Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (12): 1884-1891.doi: 10.12307/2023.043
Previous Articles Next Articles
Curcumin loaded injectable microspheres retard progression of intervertebral disc degeneration
Ye Xuwen, Gu Yong, Chen Liang
- First Affiliated Hospital of Soochow University, Suzhou 215000, Jiangsu Province, China
-
Received:2021-12-27Accepted:2022-01-25Online:2023-04-28Published:2022-07-30 -
Contact:Chen Liang, Professor, Doctoral supervisor, Chief physician, First Affiliated Hospital of Soochow University, Suzhou 215000, Jiangsu Province, China -
About author:Ye Xuwen, Master candidate, First Affiliated Hospital of Soochow University, Suzhou 215000, Jiangsu Province, China -
Supported by:the National Natural Science Foundation of China, No. 81972078, 81772312 (to CL)
CLC Number:
Cite this article
Ye Xuwen, Gu Yong, Chen Liang. Curcumin loaded injectable microspheres retard progression of intervertebral disc degeneration[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1884-1891.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
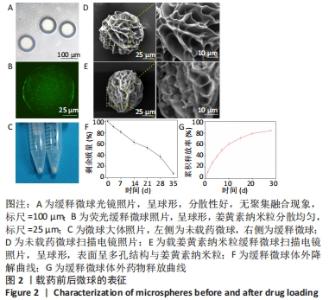
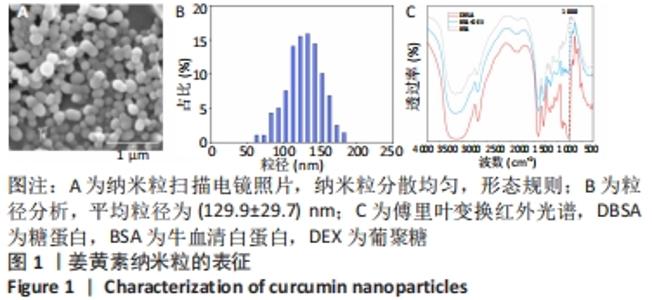
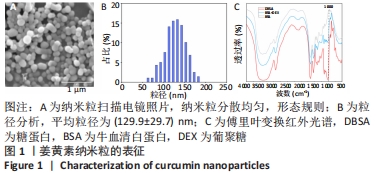
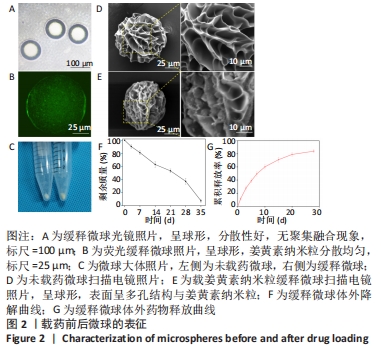
2.2 缓释微球的表征结果 光镜下缓释微球呈球形,分散性好,无聚集融合现象,见图2A。荧光显微镜下的荧光缓释微球呈球形,姜黄素纳米粒分散均匀,见图2B。图2C是冻干微球的实体图片,左侧为未载药微球,右侧缓释微球因载有姜黄素而呈现淡黄色改变。冻干后缓释微球的平均直径为(102.6±7.4) μm,依旧保持球形,表面呈多孔结构,孔径大小分布主要集中于4-9 μm,表面接触面积显著提升,且表面可观察到姜黄素纳米粒,这区别于未载药微球,见图2D,E。缓释微球在含有胶原酶的PBS中,其质量逐渐减小,至第5周时逐渐消失,见图2F。 2.3 缓释微球包封率、载药率及释放曲线 缓释微球的包封率为(76.47±5.16)%,载药率为(15.36±4.54)%。缓释微球体外药物释放曲线显示,缓释微球在最初1周内释放药物相对较快,占药物总量的(49.43±3.40)%,随后释放逐渐减缓,且释放浓度相对稳定,至28 d时释放姜黄素总量为(84.11±2.71)%,见图2G。"
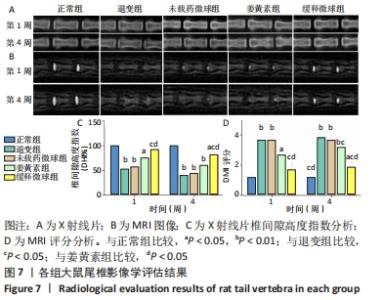
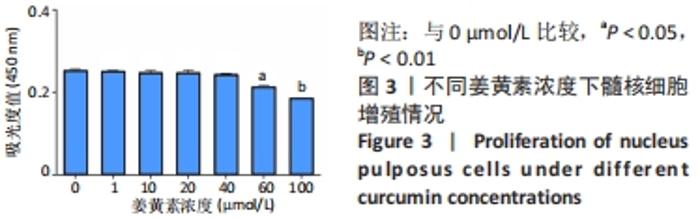
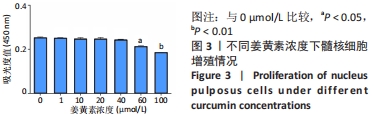
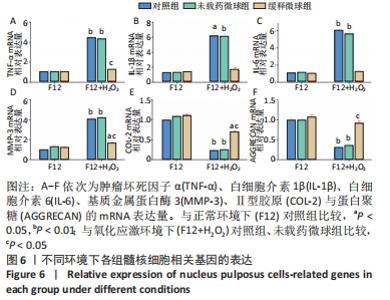
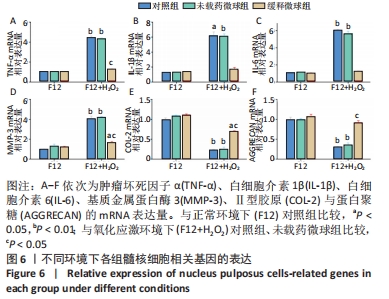
与正常环境下对照组比较,对照组、未载药微球组应激环境下的肿瘤坏死因子α、白细胞介素1β、白细胞介素6、基质金属蛋白酶3的mRNA表达量升高(P < 0.05),Ⅱ型胶原和蛋白聚糖的mRNA表达量降低(P < 0.05);缓释微球组肿瘤坏死因子α、白细胞介素1β、白细胞介素6及蛋白聚糖的mRNA表达量无明显变化(P > 0.05),基质金属蛋白酶3的mRNA表达量升高(P < 0.05),Ⅱ型胶原的mRNA表达量降低(P < 0.05),见图6。 在应激环境下,载药微球组肿瘤坏死因子α、白细胞介素1β、白细胞介素6及基质金属蛋白酶3的mRNA表达量降低(P < 0.05),Ⅱ型胶原与蛋白聚糖的mRNA表达量升高(P < 0.05),见图6。 2.5 动物体内实验结果 2.5.1 实验动物数量分析 30只大鼠全部进入结果分析。 2.5.2 影像学评估结果 见图7。 "

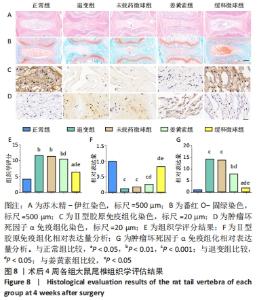
