Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (12): 1811-1816.doi: 10.12307/2023.135
Previous Articles Next Articles
Properties of force growth factor E peptide bionic bone matrix with polyethylene glycol derivative hydrogel as a carrier
Peng Kun
- Chongqing Medical and Pharmaceutical College, Chongqing 401331, China
-
Received:2022-03-14Accepted:2022-05-17Online:2023-04-28Published:2022-07-30 -
Contact:Peng Kun, MD, Associate professor, Chongqing Medical and Pharmaceutical College, Chongqing 401331, China -
About author:Peng Kun, MD, Associate professor, Chongqing Medical and Pharmaceutical College, Chongqing 401331, China -
Supported by:the Natural Science Foundation of Chongqing, No. cstc2018jcyjAX0828 (to PK); the Third Batch of Funding Plan for Young Backbone Teachers in Chongqing Institutions of Higher Learning, No. [2016]41 (to PK)
CLC Number:
Cite this article
Peng Kun. Properties of force growth factor E peptide bionic bone matrix with polyethylene glycol derivative hydrogel as a carrier[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1811-1816.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
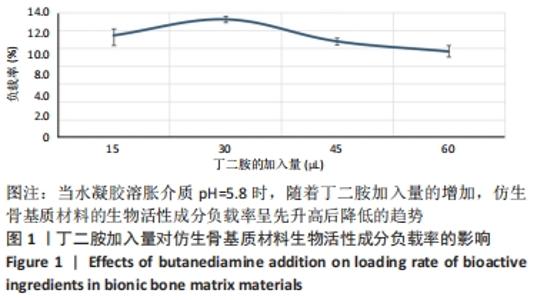
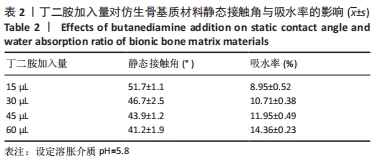
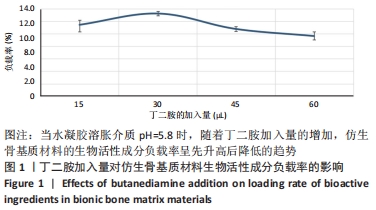
2.1 水凝胶组成对仿生骨基质材料生物活性成分负载率的影响 水凝胶的化学组成及空间结构能够决定其对生物活性物质的负载率。实验中,作者改变加入水凝胶中丁二胺的量,从而达到调节水凝胶化学组成及空间结构的目的。当丁二胺量增加时,水凝胶中包含的支链结构和物理连接点就会增加,空间结构的稳定性更好,反之则相反。当丁二胺增多时,聚乙二醇衍生物水凝胶酰亚胺环上面的羰基碳容易受到亲核试剂的攻击而发生亚胺开环反应,进而发生化学接枝,且接枝率更高,反之则相反。 实验中,利用紫外分光光度计和公式,计算PAPI-BDA15、PAPI-BDA30、PAPI-BDA45和PAPI-BDA60水凝胶中生物活性物质的负载量,见图1。实验数据显示,在pH=5.8(考虑一般骨损伤情况下,骨周围组织发生炎症反应,可出现弱酸性,故模拟弱酸性条件)的溶胀介质中,当丁二胺的加入量从15 μL增加到60 μL时,水凝胶中具备的支链结构和物理连接点增多,然而仿生骨基质材料负载的生物活性物质量呈先增多再减少的现象,仿生骨基质材料生物活性成分负载率依次从(11.43±0.02)%增加到(13.23±0.02)%,再下降到(10.77±0.01)%和(9.63±0.02)%。从实验数据看,PAPI-BDA30/MGF-Ct24E -MPLA-EG-g-HAP能够获得最优的负载率。 "
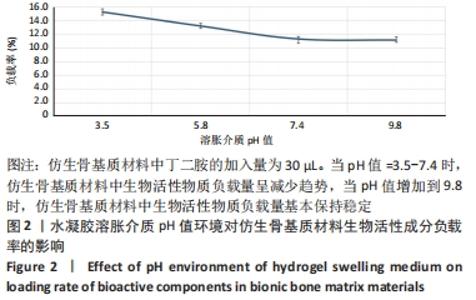
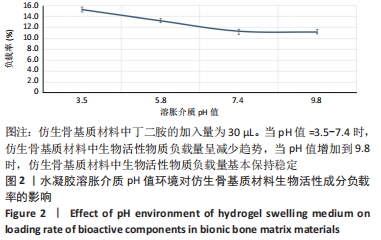
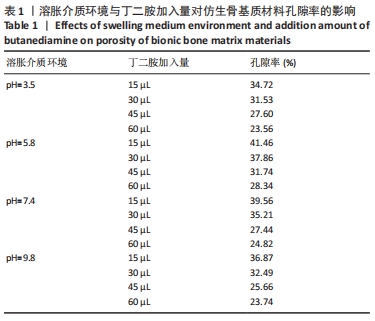
从图中数据可得出,生物活性物质的负载率与环境pH值的变化有关。从图可知,随着pH值从3.5增加到9.8,仿生骨基质材料中生物活性成分的负载率从(15.23±0.02)%下降到(11.13±0.02)%;当pH值从7.4增加到9.8时,生物活性成分负载率变化趋于平缓。当溶胀介质pH=3.5时,PAPI-BDA30凝胶因含有亚氨基而带有正电荷;又因为MGF-Ct24E-MPLA-EG-g-HAP含有碱性氨基酸残基,在同样溶胀介质环境下也可带有正电荷,由于同种电荷的排斥作用,水凝胶可形成的内部空间将更大,有利于MGF-Ct24E-MPLA-EG-g-HAP进入水凝胶,从而呈现较高的负载。两种正电荷可随着溶胀介质的pH值增大而减弱,此时电荷排斥作用被减弱,水凝胶内部空间受到挤压,不利于MGF-Ct24E-MPLA-EG-g-HAP进入水凝胶,从而呈现负载降低。当pH值达到7.4后,PAPI-BDA30凝胶基团很难再获得氢离子,即便pH值再提高,其水凝胶内部空间变化也不明显,进而导致负载情况变化不大。 2.3 仿生骨基质材料的孔隙率表征结果 在不同溶胀介质中,4种仿生骨基质材料的孔隙率见表1所示。实验数据显示,当溶胀介质保持不变时,改变材料组成,即调节加入丁二胺的量,可改变基质材料的孔隙率,随着丁二胺的加入量变化,4种仿生基质材料的孔隙率在23.56%-41.46%之间,且随着丁二胺加入量的增加,仿生骨基质材料的孔隙率逐步降低,表明材料的孔隙率与其交联度呈反向相关。当介质的pH值发生变化,各基质材料的孔隙率有所不同,当丁二胺加入量保持不变时,随着介质pH值增大,仿生骨基质材料的孔隙率呈倒“V”型变化。 "
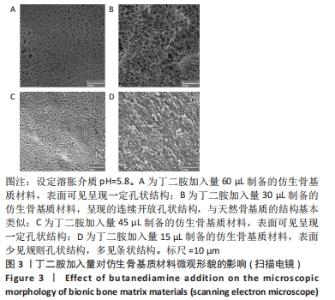
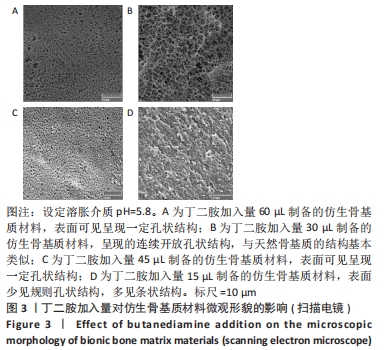
