Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (12): 1877-1883.doi: 10.12307/2023.011
Previous Articles Next Articles
Nanocellular vesicles loaded with curcumin promote wound healing in diabetic mice
Su Meng1, Wang Xin2, Zhang Jin1, Bei Ying1, Huang Yu1, Zhu Yanzhao1, Li Jiali1, Wu Yan1
- 1College of Life Sciences, Mudanjiang Medical University, Mudanjiang 157000, Heilongjiang Province, China; 2Department of Pain Management, Qingdao Municipal Hospital, Qingdao 266000, Shandong Province, China
-
Received:2021-10-28Accepted:2021-12-28Online:2023-04-28Published:2022-07-30 -
Contact:Wu Yan, Associate professor, College of Life Sciences, Mudanjiang Medical University, Mudanjiang 157000, Heilongjiang Province, China -
About author:Su Meng, Master, College of Life Sciences, Mudanjiang Medical University, Mudanjiang 157000, Heilongjiang Province, China -
Supported by:the Natural Science Foundation of Heilongjiang Province, No. LH2020H076 (to WY); the Graduate Student Innovation Research Project of Mudanjiang Medical University in 2020, No. YJSCX-MY32 (to WY); the Basic Scientific Research Project of University Belongs to Heilongjiang, No. 2019-KYYWF-0987 (to WY)
CLC Number:
Cite this article
Su Meng, Wang Xin, Zhang Jin, Bei Ying, Huang Yu, Zhu Yanzhao, Li Jiali, Wu Yan. Nanocellular vesicles loaded with curcumin promote wound healing in diabetic mice[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1877-1883.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
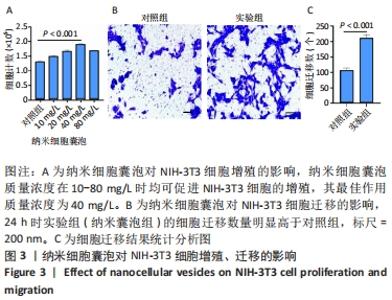
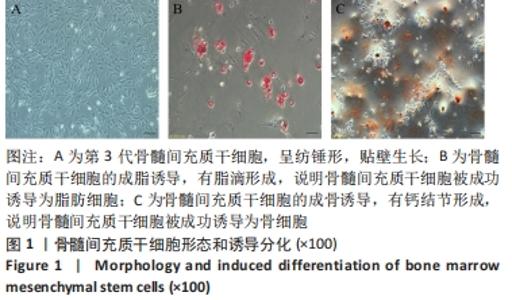
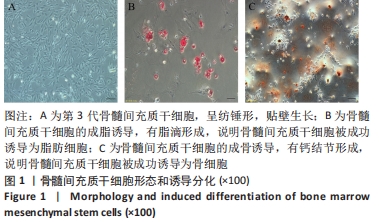
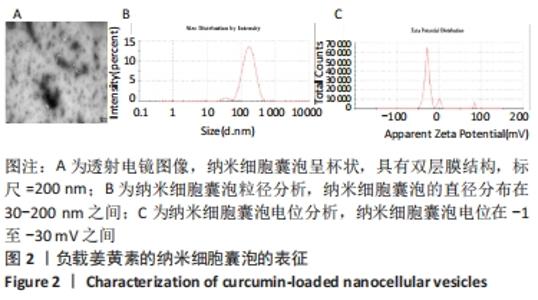
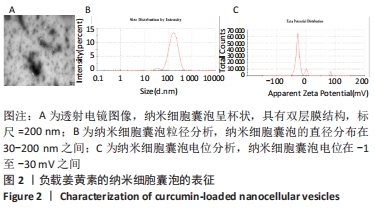
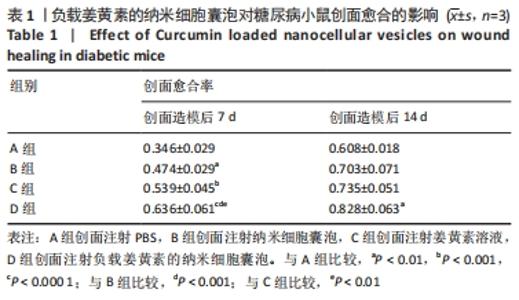
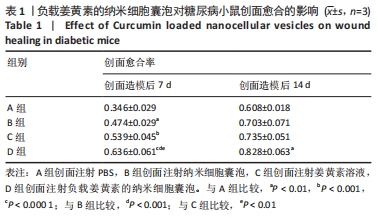
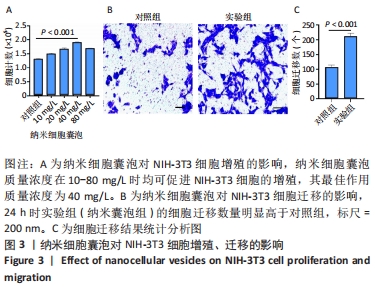
2.3 负载姜黄素的纳米细胞囊泡的包封率和载药率 首先,制作了姜黄素在甲醇溶液中的标准曲线,标准曲线拟合度达到99%以上。用甲醇溶解负载姜黄素的纳米细胞囊泡后做了连续7 d的吸光度值检测,发现纳米细胞囊泡中的姜黄素在24 h后就已完全溶解在甲醇中,所以应用24 h时姜黄素在甲醇中的浓度,根据标准曲线计算出姜黄素的量,根据公式计算出纳米细胞囊泡中姜黄素包封率为42%。通过BCA法测得纳米细胞囊泡的质量浓度为0.8 g/L。根据载药率公式计算出纳米细胞囊泡的姜黄素载药率为2.3%。 2.4 纳米细胞囊泡对NIH-3T3细胞增殖、迁移的影响 不同质量浓度纳米细胞囊泡对 NIH-3T3细胞增殖的影响,见图3A,实验结果显示,纳米细胞囊泡的质量浓度在10-80 mg/L时均可促进NIH-3T3细胞的增殖,其最佳作用质量浓度为40 mg/L。因此选择40 mg/L的纳米细胞囊泡继续后续实验。 为了研究纳米细胞囊泡是否影响NIH-3T3细胞的迁移能力,进行了Transwell实验。细胞迁移实验结果,见图3B,C,24 h时实验组(纳米囊泡组)的细胞迁移数量明显高于对照组。 "
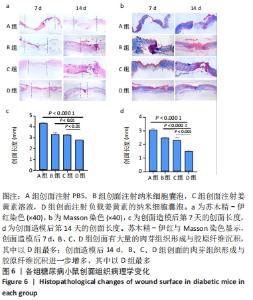
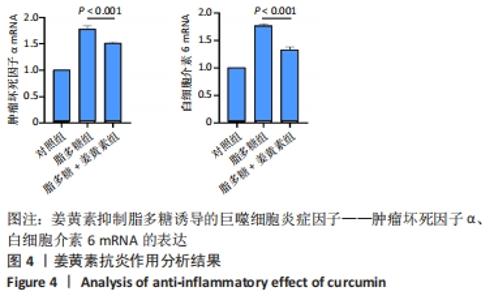
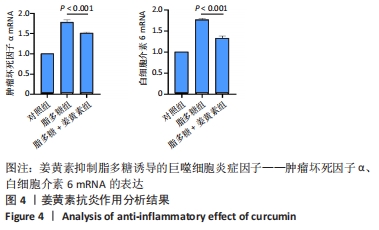
