Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (18): 2789-2794.doi: 10.3969/j.issn.2095-4344.2014.18.001
Establishment of a rabbit model of parietal critical bone defects: a reference value for identifying parietal critical bone defects
He Tong-wen, Xu Geng-chi, Han Yao-hui, Mu Lan, Ge Zhen-lin
- Department of Orthodontics, School of Stomatology, Lanzhou University, Lanzhou 730000, Gansu Province, China
-
Received:2014-03-27Online:2014-04-30Published:2014-04-30 -
Contact:Ge Zhen-lin, Professor, Department of Orthodontics, School of Stomatology, Lanzhou University, Lanzhou 730000, Gansu Province, China -
About author:He Tong-wen, Studying for master’s degree, Department of Orthodontics, School of Stomatology, Lanzhou University, Lanzhou 730000, Gansu Province, China -
Supported by:the Scientific and Technological Support Project of Gansu Province, No. 1208RJZA209
CLC Number:
Cite this article
He Tong-wen, Xu Geng-chi, Han Yao-hui, Mu Lan, Ge Zhen-lin. Establishment of a rabbit model of parietal critical bone defects: a reference value for identifying parietal critical bone defects[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(18): 2789-2794.
share this article
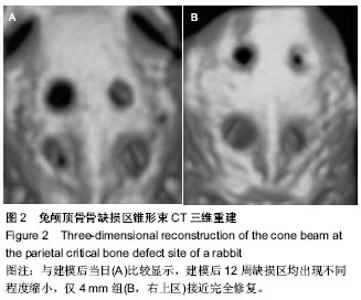
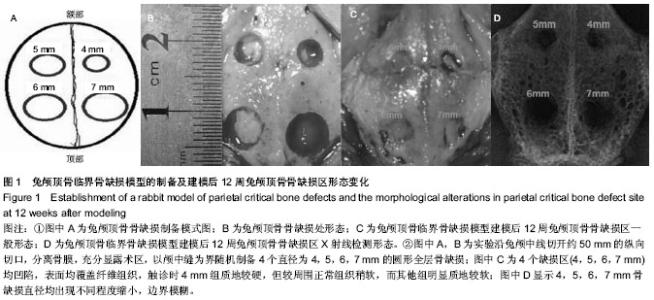
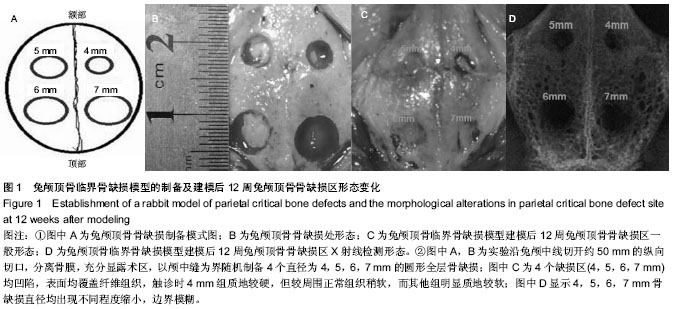
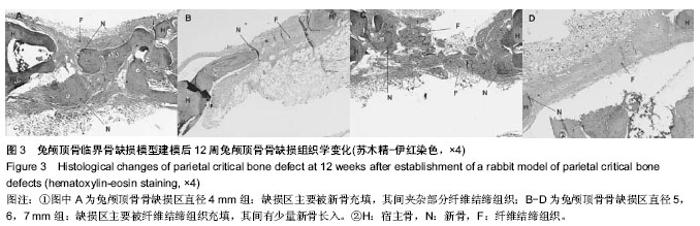
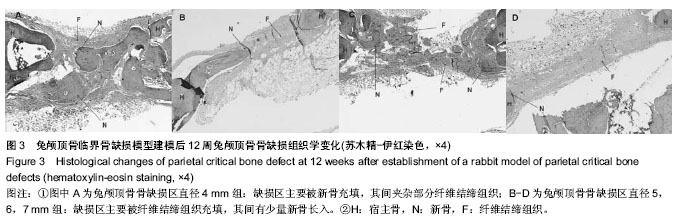
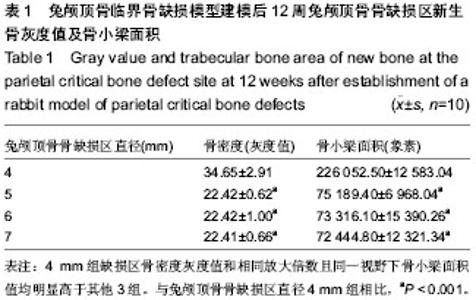
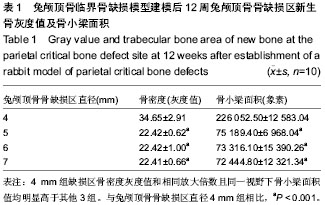
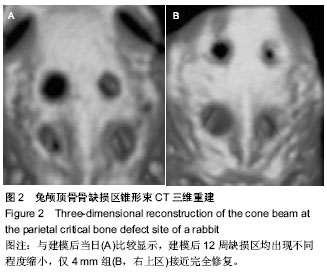
2.1 实验动物数量分析 10只兔建模后健康状况良好,无死亡及感染,均进入结果分析。 2.2 兔缺损区一般形态 建模后12周,兔颅顶骨4个缺损区均凹陷可见4个缺损均凹陷,表面均覆盖纤维组织,触诊时4 mm组质地较硬,但较周围正常组织稍软,而其他组明显质地较软(图1C,D)。 2.3 兔颅顶骨临界骨缺损模型建模缺损区影像学形态 建模后12周,锥型CT检测显示4,5,6,7 mm骨缺损直径均出现不同程度缩小,边界模糊。与建模后当日比较显示,兔缺损区均出现不同程度缩小,仅4 mm组接近完全修复(图2)。 2.4 兔颅顶骨临界骨缺损模型建模缺损区组织学变化 建模后12周,苏木精-伊红染色显示4 mm组缺损区出现排列不规则的骨小梁结构,并可见少量纤维组织嵌入其中,其他3组在骨缺损周边可见少量新生骨小梁存在,但缺损区完全被纤维组织充填(图3)。 利用软件Image-Pro Plus 6.0在相同放大倍数下测量缺损区同一视野新生骨小梁面积,结果显示,4 mm组相同放大倍数且同一视野下骨小梁面积高于其他3组(P < 0.001,表1)。"
| [1] Chiapasco M, Casentini P, Zaniboni M. Bone augmentation procedures in implant dentistry. Int J Oral Maxillofac Implants. 2009;24 Suppl:237-259. [2] Kessler P, Thorwarth M, Bloch-Birkholz A, et al. Harvesting of bone from the iliac crest-comparison of the anterior and posterior sites. Br J Oral Maxillofac Surg. 2005;43(1):51-56. [3] Moore WR, Graves SE, Bain GI. Synthetic bone graft Substitutes. Aust N Z J Surg. 2001;71(6):354-361. [4] Schmitz JP, Hollinqer JO. The critical size defect as anexperimental model for craniomandibulofacial nonunions. Clin Orthop Relat Res.1986;(205):299-308. [5] Gosain AK, Song L, Yu P, et al. Osteogenesis in cranial defects: reassessment of the concept of critical size and the expression of TGF-beta isoforms. Plast Reconstr Surg. 2000; 106(2):360-371. [6] Jiang ZQ, Liu HY, Zhang LP, et al. Repair of calvarial defects in rabbits with platelet-rich plasma as the scaffold for carrying bone marrow stromal cells. Oral Surg Oral Med Oral Pathol Oral Radiol. 