Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (12): 1817-1823.doi: 10.12307/2023.032
Previous Articles Next Articles
Characterization and photothermal effect of indocyanine green encapsulated poly lactic acid-co-glycolic acid microspheres
Fan Yaru1, 2, Li Ruixin2, Li Fengji2, Luo Rui2, Liu Hao2, Yan Yingbin2
- 1School of Stomatology, Tianjin Medical University, Tianjin 300070, China; 2Affiliated Stomatological Hospital of Nankai University, Tianjin Stomatological Hospital, Tianjin Key Laboratory of Oral and Maxillofacial Function Reconstruction, Tianjin 300041, China
-
Received:2021-12-03Accepted:2022-01-15Online:2023-04-28Published:2022-07-30 -
Contact:Liu Hao, MD, Chief physician, Affiliated Stomatological Hospital of Nankai University, Tianjin Stomatological Hospital, Tianjin Key Laboratory of Oral and Maxillofacial Function Reconstruction, Tianjin 300041, China Yan Yingbin, MD, Chief physician, Affiliated Stomatological Hospital of Nankai University, Tianjin Stomatological Hospital, Tianjin Key Laboratory of Oral and Maxillofacial Function Reconstruction, Tianjin 300041, China -
About author:Fan Yaru, Master candidate, School of Stomatology, Tianjin Medical University, Tianjin 300070, China; Affiliated Stomatological Hospital of Nankai University, Tianjin Stomatological Hospital, Tianjin Key Laboratory of Oral and Maxillofacial Function Reconstruction, Tianjin 300041, China -
Supported by:the Tianjin Science and Technology Project, No. 19ZXDBSY00070 (to LH); the National Natural Science Foundation of China, No. 11972198 (to LRX); the Special Project of Tianjin Clinical Medicine Key Discipline, No. HWZX001 (to LH)
CLC Number:
Cite this article
Fan Yaru, Li Ruixin, Li Fengji, Luo Rui, Liu Hao, Yan Yingbin. Characterization and photothermal effect of indocyanine green encapsulated poly lactic acid-co-glycolic acid microspheres[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1817-1823.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
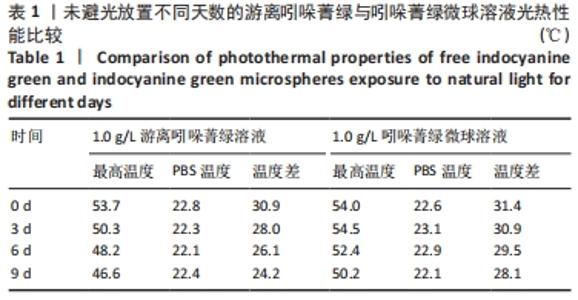
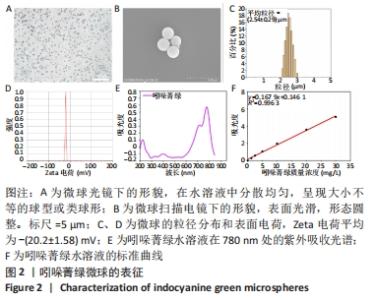
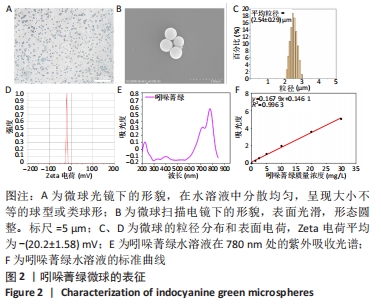
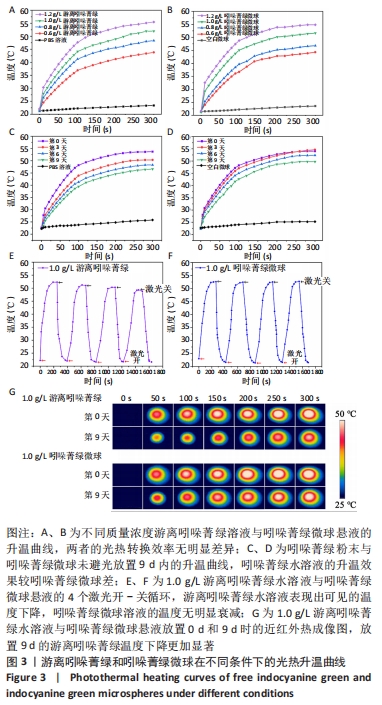
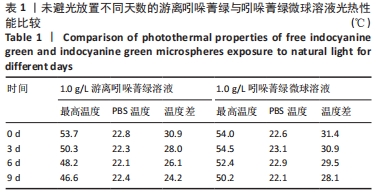
2.2 吲哚菁绿微球的光热性能检测结果 负载前后吲哚菁绿的升温曲线图,见图3A、B。随着吲哚菁绿含量的增加,两种溶液的光热温度均明显增加。当吲哚菁绿质量浓度从0.6 g/L提高到1.2 g/L时,游离吲哚菁绿的温度从43.8 ℃上升到 55.5 ℃;吲哚菁绿微球的温度从 44.3 ℃上升到54.6 ℃,负载前后吲哚菁绿的光热转换效率无明显变化。 将吲哚菁绿粉末与吲哚菁绿微球未避光放置9 d内的升温曲线,见图3C、D。吲哚菁绿水溶液的升温性能下降,升温效果较吲哚菁绿微球差。第0,3,6,9天时,1.0 g/L吲哚菁绿水溶液光照5 min的最大温度值分别为53.7,50.3,48.2,46.6 ℃,温度差为 30.9,28.0,26.1,24.2 ℃,依次衰减;1.0 g/L吲哚菁绿微球溶液同样光照条件下的最大温度值分别为54.0,54.5, 52.4,50.2 ℃,温度差分别为31.4,30.9,29.5,28.1 ℃,温度差相对没有游离吲哚菁绿明显,见表1。此处温度差定义为未避光放置不同天数时,激光照射条件下两种溶液所能达到的最高平衡温度与PBS光照后平衡温度的差值。 "

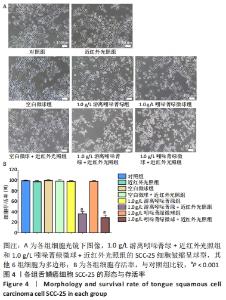
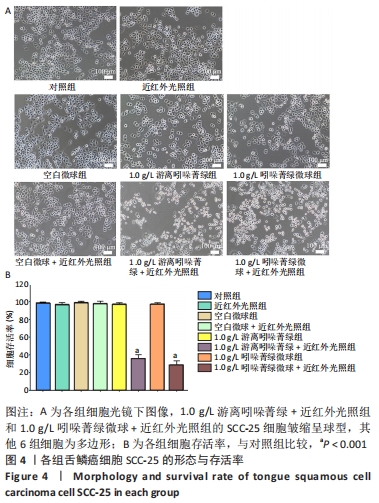