通过X射线片检查评估不同时间各组大鼠尾椎椎间隙高度变化。术后1周时,缓释微球组椎间隙高度下降不明显,与正常组比较差异无显著性意义(P > 0.05),退变组、未载药微球组、姜黄素组椎间隙高度明显降低(P < 0.05)。至第4周时,缓释微球组椎间隙高度低于正常组(P < 0.05),高于退变组、未载药微球组、姜黄素组(P < 0.05),未载药微球组与退变组类似(P > 0.05),姜黄素组较退变组、未载药微球组椎间隙高度下降有所放缓,但与缓释微球组相比仍有差距。整体而言,缓释微球组椎间隙高度下降缓慢,椎体形态较好,无明显骨赘形成。 MRI图像变化趋势与X射线片结果一致。在T2序列下,正常组术后1,4周均显示为高含水量的正常髓核信号,而退变组、未载药微球组、姜黄素组则出现了明显的信号强度下降。缓释微球组髓核信号强度在第1周时较正常组无明显下降,在第4周时信号稍弱于正常组,但强于退变组、未载药微球组、姜黄素组(P < 0.05),MRI评分变化趋势与影像学图像结果一致。 2.5.3 组织学评价结果 见图8。 "
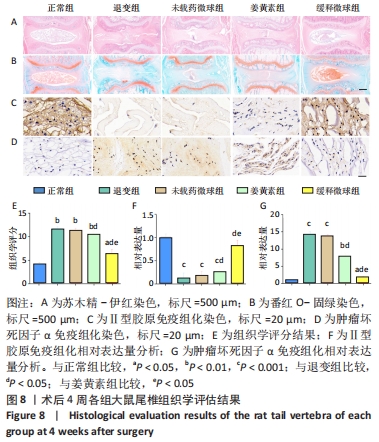

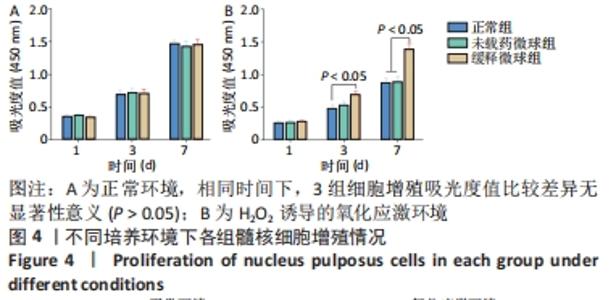
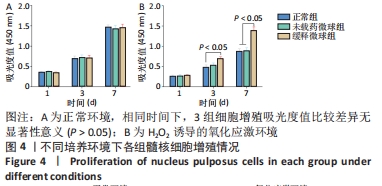
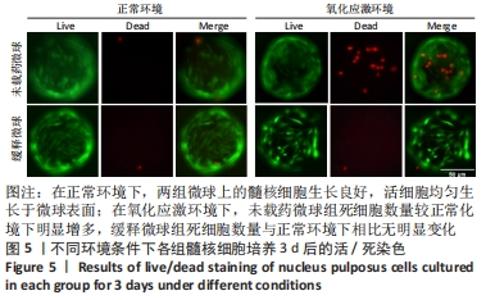
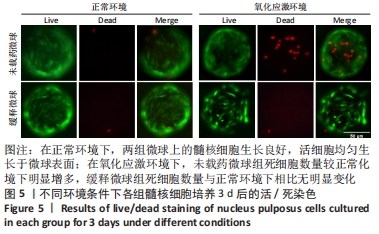
在第4周对各组椎间盘进行组织学评估。苏木精-伊红染色显示,正常组椎间盘结构完好,髓核完整性好,纤维环组织呈环状规律分布,两者界限清晰;缓释微球组出现轻度的髓核组织减少,纤维环组织轻度扭曲(< 30%),两者边界出现轻微中断;退变组、未载药微球组及姜黄素组均可见髓核细胞大量丢失,髓核组织严重萎缩,纤维环结构紊乱、断裂愈发严重,两者界限模糊,组织学评分均显著低于缓释微球组。 番红固绿染色可观察髓核组织胶原含量,胶原组织经番红O-固绿染色后呈橘红色。番红O-固绿染色显示,正常组髓核组织完好,细胞外基质丰富,呈现橘红色;缓释微球组胶原较正常组减少,并出现细胞外基质轻度凝结;退变组、未载药微球组及姜黄素组髓核细胞外基质丢失严重,胶原含量显著减少。 Ⅱ型胶原免疫组化染色结果与番红固绿染色结果一致,缓释微球组Ⅱ型胶原表达较正常组有轻度下降,但明显高于其他3组。以正常组为对照,可在退变组、未载药微球组、姜黄素组观察到高表达的肿瘤坏死因子α信号,即有较多深染髓核细胞,阳性染色面积率高,而缓释微球组的肿瘤坏死因子α信号较低,与正常组最为接近。组织学评分和半定量分析也进一步证实了上述结果。 2.6 微球的生物相容性 由体外细胞增殖实验、活/死染色、qRT-PCR,以及动物体内实验组织学评估结果,可知姜黄素缓释微球具有良好的生物相容性。 "

| [1] VOS T, ABAJOBIR AA, ABBAFATI C, et al. Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet. 2017;390(10100):1211-1259. [2] ROBERTS S, EVANS H, TRIVEDI J, et al. Histology and pathology of the human intervertebral disc. J Bone Joint Surg Am. 2006;88A:10-14. [3] FRAPIN L, CLOUET J, DELPLACE V, et al. Lessons learned from intervertebral disc pathophysiology to guide rational design of sequential delivery systems for therapeutic biological factors. Adv Drug Deliv Rev. 2019;149:49-71. [4] RISBUD MV, SHAPIRO IM. Role of cytokines in intervertebral disc degeneration: pain and disc content. Nat Rev Rheumatol. 2014;10(1):44-56. [5] GRUBER HE, HANLEY EN. Biologic strategies for the therapy of intervertebral disc degeneration. Expert Opin Biol Ther. 2003;3(8):1209-1214. [6] GOEL A, KUNNUMAKKARA AB, AGGARWAL BB. Curcumin as “Curecumin”: From kitchen to clinic. Biochem Pharmacol. 2008;75(4):787-809. [7] MA T, GUO C J, ZHAO X, et al. The effect of Curcumin on NF-kappa B expression in rat with lumbar intervertebral disc degeneration. Eur Rev Med Pharmacol Sci. 2015;19(7):1305-1314. [8] Barzegar A, Moosavi-Movahedi AA. Intracellular ROS Protection Efficiency and Free Radical-Scavenging Activity of Curcumin. Plos One. 2011;6(10):e26012. [9] ANAND P, KUNNUMAKKARA AB, NEWMAN RA, et al. Bioavailability of curcumin: Problems and promises. Mol Pharm. 