Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (24): 3931-3936.doi: 10.3969/j.issn.2095-4344.2014.24.026
Antler regeneration and its proteomics research
Dong Zhen, Chu Wen-hui, Zhao Hai-ping, Liu Zhen, Wang Gui-wu, Li Chun-yi
- State Key Laboratory for Molecular Biology of Special Animals; Institute of Special Animal and Plant Sciences, Chinese Academy of Agricultural Sciences, Changchun 130000, Jilin Province, China
-
Revised:2014-03-12Online:2014-06-11Published:2014-06-11 -
Contact:Li Chun-yi, M.D., Investigator, State Key Laboratory for Molecular Biology of Special Animals; Institute of Special Animal and Plant Sciences, Chinese Academy of Agricultural Sciences, Changchun 130000, Jilin Province, China -
About author:Dong Zhen, Studying for master’s degree, State Key Laboratory for Molecular Biology of Special Animals; Institute of Special Animal and Plant Sciences, Chinese Academy of Agricultural Sciences, Changchun 130000, Jilin Province, China -
Supported by:the National Natural Science Foundation of China, No. 31170950; the National “863” Program of China, No. 2011AA100603
CLC Number:
Cite this article
Dong Zhen, Chu Wen-hui, Zhao Hai-ping, Liu Zhen, Wang Gui-wu, Li Chun-yi. Antler regeneration and its proteomics research[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(24): 3931-3936.
share this article
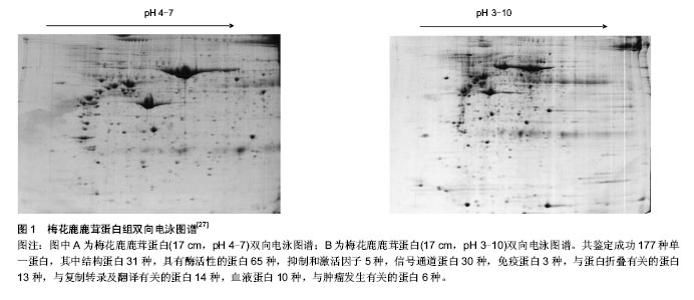
2.1 纳入文献基本情况 文献[1-10]研究了鹿茸周期性再生的过程;文献[11-21]研究了鹿茸基于干细胞再生的依据;文献[22-26]研究了鹿角柄骨膜致敏区与休眠区的发现并证实了鹿茸再生源于角柄骨膜致敏区中的干细胞;文献[27-38]研究了基于凝胶系统的鹿茸蛋白质组学进展;文献[39-43]研究了基于非凝胶电泳的鹿茸蛋白质组学进展。 2.2 鹿茸生物学 2.2.1 鹿茸基于干细胞再生 鹿茸再生的整个过程包括生长、骨化、茸皮脱落以及鹿角脱落等[11-12]。与人们所熟知的具有再生功能的物种如蝾螈(基于胚芽的再生)不同的是鹿茸的再生是基于干细胞的割处再生过程[3],是哺乳类动物中惟一有能力自切、脱落和再生的动物附属器官。 鹿茸每年从额前部永久性残桩-角柄上再生,随着年龄增长,角柄逐渐变短。实验证实,鹿茸再生源于覆盖角柄的骨膜[3]。这类细胞就是角柄骨膜细胞。鹿茸每年周期性再生都源于角柄骨膜。随后Li等[3]发现角柄骨膜细胞能表达干细胞标记物CD9抗原。与此同时,3个具有多潜能性的基因即POU结构域的家族成员Oct4,Nanog和Sox2也在其中表达[13-14]。另外发现这种细胞中的端粒酶以及核干细胞因子的表达量也较高[3]。端粒酶能增强细胞的自我更新作用而核干细胞因子具有控制干细胞的增殖作用[15-16]。Li等[17-21]还发现角柄骨膜细胞具有向其他类型细胞分化的多潜能性。 2.2.2 角柄骨膜致敏区与休眠区 鹿茸再生组织学基础表明其周期性再生来源于角柄骨膜中的干细胞。但角柄高度如梅花鹿与赤鹿在5 cm左右[22],那么是整个角柄骨膜都具有再生能力还是只有其中的部分组织承担此任务呢?针对此,Li等[23]通过组织学与形态学研究发现,角柄骨膜根据与皮肤接触的紧密程度有一个比较明显的分界,即远心端大约1/3部分骨膜与皮肤是紧密接触的;而近心端大约2/3部分骨膜与皮肤之间存在大量的疏松结缔组织使得两者没有紧密接触。发现此现象后,Li等[24]进一步进行插膜实验表明茸皮的生长是依赖角柄骨膜的刺激作用的,并且前者可以长出无皮的鹿茸组织,这表明茸皮对于该部分角柄骨膜仅起到引发其生长的作用;而后者则不会生长出鹿茸组织。由此Li等将远心端角柄骨膜与皮肤紧密接触部分称为角柄骨膜致敏区,相应的近心端角柄骨膜与皮肤非紧密接触部分称为角柄骨膜休眠区。另外,Wang等[25]曾表明没有覆盖物的体外组织是不能存活的,但此插膜实验说明鹿茸却是个例外。 