Chinese Journal of Tissue Engineering Research ›› 2013, Vol. 17 ›› Issue (32): 5877-5882.doi: 10.3969/j.issn.2095-4344.2013.32.020
Previous Articles Next Articles
Differentiation potential of umbilical cord blood-derived mesenchymal stem cells
Chen Ming-xing, Ouyang Gui-fang
- Medical School of Ningbo University, Ningbo 315000, Zhejiang Province, China
-
Received:2012-10-27Revised:2012-12-18Online:2013-08-06Published:2013-08-06 -
Contact:Ouyang Gui-fang, Master’s supervisor, Chief physician, Medical School of Ningbo University, Ningbo 315000, Zhejiang Province, China ouyangguifang@medmail.com.cn -
About author:Chen Ming-xing★, Master, Medical School of Ningbo University, Ningbo 315000, Zhejiang Province, China cmxcmx2008@yahoo.com.cn -
Supported by:the Natural Science Foundation of Ningbo City, No. 2010C50019*
CLC Number:
Cite this article
Chen Ming-xing, Ouyang Gui-fang. Differentiation potential of umbilical cord blood-derived mesenchymal stem cells[J]. Chinese Journal of Tissue Engineering Research, 2013, 17(32): 5877-5882.
share this article
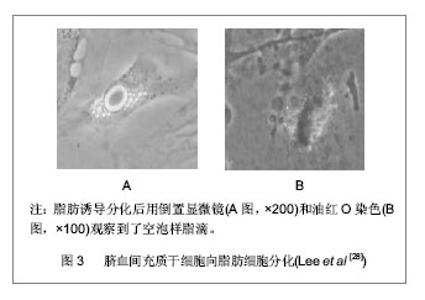
2.1 脐血来源间充质干细胞的分离培养及其生物学特性 脐血的采集:选择健康产妇,胎儿娩出后,在距胎儿5-7 cm的脐带处进行结扎,剪断脐带,穿刺脐静脉,用含抗凝保护液的采血袋抽取50 mL左右的脐血,采集后对脐血进行生物学检测、细菌污染检查、血型及HLA分型检测、血清学病毒检查以及遗传病检测。 4 ℃冰箱保存,4 h内处理标本。 脐血间充质干细胞的分离:脐血间充质干细胞的研究开展较晚,直到1999年,Erices等从脐血中分离培养出间充质干细胞后,相继有学者也分别从脐血中成功分离出间充质干细胞并对之进行鉴定。但培养成功率较低,可能与间充质干细胞在脐血中的出现频率低相关。 细胞形态呈均一的长梭形,具有贴壁生长的特点;适当诱导培养条件下,具有谱系内多向分化的特点和跨系分化的潜能;表达CD13、CD29、CD44、CD54、CD58、CD90、CD95、CD105、CD166等免疫表型,不表达CD14、CD34、CD40、CD45、CD50、CD68、CD80、CD86、CD117和CD152等免疫表型,强阳性表达抗原标记SH-2、SH-3、SH-4(人间充质干细胞特异性抗体),同时表达HLA-ABC等抗原和胶原Ⅰ,不表达HLA-DR抗原、共刺激分子B7-1(CD80)、B7-2(CD86)、CD40/ CD40L以及胶原Ⅱ、Ⅲ[9-10];细胞多处于G0/G1期,具有良好的增殖分化能力,简便易行的分离、纯化程序,而且能分泌一些造血细胞因子,具有促进造血的作用[11];作为一种原始的免疫缺陷细胞,具有良好的免疫耐受性[9],在体外培养既不能刺激也不能抑制淋巴细胞反应[12],同时具有非常好的免疫节作用,能预防移植物抗宿主病,其在造血干细胞移植中显示出了其优越性[13-14]。 2.2 脐血来源充质干细胞的分化潜能 2.2.1 脐血来源间充质干细胞的成骨分化潜能 成骨分化潜能:脐血来源间充质干细胞在含有体积分数为10%的胎牛血清+10 mmol/L β-甘油磷酸+ 10-7 mol/L地塞米松和0.2 mmol/L维生素的成骨诱导培养基培养下能发生成骨分化并表达碱性磷酸酶、Ⅰ型胶原、骨黏连蛋白、Cbfa-1和骨钙蛋白,同时高度表达成骨标记骨桥蛋白。尽管低浓度的细胞外基质层黏蛋白、纤连蛋白、胶原促进细胞贴壁和细胞生长,但是这些条件并不是成骨祖细胞生长的充分条件。 骨桥蛋白常常用来证实成骨发生的状态,它在骨中高表达;然而它同样出现在其他非骨组织中[15]。在一系列基因表达分析骨髓间充质干细胞中钙粘连蛋白mRNA水平非常高,非诱导骨髓间充质干细胞和脐血来源间充质干细胞能显著地表达糖蛋白。基质钙沉淀是成骨分化过程中的最新阶段,钙粘连蛋白称为终端分化的一个标记物[16]。Plant和Tobias等研究了成骨细胞分化,在20-24 d诱导分化后观察到了骨钙蛋白和骨黏连蛋白中等水平的表达结果。虽然碱性磷酸酶好像是一种成骨发生很好的标记,但是却存在着许多争议,Chang等[17]认为碱性磷酸酶在骨诱导下能显著增加。目前有关研究证实诱导间充质干细胞分化为成成骨系的基因是非常复杂的[18],仍然需要做进一步研究。 脐血来源间充质干细胞成骨分化时的影响因素:目前脐血间充质干细胞的培养主要建立在骨髓源间充质干细胞培养的经验上。而关于诱导分化条件的信息还是很匮乏。在脐血间充质干细胞的分化过程中,培养基的条件对其有重大影响。Hildebrandt等[19]用3种不同成分的培养基(alpha MEM+体积分数为15%胎牛血清,DMEM+体积分数为15%胎牛血清,MSCGM+10%SingleQuot),同时用两种不同浓度的地塞米松对脐血间充质干细胞培养。结果发现MSCGM+10%SingQuot能促进成骨分化;细胞外基质钙化沉淀只出现在含有10-8 mol/L地塞米松+骨形态发生蛋白2的培养基里;地塞米松浓度为10-7 mol/L能产生更高浓度的细胞外基质和在加有骨形态发生蛋白2时脐血源间充质干细胞独立表达Ⅰ型胶原产物。根据以上实验数据表明地塞米松是成骨分化诱导因子,而骨形态发生蛋白2有支持脐血间充质干细胞的作用,MSCGM+10%SingQuot+10-7 mol/L地塞米松是诱导成骨分化最好的培养条件。有人研究表明成骨诱导培养基只有加了维生素D或者维生素D+成纤维细胞生长因子9之后骨钙蛋白标记物才会明显增多[20]。此外通过碱性磷酸酶活性染色证实了瘦蛋白能加强成骨分化的能力[21],且CD105对下调脐血间充质干细胞成骨分化有非常密切的关系。