatment of symptomatic degenerative intervertebral discs with autologous platelet-rich plasma: follow-up at 5-9 years. Regen Med. 2019;14(9):831-840. [43] CHANG MC, PARK D. The effect of intradiscal platelet-rich plasma injection for management of discogenic lower back pain: a meta-analysis. J Pain Res. 2021;14:505-512. [44] BASTAMI F, VARES P, KHOJASTEH A. Healing effects of platelet-rich plasma on peripheral nerve injuries. J Craniofac Surg. 2017;28(1):e49-e57. [45] GIUSTI I, D’ASCENZO S, MACCHIARELLI G, et al. In vitro evidence supporting applications of platelet derivatives in regenerative medicine. Blood Transfus. 2020;18(2):117-129. [46] 阚厚铭,范利君,陈学泰,等.富血小板血浆在神经病理性疼痛中的应用[J].中国组织工程研究,2022,26(8):1345-1353. [47] ZAFARNIA S, BZYL-IBACH J, SPIVAK I, et al. Nilotinib enhances tumor angiogenesis and counteracts VEGFR2 blockade in an orthotopic breast cancer xenograft model with desmoplastic response. Neoplasia. 2017;19(11):896-907. [48] HANLEY EN JR, HERKOWITZ HN, KIRKPATRICK JS, et al. Debating the value of spine surgery. J Bone Joint Surg Am. 2010;92(5):1293-1304. [49] KIRCHNER F, PINAR A, MILANI I, et al. Vertebral intraosseous plasma rich in growth factor (PRGF-Endoret) infiltrations as a novel strategy for the treatment of degenerative lesions of endplate in lumbar pathology: description of technique and case presentation. J Orthop Surg Res. 2020;15(1):72. [50] 董晤讯,马勇,郭杨,等.富血小板血浆对兔腰背根神经节细胞凋亡的影响[J].安徽医药,2020,24(7):1374-1377. [51] CHANG NJ, ERDENEKHUYAG Y, CHOU PH, et al. Therapeutic effects of the addition of platelet-rich plasma to bioimplants and early rehabilitation exercise on articular cartilage repair. Am J Sports Med. 2018;46(9):2232-2241. [52] ZHANG J, WANG JH. PRP treatment effects on degenerative tendinopathy - an in vitro model study. Muscles Ligaments Tendons J. 2014;4(1):10-17. [53] MCGREGOR CE, IRWIN AM, ENGLISH AW. The val66met BDNF polymorphism and peripheral nerve injury: enhanced regeneration in mouse met-carriers is not further improved with activity-dependent treatment. Neurorehabil Neural Repair. 2019;33(6):407-418. [54] DEMBO T, BRAZ JM, HAMEL KA, et al. Primary afferent-derived BDNF contributes minimally to the processing of pain and itch. eNeuro. 2018;5(6):ENEURO.0402-18.2018. [55] ENTERRÍA-MORALES D, LÓPEZ-LÓPEZ I, LÓPEZ-BARNEO J, et al. Striatal GDNF production is independent to circulating estradiol level despite pan-neuronal activation in the female mouse. PLoS One. 2016;11(10): e0164391. [56] MOUSSA M, LAJEUNESSE D, HILAL G, et al. Platelet rich plasma (PRP) induces chondroprotection via increasing autophagy, anti-inflammatory markers, and decreasing apoptosis in human osteoarthritic cartilage. Exp Cell Res. 2017;352(1):146-156. [57] AYADI AE, ZIGMOND MJ, SMITH AD. IGF-1 protects dopamine neurons against oxidative stress: association with changes in phosphokinases. Exp Brain Res. 2016;234(7):1863-1873. [58] TEYMUR H, TIFTIKCIOGLU YO, CAVUSOGLU T, et al. Effect of platelet-rich plasma on reconstruction with nerve autografts. Kaohsiung J Med Sci. 2017 33(2):69-77. [59] EVERTS P, ONISHI K, JAYARAM P, et al. Platelet-rich plasma: new performance understandings and therapeutic considerations in 2020. Int J Mol Sci. 2020;21(20):7794. [60] ACOSTA-RODRIGUEZ EV, NAPOLITANI G, LANZAVECCHIA A, et al. Interleukins 1beta and 6 but not transforming growth factor-beta are essential for the differentiation of interleukin 17-producing human T helper cells. Nat Immunol. 2007;8(9):942-949. [61] LIN W, LIU T, WANG B, et al. The role of ocular dendritic cells in uveitis. Immunol Lett, 2019, 209: 4-10. [62] CHUDNOVSKIY A, PASQUAL G, VICTORA GD. Studying interactions between dendritic cells and T cells in vivo. Curr Opin Immunol. 2019; 58:24-30. [63] HERTER JM, ROSSAINT J, ZARBOCK A. Platelets in inflammation and immunity. J Thromb Haemost. 