2012;113(3):327-333. [7] Yun JH, Yoo JH, Choi SH, et al. Synergistic effect of bone marrow-derived mesenchymal stem cells and platelet-rich plasma on bone regeneration of calvarial defects in rabbits. Tissue Eng Regen Med. 2012;9(1):17-23. [8] Borie E, FuentesR, Del Sol M, et al. The influence of FDBA and autogenous bone particles on regeneration of calvaria defects in the rabbit: a pilot study. Ann Anat. 2011;193(5): 412-417. [9] Lin CY, Chang YH, Kao CY, et al. Augmented healing of critical-size calvarial defects by baculovirus-engineered MSCs that persistently express growth factors. Biomaterials. 2012;33(14):3682-3692. [10] Lin CY, ChangYH, Li KC, et al. The use of ASCs engineered to express BMP2 or TGF-b3 within scaffold constructs to promote calvarial bone repair. Biomaterials. 2013;34(37): 9401-9412. [11] Naitoa Y, Terukina T, Gallic S, et al. The effect of simvastatin-loaded polymeric microspheres in a critical size bone defect in the rabbit calvaria. Int J Pharm. 2014; 461(1-2): 157-162. [12] El Backly RM, Zaky SH, Canciani B, et al. Platelet rich plasma enhances osteoconductive properties of a hydroxyapatite-β-tricalcium phosphate scaffold (Skelite ) for late healing of critical size rabbit calvarial defects. J Craniomaxillofac Surg. 2013. [13] Fok TC, Jan A, Peel SA, et al. Hyperbaric oxygen results in increased vascular endothelial growth factor (VEGF) protein expression in rabbit calvarial critical-sized defects. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008;105(4): 417-422. [14] Tuusa SM, Peltola MJ, Tirri T, et al. Reconstruction of critical size calvarial bone defects in rabbits with glass-fiber-reinforced composite with bioactive glass granule coating. J Biomed Mater Res B Appl Biomater. 2008;84(2): 510-519. [15] Li G, Wang X, Cao J, et al. Coculture of peripheral blood CD34+ cell and mesenchymal stem cell sheets increase the formation of bone in calvarial critical-size defects in rabbits. Br J Craniomaxillofac Surg. 2014;52(2):134-139. [16] The Ministry of Science and Technology of the People’s Republic of China. Guidance Suggestions for the Care and Use of Laboratory Animals. 2006-09-30. [17] Rapp SJ, Jones DC, Gerety P, et al. Repairing critical-sized rat calvarial defects with progenitor cell-seeded acellularperiosteum: a novel biomimetic scaffold. Surgery. 2012;152(4):595-605. [18] Amorosa LF, Lee CH, Aydemir AB, et al. Physiologic load-bearing characteristics of autografts, allografts, and polymer-based scaffolds in a critical sized segmental defect of long bone: an experimental study. Int J Nanomedicine. 2013;8: 1637-1643. [19] Chatterjea A, van der Stok J, Danoux CB, et al. Inflammatory response and bone healing capacity of two porous calcium phosphate ceramics in critical size cortical bone defects. J Biomed Mater Res A. 2014;102(5):1399-1407. [20] Pang L, Hao W, Jiang M, et al. Bony defect repair in rabbit using hybrid rapid prototyping polylactic-co-glycolic acid/β-tricalciumphosphate collagen I/apatite scaffold and bone marrow mesenchymal stem cells. Indian J Orthop. 