Chinese Journal of Tissue Engineering Research ›› 2013, Vol. 17 ›› Issue (40): 7054-7060.doi: 10.3969/j.issn.2095-4344.2013.40.006
Previous Articles Next Articles
In vitro isolation, culture and identification of adipose-derived stem cells
Du Guo-jia, Chen Xiao-hong, Zhu Guo-hua, Fan Yan-dong, Wang Yun, Dang Mu-ren
- Department of Neurosurgery, the First Affiliated Hospital of Xinjiang Medical University, Urumqi 830054, Xinjiang Uygur Autonomous Region, China
-
Online:2013-10-01Published:2013-10-31 -
Contact:Dang Mu-ren, M.D., Chief physician, Doctoral supervisor, Department of Neurosurgery, the First Affiliated Hospital of Xinjiang Medical University, Urumqi 830054, Xinjiang Uygur Autonomous Region, China damrjab@163.com -
About author:Du Guo-jia★, Master, Attending physician, Department of Neurosurgery, the First Affiliated Hospital of Xinjiang Medical University, Urumqi 830054, Xinjiang Uygur Autonomous Region, China dgrjav@163.com -
Supported by:the Youth Science Foundation of Xinjiang Uygur Autonomous Region, No. 2011211B31*
CLC Number:
Cite this article
Du Guo-jia, Chen Xiao-hong, Zhu Guo-hua, Fan Yan-dong, Wang Yun, Dang Mu-ren. In vitro isolation, culture and identification of adipose-derived stem cells[J]. Chinese Journal of Tissue Engineering Research, 2013, 17(40): 7054-7060.
share this article
Morphology of cultured cells Under the inverted phase contrast microscope, primarily cultured adipose-derived stem cells were round or oval, adherent cells were spindle-shaped and cells were proliferated to form colonies in fish-like or spiral arrangement. After primary culture for 8-10 days, more than 90% of cells were confluent. After passage, fusiform cells appeared and grew fast with the same shape and size, and no significant difference in the morphology was found between different cell generations (Figure 1)."
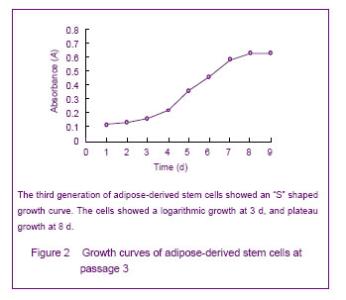
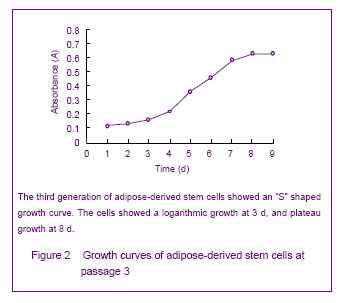
Growth curve of cultured adipose-derived stem cells Growth curves obtained by MTT assay showed active adipose-derived stem cells with strong proliferation ability were in logarithmic growth. At 1 or 2 days, the cells were in the incubation period; from the 3rd day, the cells began to enter the logarithmic phase, and reached a peak at day 8 followed by entering the plateau phase. The cell growth curve was in “S” shape, shown in Figure 2."
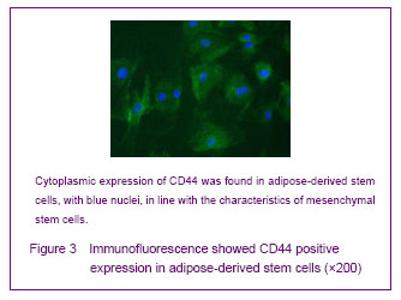
Cultured adipose-derived mesenchymal stem cells possess stem cell properties Immunofluorescence staining showed that adipose-derived stem cells strongly expressed CD44 that existed in the cytoplasm. Cell outline was clear shown by green fluorescence, nuclei were stained with 4,6-diamino-2-phenyl indole (blue), in line with mesenchymal stem cells properties (Figure 3)."