有证据表明磷酸甘油酸酯变位酶、热休克蛋白27等在脐血间充质干细胞成骨分化中是上调的[22],在体外成骨分化过程中,白细胞介素6、白细胞介素9、白细胞介素10、白细胞介素12、成纤维生长因子的含量是比较多的,随着进一步分化这些因子会减少。尤其在成骨分化过程中白细胞介素6水平的高低对成骨分化是有影响的,据报道白细胞介素6能促进骨吸收和破骨形成[23-24]。Mekala等[25]研究发现维生素C能促进脐血间充质干细胞的成骨分化,维生素的浓度与脐血间充质干细胞的增殖和分化起着至关重要作用。Liu 等[26]证实了部分脱矿物质骨基质能促进脐血间充质干细胞的成骨分化。 2.2.2 脐血来源间充质干细胞的脂肪分化潜能 脂肪分化潜能:Goodwin等[27]和Lee等[28]发现脐血间充质干细胞在脂肪诱导培养基条件下可以分化成脂肪细胞,见图3。并证明脐血中确实含有能分化成脂肪细胞的间充质干细胞。这些脂肪细胞表达过氧化物酶增殖激活受体Gamma 2(PPAR Gamma 2)、低密度脂蛋白和脂肪酸结合蛋白[1],同时能表达脂肪酸相关蛋白4(FABP-4)[29]。然而Kern等[30]研究当中未见脐血间充质干细胞发生脂肪分化,而其成骨分化、软骨分化与报道一致。一直以来研究者报道脐血间充质干细胞的脂肪分化结果不一致,其脂肪分化需要进一步研究。"
| [1] Pittenger MF, Mackay AM, Beck SC, et al. Multilineage potential of adult human mesenchymal stem cells.Science. 1999;284(5411):143-147.[2] Kim S, Min WK, Chun S, et al. Protein expression profiles during osteogenic differentiation of mesenchymal stem cells derived from human umbilical cord blood.Tohoku J Exp Med. 2010;221(2):141-150.[3] Kao IT, Yao CL, Chang YJ,et al. Chondrogenic differentiation of human mesenchymal stem cells from umbilical cord blood in chemically synthesized thermoreversible polymer.Chin J Physiol. 2008;51(4):252-258.[4] Kedong S, Xiubo F, Tianqing L,et al. Simultaneous expansion and harvest of hematopoietic stem cells and mesenchymal stem cells derived from umbilical cord blood. J Mater Sci Mater Med. 2010;21(12):3183-3193.[5] Roura S, Farré J, Hove-Madsen L, et al. Exposure to cardiomyogenic stimuli fails to transdifferentiate human umbilical cord blood-derived mesenchymal stem cells. Basic Res Cardiol. 2010;105(3):419-430.[6] Lim JY, Park SI, Oh JH, et al. Brain-derived neurotrophic factor stimulates the neural differentiation of human umbilical cord blood-derived mesenchymal stem cells and survival of differentiated cells through MAPK/ERK and PI3K/Akt- dependent signaling pathways.J Neurosci Res. 2008;86(10): 2168-2178.[7] Kang XQ, Zang WJ, Bao LJ, et al. Fibroblast growth factor-4 and hepatocyte growth factor induce differentiation of human umbilical cord blood-derived mesenchymal stem cells into hepatocytes.World J Gastroenterol. 2005;11(47): 7461-7465.[8] Phuc PV, Nhung TH, Loan DT, et al. Differentiating of banked human umbilical cord blood-derived mesenchymal stem cells into insulin-secreting cells. In Vitro Cell Dev Biol Anim. 2011; 47(1):54-63.[9] Wang M, Yang Y, Yang D,et al.The immunomodulatory activity of human umbilical cord blood-derived mesenchymal stem cells in vitro. Immunology. 2009;126(2):220-232.[10] Rojewski MT, Weber BM, Schrezenmeier H. Phenotypic Characterization of Mesenchymal Stem Cells from Various Tissues.Transfus Med Hemother. 2008;35(3):168-184.[11] Hayashi N, Takahashi K, Abe Y, et al. Placental/umbilical cord blood-derived mesenchymal stem cell-like stromal cells support hematopoietic recovery of X-irradiated human CD34+ cells. Life Sci. 2009;84(17-18):598-605.[12] Tong CK, Seow HF, Ramasamy R. Cord blood-derived mesenchymal stem cell does not stimulate nor inhibits T lymphocytes activation. Med J Malaysia. 