2013;47(4):388-394. [21] Zhang X, Xu M, Song L, et al. Effects of compatibility of deproteinized antler cancellous bone with various bioactive factors on their osteogenic potential. Biomaterials. 2013; 34(36):9103-9114. [22] Yang P, Huang X, Wang C, et al. Repair of bone defects using a new biomimetic construction fabricated by adipose-derived stem cells, collagen I, and porous beta-tricalcium phosphate scaffolds. Exp Biol Med (Maywood). 2013;238(12):1331- 1343. [23] Kang SH, Chung YG, Oh IH, et al. Bone regeneration potential of allogeneic or autogeneic mesenchymal stem cells loaded onto cancellous bone granules in a rabbit radial defect model. Cell Tissue Res. 2014;355(1):81-88. [24] Bi L, Cheng W, Fan H, et al. Reconstruction of goat tibial defects using an injectable tricalcium phosphate/chitosan in combination with autologous platelet-rich plasma. Biomaterials. 2010;31(12):3201-3211. [25] Berner A, Reichert JC, Woodruff MA, et al. Autologous vs. allogenic mesenchymal progenitor cells for the reconstruction of critical sized segmental tibial bone defects in aged sheep. Acta Biomater. 2013;9(8):7874-7884. [26] Liu T, Wu G, Wismeijer D, et al. Deproteinized bovine bone functionalized with the slow delivery of BMP-2 for the repair of critical-sized bone defects in sheep. Bone. 2013;56(1): 110-118. [27] Bergmann CJ, Odekerken JC, Welting TJ, et al. Calcium phosphate based three-dimensional cold plotted bone scaffolds for critical size bone defects. Biomed Res Int. 2014; 2014:852610. [28] Wehrhan F, Amann K, Molenberg A, et al. Critical size defect regeneration using PEG-mediated BMP-2 gene delivery and the use of cell occlusive barrier membranes–the osteopromotive principle revisited. Clin Oral Implants Res. 2013; 24(8):910-920. [29] Schubert T, Lafont S, Beaurin G, et al.Critical size bone defect reconstruction by an autologous 3D osteogenic-like tissue derived from differentiated adipose MSCs. Biomaterials. 2013; 34(18):4428-4438. [30] Asamura S, Mochizuki Y, Yamamoto M, et al. Bone regeneration using a bone morphogenetic protein-2 saturated slow-release gelatin hydrogel sheet: evaluation in a canine orbital floor fracture model. Ann Plast Surg. 2010;64(4): 496-502. [31] Chiu HC, Chiang CY, Tu HP, et al. Effects of bone morphogenetic protein-6 on periodontal wound healing/regeneration in supraalveolar periodontal defects in dogs. J Clin Periodontol. 2013;40(6):624-630. [32] Lee JS, Kim YW, Jung UW, et al. Bone regeneration and collagen fiber orientation around calcium phosphate-coated implants with machined or rough surfaces: a short-term histomorphometric study in dog mandibles. Int J Oral Maxillofac Implants. 