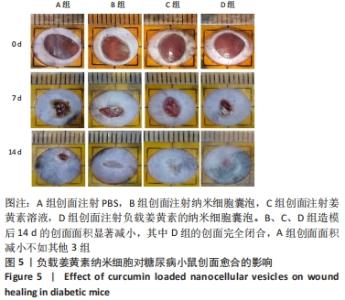
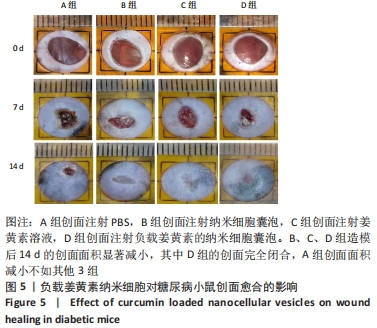
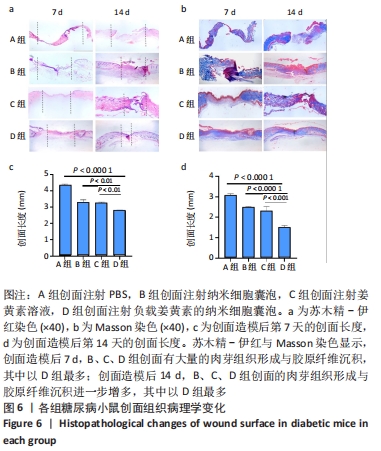
2.6.3 病理观察创面组织学变化 通过苏木精-伊红染色来评估负载姜黄素的纳米细胞囊泡的创面愈合过程中病理学变化,见图6a。A组在创面造模后第7天的肉芽组织较少,而其他3组的创面具有丰富的肉芽组织形成,其中以D组最多;各组创面长度也有显著差异,见图6c。其中D组中最短,其次分别是B、C、A组。创面造模后第14天,4组创面处肉芽组织较第7 天均有不同程度的增多,其中以D组最多,见图6d;尽管与第7天相比,4组的创面长度均有明显减少,但相比于其他组,D组创面长度最短。 适当的胶原蛋白沉积和重塑可以提高组织的抗张强度,并产生更好的愈合效果[25]。各组创面Masson染色结果,见图6b。创面造模后第7天,B、C、D组创面显示出丰富的胶原纤维,并且胶原纤维的数量随愈合时间的增加而增加,尤其是D组,A组创面处的胶原沉积较少。创面造模后第14天,4组创面都有显示出比第7天时更致密且丰富的胶原纤维沉积,其中以D组效果最好。Masson染色结果提示,负载姜黄素的纳米细胞囊泡可以改善胶原蛋白在创面部位的沉积,从而加速皮肤再生。 "
| [1] LI Y, XU TZ, TU ZL, et al. Bioactive antibacterial silica-based nanocomposites hydrogel scaffolds with high angiogenesis for promoting diabetic wound healing and skin repair. Theranostics. 2020; 10(11):4929-4943. [2] International Diabetes Federation. IDF Diabetes Atlas-10th Edition. DiabetesAtlas http://www.diabetesatlas.org/ (2021). [3] PATEL S, SRIVASTAVA S, SINGH MR, et al, Mechanistic insight into diabetic wounds: Pathogenesis, molecular targets and treatment strategies to pace wound healing. Biomed Pharmacother. 2019;112: 108615. [4] Bakker K, Apelqvist J, Lipsky BA, et al. The 2015 IWGDF guidance documents on prevention and management of foot problems in diabetes: development of an evidence-based global consensus. Diabetes Metab Res Rev. 2016;32 Suppl 1:2-6. [5] Chan YW, So C, Yau KL, et al. Adipose-derived stem cells and cancer cells fuse to generate cancer stem cell-like cells with increased tumorigenicity. J Cell Physiol. 2020;235(10):6794-6807. [6] Riedl J, Popp C, Eide C, et al. Mesenchymal stromal cells in wound healing applications: role of the secretome, targeted delivery and impact on recessive dystrophic epidermolysis bullosa treatment. Cytotherapy. 2021;23(11):961-973. [7] Choi YJ, Yi HG, Kim SW, et al. 3Dcell printedtissue analogues:anew platformfor theranostics. Theranostics. 2017;7(12):3118. [8] Yang JY, Chen ZY, Pan DY, et al. Umbilical Cord-Derived Mesenchymal Stem Cell-Derived Exosomes Combined Pluronic F127 Hydrogel Promote Chronic Diabetic Wound Healing and Complete Skin Regeneration. Int J Nanomedicine. 2020;15:5911-5926. [9] Xunian Z ,Kalluri R. The Biology and Therapeutic Potential of Mesenchymal Stem Cells derived Exosomes. Cancer Sci. 2020;111(9): 3100-3110. [10] Ferguson SW, Nguyen J. Exosomes as therapeutics: The implications of molecular composition and exosomal heterogeneity. J Control Release. 