光镜下可见,1.0 g/L游离吲哚菁绿+近红外光照组和1.0 g/L吲哚菁绿微球+近红外光照组的SCC-25细胞皱缩呈球型,其他6相细胞为多边形。空白微球组、1.0 g/L游离吲哚菁绿和1.0 g/L吲哚菁绿微球的细胞活力与对照组相似(P > 0.05),几乎达到100%,表明吲哚菁绿微球具有较高的细胞相容性;1.0 g/L游离吲哚菁绿+近红外光照组和1.0 g/L吲哚菁绿微球+近红外光照组的细胞活力分别为35.8%和28.4%,与对照组比较差异有显著性学意义(P < 0.001),表明游离吲哚菁绿与吲哚菁绿微球均具有光热消融肿瘤细胞的能力。由于实验采用的是未避光放置3 d的吲哚菁绿粉末和吲哚菁绿微球配制的溶液,而吲哚菁绿微球光照组显示出较游离吲哚菁绿光照组更低的细胞存活率,表明负载后吲哚菁绿的光能性能更加稳定。 2.4 微球的细胞相容性 由CCK8实验可知,吲哚菁绿微球具有较高的细胞相容性。 "

| [1] YANAMOTO S, DENDA Y, OTA Y, et al. Postoperative adjuvant therapy for patients with loco-regionally advanced oral squamous cell carcinoma who are at high risk of recurrence. Int J Oral Maxillofac Surg. 2020;49(7):848-853. [2] KORDZINSKA-CISEK I, GRZYBOWSKA-SZATKOWSKA L. Complications of radio- and radiochemotherapy in patients undergoing major salivary gland cancer surgery. Otolaryngol Pol. 2019;73(3):26-31. [3] LIU Y, BHATTARAI P, DAI Z, et al. Photothermal therapy and photoacoustic imaging via nanotheranostics in fighting cancer. Chem Soc Rev. 2019;48(7):2053-2108. [4] LONG S, XU Y, ZHOU F, et al. Characteristics of temperature changes in photothermal therapy induced by combined application of indocyanine green and laser. Oncol Lett. 2019;17(4):3952-3959. [5] CHEN J, LI Q, WANG F, et al. Biosafety, Nontoxic Nanoparticles for VL-NIR Photothermal Therapy Against Oral Squamous Cell Carcinoma. ACS Omega. 2021;6(17):11240-11247. [6] LAU CT, AU DM, WONG K. Application of indocyanine green in pediatric surgery. Pediatr Surg Int. 2019;35(10):1035-1041. [7] HIROHASHI K, ANAYAMA T, WADA H, et al. Lung cancer photothermal ablation by low-power near-infrared laser and topical injection of indocyanine green. Interact Cardiovasc Thorac Surg. 2019;29(5):693-698. [8] LIU C, RUAN C, SHI R, et al. A near infrared-modulated thermosensitive hydrogel for stabilization of indocyanine green and combinatorial anticancer phototherapy. Biomater Sci. 2019;7(4):1705-1715. [9] HWANG J, JIN J. Attachable Hydrogel Containing Indocyanine Green for Selective Photothermal Therapy against Melanoma. Biomolecules (Basel, Switzerland). 2020;10(8):1124. [10] SAXENA V, SADOQI M, SHAO J. Degradation kinetics of indocyanine green in aqueous solution. J Pharm Sci. 2003;92(10):2090-2097. [11] OTT P. Hepatic elimination of indocyanine green with special reference to distribution kinetics and the influence of plasma protein binding. Pharmacol Toxicol. 1998;83 Suppl 2:1-48. [12] WU L, FANG S, SHI S, et al. Hybrid polypeptide micelles loading indocyanine green for tumor imaging and photothermal effect study. Biomacromolecules. 2013;14(9):3027-3033. [13] MIRANDA D, WAN C, KILIAN HI, et al. Indocyanine green binds to DOTAP liposomes for enhanced optical properties and tumor photoablation. Biomater Sci. 2019;7(8):3158-3164. [14] WANG Y, NIU C, FAN S, et al. Indocyanine Green Loaded Modified Mesoporous Silica Nanoparticles as an Effective Photothermal Nanoplatform. Int J Mol Sci. 2020;21(13):4789. [15] MAKADIA HK, SIEGEL SJ. Poly Lactic-co-Glycolic Acid (PLGA) as Biodegradable Controlled Drug Delivery Carrier. Polymers (Basel). 2011;3(3):1377-1397. [16] TAN H, HUANG D, LAO L, et al. RGD modified PLGA/gelatin microspheres as microcarriers for chondrocyte delivery. J Biomed Mater Res B Appl Biomater. 