2007;4(6):807-818. [10] TABANELLI R, BROGI S, CALDERONE V. Improving Curcumin Bioavailability: Current Strategies and Future Perspectives. Pharmaceutics. 2021;13(10):1715. [11] AN FF, ZHANG XH. Strategies for Preparing Albumin-based Nanoparticles for Multifunctional Bioimaging and Drug Delivery. Theranostics. 2017;7(15):3667-3689. [12] SADEGHI R, MOOSAVI-MOVAHEDI AA, EMAM-JOMEH Z, et al. The effect of different desolvating agents on BSA nanoparticle properties and encapsulation of curcumin. J Nanopart Res. 2014;16(9):2565. [13] FAN Y, YI J, ZHANG Y, et al. Fabrication of curcumin-loaded bovine serum albumin (BSA)-dextran nanoparticles and the cellular antioxidant activity. Food Chem. 2018;239:1210-1218. [14] YUE K, TRUJILLO-DE SANTIAGO G, MOISES ALVAREZ M, et al. Synthesis, properties, and biomedical applications of gelatin methacryloyl (GelMA) hydrogels. Biomaterials. 2015;73:254-271. [15] SHI K, HUANG Y, HUANG L, et al. Research progress of hydrogel used for regeneration of nucleus pulposus in intervertebral disc degeneration. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2020;34(3):275-284. [16] DE GEEST BG, URBANSKI JP, THORSEN T, et al. Synthesis of monodisperse biodegradable microgels in microfluidic devices. Langmuir. 2005;21(23):10275-10279. [17] YANG J, ZHU Y, WANG F, et al. Microfluidic liposomes-anchored microgels as extended delivery platform for treatment of osteoarthritis. Chem Eng J. 2020;400:126004. [18] BIAN J, CAI F, CHEN H, et al. Modulation of Local Overactive Inflammation via Injectable Hydrogel Microspheres. Nano Lett. 2021;21(6): 2690-2698. [19] KOTHA RR, LUTHRIA DL. Curcumin: Biological, Pharmaceutical, Nutraceutical, and Analytical Aspects. Molecules. 2019;24(16):2930. [20] ZAKERIKHOOB M, ABBASI S, YOUSEFI G, et al. Curcumin-incorporated crosslinked sodium alginate-g-poly (N-isopropyl acrylamide) thermo-responsive hydrogel as an in-situ forming injectable dressing for wound healing: In vitro characterization and in vivo evaluation. Carbohydr Polym. 2021;271:118434. [21] 王厚磊,卢伟,李德芳,等.大鼠椎间盘髓核细胞体外凋亡模型的建立[J].中国实验动物学报,2015(6):607-611. [22] CALIARI SR, BURDICK JA. A practical guide to hydrogels for cell culture. Nat Methods. 2016;13(5):405-414. [23] TAYLOR SC, NADEAU K, ABBASI M, et al. The Ultimate qPCR Experiment: Producing Publication Quality, Reproducible Data the First Time. Trends Biotechnol. 