Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (34): 5525-5533.doi: 10.12307/2022.465
Previous Articles Next Articles
Hydrogel-based growth factors and drugs in the treatment of intervertebral disc degeneration: problems and prospects
Zhao Yunan1, Zhang Haobo1, Sun Tao2, Yang Xuejun3
- 1Inner Mongolia Medical University, Hohhot 010020, Inner Mongolia Autonomous Region, China; 2Department of Emergency, Second Affiliated Hospital of Inner Mongolia Medical University, Hohhot 010030, Inner Mongolia Autonomous Region, China; 3Department of Thoracic and Lumbar Spine Surgery, The Second Affiliated Hospital of Inner Mongolia Medical University, Hohhot 010030, Inner Mongolia Autonomous Region, China
-
Received:2021-06-15Accepted:2021-07-27Online:2022-12-08Published:2022-04-16 -
Contact:Yang Xuejun, Professor, Chief physician, Master’s supervisor, Department of Thoracic and Lumbar Spine Surgery, The Second Affiliated Hospital of Inner Mongolia Medical University, Hohhot 010030, Inner Mongolia Autonomous Region, China -
About author:Zhao Yunan, Master candidate, Physician, Inner Mongolia Medical University, Hohhot 010020, Inner Mongolia Autonomous Region, China
CLC Number:
Cite this article
Zhao Yunan, Zhang Haobo, Sun Tao, Yang Xuejun. Hydrogel-based growth factors and drugs in the treatment of intervertebral disc degeneration: problems and prospects[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(34): 5525-5533.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

2.1.1 椎间盘生理结构 椎间盘是一种高度水合的纤维软骨组织[17],是由中央的髓核、外层的纤维环及上下的软骨终板构成[18],髓核是椎间盘的中心部分,呈凝胶状,髓核由Ⅱ型胶原蛋白和蛋白聚糖组成,使椎间盘能够抵抗轴向压缩应变。环绕在髓核周围的纤维环富含Ⅰ型胶原蛋白,具有弹性和抵抗横向扩张功能,软骨终板位于椎间盘的上下侧,与椎体形成连接,支持人类脊柱骨骼系统,对于脊柱的稳定性和移动性至关重要[19]。椎间盘的细胞外基质由胶原蛋白和蛋白聚糖组成,其中胶原蛋白为椎间盘提供抗拉强度,蛋白聚糖则通过高渗透保持椎间盘水化和间盘高度,因而良好状态的细胞外基质对于维持椎间盘机械功能必不可少[20]。椎间盘在退变时,因其内部生化的改变,会造成蛋白聚糖的降解增加,使得椎间盘水合程度下降及高度丢失,这进一步导致了蛋白聚糖的合成减少和胶原的合成增加[21]。同时,椎间盘的退变可能导致纤维环的分层及放射状裂隙的形成[22]。 2.1.2 椎间盘血管和神经 椎间盘是人体最大的无供血组织,其营养物质及氧气供应主要来自于外侧纤维环及上下终板内的毛细血管扩散[23],故而自我再生修复能力非常差。当椎间盘开始退变时,椎间盘细胞会释放神经源性因子,促进毛细血管和神经纤维向椎间盘内部生长[24],甚至会深入退变椎间盘的放射状裂隙[20],从而导致椎间盘源性的下腰痛[25]。 2.1.3 细胞外基质的改变 正常来说,椎间盘细胞的合成代谢和分解代谢处于一个动态平衡的过程。当椎间盘开始退变时,细胞外基质的生化环境会发生变化,这种动态平衡就会被打破,在椎间盘细胞外基质微环境中,除蛋白聚糖酶增加[20],基质金属蛋白酶[26]、血小板结合蛋白基序的解聚蛋白样金属蛋白酶[27],促炎因子如肿瘤坏死因子和白细胞介素1等均会增多[28]。有研究表明,蛋白聚糖是阻止血管和神经进入椎间盘的屏障[29],而过度的基质金属蛋白酶活性和这些蛋白聚糖的基质降解会刺激血管和神经嵌入到椎间盘[20],这就进一步加速了椎间盘退变的进程。 2.2 水凝胶搭载生长因子及药物的治疗策略 "

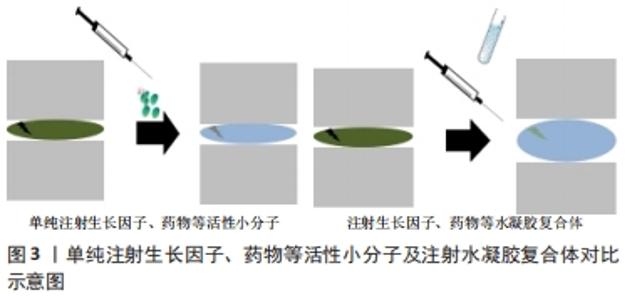
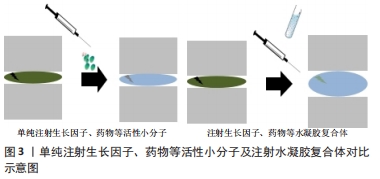
2.2.1 单纯注射生长因子及药物的不足 因为椎间盘细胞的合成代谢和分解代谢之间的平衡维持了椎间盘组织的动态平衡,所以再生退变椎间盘的潜在治疗策略之一是上调椎间盘中细胞因子诱导的合成代谢和下调分解代谢[30]。 近年来,诸多生长因子及药物已经被证明具有缓解椎间盘退变并再生修复的作用[31]。但单纯注射生长因子及药物存在几点缺陷:①缓慢修复椎间盘,无法迅速恢复椎间盘高度。椎间盘退变后,水合程度下降致使椎间盘高度降低,还会影响到周围的组织和结构,这就会导致脊柱的生物力学发生变化,造成下腰痛;若无法快速恢复椎间盘高度,下腰痛将持续存在,治疗效果不明显。②无法控制药物释放,难以实现持续缓慢给药。若单次注射大量生长因子等活性分子,无法发挥其修复椎间盘作用的最佳浓度。若少量多次注射,又会人为造成椎间盘的反复损伤,引起炎症反应,不利于椎间盘的再生和修复。 2.2.2 以水凝胶为载体的优势 水凝胶是通过物理或化学交联形成的可吸收大量水而不溶解的三维网状聚合物体系,其最主要的特性在于具有亲水性交联网络,可吸收大量的水(通常为70%-99%)并保持固体形态,可用于包裹药物、蛋白质和细胞[32]。目前来说,用于椎间盘再生修复的水凝胶根据其来源分为3大类,即天然、合成及复合水凝胶[15],其中天然水凝胶由于其优越的生物相容性和通透性,适合作为生物分子的缓释材料[33]。天然水凝胶的组成主要包括透明质酸、胶原、壳聚糖、海藻酸盐和丝素蛋白等,且其中透明质酸本身就是构成椎间盘细胞外基质的组分之一,从而在椎间盘再生修复中被广泛应用[34]。合成水凝胶则主要具备优越的力学特性和可塑性,但由于其生物相容性较差,使其应用受到限制,如聚乙二醇、聚乳酸和聚乙醇酸等。复合水凝胶如透明质酸-聚乙二醇水凝胶、纤维蛋白京尼平水凝胶等则结合了二者的优点,故而是未来研究水凝胶材料的重要方向。 以水凝胶为载体联合生长因子及药物等治疗椎间盘退变是近年来研究的热点。目前用于搭载生长因子及药物的水凝胶大多为可注射的温敏性水凝胶,即在体外为液态,注射进入椎间盘后受热凝固为固态,且具备一定的强度,起到支撑椎间盘的作用[35]。良好的生物相容性和可降解性,使其不会对椎间盘内细胞造成毒害作用。其优势在于:①治疗椎间盘退变早期即可快速恢复椎间盘高度,从而在一定程度上恢复脊柱正常的生物力学,缓解患者下腰痛的症状,早期即可见显著疗效;②在降解的过程中逐步释放包埋在内部的药物及生长因子,实现长期的治疗效果,并通过包埋密度来控制释放浓度,以达到修复椎间盘的最佳效果。单纯注射生长因子、药物等活性小分子及注射水凝胶复合体对比结果,见图3。"

