Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (15): 2369-2376.doi: 10.3969/j.issn.2095-4344.2014.15.013
Previous Articles Next Articles
Construction and identification of bicistronic eukaryotic expression vector of human brain-derived neurotrophic factor and neurotrophine-3
Li Bing-nan, Li Wei-dong, Lin Jun-tang, Feng Hui-gen
- Department of Life Sciences and Technology, Xinxiang Medical University, Xinxiang 453003, Henan Province, China
-
Online:2014-04-09Published:2014-04-09 -
Contact:Li Bing-nan, Department of Life Sciences and Technology, Xinxiang Medical University, Xinxiang 453003, Henan Province, China -
About author:Li Bing-nan, Ph.D., Lecturer, Department of Life Sciences and Technology, Xinxiang Medical University, Xinxiang 453003, Henan Province, China -
Supported by:the Tender Subject of Key Research Areas of Xinxiang Medical University in 2011, No. ZD2011-16; Key Projects in Scientific Research of Henan Provincial Education Department, No. 13A180850
CLC Number:
Cite this article
Li Bing-nan, Li Wei-dong, Lin Jun-tang, Feng Hui-gen. Construction and identification of bicistronic eukaryotic expression vector of human brain-derived neurotrophic factor and neurotrophine-3[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(15): 2369-2376.
share this article
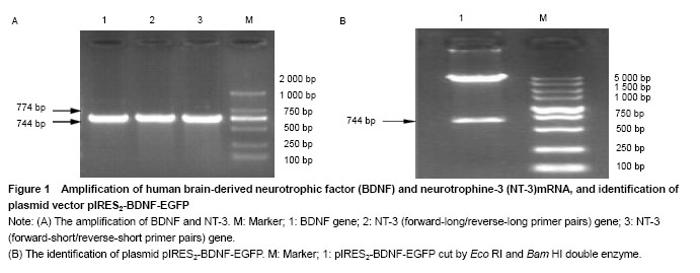
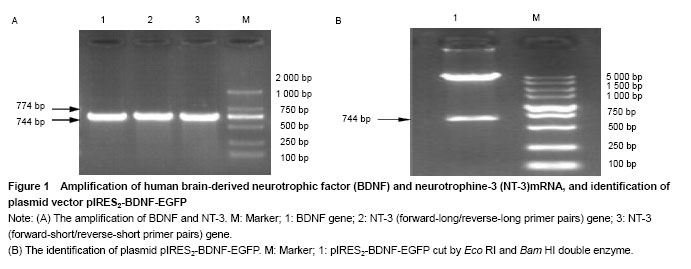
Amplification of BDNF and NT-3 genes BDNF and NT-3 genes were obtained from the genomic DNA of human peripheral blood mononuclear cells by PCR. The size of BDNF gene was 744 bp, the size of NT-3 gene was 774 bp (Figure 1A). Identification of plasmid pIRES2-BDNF-EGFP The plasmid pIRES2-BDNF-EGFP was cut by EcoRI and Bam HI double enzyme. A gene fragment with 744 bp was obtained, which was in full agreed with BDNF gene (NM-001102654.1) (Figure 1B). "