2008;63 Suppl A:77-78. [13] MacMillan ML, Weisdorf DJ, Brunstein CG, et al. Acute graft-versus-host disease after unrelated donor umbilical cord blood transplantation: analysis of risk factors. Blood. 2009; 113(11):2410-2415.[14] Nishiyama N, Miyoshi S, Hida N, et al.The significant cardiomyogenic potential of human umbilical cord blood- derived mesenchymal stem cells in vitro.Stem Cells. 2007; 25(8):2017-2024. [15] Butler WT.The nature and significance of osteopontin. Connect Tissue Res. 1989;23(2-3):123-136.[16] Rebelatto CK, Aguiar AM, Moretão MP, et al.Dissimilar differentiation of mesenchymal stem cells from bone marrow, umbilical cord blood, and adipose tissue. Exp Biol Med (Maywood). 2008;233(7):901-913.[17] Chang YJ, Shih DT, Tseng CP, et al. Disparate mesenchyme- lineage tendencies in mesenchymal stem cells from human bone marrow and umbilical cord blood.Stem Cells. 2006;24(3): 679-685.[18] Harris SE, Guo D, Harris MA, et al.Transcriptional regulation of BMP-2 activated genes in osteoblasts using gene expression microarray analysis: role of Dlx2 and Dlx5 transcription factors. Front Biosci. 2003;8:s1249-1265.[19] Hildebrandt C, Büth H, Thielecke H. Influence of cell culture media conditions on the osteogenic differentiation of cord blood-derived mesenchymal stem cells.Ann Anat. 2009; 191(1):23-32.[20] Toai TC, Thao HD, Thao NP,et al. In vitro culture and differentiation of osteoblasts from human umbilical cord blood. Cell Tissue Bank. 2010;11(3):269-280.[21] Chang YJ, Shih DT, Tseng CP, et al. Disparate mesenchyme- lineage tendencies in mesenchymal stem cells from human bone marrow and umbilical cord blood.Stem Cells. 2006; 24(3):679-685.[22] Kim JS, Lee HK, Kim MR, et al. Differentially expressed proteins of mesenchymal stem cells derived from human cord blood (hUCB) during osteogenic differentiation. Biosci Biotechnol Biochem. 2008;72(9):2309-2317.[23] de Grooth R, Kawilarang-de Haas EW, van de Sande-Rijkers CM, et al.The role of osteoblast density and endogenous interleukin-6 production in osteoclast formation from the hemopoietic stem cell line FDCP-MIX C2GM in coculture with primary osteoblasts. Calcif Tissue Int. 1998;63(1):57-62.[24] Theoleyre S, Wittrant Y, Tat SK, et al. The molecular triad OPG/RANK/RANKL: involvement in the orchestration of pathophysiological bone remodeling.Cytokine Growth Factor Rev. 2004;15(6):457-475.[25] Mekala NK, Baadhe RR, Parcha SR,et al. Enhanced Proliferation and Osteogenic Differentiation of Human Umbilical Cord Blood Stem Cells by L-Ascorbic acid, in vitro.Curr Stem Cell Res Ther. 2012. [Epub ahead of print][26] Liu G, Li Y, Sun J, et al. In vitro and in vivo evaluation of osteogenesis of human umbilical cord blood-derived mesenchymal stem cells on partially demineralized bone matrix.Tissue Eng Part A. 2010;16(3):971-982.[27] Goodwin HS, Bicknese AR, Chien SN,et al. Multilineage differentiation activity by cells isolated from umbilical cord blood: expression of bone, fat, and neural markers. Biol Blood Marrow Transplant. 