2009;91(1):228-238. [17] RAFIEI P, HADDADI A. Docetaxel-loaded PLGA and PLGA-PEG nanoparticles for intravenous application: pharmacokinetics and biodistribution profile. Int J Nanomedicine. 2017;12:935-947. [18] CHOI S, LEE SH, PARK S, et al. Indocyanine Green-Loaded PLGA Nanoparticles Conjugated with Hyaluronic Acid Improve Target Specificity in Cervical Cancer Tumors. Yonsei Med J. 2021;62(11):1042-1051. [19] CHEN HH, LU IL, LIU TI, et al. Indocyanine green/doxorubicin-encapsulated functionalized nanoparticles for effective combination therapy against human MDR breast cancer. Colloids Surf B Biointerfaces. 2019;177:294-305. [20] SAXENA V, SADOQI M, SHAO J. Indocyanine green-loaded biodegradable nanoparticles: preparation, physicochemical characterization and in vitro release. Int J Pharm. 2004;278(2):293-301. [21] LI Y, PEI Y, ZHANG X, et al. PEGylated PLGA nanoparticles as protein carriers: synthesis, preparation and biodistribution in rats. J Control Release. 2001;71(2):203-211. [22] LAN M, ZHU L, WANG Y, et al. Multifunctional nanobubbles carrying indocyanine green and paclitaxel for molecular imaging and the treatment of prostate cancer. J Nanobiotechnology. 2020;18(1):121. [23] HOUTHOOFD S, VUYLSTEKE M, MORDON S, et al. Photodynamic therapy for atherosclerosis. The potential of indocyanine green. Photodiagnosis Photodyn Ther. 2020;29:101568. [24] YOON HJ, LEE HS, LIM JY, et al. Liposomal Indocyanine Green for Enhanced Photothermal Therapy. ACS Appl Mater Interfaces. 2017; 9(7):5683-5691. [25] SAXENA V, SADOQI M, SHAO J. Enhanced photo-stability, thermal-stability and aqueous-stability of indocyanine green in polymeric nanoparticulate systems. J Photochem Photobiol B. 2004;74(1):29-38. [26] Reinhart MB, Huntington CR, Blair LJ, et al. Indocyanine Green: Historical Context, Current Applications, and Future Considerations. Surg Innov. 2016;23(2):166-175. [27] 余柳丹,罗一帆,陈小菊,等.吲哚菁绿稳定性及光敏性和声敏性研究[J].化学研究与应用,2016,28(3):400-403. [28] LIM HJ, CHIOW A, LEE LS, et al. Novel method of intraoperative liver tumour localisation with indocyanine green and near-infrared imaging. Singapore Med J. 2021;62(4):182-189. [29] WANG Q, SHEN M, LI W, et al. Controlled-release of fluazinam from biodegradable PLGA-based microspheres. J Environ Sci Health B. 2019; 54(10):810-816. [30] LIU Z, YE W, ZHENG J, et al. Hierarchically electrospraying a PLGA@chitosan sphere-in-sphere composite microsphere for multi-drug-controlled release. Regen Biomater. 2020;7(4):381-390. [31] GAIGNAUX A, REEFF J, DE VRIESE C, et al. Evaluation of the degradation of clonidine-loaded PLGA microspheres. J Microencapsul. 2013;30(7): 681-691. [32] HAN S, ZHANG X, LI M. Progress in research and application of PLGA embolic microspheres. Front Biosci (Landmark Ed). 