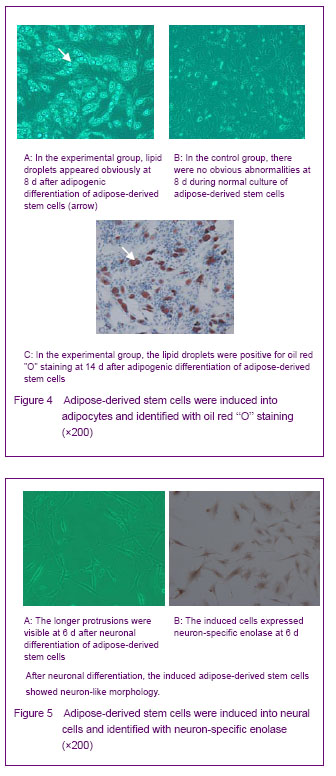
Adipogenic differentiation of cultured adipose-derived stem cells After adipogenic differentiation, the polygonal cells were seen. Small intracellular lipid droplets were visible at day 3, and became significantly larger at day 7, which are positive for oil red “O” staining. In the control group, no lipid droplets appeared (Figure 4). Neuronal differentiation of cultured adipose-derived stem cells After neuronal differentiation, the cells were extended, and the longer protrusions formed and were mutually connected, all of which showed neuron-like cell morphology. Immunohistochemistry examination showed that the induced cells expressed neurons specific enolase (Figure 5)."
| [1]Rodbell M. Metabolism of isolated fat cells. I. Effects of hormones on glucose metabolism and lipolysis. J Biol Chem. 1964;239:375-380. [2]Zuk PA, Zhu M, Mizuno H, et al. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001;7(2):211-228. [3]Zuk PA, Zhu M, Ashjian P, et al. Human adipose tissue is a source of multipotent stem cells. Mol Biol Cell. 2002;13(12): 4279-4295. [4]Lin XH, Liu N, Xiao YC, et al. Effects of adipose-derived stem cells transplantation on the neuronal apoptosis and the expression of Bcl-2 and caspase-12 in the brain post focal cerebral ischemia in rats. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2011;27(1):40-43. [5]Graham SH, Chen J, Clark RS. Bcl-2 family gene products in cerebral ischemia and traumatic brain injury. J Neurotrauma. 2000;17(10):831-841. [6]Plesnila N. Role of mitochondrial proteins for neuronal cell death after focal cerebral ischemia. Acta Neurochir Suppl. 2004;89:15-19. [7]Ferrer I, Planas AM. Signaling of cell death and cell survival following focal cerebral ischemia: life and death struggle in the penumbra. J Neuropathol Exp Neurol. 2003;62(4): 329-339. [8]Cai YN, Yuan XD, Ou Y, et al. Apoptosis duringβ-mercaptoethanol-induced differentiation of adult adipose-derived stromal cells into neurons. Neural Regen Res. 2011;6(10):750-755. [9]Gao X, Dong T, Li Z Q, et al. Survival of human adipose-derived mesenchymal stem cells transplanted via the tail vein in rat brain with cerebral ischemia. Zhongguo Zuzhi Gongchang Yanjiu. 2012;16(27):5067-5071. [10]The Ministry of Science and Technology of the People’s Republic of China. Guidance Suggestions for the Care and Use of Laboratory Animals. 2006-09-30. [11]Stanford WL, Caruana G, Vallis KA, et al. Expression trapping: identification of novel genes expressed in hematopoietic and endothelial lineages by gene trapping in ES cells. Blood. 1998;92(12):4622-4631. [12]Forrai A, Robb L. The gene trap resource: a treasure trove for hemopoiesis research. Exp Hematol. 2005;33(8): 845-856. [13]Stanford WL, Cohn JB, Cordes SP. Gene-trap mutagenesis: past, present and beyond. Nat Rev Genet. 2001;2(10): 756-768. [14]Joyner AL, Auerbach A, Skarnes WC. The gene trap approach in embryonic stem cells: the potential for genetic screens in mice. Ciba Found Symp. 1992;165:277-297. [15]Clay TM, Custer MC, Spiess PJ, et al. Potential use of T cell receptor genes to modify hematopoietic stem cells for the gene therapy of cancer. Pathol Oncol Res. 1999;5(1):3-15. [16]Filippi MD, Sainteny F. Hematopoietic differentiation of embryonic stem cells in mice: a model to study the biology of hematopoiesis. Transfus Clin Biol. 2001;8(1):6-16. [17]Kitajima K, Takeuchi T. Mouse gene trap approach: identification of novel genes and characterization of their biological functions. Biochem Cell Biol. 1998;76(6): 1029-1037. [18]Conquet F. Inactivation in vivo of metabotropic glutamate receptor 1 by specific chromosomal insertion of reporter gene lacZ. Neuropharmacology. 