2001;7(11):581-588.[28] Lee OK, Kuo TK, Chen WM,et al. Isolation of multipotent mesenchymal stem cells from umbilical cord blood. Blood. 2004;103(5):1669-1675.[29] Hussain I, Magd SA, Eremin O, et al. New approach to isolate mesenchymal stem cell (MSC) from human umbilical cord blood. Cell Biol Int. 2012;36(7):595-600.[30] Kern S, Eichler H, Stoeve J,et al. Comparative analysis of mesenchymal stem cells from bone marrow, umbilical cord blood, or adipose tissue.Stem Cells. 2006;24(5):1294-1301.[31] Zhang X, Hirai M, Cantero S, et al. Isolation and characterization of mesenchymal stem cells from human umbilical cord blood: reevaluation of critical factors for successful isolation and high ability to proliferate and differentiate to chondrocytes as compared to mesenchymal stem cells from bone marrow and adipose tissue.J Cell Biochem. 2011;112(4):1206-1218.[32] Sibov TT, Severino P, Marti LC, et al. Mesenchymal stem cells from umbilical cord blood: parameters for isolation, characterization and adipogenic differentiation. Cytotechnology. 2012;64(5):511-521.[33] J Jiang Y, Jahagirdar BN, Reinhardt RL,et al. Pluripotency of mesenchymal stem cells derived from adult marrow.Nature. 2002;418(6893):41-49.[34] Toma C, Pittenger MF, Cahill KS, et al. Human mesenchymal stem cells differentiate to a cardiomyocyte phenotype in the adult murine heart.Circulation. 2002;105(1):93-98.[35] Kadivar M, Khatami S, Mortazavi Y,et al. In vitro cardiomyogenic potential of human umbilical vein-derived mesenchymal stem cells.Biochem Biophys Res Commun. 2006;340(2):639-647. [36] Rosca AM, Burlacu A. Effect of 5-azacytidine: evidence for alteration of the multipotent ability of mesenchymal stem cells.Stem Cells Dev. 2011;20(7):1213-1221.[37] Yan X, Lv A, Xing Y, et al. Inhibition of p53-p21 pathway promotes the differentiation of rat bone marrow mesenchymal stem cells into cardiomyocytes. Mol Cell Biochem. 2011;354 (1-2):21-28.[38] Qian Q, Qian H, Zhang X,et al. 5-Azacytidine induces cardiac differentiation of human umbilical cord-derived mesenchymal stem cells by activating extracellular regulated kinase.Stem Cells Dev. 2012;21(1):67-75.[39] Rosca AM, Burlacu A. Effect of 5-azacytidine: evidence for alteration of the multipotent ability of mesenchymal stem cells.Stem Cells Dev. 2011;20(7):1213-1221.[40] Leor J, Guetta E, Chouraqui P, et al. Human umbilical cord blood cells: a new alternative for myocardial repair. Cytotherapy. 2005;7(3):251-257.[41] Shetty P, Cooper K, Viswanathan C. Comparison of proliferative and multilineage differentiation potentials of cord matrix, cord blood, and bone marrow mesenchymal stem cells. Asian J Transfus Sci. 2010;4(1):14-24.[42] Henning RJ, Abu-Ali H, Balis JU, et al. Human umbilical cord blood mononuclear cells for the treatment of acute myocardial infarction. Cell Transplant. 2004;13(7-8):729-739.