2014;12(11):1764-1775. [64] COGNASSE F, LARADI S, BERTHELOT P, et al. Platelet Inflammatory response to stress. Front Immunol. 2019;10:1478. [65] 赵雪颖,张立媛,王育文.血小板与中性粒细胞的配-受体结合及信号传导[J].中国免疫学杂志,2021,37(22):2730-2733. [66] HUANG J, LI X, SHI X, et al. Platelet integrin αIIbβ3: signal transduction, regulation, and its therapeutic targeting. J Hematol Oncol. 2019;12(1):26. [67] IBERG CA, HAWIGER D. Natural and induced tolerogenic dendritic cells. J Immunol. 2020;204(4):733-744. [68] SCHIRA J, HEINEN A, POSCHMANN G, et al. Secretome analysis of nerve repair mediating Schwann cells reveals smad-dependent trophism. FASEB J. 2019;33(4):4703-4715. [69] PATEL NP, LYON KA, HUANG JH. An update-tissue engineered nerve grafts for the repair of peripheral nerve injuries. Neural Regen Res. 2018;13(5):764-774. [70] LAI F, KAKUDO N, MORIMOTO N, et al. Platelet-rich plasma enhances the proliferation of human adipose stem cells through multiple signaling pathways. Stem Cell Res Ther. 2018;9(1):107. [71] OYA T, ZHAO YL, TAKAGAWA K, et al. Platelet-derived growth factor-b expression induced after rat peripheral nerve injuries. Glia. 2002;38(4):303-312. [72] ZHENG C, ZHU Q, LIU X, et al. Effect of platelet-rich plasma (PRP) concentration on proliferation, neurotrophic function and migration of Schwann cells in vitro. J Tissue Eng Regen Med. 2016;10(5):428-436. [73] AL-MASSRI KF, AHMED LA, EL-ABHAR HS. Mesenchymal stem cells therapy enhances the efficacy of pregabalin and prevents its motor impairment in paclitaxel-induced neuropathy in rats: Role of Notch1 receptor and JAK/STAT signaling pathway. Behav Brain Res. 2019;360: 303-311. [74] YE F, LI H, QIAO G, et al. Platelet-rich plasma gel in combination with schwann cells for repair of sciatic nerve injury. Neural Regen Res. 2012; 7(29):2286-2292. [75] OGUNDELE OM, PARDO J, FRANCIS J, et al. A putative mechanism of age-related synaptic dysfunction based on the impact of IGF-1 receptor signaling on synaptic CaMKIIα phosphorylation. Front Neuroanat. 2018; 12:35. [76] CASTRO MV, SILVA MVRD, CHIAROTTO GB, et al. Reflex arc recovery after spinal cord dorsal root repair with platelet rich plasma (PRP). Brain Res Bull. 2019;152:212-224. [77] SINGH S, PATEL AA, SINGH JR. Intervertebral Disc degeneration: the role and evidence for Nnon-stem-cell-based regenerative therapies. Int J Spine Surg. 2021;15(s1):54-67. [78] RAWSON B. Platelet-Rich Plasma and Epidural Platelet lysate: novel treatment for lumbar disk herniation. J Am Osteopath Assoc. 2020; 120(3):201-207. [79] ANITUA E, PRADO R, MURUZABAL F. Why dilute the regenerative power of platelet-rich plasma? J Craniomaxillofac Surg. 2019;47(3):530-531. [80] 杜薇,丁宇,崔洪鹏,等.经皮内窥镜下椎间盘摘除联合富血小板血浆凝胶微球置入治疗腰椎间盘突出症的疗效观察[J].中国脊柱脊髓杂志,2020,30(11):1001-1006. [81] LIU C, CHAN C. An Approach to enhance alignment and myelination of dorsal root ganglion neurons. J Vis Exp. 2016;(114):54085. [82] HUANG H, LIU H, YAN R, et al. PI3K/Akt and ERK/MAPK signaling promote different aspects of neuron survival and axonal regrowth following rat facial nerve axotomy. Neurochem Res. 2017;42(12):3515-3524. [83] MUTHU S, JEYARAMAN M, CHELLAMUTHU G, et al. Does the intradiscal injection of platelet rich plasma have any beneficial role in the management of lumbar disc disease? Global Spine J. 2021: 2192568221998367. [84] NAZARI L, SALEHPOUR S, HOSSEINI MS, et al. The effects of autologous platelet-rich plasma in repeated implantation failure: a randomized controlled trial. Hum Fertil (Camb). 2020;23(3):209-213. [85] GAVRIIL L, DETOPOULOU M, PETSINI F, et al. Consumption of plant extract supplement reduces platelet activating factor-induced platelet aggregation and increases platelet activating factor catabolism: a randomised, double-blind and placebo-controlled trial. Br J Nutr. 2019; 121(9):982-991. [86] MIROSHNYCHENKO O, CHALKLEY RJ, LEIB RD, et al. Proteomic analysis of platelet-rich and platelet-poor plasma. Regen Ther. 2020;15:226-235. |