Hsueh等[26]研究表明,干细胞的更新,分化与成熟均与其周围的微环境有关。Li等[24]认为鹿角柄皮肤应该为角柄骨膜干细胞提供了这个微环境。目前的实验证明是骨组织相应成分包括角柄骨膜引起的鹿茸再生。骨组织在与其所覆盖的皮肤相互作用后,会被诱导并具有潜在的再生能力[3]。 2.3 鹿茸蛋白质组学研究进展 蛋白质组学的研究是生命科学进入后基因组时代的特征。蛋白质组学对于治疗人类疾病,寻找特效药物等解决方案上起着至关重要的作用。 再生生物学特别是割处再生是近年来研究的重点,鹿茸是迄今为止所发现的惟一可以割处完全再生的哺乳动物附属器官,以鹿茸为模型研究哺乳动物器官再生机制具有明显优势[1]。而在整个鹿茸再生的过程中,大量具有生物活性的蛋白类物质与小分子多肽类物质承担着重要的生物学作用。所以为了能够全面了解在鹿茸加速生长过程中所涉及的分子机制并找到潜在的信号分子,对于鹿茸蛋白质谱的分析鉴定就显得十分必要了。 目前国内外对鹿茸蛋白质组学的研究从分离方法上可分为两类,其一是基于凝胶系统进行蛋白分离(如双向电泳等);其二是是基于非凝胶系统进行蛋白分离(如在线二维色谱技术等)。 2.3.1 基于凝胶系统的鹿茸蛋白质组学研究 赵东[27-28]与刘华淼等[29]利用双向电泳技术分别对梅花鹿二杠茸与梅花鹿茸的不同生长部位进行了蛋白质组的分析。赵东[27]通过整理质谱鉴定结果共得到了177种单一蛋白。其中结构蛋白31种,具有酶活性的蛋白65种,抑制和激活因子5种,信号通道蛋白30种,免疫蛋白3种,与蛋白折叠有关的蛋白13种,与复制转录及翻译有关的蛋白14种,血液蛋白10种,与肿瘤发生有关的蛋白6种(图1)。因为器官再生和肿瘤发生相类似,从结果中可以看出鹿茸中有多个与信号通路和肿瘤发生相关的蛋白,这些蛋白有可能在促使鹿茸具有甚至超过癌症细胞的生长速度的同时又被精确的进行调控生长的整个过程中起到重要作用。 丁倩南等[30]同样对不同部位的鹿茸进行了双向电泳最终共找到了18个蛋白差异点,实验结果为区分鉴定鹿茸的级别建立了有效的方法。同时因鹿茸顶端活性较强,其中特有的蛋白点可能是潜在的生物活性物质,具体机制有待进一步验证。 Park等[31]利用双向电泳技术研究赤鹿鹿茸蛋白质组。他将鹿茸尖端与血浆组织进行比对发现两者蛋白质组的匹配度高达43%,这个结果证实了先前的组织学实验即鹿茸组织中确实包含了一个发达的血管系统来为其提供充足的血液以及供给养分(足以支撑其比癌细胞发育还要快速的生长速度)直到鹿茸脱落[32]。并且鹿茸中含有大量的新陈代谢酶类,与基因表达调控有关的蛋白以及信号通路相关蛋白等。其中发现了调控细胞生长的信号通路因子如PKCβ与MAPK1;也发现了一些核受体蛋白如雌激素受体,维生素D3受体以及甲状腺激素受体等。与此同时,鹿茸尖端的蛋白质组一共发现了800多个蛋白点,但质谱鉴定等只得到了130个左右的蛋白,可以看出鹿茸中还含有大量的未知蛋白是目前数据库中所没有收录的,这部分蛋白很有可能是鹿茸所独有的 或特异性很强的且具有重要生物活性的蛋白,有必要对这些未知蛋白进一步分析研究。但需注意的是,通过组织提取得到的鹿茸蛋白质组可能有来源于血浆中的蛋白进而干扰实验结果,所以需要考虑血浆蛋白的纯化以还原最真实的鹿茸蛋白质谱。 以上是二维凝胶电泳技术在鹿茸中的研究进展,这些研究有助于发现鹿茸中的生物活性物质。而对于探究鹿茸的再生机制需对鹿角柄骨膜(细胞)进行相应的蛋白质组学研究。 徐代勋等[33-34]对角柄骨膜休眠区与致敏区的蛋白质组进行比较分析与鉴定,共检测出47个蛋白,其中有6个与再生相关,即PKM2、MAPK1、PEDF、peroxiredoxin 4、Hsp90α、fibulin5,这些蛋白可能参与了鹿茸再生的相关分子调节与信号通路转导。值得注意的是,其中PEDF具有促细胞分化、抑制肿瘤生长、抑制新生血管形成等作用[35];fibulin 5是血管生成抑制剂[36],并与原弹性蛋白表达有关,他们在肿瘤所需血管生成中扮演重要角色。由此可知PEDF与fibulin 5可能对鹿茸快速生长而不引发癌变起到调节作用。另外皮肤与角柄骨膜之间存在相互作用而促使角柄骨膜干细胞分化[3],而Lord等[37]研究发现PEDF在鹿茸真皮的皮脂腺与毛囊等部位中不能检测到,仅能在成纤维细胞中检测到。PEDF存在于角柄骨膜却不存在于皮肤某些关键组织中,那么其是否在两者相互作用中起到信使作用,具体机制有待进一步研究验证。 Li等[11]利用双向电泳分别对鹿生茸区骨膜、角柄骨膜以及面部骨膜三种骨膜细胞的蛋白质组进行分析。结果表明生茸区骨膜细胞中鉴定得到了66种蛋白而角柄骨膜细胞中鉴定得到了98种蛋白。同时对结果进行生物信息学分析发现PI3K/Akt,ERK/MAPK,p38 MAPK等细胞信号通路在鹿茸干细胞增殖时起作用。并找到了胚胎干细胞的特异性标记物POU5F1,SOX2,NANOG和MYC,从而推测鹿茸干细胞可能是一种介于成体干细胞与胚胎干细胞之间的间性干细胞类型。其中值得注意的是Li等[11]与徐代勋等[33]均在角柄骨膜中发现了Hsp90α。而近期清华大学罗永章等研究发现分泌型Hsp90α能促进肿瘤侵袭与转移,且其在血液中含量与肿瘤恶性程度成正相关。该蛋白在细胞外促进了肿瘤细胞的侵袭与转移,并在癌症发生与转移过程中几乎参与了全部活动。该蛋白在肿瘤细胞中的含量达到2%-7%[38]。罗永章认为该蛋白可作为一种稳定的肿瘤标志物并研制出了相应的Hsp90α定量试剂盒用于临床诊断。那么在角柄骨膜中发现相关蛋白到底意味着什么,鹿茸组织与肿瘤到底有怎样的关系还有待进一步的研究。另外罗永章所发现的蛋白是一种分泌型蛋白,在角柄骨膜细胞中该类蛋白是否为分泌型还需要对其培养基中的蛋白进行检测,所以下一步应该找角柄骨膜细胞培养基中是否存在相关蛋白。"