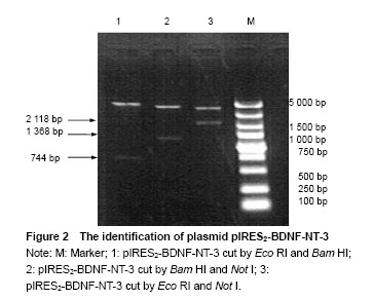
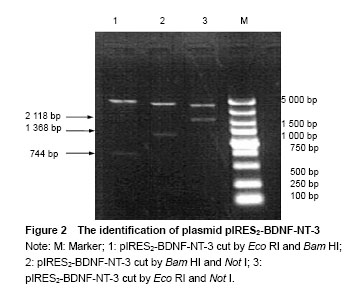
Identification of plasmid pIRES2-BDNF-NT-3 The plasmid pIRES2-BDNF-NT-3 was cut by Eco RI and Bam HI double enzyme. And a fragment about 744 bp would be obtained. This indicated BDNF gene was inserted into the plasmid pIRES2-BDNF-NT-3. The plasmid pIRES2-BDNF-NT-3 was cut by Bam HI and Not I double enzyme. And a fragment about 1 368 bp would be obtained. This indicated IRES-NT-3 gene was inserted into the plasmid pIRES2-BDNF-NT-3. The plasmid pIRES2-BDNF-NT-3 was cut by Eco RI and Not I double enzyme. And a fragment about 2 118 bp would be obtained. This indicated that BDNF-IRES-NT-3 gene was inserted into the plasmid pIRES2-BDNF-NT-3. The sequence of the plasmid pIRES2-BDNF-NT-3 was in accordance with gene sequence in Gene Bank (Figure 2). "
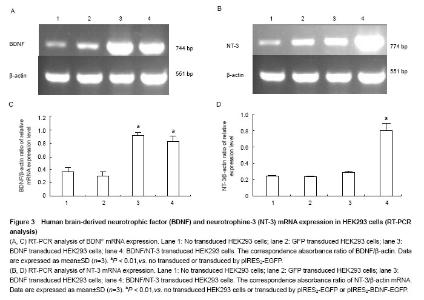
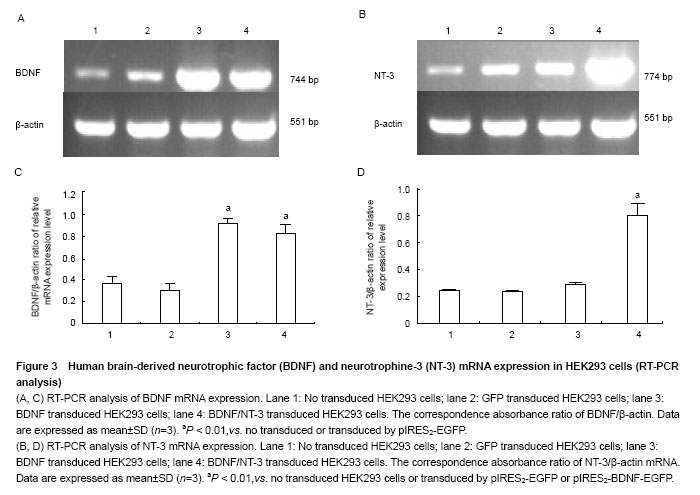
RT-PCR analysis of BDNF and NT-3 mRNA expression in HEK293 cells To illustrate mRNA expression by pIRES2-EGFP, pIRES2-BDNF/EGFP and pIRES2-BDNF/NT-3 transduced HEK293 cells, we evaluated the expression of BDNF and NT-3 by RT-PCR analysis. RT-PCR was performed using BDNF-specific primers and the β-actin sequence served as an internal standard. GFP expression was monitored in pIRES2-EGFP and pIRES2-BDNF/EGFP transduced HEK293 cells by inverted fluorescence microscopy. Expression of BDNF mRNA was higher in either pIRES2-BDNF/EGFP or pIRES2-BDNF/NT-3 transduced HEK293 cells than that pIRES2-EGFP transduced HEK293 cells or negative control (P < 0.01; Figure 3A, C). As shown above, expression of NT-3 mRNA was higher in pIRES2-BDNF/NT-3-transduced HEK293 cells than the other three (P < 0.01; Figure 3B, D). These results demonstrated that the BDNF and NT-3 had been introduced successfully into HEK293 cells by pIRES2-BDNF/EGFP and pIRES2-BDNF/NT-3."