[43] Ma N, Stamm C, Kaminski A, et al. Human cord blood cells induce angiogenesis following myocardial infarction in NOD/scid-mice.Cardiovasc Res. 2005;66(1):45-54.[44] Henning RJ, Abu-Ali H, Balis JU, et al. Human umbilical cord blood mononuclear cells for the treatment of acute myocardial infarction.Cell Transplant. 2004;13(7-8):729-739.[45] Jäger M, Degistirici O, Knipper A, et al. Bone healing and migration of cord blood-derived stem cells into a critical size femoral defect after xenotransplantation.J Bone Miner Res. 2007;22(8):1224-1233.[46] Liu GP, Li YL, Sun J, et al. Repair of calvarial defects with human umbilical cord blood derived mesenchymal stem cells and demineralized bone matrix in athymic rats. Zhonghua Zheng Xing Wai Ke Za Zhi. 2010;26(1):34-38.[47] Rosada C, Justesen J, Melsvik D, et al. The human umbilical cord blood: a potential source for osteoblast progenitor cells.Calcif Tissue Int. 2003;72(2):135-142.[48] Jang BJ, Byeon YE, Lim JH, et al. Implantation of canine umbilical cord blood-derived mesenchymal stem cells mixed with beta-tricalcium phosphate enhances osteogenesis in bone defect model dogs.J Vet Sci. 2008;9(4):387-393.[49] Rios CG,Beth McCarthy M,Arciero C,et al.Biologics in Shoulder Surgery: The Role of Adult Mesenchymal Stem Cells in Tendon Repair.Techniques in Orthopaedics. 2007; 22(1):2-9.[50] Tisato V, Naresh K, Girdlestone J, et al. Mesenchymal stem cells of cord blood origin are effective at preventing but not treating graft-versus-host disease. Leukemia. 2007;21(9): 1992-1999.[51] Hayashi N, Takahashi K, Abe Y,et al. Placental/umbilical cord blood-derived mesenchymal stem cell-like stromal cells support hematopoietic recovery of X-irradiated human CD34+ cells.Life Sci. 2009;84(17-18):598-605.[52] Cornetta K, Laughlin M, Carter S,et al. Umbilical cord blood transplantation in adults: results of the prospective Cord Blood Transplantation (COBLT). Biol Blood Marrow Transplant. 2005;11(2):149-160. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [3] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [4] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [5] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [6] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [7] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| [8] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [9] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [10] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [11] | Kong Desheng, He Jingjing, Feng Baofeng, Guo Ruiyun, Asiamah Ernest Amponsah, Lü Fei, Zhang Shuhan, Zhang Xiaolin, Ma Jun, Cui Huixian. Efficacy of mesenchymal stem cells in the spinal cord injury of large animal models: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1142-1148. |
| [12] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [13] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [14] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [15] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||