2016;228:179-190. [11] Hu YQ, Wu B, Xiong Y, et al. Cryogenic 3D Printed Hydrogel Scaffolds Loading Exosomes Accelerate Diabetic Wound Healing. Chem Eng J. 2021;(9):130634. [12] Huang JH, Yu M , Yin WJ, et al. Development of a novel RNAi therapy: Engineered miR-31 exosomes promoted the healing of diabetic wounds. Bioact Mater. 2021;6(9):2841-2853. [13] Guo SC, Tao SC, Yin WJ, et al. Exosomes derived from platelet-rich plasma promote the re-epithelization of chronic cutaneous wounds via activation of YAP in a diabetic rat model. Theranostics. 2017;7(1):81-96. [14] Jang SC, Kim OY, Yoon CM, et al. Bioinspired exosome-mimetic nanovesicles for targeted delivery of chemotherapeutics to malignant tumors. ACS Nano. 2013;7(9):7698-7710. [15] AlQahtani SA, Harisa GI, Badran MM, et al. Nano-erythrocyte membrane-chaperoned 5-fluorouracil liposomes as biomimetic delivery platforms to target hepatocellular carcinoma cell lines. Artif Cells Nanomed Biotechnol. 2019;47(1):989-996. [16] Fereydouni N, Darroudi M, Movaffagh J, et al. Curcumin nanofibers for the purpose of wound healing. J Cell Physiol. 2019; 234(5):5537-5554. [17] Mokhtari M, Razzaghi R, Mansooreh MH. The effects of Curcumin intake on wound healing and metabolic status in patients with diabetic foot ulcer: A randomized, double-blind, placebo-controlled trial. Phytother Res. 2021;35(4):2099-2107. [18] Mohanty C, Pradhan J. A human epidermal growth factor-Curcumin bandage bioconjugate loaded with mesenchymal stem cell for in vivo diabetic wound healing. Mater Sci Eng C Mater Biol Appl. 2020;111:110751. [19] Li F, Shi YJ, Liang J, et al. Curcumin-loaded chitosan nanoparticles promote diabetic wound healing via attenuating inflammation in a diabetic rat model. J Biomater Appl. 2019;34(4):476-486. [20] Feng T, Wei YM, Lee RJ, et al. Liposomal Curcumin and its application in cancer. Int J Nanomedicine. 2017;12:6027-6044. [21] Ghufran H, Mehmood A, Azam M, et al. Curcumin preconditioned human adipose derived stem cells co-transplanted with platelet rich plasma improve wound healing in diabetic rats. Life Sci. 2020;257: 118091. [22] Liu YF, Li Z, Li JJ, et al. Stiffness-mediated mesenchymal stem cell fate decision in 3D-bioprinted hydrogels. Burns Trauma. 2020;8:tkaa029. [23] Fan JB, Lee CS, Kim S, et al. Generation of Small RNA-Modulated Exosome Mimetics for Bone Regeneration. ACS Nano. 2020;14(9): 11973-11984. [24] Wang QR, Ye CQ, Sun SK, et al. Curcumin attenuates collagen-induced rat arthritis via anti-inflammatoryand apoptotic effects. Int Immunopharmaco. 