| [1] | Cao Sheng, Kong Lingwei, Xu Kun, Sun Zhijie. Correlation of cervical sagittal force line parameters with degenerative segment and Pfirrmann classification in patients with cervical intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1319-1324. |
| [2] | Zheng Hongrui, Zhang Wenjie, Wang Yunhua, He Bin, Shen Yajun, Fan Lei. Femoral neck system combined with platelet-rich plasma in the treatment of femoral neck fracture [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1390-1395. |
| [3] | Lian Shilin, Zhang Yan, Jiang Qiang, Zhang Hanshuo, Li Tusheng, Ding Yu. Interventional effects of whole blood and platelet-rich plasma with different preparation methods on nucleus pulposus cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1199-1204. |
| [4] | Ning Ziwen, Wang Xu, Shi Zhengliang, Qin Yihua, Wang Guoliang, Jia Di, Wang Yang, Li Yanlin. Meniscal injury repair methods for non-blood supply area [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 420-426. |
| [5] | Gao Xu, Xing Wenhua. Application of finite element analysis in spine surgery [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(18): 2921-2927. |
| [6] | Ye Xuwen, Gu Yong, Chen Liang. Curcumin loaded injectable microspheres retard progression of intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1884-1891. |
| [7] | Zhu Biwen, Wang Dongzhi, Wu Di, Gong Tiancheng, Pan Haopeng, Lu Yuhua, Guo Yibing, Wang Zhiwei, Huang Yan. Biomimetic microenvironment constructed from gelatin methacrylamide/platelet-rich plasma hydrogel promotes the function of insulinoma cell line MIN6 in mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1824-1831. |
| [8] | Feng Junming, Xiong Xianmei, Ma Liqiong, Zhang Yan, Chen Zijie, Li Shijie, Chen Baixing, Jiang Ziwei, Zeng Zhanpeng, Gao Yijia . Comparison of platelet-rich plasma, concentrated growth factor and 3D micro-nanostructure composite scaffolds in repair of rabbit radius defects [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1906-1913. |
| [9] | Lu Jialin, Gao Yao, Li Han, Zhang Ziyu, Wang Yiming, Xu Rui, Wang Zhonghan, Jin Hui. Influencing factors and mechanisms of platelet-rich plasma in tendinopathy [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1944-1953. |
| [10] | Zhang Haobo, Zhao Yunan, Yang Xuejun. Role and therapeutic implications of pyroptosis in intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1445-1451. |
| [11] | Huang Fan, Di Anqi, Qiu Mingwang, Huang Chuyu, Li Xiaohui, Zhao Siyi, Fan Zhiyong, Wu Shan. Establishing a rat model of intervertebral disc degeneration using X-ray guidance [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(35): 5652-5657. |
| [12] | Zhao Yunan, Zhang Haobo, Sun Tao, Yang Xuejun. Hydrogel-based growth factors and drugs in the treatment of intervertebral disc degeneration: problems and prospects [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(34): 5525-5533. |
| [13] | Huang Gao, Xu Jun, Chen Wenge. Implantation of bone marrow mesenchymal stem cells-loaded platelet-rich plasma combined with extracorporeal shock wave in the repair of bone defects [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4812-4818. |
| [14] | Hu Pengyu, Yu Zhiping, Jia Guanghou, Cong Zhichao, Cong Haibo. Adjuvant treatment of nonunion of tibial fractures with platelet-rich plasma evaluated by bone turnover markers [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(27): 4367-4373. |
| [15] | Liu Chao, Zhang Lijun, Du Xinjie, Xu Qian, Lü Hongjuan, Fan Dongmei, Tian Huanling, Huang Jian, Huang Yuxiang. Mechanism of human umbilical cord mesenchymal stem cells to promote platelet plasma coagulation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(25): 4010-4015. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||