1995;34(8):865-870. [19]Guan C, Ye C, Yang X, et al. A review of current large-scale mouse knockout efforts. Genesis. 2010;48(2):73-85. [20]Araki K, Yamamura K. Genetic screening for novel genes by insertional mutagenesis with gene trap method in ES cells. Tanpakushitsu Kakusan Koso. 1995;40(14): 2017- 2024. [21]Vasileiskii SS. Fetal and embryonic hemoglobins. Sov J Dev Biol. 1972;3(1):27-32. [22]Schnütgen F. Generation of multipurpose alleles for the functional analysis of the mouse genome. Brief Funct Genomic Proteomic. 2006;5(1):15-18. [23]Reubinoff BE, Pera MF, Fong CY, et al. Embryonic stem cell lines from human blastocysts: somatic differentiation in vitro. Nat Biotechnol. 2000;18(4):399-404. [24]Kørbling M, Estrov Z. Adult stem cells for tissue repair - a new therapeutic concept? N Engl J Med. 2003;349(6): 570-582. [25]Mitchell JB, McIntosh K, Zvonic S, et al. Immunophenotype of human adipose-derived cells: temporal changes in stromal-associated and stem cell-associated markers. Stem Cells. 2006;24(2):376-385. [26]Qin YW, Zhao JM, Su W, et al. Isolation, culture and identification of rat adipose-derived stem cells. Zhongguo Zuzhi Gongcheng Yanjiu yu Linchuang Kangfu. 2011; 15(23):4242-4246. [27]Choi YS, Dusting GJ, Stubbs S, et al. Differentiation of human adipose-derived stem cells into beating cardiomyocytes. J Cell Mol Med. 2010;14(4):878-889. [28]Fang B, Song Y, Lin Q, et al. Human adipose tissue-derived mesenchymal stromal cells as salvage therapy for treatment of severe refractory acute graft-vs.-host disease in two children. Pediatr Transplant. 2007;11(7):814-817. [29]Campagnoli C, Roberts IA, Kumar S, et al. Identification of mesenchymal stem/progenitor cells in human first-trimester fetal blood, liver, and bone marrow. Blood. 2001;98(8): 2396-2402. [30]Schieker M, Pautke C, Haasters F, et al. Human mesenchymal stem cells at the single-cell level: simultaneous seven-colour immunofluorescence. J Anat. 2007;210(5):592-599. [31]Zhou MH, Zhang XW, Li L. Research of rat's adipose-derived stem cells induced differentiation into adipogenic cells. Shiyong Fuchan Ke Zazhi. 2011;27(7): 515-518. [32]Cheng WD, Zhao Y, He JS, et al. Adipogenic and osteogenic potential of rat adipose tissue-derived stromal cells in vitro. Anhui Yike Daxue Xuebao. 2007;42(5): 501-504. [33]Yang XF, HE X, He J, et al. Human adipose-derived stem cells into adipocytes directed differentiation. Jilin Daxue Xuebao: Yixue Ban. 2009;35(6):987-991. [34]Kirchner S, Kieu T, Chow C, et al. Prenatal exposure to the environmental obesogen tributyltin predisposes multipotent stem cells to become adipocytes. Mol Endocrinol. 2010; 24(3):526-539. [35]Li HX, Luo X, Liu RX, et al. Roles of Wnt/beta-catenin signaling in adipogenic differentiation potential of adipose-derived mesenchymal stem cells. Mol Cell Endocrinol. 2008;291(1-2):116-124. [36]Liu Q, Cen L, Zhou H, et al. The role of the extracellular signal-related kinase signaling pathway in osteogenic differentiation of human adipose-derived stem cells and in adipogenic transition initiated by dexamethasone. Tissue Eng Part A. 2009;15(11):3487-3497. [37]Ashjian PH, Elbarbary AS, Edmonds B, et al. In vitro differentiation of human processed lipoaspirate cells into early neural progenitors. Plast Reconstr Surg. 2003;111(6): 1922-1931. |