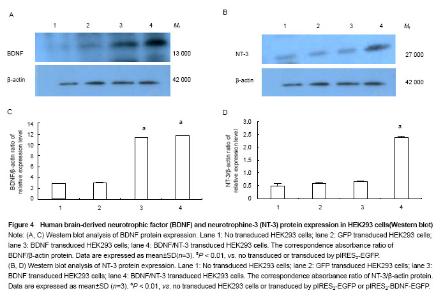
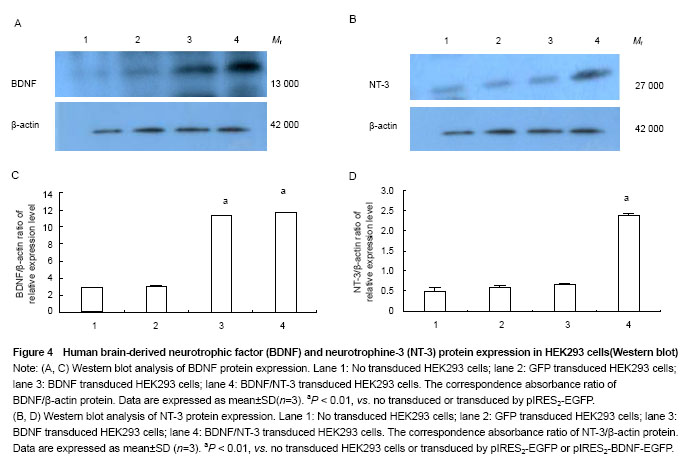
Western blot analysis of BDNF and NT-3 expression in HEK293 cells To illustrate protein expression by pIRES2-EGFP, pIRES2-BDNF/EGFP, and pIRES2-BDNF/NT-3 transduced HEK293 cells, we evaluated the expression of BDNF and NT-3 by western blot assay. After 72 hours of transduction, we collected the culture supernatants of infected HEK293 cells without serums which were processed for western blot analysis using an anti-BDNF and anti-NT-3 antibody. The β-actin served as an internal standard. The results suggested that exogenous BDNF protein was strongly expressed in pIRES2-BDNF/EGFP and pIRES2-BDNF/NT-3 transduced HEK293 cells, but the pIRES2-EGFP transduced HEK293 cells and no transduced expression of endogenous BDNF was very low (P < 0.01; Figure 4A, C). As shown above, western blot analysis also revealed that exogenous NT-3 protein was strongly expressed in pIRES2-BDNF/NT-3 transduced HEK293 cells, and the pIRES2-EGFP, pIRES2-BDNF/EGFP transduced HEK293 cells expression of endogenous NT-3 was very low (P < 0.01; Figure 4B, D). These results also demonstrated that BDNF and NT-3 had been introduced successfully into HEK293 cells by pIRES2-BDNF/NT-3. "
| [1]Tancredo JR, Maria RM, Azevedo ER, et al. Clinical assessment of spasticity in individuals with spinal cord injury. Acta Ortop Bras. 2013;21(6):310-314. [2]Fouad K, Bennett DJ, Vavrek R, et al. Long-term viral brain-derived neurotrophic factor delivery promotes spasticity in rats with a cervical spinal cordhemisection. Front Neurol. 2013;19(4):187-197. [3]Hou S, Nicholson L, van Niekerk E, et al. Dependence of regenerated sensory axons on continuous neurotrophin-3 delivery. J Neurosci. 2012;32(38): 13206-13220. [4]Blesch A, Fischer I, Tuszynski MH. Gene therapy, neurotrophic factors and spinal cord regeneration. Handb Clin Neurol. 2012;109(1):563-574. [5]Franz S, Weidner N, Blesch A. Gene therapy approaches to enhancing plasticity and regeneration after spinal cord injury. Exp Neurol. 2012;235(1):62-69. [6]Rauskolb S, Zagrebelsky M, Dreznjak A, et al. Global deprivation of brain-derived neurotrophic factor in the CNS reveals an area-specific requirement for dendritic growth. J Neurosci. 2010;30(5):1739-1749. [7]Huang T, Krimm RF. BDNF and NT4 play interchangeable roles in gustatory development. Dev Biol. 2014;386(2): 308-320. [8]Angelucci F, Ricci V, Gelfo F, et al. BDNF serum levels in subjects developing or not post-traumatic stress disorder after trauma exposure. Brain Cogn. 2014;84(1):118-122. [9]Fon D, Zhou K, Ercole F, et al. Nanofibrous scaffolds releasing a small molecule BDNF-mimetic for the re-direction of endogenous neuroblast migration in the brain. Biomaterials. 2014;35(9):2692-2712. [10]Li ST, Pan J, Hua XM, et al. Endothelial nitric oxide synthase protects neurons against ischemic injury through regulation of brain-derived neurotrophic factor expression. CNS Neurosci Ther. 2014;20(2):154-164. [11]Franz S, Weidner N, Blesch A. Gene therapy approaches to enhancing plasticity and regeneration after spinal cord injury. Exp Neurol. 2012;235(1):62-69. [12]Andero R, Choi DC, Ressler KJ. BDNF-TrkB receptor regulation of distributed adult neural plasticity, memory formation, and psychiatric disorders. Prog Mol Biol Transl Sci. 2014;122(1):169-192. [13]Morcuende S, Muñoz-Hernández R, Benítez-Temiño B, et al. Neuroprotective effects of NGF, BDNF, NT-3 and GDNF on axotomized extraocular motoneurons in neonatal rats. Neuroscience. 