| [1] Sunwoo HH, Nakano T, Hudson RJ, et al. Isolation, characterization and localization of glycosaminoglycans in growing antlers of wapiti (Cervus elaphus). Comp Biochem Physiol B Biochem Mol Biol. 1998;120(2):273-283. [2] Sunwoo HH, Nakano T, Sim JS.Isolation and characterization of proteoglycans from growing antlers of wapiti (Cervus elaphus). 1998;121(4):437-442. [3] Li C, Yang F, Sheppard A. Adult stem cells and mammalian epimorphic regeneration-insights from studying annual renewal of deer antlers. Curr Stem Cell Res Ther. 2009; 4(3): 237-251. [4] Li C, Pearson A, McMahon C.Morphogenetic mechanisms in the cyclic regeneration of hair follicles and deer antlers from stem cells.Biomed Res Int. 2013;2013:643601. [5] Gao Z, Li C. The study on the relationship between antler’s growth rate, relative bone mass and circulation testosterone, estradiol, AKP in sika deer. Acta Vet Zootech Sin. 1988;19: 224-231. [6] Unsal C, Oran M, Tureli HO, et al. Detection of subclinical atherosclerosis and diastolic dysfunction in patients with schizophrenia. Neuropsychiatr Dis Treat. 2013;9:1531-1537. [7] Garcia M, Charlton BD, Wyman MT, et al. Do Red Deer Stags (Cervus elaphus) Use Roar Fundamental Frequency (F0) to Assess Rivals? PLoS One. 2013;8(12):e83946. [8] Clark DE, Li C, Wang W, et al. Vascular localization and proliferation in the growing tip of the deer antler. Anat Rec A Discov Mol Cell Evol Biol. 2006;288(9):973-981. [9] Li C. Deer antler regeneration: a stem cell-based epimorphic process. Birth Defects Res C Embryo Today. 2012;96(1): 51-62. [10] Kierdorf U, Kierdorf H, Szuwart T.Deer antler regeneration: cells, concepts, and controversies.J Morphol. 2007;268(8): 726-738. [11] Li C, Harper A, Puddick J, et al. Proteomes and signalling pathways of antler stem cells.PLoS One. 2012;7(1): e30026. [12] Price JS, Allen S, Faucheux C,et al.Deer antlers: a zoological curiosity or the key to understanding organ regeneration in mammals?J Anat. 2005;207(5):603-618. [13] Li W, Ding S. Converting mouse epiblast stem cells into mouse embryonic stem cells by using small molecules. Methods Mol Biol. 2013;1074:31-37. [14] Zhou H, Li W, Zhu S, Joo JY,et al.Conversion of mouse epiblast stem cells to an earlier pluripotency state by small molecules.J Biol Chem. 2010;285(39):29676-2980. [15] Yang C, Przyborski S, Cooke MJ, et al. A key role for telomerase reverse transcriptase unit in modulating human embryonic stem cell proliferation, cell cycle dynamics, and in vitro differentiation. Stem Cells. 