2016;21:931-940. [33] ABULATEEFEH SR, ALKAWAREEK MY, ABDULLAH FR, et al. Preparation of Aqueous Core-Poly(d,l-Lactide-co-Glycolide) Shell Microcapsules With Mononuclear Cores by Internal Phase Separation: Optimization of Formulation Parameters. J Pharm Sci. 2017;106(4):1136-1142. [34] PALAZZO I, LAMPARELLI EP, CIARDULLI MC, et al. Supercritical emulsion extraction fabricated PLA/PLGA micro/nano carriers for growth factor delivery: Release profiles and cytotoxicity. Int J Pharm. 2021;592:120108. [35] SHI NQ, ZHOU J, WALKER J, et al. Microencapsulation of luteinizing hormone-releasing hormone agonist in poly (lactic-co-glycolic acid) microspheres by spray-drying. J Control Release. 2020;321:756-772. [36] Ohta S, Matsuura M, Kawashima Y, et al. Facile fabrication of PEG-coated PLGA microspheres via SPG membrane emulsification for the treatment of scleroderma by ECM degrading enzymes. Colloids Surf B Biointerfaces. 2019;179:453-461. [37] WU B, WU L, HE Y, et al. Engineered PLGA microspheres for extended release of brexpiprazole: in vitro and in vivo studies. Drug Dev Ind Pharm. 2021;47(6):1001-1010. [38] GASPAR MC, PAIS A, SOUSA J, et al. Development of levofloxacin-loaded PLGA microspheres of suitable properties for sustained pulmonary release. Int J Pharm. 2019;556:117-124. [39] BUTREDDY A, GADDAM RP, KOMMINENI N, et al. PLGA/PLA-Based Long-Acting Injectable Depot Microspheres in Clinical Use: Production and Characterization Overview for Protein/Peptide Delivery. Int J Mol Sci. 2021;22(16):8884. [40] ZHAI P, CHEN XB, SCHREYER DJ. PLGA/alginate composite microspheres for hydrophilic protein delivery. Mater Sci Eng C Mater Biol Appl. 2015; 56:251-259. [41] PARK JS, YANG HN, JEON SY, et al. The use of anti-COX2 siRNA coated onto PLGA nanoparticles loading dexamethasone in the treatment of rheumatoid arthritis. Biomaterials. 2012;33(33):8600-8612. [42] LIU R, MA GH, WAN YH, et al. Influence of process parameters on the size distribution of PLA microcapsules prepared by combining membrane emulsification technique and double emulsion-solvent evaporation method. Colloids Surf B Biointerfaces. 2005;45(3-4):144-153. [43] KONAN YN, CERNY R, FAVET J, et al. Preparation and characterization of sterile sub-200 nm meso-tetra(4-hydroxylphenyl)porphyrin-loaded nanoparticles for photodynamic therapy. Eur J Pharm Biopharm. 2003; 55(1):115-124. [44] JAQUE D, MARTINEZ ML, DEL RB, et al. Nanoparticles for photothermal therapies. Nanoscale. 2014;6(16):9494-9530. |
| [1] | Liu Xiaolin, Mu Xinyue, Ma Ziyu, Liu Shutai, Wang Wenlong, Han Xiaoqian, Dong Zhiheng. Effect of hydrogel-loaded simvastatin microspheres on osteoblast proliferation and differentiation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 998-1003. |
| [2] | Huang Yanni, Yang Hua, Yang Dongmei, Hu Xulin, Gao Hong, Huang Yina. Rat bone defect repaired with polytrimethylene carbonate/beta-tricalcium phosphate microsphere scaffold [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1856-1862. |
| [3] | Ye Xuwen, Gu Yong, Chen Liang. Curcumin loaded injectable microspheres retard progression of intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1884-1891. |