2.2.3 搭载生长因子等活性分子的水凝胶治疗策略 转化生长因子β:转化生长因子β分子属于生长分化因子家族,代表着一组在组织发育和修复中起关键作用的同源二聚体细胞因子,到目前为止,已经发现和描述了5种亚型(转化生长因子β1-β5),它们在结构和功能上密切相关[36],有研究表明胚胎时期,转化生长因子β信号能促进脊索内髓核组织的形成[37],同时在椎间盘细胞分化后其生长过程仍受到转化生长因子β信号的影响[38],特别是转化生长因子β3在组织培养中已被证明能促进髓核细胞的活性[39]。由于石墨烯含有纳米尺度亲水和疏水性的基团,使其具有良好的吸附、控制和释放生长因子的能力,LIGORIO等[40]研究了一种搭载了转化生长因子β3的氧化石墨烯-自组装肽杂化水凝胶,证明了此种水凝胶中石墨烯片层作为载体来隔离和控制输送转化生长因子β3以促进椎间盘修复和再生的能力且具备良好的生物相容性。GAN等[41]构建了聚乳酸-乙醇酸(poly D,L-lactideco-glycolide,PLGA)纳米微粒,作为搭载转化生长因子β3的载体,并建立其与葡聚糖/明胶水凝胶的共给药系统,将转化生长因子β3的纳米粒子均匀种植到水凝胶中。结果表明共传递系统具有良好的细胞相容性,释放的转化生长因子β3可诱导骨髓间充质干细胞分化为髓核样细胞,同时促进椎间盘细胞外基质相关的生物合成。JIANG等[42]利用温敏性壳聚糖水凝胶作为碱性成纤维生长因子及转化生长因子β1的载体,验证了该种水凝胶对大鼠椎间盘退变模型具有修复作用。GUILLAUME等[43]开发了一种具有形状记忆功能的藻酸盐水凝胶支架,并在其上搭载转化生长因子β3以刺激纤维环细胞增殖和细胞外基质的积淀,从而实现退变椎间盘的再生修复。 骨形态发生蛋白:是转化生长因子β超家族的成员,已有研究表明,骨形态发生蛋白2/7在体外和体内都被证明能促进椎间盘再生[44]。LI等[45]制备了一种纤维蛋白原-透明质酸共轭水凝胶,并以其为载体搭载了骨形态发生蛋白2/7异源二聚体,可上调髓核细胞聚集素和Ⅱ型胶原的表达及糖胺聚糖的合成,而对Ⅰ型胶原及碱性磷酸酶基因表达水平无明显影响,提示该种水凝胶对椎间盘组织无不良成纤维或者成骨的作用。这表明骨形态发生蛋白2/7异源二聚体可能是治疗椎间盘再生的一种有前途的方法。但是,PEETERS等[46]却得出了相反的结论,他们探究了骨形态发生蛋白2及骨形态发生蛋白2/7偶联到纤维蛋白/透明质酸水凝胶上,并在山羊轻度椎间盘退变模型上进行椎间盘内注射,以研究其安全性和有效性,但结果并没有观察到椎间盘再生,他们探究可能的原因包括结合的骨形态发生蛋白剂量低,随访时间短或释放不足等。 HINGERT等[47]在水凝胶模型中证明了骨形态发生蛋白3能够促进椎间盘细胞的生长,认为在10 μg/L的质量浓度更能刺激软骨和细胞外基质的合成。基质金属蛋白酶组织抑制因子的作用是对抗基质金属蛋白酶降解细胞外基质[48]。GANDHI等[49]比较了搭载了转化生长因子β3、骨形态发生蛋白4及基质金属蛋白酶组织抑制因子的壳聚糖水凝胶对兔急性椎间盘损伤模型的作用,结果发现壳聚糖水凝胶转化生长因子β3和骨形态发生蛋白4是在急性椎间盘损伤兔模型促进再生有效,而基质金属蛋白酶组织抑制因子不具有显著效果。有研究构建了搭载骨形态发生蛋白7的自主装多肽纳米纤维水凝胶RADKPS,并验证了该种水凝胶支架具有良好的安全性和生物相容性[50]。 富血小板血浆:是由全血通过特殊的离心过程制成的血小板的浓缩溶液。富血小板血浆中的血小板浓度是血循环中血小板浓度的5-7倍,激活后的血小板包含有300多种生物分子,包括免疫介质(如补体C3,C4前体),各种蛋白质和多种生长因子。许多这些因子在椎间盘再生恢复中起着重要作用,包括细胞增殖和合成增强因子,如转化生长因子β1、血小板衍生生长因子、碱性成纤维细胞生长因子、血管内皮生长因子、胰岛素样生长因子1、表皮生长因子和结缔组织生长因子等[51],其内部的趋化因子更是可以诱导椎间盘内源性干细胞迁移分化,修复和再生受损的椎间盘[31]。 富血小板血浆在体外具备优良的治疗效果,但其在体内由于降解作用,多种搭载富血小板血浆的载体也被设计出来。VADALà等[52]设计了一种由富血小板血浆、巴曲酶和透明质酸混合而成的水凝胶,形成了一个有利于细胞增殖、分化和基质合成的分子网络,能够刺激干细胞增殖分化为椎间盘细胞。而GROWNEY等[51]将富血小板血浆与海藻酸盐应用碳二亚胺进行了化学偶联,得到了一种富血小板血浆功能化的海藻酸盐水凝胶,证明了偶联后的水凝胶相比于单纯将富血小板血浆混合于海藻酸盐所形成的水凝胶,除了在刺激椎间盘细胞增殖和合成细胞外基质上具有更优越的性能外,还提高了水凝胶的机械应力。而针对富血小板血浆在体内的释放时间,有研究开发了一种聚乙二醇水凝胶微球[53],作为搭载富血小板血浆的缓释载体,他们对比了冻干的富血小板血浆粉末和溶解富血小板血浆粉末后除凝得到的液体富血小板血浆分别装载进聚乙二醇水凝胶微球后的释放效果,其中液体富血小板血浆由于在在制备是加入了纤溶酶而降低了水凝胶微球的降解,粉末富血小板血浆则因为微凝块的存在而减缓降解,且粉末富血小板血浆因降解时间延长而获得了更久的缓释效果。SAWAMURA等[54]将富血小板血浆浸渍到明胶水凝胶微球之中,形成了富血小板血浆-明胶水凝胶微球,证明了该种水凝胶无细胞毒性且其对退行型椎间盘的具备治疗修复效果。 血小板衍生生长因子BB:是富血小板血浆的主要成分,已被证明在多种骨科应用中有效。体外研究表明,相较于血小板衍生生长因子、碱性成纤维细胞生长因子和胰岛素样生长因子1,血小板衍生生长因子BB对诱导椎间盘细胞的增殖合成的作用最强。PAGLIA等[55]探究了巯基修饰的透明质酸水凝胶中血小板衍生生长因子BB对椎间盘退变的体内作用,发现装载了血小板衍生生长因子BB的巯基修饰的透明质酸水凝胶具备细胞相容性,能够抑制椎间盘细胞的凋亡和Ⅲ型胶原基质的沉积,阻止退变的进展,并保留了椎间盘的生物力学特性。 生长分化因子:生长分化因子5被证明对促进髓核细胞的存活和增殖有显著的作用[56],HENRY等[57]制备了一种由支链淀粉微球和硅烷化羟丙基甲基纤维素水凝胶(silanized hydroxypropyl methylcellulose,Si-HPMC)联合制成的(PMBs/Si-HPMC)双相可注射水凝胶,用于装载转化生长因子β1和生长分化因子5,在受损的椎间盘中持续释放,能够刺激椎间盘的再生和修复。生长分化因子6是骨形态发生蛋白成员之一,亦被称为骨形态发生蛋白13,在包括椎间盘在内的软骨组织的形成中发挥重要的发育作用,是最近提出的用于髓核再生的一种生长因子[58]。相较于生长分化因子5和转化生长因子β,生长分化因子6对干细胞转化为椎间盘细胞的作用更加显著。HODGKINSON等[59]制备了一种PLGA与聚乙二醇共混的水凝胶(PLGA-PEG-PLGA),能够包裹生长分化因子6并持续释放,该种水凝胶作为一种合成水凝胶,虽然对搭载物质具有较好的控释效果,但其较差的生物相容性可能会限制其进一步被应用。 成纤维细胞生长因子18:由于椎间盘组织类似软骨的特性[60],故而许多可促进软骨形成的生长因子同样可对退变的椎间盘起到修复的作用,但这并不绝对。研究表明成纤维细胞生长因子18可刺激软骨细胞的合成和代谢过程,可促进软骨形成,目前被作为治疗膝关节骨性关节炎的药物之一[61]。为了探究成纤维细胞生长因子18对退变椎间盘的治疗效果,H?CKEL等[62]构建了纤维蛋白透明质酸水凝胶(fibrin-hyaluronic acid hydrogel,FBG-HA),并分别对比了FBG-HA和搭载了成纤维细胞生长因子18的FBG-HA与人和牛的髓核细胞共培养的结果,最终发现FBG-HA可促进髓核细胞的增殖与合成,但成纤维细胞生长因子18对其却无明显作用。由此在退变椎间盘治疗的方面,可将成纤维细胞生长因子排除在外。 基质细胞衍生因子1:是一种众所周知的趋化因子,在骨髓中组成性产生,对淋巴细胞、单核细胞、内皮祖细胞和造血干细胞有很强的趋化作用,基质细胞衍生因子1也被证明可以招募人间充质干细胞[63]。PEREIRA等[64]设计了一种热可逆透明质酸-聚(N-异丙基丙烯酰胺)(thermoreversible hyaluronan-poly N-isopropylacrylamide,HAP)水凝胶,并将基质细胞衍生因子1搭载其中,最终发现此复合物对椎间盘中干细胞的招募是有效的,从而为基于内源性细胞迁移的再生疗法的发展打开了新的可能性。 趋化因子:是修复过程的中介因子,是能够刺激和引导再生机制的潜在分子靶点,趋化因子配体5可募集大量细胞,包括骨髓间充质干细胞。在椎间盘中,趋化因子配体5是由椎间盘细胞分泌的,趋化因子配体5受体表达在纤维环和髓核细胞的表面,因此表明趋化因子配体5可能在椎间盘的愈合过程中发挥作用[65-66]。FRAPIN等[65]开发了一种普鲁兰多糖微珠水凝胶的递送系统,并证明了其生物相容性,用于顺序释放趋化因子配体5,将椎间盘内源性祖细胞招募至髓核之中,在依次释放转化生长因子β1和生长分化因子5以诱导椎间盘细胞外基质的合成。 文章总结了水凝胶搭载生长因子的主要特点,见表1。"