2.5 仿生骨基质材料的表面形貌表征观察结果 图3为4种仿生骨基质材料的扫描电镜图。其中,PAPI-BDA60/MGF-Ct24E-MPLA-EG-g-HAP与PAPI-BDA45/MGF-Ct24E-MPLA-EG-g-HAP的表面结构可见呈现一定孔状结构,但两者整体表面形貌差异不大。PAPI-BDA15/MGF-Ct24E-MPLA-EG-g-HAP的表面少见规则孔状结构,多见条状结构,可能是丁二按的引入量不足,水凝胶交联不够充分而导致。需要注意的是,PAPI-BDA30/MGF-Ct24E-MPLA-EG-g-HAP基质材料呈现的连续开放孔状结构较优,与天然骨基质的结构基本类似。利用分析软件,估算PAPI-BDA/MGF-Ct24E-MPLA-EG-g-HAP系列材料的孔径大小,其有效分布范围为230-690 nm,且孔径大小与丁二胺的加入量呈反向相关。"
| [1] CAMPANA V, MILANO G, PAGANO E, et al. Bone substitutes in orthopaedic surgery: from basic science to clinical practice. J Mater Sci Mater Med. 2014;25:2445-2461. [2] QUARTO R, GIANNONI P. Bone tissue engineering: past-present-future Methods. Mol Biol. 2016;1416:21-33. [3] SADEGHINIA A, SOLTANI S, AGHAZADEH M, et al. Design and fabrication of clinoptilolite-nanohydroxyapatite/chitosan-gelatin composite scaffold and evaluation of its effects on bone tissue engineering. J Biomed Mater Res A. 2020;108(2):221-233. [4] CHEN P, LIU L, PAN J, et al. Biomimetic composite scaffold of hydroxyapatite/gelatin-chitosan core-shell nanofibers for bone tissue engineering. Mater Sci Eng C Mater Biol Appl. 2019;97:325-335. [5] MUSTAFA R, RAINER L, PHILIPP R, et al. Bone response to biomimetic implants delivering BMP-2 and VEGF:An immunohistochemical study. J Craniomaxillofac Surg. 2013;41:826-835. [6] STOCKMANN P, PARK J, VON WILMOWSKY C, et al. Guided bone regeneration in pig calvarial bone defects using autologous mesenchymal stem/progenitor cells-a comparison of different tissue sources. J Craniomaxillofac Surg. 2012;40:310-320. [7] 赵鸣岐,黄威嫔,胡米,等.生物医用材料表面高分子基涂层的功能化构筑[J].材料导报,2019,33(1):27-33. [8] YU W, LI R, LONG J, et al. Use of a three-dimensional printed polylactide-coglycolide/tricalcium phosphate composite scaffold incorporating magnesium powder to enhance bone defect repair in rabbits. J Orthop Transl. 2019;16:62-70. [9] GUGLIELMOTTI MB, OLMEDO DG, CABRINI RL. Research on implants and osseointegration. Periodontology 2000. 2019;79(1):178-189. [10] JANUARIYASA IK, ANA ID, YUSUF Y. Nanofibrous poly(vinyl alcohol)/chitosan contained carbonated hydroxyapatite nanoparticles scaffold for bone tissue engineering. Mater Sci Eng C Mater Biol Appl. 2020; 107:110347. [11] HU Y, CHEN J, FAN T, et al. Biomimetic mineralized hierarchical hybrid scaffolds based on in situ synthesis of nano-hydroxyapatite/chitosan/chondroitin sulfate/hyaluronic acid for bone tissue engineering. Colloids Surf B. 2017;157:93-100. [12] COJOCARU FD, BALAN V, POPA MI, et al. Biopolymer calcium phosphates composites with inclusions of magnetic nanoparticles for bone tissue engineering. Int J Biol Macromol. 2019;125:612-620. [13] KUTTAPPAN S, MATHEW D, NAIR MB. Biomimetic composite scaffolds containing bioceramics and collagen/gelatin for bone tissue engineering - A mini review. Int J Biol Macromol. 