2019;37(7):761-774. [24] ISSY AC, CASTANIA V, CASTANIA M, et al. Experimental model of intervertebral disc degeneration by needle puncture in Wistar rats. Braz J Med Biol Res. 2013;46(3):235-244. [25] XIA K, ZHU J, HUA J, et al. Intradiscal Injection of Induced Pluripotent Stem Cell-Derived Nucleus Pulposus-Like Cell-Seeded Polymeric Microspheres Promotes Rat Disc Regeneration. Stem Cells Int. 2019; 2019:6806540. [26] MASUDA K, AOTA Y, MUEHLEMAN C, et al. A novel rabbit model of mild, reproducible disc degeneration by an anulus needle puncture: Correlation between the degree of disc injury and radiological and histological appearances of disc degeneration. Spine. 2005;30(1):5-14. [27] BAI JY, ZHANG YZ, FAN Q, et al. Reactive Oxygen Species-Scavenging Scaffold with Rapamycin for Treatment of Intervertebral Disk Degeneration. Adv Healthc Mater. 2020;9(3):e1901186. [28] XU H, SUN M, WANG C, et al. GDF5-GelMA injectable microspheres laden with adipose-derived stem cells for disc degeneration repair. Biofabrication. 2020;10.1088/1758-5090/abc4d3. [29] XU Y, GU Y, CAI F, et al. Metabolism Balance Regulation via Antagonist-Functionalized Injectable Microsphere for Nucleus Pulposus Regeneration. Adv Funct. Mater. 2020;30(52):2006333. [30] ROHIWAL SS, ELLEDEROVA Z, TIWARI AP, et al. Self-assembly of bovine serum albumin (BSA)-dextran bio-nanoconjugate: structural, antioxidant and in vitro wound healing studies. Rsc Adv. 2021;11(8): 4308-4317. [31] KLAWITTER M, QUERO L, KLASEN J, et al. Curcuma DMSO extracts and curcumin exhibit an anti-inflammatory and anti-catabolic effect on human intervertebral disc cells, possibly by influencing TLR2 expression and JNK activity. J Inflamm (Lond). 2012;9(1):29. [32] MAHESHWARI RK, SINGH AK, GADDIPATI J, et al. Multiple biological activities of curcumin: A short review. Life Sci. 2006;78(18):2081-2087. [33] LAO CD, RUFFIN MTT, NORMOLLE D, et al. Dose escalation of a curcuminoid formulation. BMC Complement Altern Med. 2006;6:10. [34] 任爽,董文霞,刘锦芳,等.食品运载体系包埋姜黄素的研究进展[J].食品科学,2021,42(9):264-274. [35] EDWARDS RL, LUIS PB, VARUZZA PV, et al. The anti-inflammatory activity of curcumin is mediated by its oxidative metabolites. J Biol Chem. 2017;292(52):21243-21252. [36] MARTINS C, SOUSA F, ARAUJO F, et al. Functionalizing PLGA and PLGA Derivatives for Drug Delivery and Tissue Regeneration Applications. Adv Healthc Mater. 