2.2.4 搭载药物及活性分子的水凝胶治疗策略 阿司匹林:解决椎间盘退变相关炎症的解决是组织再生的先决条件,可以通过药物治疗到细胞治疗等多种策略来实现。有研究报道,调节因子高迁移率族蛋白1在细胞外存在时是有效的促炎介质,决定了慢性炎症的进程[67]。阿司匹林作为一种非类固醇抗炎药物被广泛应用于解热镇痛抗炎等方面。LIU等[68]将阿司匹林脂质体与光交联明胶-甲基丙烯酰基混合制备成水凝胶,这种水凝胶在体外能够抑制调节因子高迁移率族蛋白1的释放,从而显著降低炎性细胞因子的表达,在一定程度上延缓了椎间盘的退变。 塞来昔布:塞来昔布作为一种COX-2抑制剂,常被广泛应用于镇痛治疗。但目前塞来昔布主要是通过口服进入体内,不但存在不良反应,一些特殊部位的疼痛也无法得到有效缓解,如椎间盘源性的下腰痛。WILLEMS等[69]设计了一种新型热可逆聚N-异丙基丙烯酰胺镁铁层状双氢氧化物(poly-N-isopropylacrylamide MgFe-layered double hydroxide, pNIPAAM MgFe-LDH)水凝胶,作为控释塞来昔布的载体,并证明了其安全性和有效性并评估了其抑制前列腺素E2的能力,结果发现塞来昔布控释对椎间盘轻度退变动物模型前列腺素E2的抑制作用有限。TELLEGEN等[70]则为此制备了一种丙酰化聚乳酸-聚乙二醇-聚乳酸水凝胶(PCLA-PEG-PCLA)水凝胶用于搭载塞来昔布并注入椎间盘中来缓解下腰痛,目前已在小鼠和犬体内证实了其安全性和有效性。但该种治疗方式仅能用于缓解疼痛而无法恢复和再生已经退变的椎间盘,因而存在较多的限制性。 SHP099:WANG等[19]发现含SRC同源区2的蛋白酪氨酸磷酸酶2(SHP2)同样参与了椎间盘退变的病理生理过程,SHP2的过度表达是椎间盘退变的危险因素,而SHP099是最近发现的一种小分子化合物,能抑制SHP2。证明了SHP099以可注射温敏性聚N-异丙基丙烯酰胺水凝胶为载体时,可以作为一种潜在的治疗椎间盘退变的小分子药物,能够一定程度上恢复和再生退变的椎间盘。 雷帕霉素:有研究表明,慢性炎症微环境特点是活性氧和巨噬细胞M1/M2升高,而这种慢性炎症与椎间盘的退变的发病过程密切相关。其中M1巨噬细胞能分泌促炎性因子,M2巨噬细胞能够分泌抑制因子如转化生长因子β等[71]。雷帕霉素通常被用于通过抑制免疫系统来预防器官移植排斥反应,同时雷帕霉素可以诱导自噬,保护软骨细胞外基质免受降解。BAI等[72]为此而开发了一种负载雷帕霉素的清除活性氧支架水凝胶,在活性氧丰富的环境下,水凝胶支架不断消耗清除活性氧且逐步释放雷帕霉素。当用该种水凝胶处理大鼠时,炎症环境中的M2细胞增多,M1细胞减少,使得炎症减轻,从而阻止椎间盘的退变。 硫化氢:XU等[73]首次提出了硫化氢可防止髓核细胞内质网应力和线粒体损伤,并改善椎间盘退变。但硫化氢在病变组织内突发性释放和快速消失的限制,ZHENG等[74]根据此特点开发了一种pH和酶双响应型硫化氢释放水凝胶体系来治疗椎间盘退变疾病。该种水凝胶被命名为COL-JK1,在中性条件下十分稳定,但在酸性pH值和高水平基质金属蛋白酶的退变椎间盘环境中,会迅速释放硫化氢。目前,在体外试验中COL-JK1通过抑制髓核细胞的凋亡和减轻椎间盘细胞外基质的降解来保护椎间盘免于退变,并在大鼠椎间盘退变模型中证实其安全性及能够抑制椎间盘的退变。 吉非替尼:表皮生长因子受体信号对组织内环境平衡非常重要,有研究表明在退变相对较晚的阶段,表皮生长因子受体在大鼠和人类椎间盘中都有高表达,故而可能在大鼠和人类的软骨退变过程中发挥作用[75]。吉非替尼是小分子表皮生长因子受体抑制剂,通过竞争表皮生长因子受体上的ATP结合位点而抑制表皮生长因子受体,可以保护椎间盘免于退变。PAN等[76]探究吉非替尼对椎间盘的保护作用而设计了一种搭载吉非替尼的热敏性水凝胶,最终发现吉非替尼不仅能抑制软骨基质的降解,而且能促进Ⅱ型胶原的合成,揭示了吉非替尼在治疗椎间盘退变上的应用前景。 Kartogenin (KGN):KGN是一种小分子的生物化合物,可以促进间充质干细胞向软骨细胞的选择性分化,是一种促软骨形成和软骨保护剂。ZHU等[77]将壳聚糖、透明质酸与磷酸甘油按不同比例交联而成可注射水凝胶,并将其作为能激活软骨细胞的生物化合物KGN的缓释系统,发现以5∶3∶2的比例制备的KGN偶联水凝胶具有良好的力学性能,是能促进椎间盘细胞增殖和分化的最佳凝胶配比。 英夫利昔单抗:肿瘤坏死因子α与人类受损的椎间盘密切相关,因为它增加了促炎细胞因子白细胞介素1β、白细胞介素6、白细胞介素8的产生,并促进基质降解酶和疼痛介质的产生[78]。英夫利昔单抗是抗肿瘤坏死因子α的药物之一。LIKHITPANICHKUL等[79]设计了京尼平交联纤维蛋白水凝胶与Ⅰ型胶原空心球分别为载体搭载英夫利昔单抗与人类纤维环细胞共培养,明确药物释放的动力学及有效性,结果发现英夫利昔单抗与京尼平纤维蛋白的直接混合是能够持续释放的最简单的载药方案,且持续释放 20 d以上的英夫利昔单抗保持了生物活性,纤维环细胞产生的白细胞介素1β、白细胞介素6、白细胞介素8和肿瘤坏死因子α浓度持续降低,揭示了该种水凝胶在封堵纤维环损伤和部分恢复椎间盘生物力学的潜力。 辛伐他汀:骨形态发生蛋白2在椎间盘的软骨形成中起重要作用,辛伐他汀,一种3-羟基3-甲基戊二酰辅酶A还原酶抑制剂,已被证明在体外可内源性上调椎间盘细胞中的骨形态发生蛋白2。THAN等[80]制备了一种可交联明胶聚乙二醇酪胺水凝胶,并以其为载体搭载了辛伐他汀,证明了质量浓度5 g/L辛伐他汀水凝胶处理的椎间盘表现出比对照更高的聚集素和Ⅱ型胶原基因表达。 戊聚糖多硫酸盐:FRITH等[81]发现戊聚糖多硫酸盐是一种高度硫酸化的半合成多糖, 具有诱导软骨合成的潜力。他们将戊聚糖多硫酸盐共价结合到透明质酸上,再添加到聚乙二醇/透明质酸的水凝胶中,能诱导椎间盘内源性祖细胞分化,修复退变的椎间盘。 阿魏酸:活性氧诱导的氧化应激参与髓核细胞的衰老和凋亡,而阿魏酸具有治疗活性氧所致疾病的能力,CHENG等[82]研制了温敏性阿魏酸-明胶/壳聚糖/磷酸甘油(thermosensitive FA–gelatin/chitosan/glycerol phosphate,FA-G/C/GP)水凝胶,作为阿魏酸的缓释体系,结果发现氧化受损的髓核细胞,经FA-G/C/GP处理后,基质金属蛋白酶3表达下调,聚集素和Ⅱ型胶原表达上调,可减轻氧化应激对髓核细胞的损伤。 地塞米松:是一种人工合成的糖皮质激素,因其抗炎作用而被广泛应用,已经证明它可以刺激软骨细胞中的软骨生成[83]。BERTOLO等[84]以藻酸盐微珠水凝胶为载体,对比测试了地塞米松、三碘甲状腺原氨酸(T3)以及胰岛素3种药物分子对于椎间盘细胞作用,结果发现仅有地塞米松是一种在常氧和低氧条件下诱导椎间盘细胞成软骨和存活的有价值且经济有效的分子,而胰岛素和T3无明显作用。 硫酸软骨素:硫酸软骨素被用于防治心脑血管疾病、骨关节炎及神经保护等,NAKASHIMA等[85]测试了透明质酸及硫酸软骨素交联而成的水凝胶对于兔的椎间盘损伤模型的影响,结果发现该种水凝胶能够抑制椎间盘退变的级联过程,从而抑制椎间盘退变的进程。 文章总结了水凝胶搭载药物及活性分子的主要特点,见表2。"