2016;93:1390-1401. [14] PATEL PP, BUCKLEY C, TAYLOR BL, et al. Mechanical and biological evaluation of a hydroxyapatite-reinforced scaffold for bone regeneration. J Biomed Mater Res A. 2019;107(4):732-741. [15] HU J, HOU Y, PARK H, et al. Visible light crosslinkable chitosan hydrogels for tissue engineering. Acta Biomater. 2012;8:1730-1738. [16] NAZEER MA, YILGOR E, YILGOR I. Intercalated chitosan/hydroxyapatite nanocomposites: promising materials for bone tissue engineering applications. Carbohydr Polym. 2017;175:38-46. [17] ZHU W, WANG M, FU YB, et al. Engineering a biomimetic three-dimensional nanostructured bone model for breast cancer bone metastasis study. Acta Biomater. 2015;14:164-174. [18] IM O, LI J, WANG M, et al. Biomimetic three-dimensional nanocrystalline hydroxyapatite and magnetically synthesized single-walled carbon nanotube chitosan nanocomposite for bone regeneration. Int J Nanomed. 2012;7:2087-2099. [19] SUBHADRA G, ARVIND SM. Biomimetic nanocomposites of carboxymethylcellulose-hydroxyapatite: Novel three dimensional load bearingbone grafts. Colloids Surf B Biointerfaces. 2014;115:182-190. [20] WANG JC, LIU CZ. Biomimetic Collagen/Hydroxyapatite Composite Scaffolds: Fabrication and Characterizations. J Bionic Eng. 2014;11:600-609. [21] RONG CB, NAOKI K, CHEN GP. Influence of surfaces modified with biomimetic extracellular matriceson adhesion and proliferation of mesenchymal stem cells andosteosarcoma cells. Colloids Surf B Biointerfaces. 2015;126:381-386. [22] SARKAR SK, LEE BT. Hard tissue regeneration using bone substitutes: an update on innovations in materials. Korean J Intern Med. 2015;30:279-293. [23] DE WITTE TM, FRATILA-APACHITEI LE, ZADPOOR AA, et al. Bone tissue engineering via growth factor delivery: from scaffolds to complex matrices. Regen Biomater. 2018;5:197-211. [24] ARABNEJAD S, BURNETT JOHNSTON R, PURA JA, et al. High-strength porous biomaterials for bone replacement: a strategy to assess the interplay between cell morphology, mechanical properties, bone ingrowth and manufacturing constraints. Acta Biomater. 2016;30:345-356. [25] LIU F, MAO Z, ZHANG P, et al. Functionally graded porous scaffolds in multiple patterns: new design method, physical and mechanical properties. Mater Des. 2018;160: 849-860. [26] WU S, LIU XM, KELVIN WK, et al. Biomimetic porous scaffolds for bone tissue engineering. Mater Sci Eng R Rep. 2014;80:1-36. [27] WANG T, ZHAI Y, NUZZO M, et al. Layer-by-layer nanofiber-enabled engineering of biomimetic periosteum for bone repair and reconstruction. Biomaterials. 2018;182:279-288. [28] JIANG H, ZUO Y, ZOU Q, et al. Biomimetic spiral-cylindrical scaffold based on hybrid chitosan/cellulose/nano-hydroxyapatite membrane for bone regeneration. ACS Appl Mater Interfaces. 2013;5(22):12036-12044. [29] AMIRTHALINGAM S, RAMESH A, LEE SS, et al. Injectable in Situ Shape-Forming Osteogenic Nanocomposite Hydrogel for Regenerating Irregular Bone Defects. ACS Appl Bio Mater. 