2008;26(4):850-863. [16] Beekman C, Nichane M, De Clercq S, et al. Evolutionarily conserved role of nucleostemin: controlling proliferation of stem/progenitor cells during early vertebrate development. Mol Cell Biol. 2006;26(24):9291-9301. [17] Li C, Yang F, Sheppard A. Adult stem cells and mammalian epimorphic regeneration-insights from studying annual renewal of deer antlers. Curr Stem Cell Res Ther. 2009; 4(3):237-251. [18] Rolf HJ, Kierdorf U, Kierdorf H, et al. Localization and characterization of STRO-1 cells in the deer pedicle and regenerating antler. PLoS One. 2008;3(4):e2064. [19] Berg DK, Li C, Asher G, et al. Red deer cloned from antler stem cells and their differentiated progeny. Biol Reprod. 2007;77(3):384-394. [20] Kierdorf U, Kierdorf H. Deer antlers - a model of mammalian appendage regeneration: an extensive review. Gerontology. 2011;57(1):53-65. [21] Kierdorf U, Li C, Price JS. Improbable appendages: Deer antler renewal as a unique case of mammalian regeneration. Semin Cell Dev Biol. 2009;20(5):535-542. [22] Li C.Exploration of the mechanism underlying neogenesis and regeneration of postnatal mammalian skin-deer antler velvet. Int J Med Biol Front. 2010;11/12(16):1-19. [23] Li C, Suttie JM. Tissue collection methods for antler research. Eur J Morphol. 2003;41(1):23-30. [24] Li C, Yang F, Li G, et al. Antler regeneration: a dependent process of stem tissue primed via interaction with its enveloping skin. J Exp Zool A Ecol Genet Physiol. 2007; 307(2):95-105. [25] Wang P, Zhang C, Zhu Y, et al. The resolution and regeneration of a cointegrate plasmid reveals a model for plasmid evolution mediated by conjugation and oriT site-specific recombination. Environ Microbiol. 2013;15(12): 3305-3318. [26] Hsueh YY, Chang YJ, Huang TC, et al. Functional recoveries of sciatic nerve regeneration by combining chitosan-coated conduit and neurosphere cells induced from adipose-derived stem cells. Biomaterials. 2014;35(7):2234-2244. [27] 赵东.梅花鹿鹿茸双向电泳体系建立及蛋白质组学的研究[D].江苏科技大学,2012. [28] 赵东.梅花鹿鹿茸总蛋白提取方法对双向电泳图谱的影响[J].中国畜牧兽医,2012,39(7):25-28. [29] 刘华淼,邢秀梅,杨福合. 梅花鹿鹿茸双向电泳蛋白提取条件筛选[J]. 特产研究,2013,(02):5-6+22. [30] 丁倩南,王春梅,戴俊东,等. 梅花鹿鹿茸不同部位蛋白双向电泳谱图比较[J]. 中药材,2013,36(4):521-525. [31] Park HJ, Lee DH, Park SG, et al. Proteome analysis of red deer antlers. Proteomics. 