| [1] HOY D, MARCH L, BROOKS P, et al. The global burden of low back pain: estimates from the Global Burden of Disease 2010 study. Ann Rheum Dis. 2014;73(6):968-974. [2] VOS T, FLAXMAN A D, NAGHAVI M, et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990-2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380 (9859):2163-2196. [3] Global, regional, and national disability-adjusted life-years (DALYs) for 333 diseases and injuries and healthy life expectancy (HALE) for 195 countries and territories, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet. 2017;390(10100):1260-1344. [4] MANCHIKANTI L, SINGH V, DATTA S, et al. Comprehensive review of epidemiology, scope, and impact of spinal pain. Pain Physician. 2009; 12(4):E35-E70. [5] GORTH DJ, SHAPIRO IM, RISBUD MV. Discovery of the drivers of inflammation induced chronic low back pain: from bacteria to diabetes. Discov Med. 2015;20(110):177-184. [6] HUGHES SP, FREEMONT AJ, HUKINS DW, et al. The pathogenesis of degeneration of the intervertebral disc and emerging therapies in the management of back pain. J Bone Joint Surg Br. 2012;94(10):1298-304. [7] CHEN Z, LI X, PAN F, et al. A retrospective study: does cigarette smoking induce cervical disc degeneration? Int J Surg. 2018;53:269-273. [8] WANG F, CAI F, SHI R, et al. Aging and age related stresses: a senescence mechanism of intervertebral disc degeneration. Osteoarthritis Cartilage. 2016;24(3):398-408. [9] JIN LY, SONG XX, LI XF. The role of estrogen in intervertebral disc degeneration. Steroids. 2020;154:108549. [10] CLOUET J, FUSELLIER M, CAMUS A, et al. Intervertebral disc regeneration:From cell therapy to the development of novel bioinspired endogenous repair strategies. Adv Drug Deliv Rev. 2019; 146:306-324. [11] 李秋江,房晓敏,王胤斌,等.干细胞治疗椎间盘退变研究现状和趋势的文献计量学分析[J].中国组织工程研究,2021,25(31):5000-5011. [12] 余城墙,张宇,谢程欣,等.椎间盘退变分子生物学机制及再生治疗的优势与未来[J].中国组织工程研究,2019,23(30):4889-4896. [13] FRAUCHIGER DA, TEKARI A, WöLTJE M, et al. A review of the application of reinforced hydrogels and silk as biomaterials for intervertebral disc repair. Eur Cell Mater. 2017;34:271-290. [14] 孟真,张永兴,赵庆华.水凝胶人工髓核应用于椎间盘退变的研究进展[J].中国矫形外科杂志,2015,23(19):1790-1792. [15] 石坤,黄勇,黄雷震,等.水凝胶再生修复退变椎间盘的研究进展[J].中国修复重建外科杂志,2020,34(3):275-284. [16] 陈亮,姚峰.载细胞水凝胶在椎间盘修复中的应用[J].医学综述, 2017,23(18):3651-3655. [17] BORRELLI C, BUCKLEY CT. Injectable disc-derived ECM hydrogel functionalised with chondroitin sulfate for intervertebral disc regeneration. Acta Biomater. 2020;117:142-155. [18] GHANNAM M, JUMAH F, MANSOUR S, et al. Surgical anatomy, radiological features, and molecular biology of the lumbar intervertebral discs. Clin Anat. 2017;30(2):251-266. [19] WANG J, HUANG L, HUANG Y, et al. Therapeutic effect of the injectable thermosensitive hydrogel loaded with SHP099 on intervertebral disc degeneration. Life Sci. 2021;266:118891. [20] KHAN AN, JACOBSEN HE, KHAN J, et al. Inflammatory biomarkers of low back pain and disc degeneration: a review. Ann N Y Acad Sci. 2017; 1410(1):68-84. [21] URBAN JP, ROBERTS S. Degeneration of the intervertebral disc. Arthritis Res Ther. 2003;5(3):120-130. [22] ROUGHLEY PJ, ALINI M, ANTONIOU J. The role of proteoglycans in aging, degeneration and repair of the intervertebral disc. Biochem Soc Trans. 2002;30(Pt 6):869-874. [23] MALANDRINO A, LACROIX D, HELLMICH C, et al. The role of endplate poromechanical properties on the nutrient availability in the intervertebral disc. Osteoarthritis Cartilage. 2014;22(7):1053-1060. [24] GARCíA-COSAMALóN J, DEL VALLE ME, CALAVIA MG, et al. Intervertebral disc, sensory nerves and neurotrophins: who is who in discogenic pain? J Anat. 2010;217(1):1-15. [25] RAJ PP. Intervertebral disc: anatomy-physiology-pathophysiology-treatment. Pain Prac. 2008;8(1):18-44. [26] XU H, MEI Q, XU B, et al. Expression of matrix metalloproteinases is positively related to the severity of disc degeneration and growing age in the East Asian lumbar disc herniation patients. Cell Biochem Biophys. 2014;70(2):1219-1225. [27] KAMEDA T, ZVICK J, VUK M, et al. Expression and Activity of TRPA1 and TRPV1 in the Intervertebral Disc: association with Inflammation and Matrix Remodeling. Int J Mol Sci. 2019;20(7):1767. [28] 余盈,叶霖,叶菲.