2018;1 (4):1037-1046. [30] ROSETI L, PARISI V, PETRETTA M, et al. Scaffolds for Bone Tissue Engineering: State of the art and new perspectives. Mater Sci Eng. 2017;78:1246-1262. [31] LEE SS, KIM JH, JEONG J, et al. Sequential growth factor releasing double cryogel system for enhanced bone regeneration. Biomaterials. 2020;257:120223. [32] YAO Q, COSME JG, XU T, et al. Three dimensional electrospun PCL/PLA blend nanofibrous scaffolds with significantly improved stem cells osteogenic differentiation and cranial bone formation. Biomaterials. 2017;115:115-127. [33] HUANG YZ, JI YR, KANG ZW, et al. Integrating eggshell-derived CaCO3/MgO nanocomposites and chitosan into a biomimetic scaffold for bone regeneration. Chem Eng J. 2020;395:125098. [34] CHEN X, ZHU L, WEN W, et al. Biomimetic mineralisation of eggshell membrane featuring natural nanofiber network structure for improving its osteogenic activity. Colloid Surf B-Biointerfaces. 2019;179:299-308. [35] ZHAO PP, GE YW, LIU XL, et al. Ordered arrangement of hydrated GdPO4 nanorods in magnetic chitosan matrix promotes tumor photothermal therapy and bone regeneration against breast cancer bone metastases. Chem Eng J. 2020;381:122694. [36] OU Q, HUANG K, FU C, et al. Nanosilverincorporated halloysite nanotubes/gelatin methacrylate hybrid hydrogel with osteoimmunomodulatory and antibacterial activity for bone regeneration. Chem Eng J. 2019;382:123019. [37] 彭坤,孙姣霞,李玉筱,等. MGF-Ct24E功能化聚乳酸仿生骨基质材料的制备与表征[J].功能材料,2014,14(45):14156-14160. [38] 张通,蔡金池,袁志发,等.基于透明质酸的复合水凝胶修复骨关节炎软骨损伤: 应用与机制[J].中国组织工程研究,2022,26(4):617-625. [39] PING H, WAGERMAIER WG, HORBELT N, et al. Mineralization generates megapascal contractile stresses in collagen fibrils. Science. 2022;376:188-192. |
| [1] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [2] | Liu Xiaolin, Mu Xinyue, Ma Ziyu, Liu Shutai, Wang Wenlong, Han Xiaoqian, Dong Zhiheng. Effect of hydrogel-loaded simvastatin microspheres on osteoblast proliferation and differentiation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 998-1003. |
| [3] | Dai Xianglin, Zhang Wenfeng, Yao Xijun, Shang Jiaqi, Huang Qiujin, Ren Yifan, Deng Jiupeng. Barium titanate/polylactic acid piezoelectric composite film affects adhesion, proliferation, and osteogenic differentiation of MC3T3-E1 cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 367-373. |
| [4] | Cheng Mingguang, Zhang Chaoyu, Zhuang Kangle, Ruan Peng, Zuo Yi, Zhou Zhengchun, Kong Xiang, Ge Jianjun, Cheng Guangcun. In vitro construction of Stanford type A aortic dissection 3D dynamic simulation diagram and individual tissue-engineered blood vessels [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 335-338. |