| [1] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [2] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [3] | Tian Chuan, Zhu Xiangqing, Yang Zailing, Yan Donghai, Li Ye, Wang Yanying, Yang Yukun, He Jie, Lü Guanke, Cai Xuemin, Shu Liping, He Zhixu, Pan Xinghua. Bone marrow mesenchymal stem cells regulate ovarian aging in macaques [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 985-991. |
| [4] | Hui Xiaoshan, Bai Jing, Zhou Siyuan, Wang Jie, Zhang Jinsheng, He Qingyong, Meng Peipei. Theoretical mechanism of traditional Chinese medicine theory on stem cell induced differentiation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1125-1129. |
| [5] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [6] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1137-1142. |
| [7] | Hou Jingying, Guo Tianzhu, Yu Menglei, Long Huibao, Wu Hao. Hypoxia preconditioning targets and downregulates miR-195 and promotes bone marrow mesenchymal stem cell survival and pro-angiogenic potential by activating MALAT1 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1005-1011. |
| [8] | Zhou Ying, Zhang Huan, Liao Song, Hu Fanqi, Yi Jing, Liu Yubin, Jin Jide. Immunomodulatory effects of deferoxamine and interferon gamma on human dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1012-1019. |
| [9] | Liang Xuezhen, Yang Xi, Li Jiacheng, Luo Di, Xu Bo, Li Gang. Bushen Huoxue capsule regulates osteogenic and adipogenic differentiation of rat bone marrow mesenchymal stem cells via Hedgehog signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1020-1026. |
| [10] | Wang Jifang, Bao Zhen, Qiao Yahong. miR-206 regulates EVI1 gene expression and cell biological behavior in stem cells of small cell lung cancer [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1027-1031. |
| [11] | Liu Feng, Peng Yuhuan, Luo Liangping, Wu Benqing. Plant-derived basic fibroblast growth factor maintains the growth and differentiation of human embryonic stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1032-1037. |
| [12] | Wen Dandan, Li Qiang, Shen Caiqi, Ji Zhe, Jin Peisheng. Nocardia rubra cell wall skeleton for extemal use improves the viability of adipogenic mesenchymal stem cells and promotes diabetes wound repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1038-1044. |
| [13] | Zhu Bingbing, Deng Jianghua, Chen Jingjing, Mu Xiaoling. Interleukin-8 receptor enhances the migration and adhesion of umbilical cord mesenchymal stem cells to injured endothelium [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1045-1050. |
| [14] | Luo Xiaoling, Zhang Li, Yang Maohua, Xu Jie, Xu Xiaomei. Effect of naringenin on osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1051-1056. |
| [15] | Wang Xinmin, Liu Fei, Xu Jie, Bai Yuxi, Lü Jian. Core decompression combined with dental pulp stem cells in the treatment of steroid-associated femoral head necrosis in rabbits [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1074-1079. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||