椎间盘再生治疗策略的研究进展[J].华南国防医学杂志,2019,33(4):292-295. [29] JOHNSON WE, CATERSON B, EISENSTEIN SM, et al. Human intervertebral disc aggrecan inhibits nerve growth in vitro. Arthritis Rheum. 2002;46(10):2658-2664. [30] MASUDA K. Biological repair of the degenerated intervertebral disc by the injection of growth factors. Eur Spine J. 2008;17 Suppl 4(Suppl 4): 441-451. [31] 孙阳,李小飞,马胜山,等.富血小板血浆治疗椎间盘退变的研究进展[J].中国脊柱脊髓杂志,2020,30(8):762-766. [32] ZHANG YS, KHADEMHOSSEINI A. Advances in engineering hydrogels. Science. 2017;356(6337):eaaf3627. [33] ANNABI N, TAMAYOL A, UQUILLAS JA, et al. 25th anniversary article: rational design and applications of hydrogels in regenerative medicine. Adv Mater. 2014;26(1):85-123. [34] MOHD ISA IL, ABBAH SA, KILCOYNE M, et al. Implantation of hyaluronic acid hydrogel prevents the pain phenotype in a rat model of intervertebral disc injury. Sci Adv. 2018;4(4):eaaq0597. [35] 俞磊.新型温敏脱细胞髓核基质凝胶的制备及治疗椎间盘退变的研究[D].上海:中国人民解放军海军军医大学,2020. [36] MITTL PR, PRIESTLE JP, COX DA, et al. The crystal structure of TGF-beta 3 and comparison to TGF-beta 2: implications for receptor binding. Protein Sci. 1996;5(7):1261-1271. [37] CHEN S, LIU S, MA K, et al. TGF-β signaling in intervertebral disc health and disease. Osteoarthritis Cartilage. 2019;27(8):1109-1117. [38] JIN H, SHEN J, WANG B, et al. TGF-β signaling plays an essential role in the growth and maintenance of intervertebral disc tissue. FEBS Lett. 2011;585(8):1209-1215. [39] REZA AT, NICOLL SB. Serum-free, chemically defined medium with TGF-beta(3) enhances functional properties of nucleus pulposus cell-laden carboxymethylcellulose hydrogel constructs. Biotechnol Bioeng. 2010;105(2):384-395. [40] LIGORIO C, O’BRIEN M, HODSON NW, et al. TGF-β3-loaded graphene oxide -self-assembling peptide hybrid hydrogels as functional 3D scaffolds for the regeneration of the nucleus pulposus. Acta Biomater. 2021;127:116-130. [41] GAN Y, LI S, LI P, et al. A controlled release codelivery system of MSCs encapsulated in dextran/gelatin hydrogel with TGF-beta3-loaded nanoparticles for nucleus pulposus regeneration. Stem Cells Int. 2016; 2016:9042019. [42] JIANG C, LI DP, ZHANG ZJ, et al. Effect of basic fibroblast growth factor and transforming growth factor-β1 combined with bone marrow mesenchymal stem cells on the repair of degenerated intervertebral discs in rat models. Zhongguo Yi Xue Ke Xue Yuan Xue Bao. 2015;37(4): 456-465. [43] GUILLAUME O, DALY A, LENNON K, et al. Shape-memory porous alginate scaffolds for regeneration of the annulus fibrosus: effect of TGF-β3 supplementation and oxygen culture conditions. Acta Biomater. 2014;10(5):1985-1995. [44] OKADA M, KIM JH, YOON ST, et al. Pulsed Electromagnetic Field (PEMF) plus BMP-2 upregulates intervertebral disc-cell matrix synthesis more than either BMP-2 alone or PEMF alone. J Spinal Disord Tech. 2013; 26(6):E221-E226. [45] LI Z, LANG G, KARFELD-SULZER LS, et al. Heterodimeric BMP-2/7 for nucleus pulposus regeneration-In vitro and ex vivo studies. J Orthop Res. 2017;35(1):51-60. [46] PEETERS M, DETIGER SE, KARFELD-SULZER LS, et al. BMP-2 and BMP-2/7 heterodimers conjugated to a fibrin/hyaluronic acid hydrogel in a large animal model of mild intervertebral disc degeneration. Biores Open Access. 2015;4(1):398-406. [47] HINGERT D, BARRETO HENRIKSSON H, BARANTO A, et al. BMP-3 promotes matrix production in co-cultured stem cells and disc cells from low back pain patients. Tissue Eng Part A. 2020;26(1-2):47-56. [48] CAWSTON TE. Metalloproteinase inhibitors and the prevention of connective tissue breakdown. Pharmacol Ther. 1996;70(3):163-182. [49] GANDHI SD, MAERZ T, MITCHELL S, et al. Intradiscal delivery of anabolic growth factors and a metalloproteinase inhibitor in a rabbit acute lumbar disc injury model. Int J Spine Surg. 2020;14(4):585-589. [50] 刘龙刚,伍耀宏,陶晖,等.