2019;72:292-300. [25] Wang CG, Wang M, Xu TZ, et al. Engineering Bioactive Self-Healing Antibacterial Exosomes Hydrogel for Promoting Chronic Diabetic Wound Healing and Complete Skin Regeneration. Theranostics. 2019; 9(1):65-76. [26] Ahmadi H, Amini A, Fathabady FF, et al.Transplantation of photobiomodulation-preconditioned diabetic stem cells accelerates ischemic wound healing in diabetic rats. Stem Cell Res Ther. 2020; 11(1):494. [27] Alamdari NM, Mehraneroodi B, Gholizadeh B, et al. The efficacy of negative pressure wound therapy compared with conventional dressing in treating infected diabetic foot ulcers: a randomized controlled trial. Int J Diabetes Dev C. 2021(9).doi:10.1007/s13410-021-00941-9 [28] Abolgheit S, Abdelkader S, Aboushelib M, et al. Bone marrow-derived mesenchymal stem cells and extracellular vesicles enriched collagen chitosan scaffold in skin wound healing (a rat model). J Biomater Appl. 2021;36(1):128-139. [29] Nikfarjam S, Rezaie J , Zolbanin NM, et al. Mesenchymal Stem Cell Derived-Exosomes: A Modern Approach in Translational Medicine. J Transl Med. 2020;18(1):449. [30] Adalberto EJ, Argenta DF, Bazzo GC, et al. Development of Curcumin-loaded chitosan/pluronic membranes for wound healing applications. Int J Biol Macromol. 2020;163:167-179. [31] Walter MNM, Wright KT, Fuller HR, et al. Mesenchymal stem cell-conditioned medium accelerates skin wound healing: an in vitro study of fibroblast and keratinocyte scratch assays. Exp Cell Res. 2010; 316(7):1271-1281. [32] Rong XL, Chu WH, Zhang HY, et al. Antler stem cell-conditioned medium stimulates regenerative wound healing in rats. Stem Cell Res Ther. 2019;10(1):326. [33] Huang YZ, Gou M, Da LC, et al. Mesenchymal Stem Cells for Chronic Wound Healing: Current Status of Preclinical and Clinical Studies. Tissue Eng Part B Rev. 2020;26(6):555-570. [34] PELIZZO G, AVANZINI MA, ICARO CORNAGLIA A, et al. Mesenchymal stromal cells for cutaneous wound healing in a rabbit model: pre-clinical study applicable in the pediatric surgical setting. J Transl Med. 2015;13(1):1-14. [35] MURPHY KC, WHITEHEAD J, FALAHEE PC, et al. MμLtifactorial experimental design to optimize the anti-inflammatory and proangiogenic potential of mesenchymal stem cell spheroids. Stem Cells. 