2004;4(11):3642-3653. [32] Price J, Faucheux C, Allen S. Deer antlers as a model of Mammalian regeneration. Curr Top Dev Biol. 2005;67:1-48. [33] 徐代勋. 梅花鹿鹿茸角柄骨膜不同部位差异蛋白的筛选[D].江苏科技大学,2011. [34] 徐代勋,王桂武,赵海,等. 梅花鹿角柄骨膜蛋白质组学初步研究[J]. 特产研究,2011,33(1):1-4. [35] 祁伟祥,姚阳,沈赞. 色素上皮衍生因子抗肿瘤治疗的研究进展[J]. 中国肿瘤临床,2011,38(8):477-480. [36] 文灿,余汇洋. Fibulin-5与肿瘤的研究进展[J]. 局解手术学杂志, 2006,15(5):337-338. [37] Lord EA, Martin SK, Gray JP, et al. Cell cycle genes PEDF and CDKN1C in growing deer antlers. Anat Rec (Hoboken). 2007;290(8):994-1004. [38] Wang X, Song X, Zhuo W, et al. The regulatory mechanism of Hsp90alpha secretion and its function in tumor malignancy. Proc Natl Acad Sci U S A. 2009;106(50):21288-21293. [39] 高亮,乔晓强,梁振,等. 基于在线二维色谱的不同生长时期鹿茸的比较蛋白质组分析[J].色谱,2010,28(2):146-151. [40] Gao L, Tao D, Shan Y, et al. HPLC-MS/MS shotgun proteomic research of deer antlers with multiparallel protein extraction methods. J Chromatogr B Analyt Technol Biomed Life Sci. 2010;878(32):3370-3374. [41] Sui Z, Yuan H, Liang Z, et al. An activity-maintaining sequential protein extraction method for bioactive assay and proteome analysis of velvet antlers. Talanta. 2013;107: 189-194. [42] 高亮,鹿茸蛋白质的提取及差异研究[D].中国科学院研究生院, 2010. [43] Unlü M, Morgan ME, Minden JS. Difference gel electrophoresis: a single gel method for detecting changes in protein extracts. Electrophoresis. 1997;18(11):2071-2077. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [4] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [5] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [6] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [7] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [8] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [9] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [10] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [11] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [12] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [13] | Guan Qian, Luan Zuo, Ye Dou, Yang Yinxiang, Wang Zhaoyan, Wang Qian, Yao Ruiqin. Morphological changes in human oligodendrocyte progenitor cells during passage [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1045-1049. |
| [14] | Wang Feng, Zhou Liyu, Saijilafu, Qi Shibin, Ma Yanxia, Wei Shanwen. CaMKII-Smad1 promotes axonal regeneration of peripheral nerves [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1064-1068. |
| [15] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||