2018;7(1):10.1002. [37] DALY AC, RILEY L, SEGURA T, et al. Hydrogel microparticles for biomedical applications. Nat Rev Mater. 2020;5(1):20-43. [38] 马晟,贾育松,孙旗.体内腰椎间盘退变动物模型的研究与进展[J].中国组织工程研究,2017,21(11):1790-1797. [39] ZHOU HY, BEEVERS CS, HUANG SL. The Targets of Curcumin. Curr Drug Targets. 2011;12(3):332-347. [40] PENG Y, AO M, DONG B, et al. Anti-Inflammatory Effects of Curcumin in the Inflammatory Diseases: Status, Limitations and Countermeasures. Drug Des Devel Ther. 2021;15:4503-4525. [41] WANG C, YU XH, YAN YG, et al. Tumor necrosis factor-alpha: a key contributor to intervertebral disc degeneration. Acta Biochim Biophys Sin (Shanghai). 2017;49(1):1-13. [42] LIANG H, YANG XJ, LIU C, et al. Effect of NF-kB signaling pathway on the expression of MIF, TNF-alpha, IL-6 in the regulation of intervertebral disc degeneration. J Musculoskelet Neuronal Interact. 2018;18(4):551-556. [43] WANG YJ, CHE MX, XIN JG, et al. The role of IL-1 beta and TNF-alpha in intervertebral disc degeneration. Biomed Pharmacother. 2020;131:110660. [44] 白荣飞,张震,林一峰,等.三种方法建立大鼠腰椎间盘退变模型[J].中国组织工程研究,2018,22(16):2514-2519. |
| [1] | Cao Sheng, Kong Lingwei, Xu Kun, Sun Zhijie. Correlation of cervical sagittal force line parameters with degenerative segment and Pfirrmann classification in patients with cervical intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1319-1324. |
| [2] | Lian Shilin, Zhang Yan, Jiang Qiang, Zhang Hanshuo, Li Tusheng, Ding Yu. Interventional effects of whole blood and platelet-rich plasma with different preparation methods on nucleus pulposus cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1199-1204. |
| [3] | Liu Xiaolin, Mu Xinyue, Ma Ziyu, Liu Shutai, Wang Wenlong, Han Xiaoqian, Dong Zhiheng. Effect of hydrogel-loaded simvastatin microspheres on osteoblast proliferation and differentiation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 998-1003. |
| [4] | Gao Ting, Ma Xiaohong, Li Xiaorong. Extraction and identification of exosomes from three different sources of ovarian granulosa cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 860-865. |
| [5] | Tian Qinyu, Tian Xinggui, Tian Zhuang, Sui Xiang, Liu Shuyun, Lu Xiaobo, Guo Quanyi. Protection of manganese oxide nanoparticles for bone marrow mesenchymal stem cell spreading against oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 821-826. |