2013;28(5):1395-1402. [33] Choi S, Liu IL, Yamamoto K, et al. Implantation of tetrapod-shaped granular artificial bones or β-tricalcium phosphate granules in a canine large bone-defect model. J Vet Med Sci. 2014;76(2):229-235. [34] Seeherman HJ, Li XJ, Smith E, et al. rhBMP-2/calcium phosphate matrix induces bone formation while limiting transient bone resorption in a nonhuman primate core defect model. J Bone Joint surg Am. 2012;94(19):1765-1776. [35] Schlegel KA, Lang FJ, DonathK, et al. The monocortical critical size bone defect as an alternative experimental model in testing bone substitute materials. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;102(1):7-13. [36] Hollinger JO, Kleinschmidt JC. The critical size defect as an experimental model to test bone repair materials. J Craniofac Surg. 1990;1(1):60-68. [37] Lee HE, Kim JY, Kweon HY, et al. A combination graft of low-molecular-weight silk fibroin with Choukroun platelet-rich fibrin for rabbit calvarialdefect. OralSurg Oral Med Oral Pathol Oral Radiol Endod. 2010;109(5):e33-e38. [38] Dumas JE, Brown Baer PB, Prieto EM, et al. Injectable reactive biocomposites for bone healing in critical-size rabbit calvarial defects. Biomed Mater. 2012;7(2):024112. [39] Liu Y, Lu Y, TianX, et al. Segmental bone regeneration using an rhBMP-2-loaded gelatin/nanohydroxyapatite/fibrin scaffold in a rabbit model. Biomaterials. 2009;30(31):6276-6285. [40] Humber CC, Sandor GK, Davis JM, et al. Bone healing with an in situ–formed bioresorbable polyethylene glycol hydrogel membrane in rabbit calvarial defects. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2010;109(3):372-384. [41] Pripatnanont P, Nuntanaranont T, Vongvatcharanon S, et al. The primacy of platelet-rich fibrin on bone regeneration of various grafts in rabbit’s calvarial defects. J Craniomaxillofac Surg. 2013;41(8):e191-200. [42] Biguetti CC, Filho EJ, de Andrade Holgado L, et al. Effect of low-level laser therapy on intramembranous and endochondral autogenous bone grafts healing. Microse Res Tech. 2012;75(9):1237-1244. [43] Guo Z, Iku S, Mu L, et al. Implantation with new three-dimensional porous titanium web for treatment of parietal bone defect in rabbit. Artif Organs. 2013;37(7): 623-628. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Yang Weiqiang, Ding Tong, Yang Weike, Jiang Zhengang. Combined variable stress plate internal fixation affects changes of bone histiocyte function and bone mineral density at the fractured end of goat femur [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 890-894. |
| [4] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [5] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [6] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [7] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [8] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [9] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [10] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [11] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [12] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [13] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [14] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [15] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||