2017;35(6):1493-1504. [36] CAPPUZZELLO C, DONI A, DANDER E, et al. Mesenchymal stromal cell-derived PTX3 promotes wound healing via fibrin remodeling. J Invest Dermatol. 2016; 136(1):293-300. [37] WANG C, WANG M, XU T, et al. Engineering bioactive self-healing antibacterial exosomes hydrogel for promoting chronic diabetic wound healing and complete skin regeneration. Theranostics. 2019;9(1):65. [38] Zhang W, Bai XZ, Zhao B, et al. Cell-free therapy based on adipose tissue stem cell-derived exosomes promotes wound healing via the PI3K/Akt signaling pathway. Exp Cell Res. 2018;370(2):333-342. [39] Liu HM, Deng SC, Han L, et al. Mesenchymal stem cells, exosomes and exosome-mimics as smart drug carriers for targeted cancer therapy. Colloids Surf B Biointerfaces. 2021;209(Pt 1):112163. [40] van der Meel R, Fens MHAM, Vader P, et al. Extracellular vesicles as drug delivery systems: Lessons from the liposome field J Control Release. 2014;195:72-85. [41] Li F, Shi YJ, Liang J, et al. Curcumin-loaded chitosan nanoparticles promote diabetic wound healing via attenuating inflammation in a diabetic rat model. J Biomater Appl. 2019;34(4):476-486. [42] Panchatcharam M, Miriyala S, Gayathri VS, et al. Curcumin improves wound healing by modulating collagen and decreasing reactive oxygen species. Mol Cell Biochem. 2006;290(1-2):87-96. [43] Aggarwal BB, Sung B. Pharmacological basis for the role of curcumin in chronic diseases: an age-old spice with modern targets. Trends Pharmacol Sci. 2009;30(2):85-94. [44] Lakhal S, Wood MJA. Exosome nanotechnology: An emerging paradigm shift in drug delivery : Exploitation of exosome nanovesicles for systemic in vivo delivery of RNAi heralds new horizons for drug delivery across biological barriers. Bioessays. 2011;33(10):737-741. |
| [1] | He Xi, Wan Yu, Tang Yuting, Yang Anning, Wu Kai, Jiao Yun, Bai Zhigang, Jiang Yideng, Shen Jiangyong. Erastin inhibits proliferation of hypertrophic scar fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-. |
| [2] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [3] | Ke Weiqiang, Chen Xianghui, Chen Xiaoling, Meng Jie, Ma Yanlin. Rituximab combined with autologous peripheral blood stem cell transplantation in the treatment of diffuse large B-cell lymphoma and the expression of related factors [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 915-920. |
| [4] | Hu Xinming, Qiao Yanhua, Wang Xiaofan, Li Linyu, Zhao Bing. Mechanism of long non-coding RNA plasmacytoma variant translocation 1 involved in pelvic organ prolapse [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 669-675. |