2013;250(1):31-48. [14]Zanin JP, Battiato NL, Rovasio RA. Neurotrophic factor NT-3 displays a non-canonical cell guidance signaling function for cephalic neural crest cells. Eur J Cell Biol. 2013;92(8-9): 264-279. [15]Sun Z, Guo Q, Xu X, et al. Protective effects of pretreatment with neurotrophin-3 on intrathecal ropivacaine in rats. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2014;39(1):17-22. [16]Tang Y, He H, Cheng N, et al. PDGF, NT-3 and IGF-2 in combination induced transdifferentiation of muscle-derived stem cells into schwann cell-like cells. PLoS One. 2014;9(1): 73402-73410. [17]Yang J, Yan Y, Xia Y, et al. Neurotrophin 3 transduction augments remyelinating and immunomodulatory capacity of neural stem cells. Mol Ther. 2013;22(2):440-450. [18]Ishimaru N, Fukuchi M, Hirai A, et al. Differential epigenetic regulation of BDNF and NT-3 genes by trichostatin A and 5-aza-2'-deoxycytidine in Neuro-2a cells. Biochem Biophys Res Commun. 2010;394(1):173-177. [19]Xie BH, Xie YF. Twin PCRs: a simple and efficient method for directional cloning of PCR products. World J Microbiol Biotechnol. 2011;27(9):2223-2225. [20]Sakai K, Yamamoto A, Matsubara K, et al. Human dental pulp-derived stem cells promote locomotor recovery after complete transection of the rat spinalcord by multiple neuro-regenerative mechanisms. J Clin Invest. 2012;122(1): 80-90. [21]Massa SM, Yang T, Xie Y, et al. Small molecule BDNF mimetics activate TrkB signaling and prevent neuronal degeneration in rodents. J Clin Invest. 2010;120(5): 1774-1785. [22]Deogracias R, Yazdani M, Dekkers MP, et al. Fingolimod, a sphingosine-1 phosphate receptor modulator, increases BDNF levels and improves symptoms of amouse model of Rett syndrome. Proc Natl Acad Sci U S A. 2012;109(35): 14230-14235. [23]Bonner JF, Blesch A, Neuhuber B, et al. Promoting directional axon growth from neural progenitors grafted into the injured spinal cord. J Neurosci Res. 2010;88(6):1182-1192. [24]Franz S, Weidner N, Blesch A. Gene therapy approaches to enhancing plasticity and regeneration after spinal cord injury. Exp Neurol. 2012;235(1):62-69. [25]Ding Y, Yan Q, Ruan JW, et al. Electroacupuncture promotes the differentiation of transplanted bone marrow mesenchymal stem cells overexpressing TrkC into neuron-like cells in transected spinal cord of rats. Cell Transplant. 2013;22(1): 65-86. [26]Gelain F, Panseri S, Antonini S, et al. Transplantation of nanostructured composite scaffolds results in the regeneration of chronically injured spinalcords. ACS Nano. 2011;5(1):227-236. [27]Overman JJ, Carmichael ST. Plasticity in the injured brain: more than molecules matter. Neuroscientist. 2014;20(1): 15-28. [28]García-Alías G, Petrosyan HA, Schnell L, et al. Chondroitinase ABC combined with neurotrophin NT-3 secretion and NR2D expression promotes axonalplasticity and functional recovery in rats with lateral hemisection of the spinal cord. J Neurosci. 2011;31(49):17788-17799. [29]Jenkins SI, Pickard MR, Granger N, et al. Magnetic nanoparticle-mediated gene transfer to oligodendrocyte precursor cell transplant populations isenhanced by magnetofection strategies. ACS Nano. 2011;5(8):6527-6538. [30]Koo HM, Lee SM, Kim MH. Spontaneous wheel running exercise induces brain recovery via neurotrophin-3 expression following experimental traumatic brain injury in rats. J Phys Ther Sci. 2013;25(9):1103-1107. [31]Beggs S, Alvares D, Moss A, et al. A role for NT-3 in the hyperinnervation of neonatally wounded skin. Pain. 2012; 153(10):2133-2139. [32]Smith GM, Falone AE, Frank E. Sensory axon regeneration: rebuilding functional connections in the spinal cord. Trends Neurosci. 2012;35(3):156-163. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||