| [5] | Li Yue, Lyu Yan, Feng Wanying, Song Yang, Yan Yu, Guan Yongge. Preparation of hyperoside nanoparticles to repair endometrial injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 360-366. |
| [6] | Zhu Biwen, Wang Dongzhi, Wu Di, Gong Tiancheng, Pan Haopeng, Lu Yuhua, Guo Yibing, Wang Zhiwei, Huang Yan. Biomimetic microenvironment constructed from gelatin methacrylamide/platelet-rich plasma hydrogel promotes the function of insulinoma cell line MIN6 in mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1824-1831. |
| [7] | Ling Huajun, Cui Ruiwen, Wang Qiyou. 3D extracellular matrix hydrogel loaded with exosomes promotes wound repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1900-1905. |
| [8] | Hu Jinlong, Quan Huahong, Wang Jingcheng, Zhang Pei, Zhang Jiale, Chen Pengtao, Liang Yuan. Effect of copper sulfide nanoparticles loaded thermosensitive hydrogel Pluronic F127 on infected wound healing in rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1927-1931. |
| [9] | Wang Xinxin, Wang Jingxin. Mesenchymal stem cells-derived exosomes in treatment of secondary lymphedema [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(10): 1603-1609. |
| [10] | Liu Siqi, Wu Mingrui, Qiao Lingran, Xie Liying, Chen Siyu, Han Zhibo, Zuo Lin. Effects of hydrogel loaded with human umbilical cord mesenchymal stem cells on diabetic wound repair in mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(1): 21-27. |
| [11] | Zhang Tong, Cai Jinchi, Yuan Zhifa, Zhao Haiyan, Han Xingwen, Wang Wenji. Hyaluronic acid-based composite hydrogel in cartilage injury caused by osteoarthritis: application and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 617-625. |
| [12] | Huang Chuanjun, Zou Yu, Zhou Xiaoting, Zhu Yangqing, Qian Wei, Zhang Wei, Liu Xing. Transplantation of umbilical cord mesenchymal stem cells encapsulated in RADA16-BDNF hydrogel promotes neurological recovery in an intracerebral hemorrhage rat model [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 510-515. |
| [13] | Le Guoping, Zhang Ming, Xi Licheng, Luo Hanwen. Preparation and in vitro evaluation of vancomycin hydrochloride@polylactic acid-glycolic acid copolymer-chitosan-hyaluronic acid composite sustained-release microspheres [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 528-534. |
| [14] | Lu Dongdong, Zhu Tianfeng, Zhang Yijian, Zhao Zhijian, Liu Yang, Shen Xu, Zhu Xuesong. 3D bio-printing methylacrylated gelatin hydrogel scaffolds promote the repair of subchondral bone defects [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(34): 5454-5460. |
| [15] | Zhao Yunan, Zhang Haobo, Sun Tao, Yang Xuejun. Hydrogel-based growth factors and drugs in the treatment of intervertebral disc degeneration: problems and prospects [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(34): 5525-5533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||