| [5] | Li Rui, Liu Zhen, Guo Zige, Lu Ruijie, Wang Chen. Aspirin-loaded chitosan nanoparticles and polydopamine modified titanium sheets improve osteogenic differentiation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 374-379. |
| [6] | Ye Xuwen, Gu Yong, Chen Liang. Curcumin loaded injectable microspheres retard progression of intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1884-1891. |
| [7] | Ma Hongfeng, Ren Qing, Cheng Wanmin, Wang Jiguo, Meng Qingyan. Effects of Sanhuang gel on wound healing, basic fibroblast growth factor and interleukin 17 levels of auricular skin defect in rabbits [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1842-1847. |
| [8] | He Xi, Ma Fang, Wan Yu, Tang Yuting, Ma Shengchao, Jiang Yideng, Shen Jiangyong. miR-382-3p inhibits the proliferation of human hypertrophic scar fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(11): 1758-1764. |
| [9] | Ma Munan, Xie Jun, Sang Yuchao, Huang Lei, Zhang Guodong, Yang Xiaoli, Fu Songtao. Electroacupuncture combined with bone marrow mesenchymal stem cells in the treatment of chemotherapy-induced premature ovarian insufficiency in rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(1): 1-7. |
| [10] | Huang Bin, Zheng Jinxu, Zhang Jun. Progress of non-coding RNAs in stem cell abnormality of idiopathic pulmonary fibrosis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(1): 130-137. |
| [11] | Liu Wentao, Feng Xingchao, Yang Yi, Bai Shengbin. Effect of M2 macrophage-derived exosomes on osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(在线): 1-6. |
| [12] | Li Qin, Mao Shuangfa, Li Min, Cheng Jiyan. Protective effect and mechanism of dendrobium on fibroblasts damaged by ultraviolet B [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1228-1233. |
| [13] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [14] | Liu Feng, Peng Yuhuan, Luo Liangping, Wu Benqing. Plant-derived basic fibroblast growth factor maintains the growth and differentiation of human embryonic stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1032-1037. |
| [15] | Fang Xiaolei, Leng Jun, Zhang Chen, Liu Huimin, Guo Wen. Systematic evaluation of different therapeutic effects of mesenchymal stem cell transplantation in the treatment of ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1085-1092. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||