修饰有BMP-7功能片段的功能化自组装多肽纳米纤维水凝胶RADKPS制备及其生物相容性研究[J].中国修复重建外科杂志,2016,30(4):491-498. [51] GROWNEY EA, LINDER HR, GARG K, et al. Bio-conjugation of platelet-rich plasma and alginate through carbodiimide chemistry for injectable hydrogel therapies. J Biomed Mater Res B Appl Biomater. 2020;108(5):1972-1984. [52] VADALà G, RUSSO F, MUSUMECI M, et al. Clinically relevant hydrogel-based on hyaluronic acid and platelet rich plasma as a carrier for mesenchymal stem cells: rheological and biological characterization. J Orthop Res. 2017;35(10):2109-2116. [53] CHOI MH, BLANCO A, STEALEY S, et al. Micro-clotting of platelet-rich plasma upon loading in hydrogel microspheres leads to prolonged protein release and slower microsphere degradation. Polymers (Basel). 2020;12(8):1712. [54] SAWAMURA K, IKEDA T, NAGAE M, et al. Characterization of in vivo effects of platelet-rich plasma and biodegradable gelatin hydrogel microspheres on degenerated intervertebral discs. Tissue Eng Part A. 2009;15(12):3719-3727. [55] PAGLIA DN, SINGH H, KARUKONDA T, et al. PDGF-BB delays degeneration of the intervertebral discs in a rabbit preclinical model. Spine (Phila Pa 1976). 2016;41(8):E449-E458. [56] GANTENBEIN-RITTER B, BENNEKER LM, ALINI M, et al. Differential response of human bone marrow stromal cells to either TGF-β(1) or rhGDF-5. Eur Spine J. 2011;20(6):962-971. [57] HENRY N, CLOUET J, FRAGALE A, et al. Pullulan microbeads/Si-HPMC hydrogel injectable system for the sustained delivery of GDF-5 and TGF-β1: new insight into intervertebral disc regenerative medicine. Drug Deliv. 2017;24(1):999-1010. [58] WEI A, SHEN B, WILLIAMS LA, et al. Expression of growth differentiation factor 6 in the human developing fetal spine retreats from vertebral ossifying regions and is restricted to cartilaginous tissues. J Orthop Res. 2016;34(2):279-289. [59] HODGKINSON T, STENING JZ, WHITE LJ, et al. Microparticles for controlled growth differentiation factor 6 delivery to direct adipose stem cell-based nucleus pulposus regeneration. J Tissue Eng Regen Med. 2019;13(8):1406-1417. [60] MWALE F, ROUGHLEY P, ANTONIOU J. Distinction between the extracellular matrix of the nucleus pulposus and hyaline cartilage:a requisite for tissue engineering of intervertebral disc. Eur Cell Mater. 2004;8:58-63. [61] ELLMAN MB, YAN D, AHMADINIA K, et al. Fibroblast growth factor control of cartilage homeostasis. J Cell Biochem. 2013;114(4):735-742. [62] HäCKEL S, ZOLFAGHAR M, DU J, et al. Fibrin-hyaluronic acid hydrogel (RegenoGel) with fibroblast growth factor-18 for in vitro 3D culture of human and bovine nucleus pulposus cells. Int J Mol Sci. 2019;20(20): 5036. [63] GONçALVES RM, ANTUNES JC, BARBOSA MA. Mesenchymal stem cell recruitment by stromal derived factor-1-delivery systems based on chitosan/poly(γ-glutamic acid) polyelectrolyte complexes. Eur Cell Mater. 2012;23:249-260. [64] PEREIRA CL, GONçALVES RM, PEROGLIO M, et al. The effect of hyaluronan-based delivery of stromal cell-derived factor-1 on the recruitment of MSCs in degenerating intervertebral discs. Biomaterials. 2014;35(28):8144-8153. [65] FRAPIN L, CLOUET J, CHéDEVILLE C, et al. Controlled release of biological factors for endogenous progenitor cell migration and intervertebral disc extracellular matrix remodelling. Biomaterials. 2020; 253:120107. [66] PATTAPPA G, PEROGLIO M, SAKAI D, et al. CCL5/RANTES is a key chemoattractant released by degenerative intervertebral discs in organ culture. Eur Cell Mater. 