| [6] | Li Rui, Liu Zhen, Guo Zige, Lu Ruijie, Wang Chen. Aspirin-loaded chitosan nanoparticles and polydopamine modified titanium sheets improve osteogenic differentiation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 374-379. |
| [7] | Li Yue, Lyu Yan, Feng Wanying, Song Yang, Yan Yu, Guan Yongge. Preparation of hyperoside nanoparticles to repair endometrial injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 360-366. |
| [8] | Li Zhen, Liu Hongbao. Influencing factors and mechanism of nanoparticle renal targeting [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 453-460. |
| [9] | Xu Dayong, Li Yunpeng, Wei Jingmei, Liu Ruyin. Rutin attenuates the progression of intervertebral disc degeneration in rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(14): 2139-2145. |
| [10] | Huang Yanni, Yang Hua, Yang Dongmei, Hu Xulin, Gao Hong, Huang Yina. Rat bone defect repaired with polytrimethylene carbonate/beta-tricalcium phosphate microsphere scaffold [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1856-1862. |
| [11] | Ma Ziyu, Zhang Bin, Zhang Yuntao, Liu Xiaolin, Bian Zhihong, Qiao Luhui, Hou Yudong. Effects of quercetin sustained release system on osteogenic properties of MC3T3-E1 cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1870-1876. |
| [12] | Fan Yaru, Li Ruixin, Li Fengji, Luo Rui, Liu Hao, Yan Yingbin. Characterization and photothermal effect of indocyanine green encapsulated poly lactic acid-co-glycolic acid microspheres [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1817-1823. |
| [13] | Hu Jinlong, Quan Huahong, Wang Jingcheng, Zhang Pei, Zhang Jiale, Chen Pengtao, Liang Yuan. Effect of copper sulfide nanoparticles loaded thermosensitive hydrogel Pluronic F127 on infected wound healing in rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1927-1931. |
| [14] | Gan Tian, Wang Wenyuan, Yan Shujin, Hao Lan, Ran Haitao, Wang Zhigang, Xia Jizhu. Near infrared photoresponsive nanoparticles loaded with LXR agonists for photothermal immunotherapy [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1863-1869. |
| [15] | Su Meng, Wang Xin, Zhang Jin, Bei Ying, Huang Yu, Zhu Yanzhao, Li Jiali, Wu Yan. Nanocellular vesicles loaded with curcumin promote wound healing in diabetic mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1877-1883. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||