| [4] | Gan Tian, Wang Wenyuan, Yan Shujin, Hao Lan, Ran Haitao, Wang Zhigang, Xia Jizhu. Near infrared photoresponsive nanoparticles loaded with LXR agonists for photothermal immunotherapy [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1863-1869. |
| [5] | FAN Yaru, LI Ruixin , LI Fengji, LUO Rui, LIU Hao, YAN Yingbin. Characterization and photothermal effect of indocyanine green encapsulated poly lactic acid-co-glycolic acid microspheres [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(在线): 1-6. |
| [6] | Le Guoping, Zhang Ming, Xi Licheng, Luo Hanwen. Preparation and in vitro evaluation of vancomycin hydrochloride@polylactic acid-glycolic acid copolymer-chitosan-hyaluronic acid composite sustained-release microspheres [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 528-534. |
| [7] | Liu Tongbin, Lin Peng, Zhang Xiaoming, Dong Xiling, Cao Fei, Wang Le, Guo Xinxing. Optimization of preparation method of atorvastatin calcium sustained-release microspheres [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 535-539. |
| [8] | Zhang Yixin, Luo Qian, Liang Hanwen, Chen Jianlin, Zhao Nan, He Bin. In vivo neocollagenesis of injectable poly(L-lactic acid) microspheres [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(34): 5448-5453. |
| [9] | Jiang Haoran, Gao Jianming, Lin Wancheng, Li Ting, Li Huo, Wang Peng, Feng Jing, Meng Haoye, Peng Jiang, Ding Lixiang. Gelatin-alginate composite microspheres and gels for cartilage damage repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(28): 4452-4457. |
| [10] | Liu Ming, Wang Kai. Chitosan/alginate composite scaffold combined with hawthorn leaf flavonoids for spinal cord injury repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(28): 4466-4471. |
| [11] | He Jiachen, Liu Chang, Chen Chichi, Shi Qin. Preparation and in vitro evaluation of injectable microspheres loaded with cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(28): 4483-4488. |
| [12] | Li Xikai, Meng Yongchun, Fu Li, Liu Qing, Wang Xiao, Gu Chengxu, Huang Fei. Preparation and in vitro release of curcumin sustained-release microspheres [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(28): 4489-4493. |
| [13] | Zhu Meiying, Jiang Liming, Liu Shuangwei, Ma Mingqi, Yang Yutong. Application of drug delivery system in the field of periodontal tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(27): 4423-4428. |
| [14] | Chen Sheng, Yang Yan, Ding Liang, Ye Chuanjin, Zhang Lei, Xia Shu, Li Huiling, Huang Xiaofeng. Establishment and identification of an immortalized oral cancer-related fibroblast line [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(24): 3808-3813. |
| [15] | Xu Xinzhi, Zhang Yue, Jin Ying, Jin Chunxiang. Preparation and imaging experiment of a new type of safe near-infrared luminescent nanoparticles [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(22): 3450-3454. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||