2014;27:124-136. [67] OTOSHI K, KIKUCHI S, KATO K, et al. Anti-HMGB1 neutralization antibody improves pain-related behavior induced by application of autologous nucleus pulposus onto nerve roots in rats. Spine (Phila Pa 1976). 2011;36(11):E692-E698. [68] LIU Y, DU J, PENG P, et al. Regulation of the inflammatory cycle by a controllable release hydrogel for eliminating postoperative inflammation after discectomy. Bioact Mater. 2021;6(1):146-157. [69] WILLEMS N, YANG HY, LANGELAAN ML, et al. Biocompatibility and intradiscal application of a thermoreversible celecoxib-loaded poly-N-isopropylacrylamide MgFe-layered double hydroxide hydrogel in a canine model. Arthritis Res Ther. 2015;17(1):214. [70] TELLEGEN AR, WILLEMS N, BEUKERS M, et al. Intradiscal application of a PCLA-PEG-PCLA hydrogel loaded with celecoxib for the treatment of back pain in canines: what’s in it for humans? J Tissue Eng Regen Med. 2018;12(3):642-652. [71] HWANG MH, KIM KS, YOO CM, et al. Photobiomodulation on human annulus fibrosus cells during the intervertebral disk degeneration: extracellular matrix-modifying enzymes. Lasers Med Sci. 2016;31(4):767-777. [72] BAI J, ZHANG Y, FAN Q, et al. Reactive oxygen species-scavenging scaffold with rapamycin for treatment of intervertebral disk degeneration. Adv Healthc Mater. 2020;9(3):e1901186. [73] XU D, JIN H, WEN J, et al. Hydrogen sulfide protects against endoplasmic reticulum stress and mitochondrial injury in nucleus pulposus cells and ameliorates intervertebral disc degeneration. Pharmacol Res. 2017;117:357-369. [74] ZHENG Z, CHEN A, HE H, et al. pH and enzyme dual-responsive release of hydrogen sulfide for disc degeneration therapy. J Mater Chem B. 2019;7(4):611-618. [75] CITRI A, YARDEN Y. EGF-ERBB signalling: towards the systems level. Nat Rev Mol Cell Biol. 2006; 7(7):505-516. [76] PAN Z, SUN H, XIE B, et al. Therapeutic effects of gefitinib-encapsulated thermosensitive injectable hydrogel in intervertebral disc degeneration. Biomaterials. 2018;160:56-68. [77] ZHU Y, TAN J, ZHU H, et al. Development of kartogenin-conjugated chitosan-hyaluronic acid hydrogel for nucleus pulposus regeneration. Biomater Sci. 2017;5(4):784-791. [78] SCHROEDER M, VIEZENS L, SCHAEFER C, et al. Chemokine profile of disc degeneration with acute or chronic pain. J Neurosurg Spine. 2013; 18(5):496-503. [79] LIKHITPANICHKUL M, KIM Y, TORRE OM, et al. Fibrin-genipin annulus fibrosus sealant as a delivery system for anti-TNFα drug. Spine J. 2015; 15(9):2045-2054. [80] THAN KD, RAHMAN SU, WANG L, et al. Intradiscal injection of simvastatin results in radiologic, histologic, and genetic evidence of disc regeneration in a rat model of degenerative disc disease. Spine J. 2014;14(6):1017-1028. [81] FRITH JE, MENZIES DJ, CAMERON AR, et al. Effects of bound versus soluble pentosan polysulphate in PEG/HA-based hydrogels tailored for intervertebral disc regeneration. Biomaterials. 2014;35(4):1150-1162. [82] CHENG YH, YANG SH, LIN FH. Thermosensitive chitosan-gelatin-glycerol phosphate hydrogel as a controlled release system of ferulic acid for nucleus pulposus regeneration. Biomaterials. 2011;32(29):6953-6961. [83] NA K, PARK JH, KIM SW, et al. Delivery of dexamethasone, ascorbate, and growth factor (TGF beta-3) in thermo-reversible hydrogel constructs embedded with rabbit chondrocytes. Biomaterials. 2006; 27(35):5951-5957. [84] BERTOLO A, ETTINGER L, AEBLI N, et al. The in vitro effects of dexamethasone, insulin and triiodothyronine on degenerative human intervertebral disc cells under normoxic and hypoxic conditions. Eur Cell Mater. 2011;21:221-229. [85] NAKASHIMA S, MATSUYAMA Y, TAKAHASHI K, et al. Regeneration of intervertebral disc by the intradiscal application of cross-linked hyaluronate hydrogel and cross-linked chondroitin sulfate hydrogel in a rabbit model of intervertebral disc injury. Biomed Mater Eng. 2009; 19(6):421-429. [86] 杨帅,吴晓淋,刘畅,等.干细胞治疗退变性椎间盘疾病的研究进展[J].中国骨与关节杂志,2020,9(7):502-507. [87] 王宇翔,徐海栋,赵建宁.细胞移植治疗椎间盘退变的研究进展[J].中国矫形外科杂志,2021,29(11):1009-1012. |
| [1] | Zhang Haobo, Zhao Yunan, Yang Xuejun. Role and therapeutic implications of pyroptosis in intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1445-1451. |
| [2] | Wu Weiyue, Guo Xiaodong, Bao Chongyun. Application of engineered exosomes in bone repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1102-1106. |
| [3] | Zhou Hongqin, Wu Dandan, Yang Kun, Liu Qi. Exosomes that deliver specific miRNAs can regulate osteogenesis and promote angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1107-1112. |
| [4] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [5] | Tian Chuan, Zhu Xiangqing, Yang Zailing, Yan Donghai, Li Ye, Wang Yanying, Yang Yukun, He Jie, Lü Guanke, Cai Xuemin, Shu Liping, He Zhixu, Pan Xinghua. Bone marrow mesenchymal stem cells regulate ovarian aging in macaques [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 985-991. |
| [6] | Gao Yujin, Peng Shuanglin, Ma Zhichao, Lu Shi, Cao Huayue, Wang Lang, Xiao Jingang. Osteogenic ability of adipose stem cells in diabetic osteoporosis mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 999-1004. |
| [7] | Zhang Tong, Cai Jinchi, Yuan Zhifa, Zhao Haiyan, Han Xingwen, Wang Wenji. Hyaluronic acid-based composite hydrogel in cartilage injury caused by osteoarthritis: application and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 617-625. |
| [8] | Li Hui, Chen Lianglong. Application and characteristics of bone graft materials in the treatment of spinal tuberculosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 626-630. |
| [9] | Hou Wanxing, Li Hongwei, Zheng Xin, Zhu Xianren. Correlation between preoperative magnetic resonance imaging findings and bone cement leakage after percutaneous vertebral augmentation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 504-509. |
| [10] | Huang Chuanjun, Zou Yu, Zhou Xiaoting, Zhu Yangqing, Qian Wei, Zhang Wei, Liu Xing. Transplantation of umbilical cord mesenchymal stem cells encapsulated in RADA16-BDNF hydrogel promotes neurological recovery in an intracerebral hemorrhage rat model [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 510-515. |
| [11] | He Guanyu, Xu Baoshan, Du Lilong, Zhang Tongxing, Huo Zhenxin, Shen Li. Biomimetic orientated microchannel annulus fibrosus scaffold constructed by silk fibroin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 560-566. |
| [12] | Li Maoxue, Ding Yi, Guo Shujuan. Understanding of periodontal biomaterials based on the concept of bone homeostasis control and the effects on bone formation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(34): 5562-5568. |
| [13] | Liu Chang, Xu Ling. Modified coaxial 3D-printed akermanite scaffold with hollow-pipe structure [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(34): 5425-5429. |
| [14] | Liu Pei, Zhang Guanying, Yu Quanfeng, Li Zeyu, Han Guangye, Wu Chunlei. Basic fibroblast growth factor combined with poly(lactic acid)/collagen scaffold for urethral defect in rabbits [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(34): 5468-5474. |
| [15] | Lin Lingqi, Chen Jin, Qian Kun, Zhao Liang, Shi Yijie. Preparation and in vitro release of manganese-based metal-organic framework materials loaded with baicalin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(34): 5475-5481. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

